Abstract
Objectives:
Tracking trends in the testing of latent tuberculosis infection (LTBI) can help measure tuberculosis elimination efforts in the United States. The objectives of this study were to estimate (1) the annual number of persons tested for LTBI and the number of LTBI tests conducted, by type of test and by public, private, and military sectors, and (2) the cost of LTBI testing in the United States.
Methods:
We searched the biomedical literature for published data on private-sector and military LTBI testing in 2013, and we used back-calculation to estimate public-sector LTBI testing. To estimate costs, we applied Medicare-allowable reimbursements in 2013 by test type.
Results:
We estimated an average (low-high) 13.3 million (11.3-15.4 million) persons tested for LTBI and 15.3 million (12.9-17.7 million) LTBI tests, of which 13.2 million (11.1-15.3 million) were tuberculin skin tests and 2.1 million (1.8-2.4 million) were interferon-γ release assays (IGRAs). Eighty percent of persons tested were in the public sector, 18% were in the private sector, and 2% were in the military. Costs of LTBI tests and of chest radiography totaled $314 million (range, $256 million to $403 million).
Conclusions:
To achieve tuberculosis elimination, millions more persons will need to be tested in all sectors. By targeting testing to only those at high risk of tuberculosis and by using more specific IGRA tests, the incidence of tuberculosis in the United States can be reduced and resources can be more efficiently used.
The reported number of tuberculosis disease cases in 2017 (n = 9105) was the lowest since national tuberculosis surveillance began in 1953 in the United States. 1 However, the tuberculosis case rate was 28 times that needed to eliminate tuberculosis, defined as <1 new case per 1 million population annually, which is a goal of the Centers for Disease Control and Prevention (CDC). 2 Mathematical models indicate that greatly expanded treatment of latent tuberculosis infection (LTBI) is needed to make substantial progress toward tuberculosis elimination. 3,4
Recent improvements in testing for LTBI include the use of interferon-γ release assays (IGRAs), which are conducted by laboratories from a whole blood specimen, 5 rather than tuberculin skin tests (TSTs), which require 1 visit with a health care worker for the test to be placed and another visit 2 or 3 days later to have the test read. Improvements in treatment include a treatment regimen of 12 weekly doses of isoniazid and rifapentine 6 rather than 9 months of isoniazid (240 doses). It is important to track data on the volume, types, and costs of LTBI testing over time so that national, state, and local tuberculosis programs can plan activities for, estimate resources for, and assess progress toward tuberculosis elimination. Three major sectors of the US health care system conduct LTBI testing: the private sector, the public sector, and the military. Estimating LTBI testing by these 3 sectors can help tuberculosis programs tailor tuberculosis elimination interventions for the populations served by these sectors.
The objective of our analysis was to estimate the annual number of persons tested for tuberculosis and the number of LTBI tests conducted, by type of test, sector (public, private, and military), and testing cost, in the United States.
Methods
We used data on LTBI testing in the United States from 3 sources. We chose calendar year 2013 because that was the most recent common year for which data on published estimates for the private sector and military sector were available.
We obtained data on private-sector LTBI testing from persons who submitted claims through private insurance by type of test from the 2013 IBM MarketScan Commercial Claims database. 7 Because the MarketScan database included approximately 25% of the privately insured US population in 2013, we extrapolated the results to the United States by multiplying published estimates by 4.
We obtained published data on the number of persons tested and the number of LTBI tests conducted in 2013 among active-component military service members from a report by the Armed Forces Health Surveillance Center. 8
We obtained data on public-sector LTBI testing by using a study that surveyed 244 clinics (general public health, HIV, pediatric, refugee, migrant, jail/prison, homeless, drug/alcohol, and private practice) where ≥10 patients were treated for LTBI in 2002 in the catchment area of 19 sites of the Tuberculosis Epidemiologic Studies Consortium. 9 The Tuberculosis Epidemiologic Studies Consortium is a CDC-funded consortium of public health tuberculosis programs and universities that conducts programmatically relevant research to prevent and control tuberculosis. Sterling et al 9 derived low and high estimates of persons who initiated LTBI treatment, giving greater credibility to the low estimate because it was based on the number of tuberculosis cases. We assumed that the nearly 80% of testing in general public health clinics plus the remaining clinic categories minus 2.1% in private-practice clinics represented public-sector testing regardless of type of health insurance. We then back-calculated public-sector testing by using statistics of contacts of tuberculosis patients, reported to CDC by US tuberculosis programs for all 50 states and the District of Columbia (unpublished data, CDC, Aggregate Reports for Program Evaluation, December 18, 2017), and from a US representative sample tested for LTBI during 2011-2012 that had TST results from the National Health and Nutrition Examination Survey (NHANES). 10
Number tested for LTBI =
Where:
X = US estimate of number treated for LTBI (low: 291 000; high: 433 000)
Y = estimated proportion of persons who tested LTBI positive who started LTBI treatment
Z = estimated proportion of persons who tested positive for LTBI
Low number tested for LTBI =
We calculated the average of low and high estimates, and we conducted a sensitivity analysis by applying the 95% confidence intervals (CIs) from NHANES TST positivity results (95% CI, 3.4%-6.3%). 10 Lastly, we estimated the number of public-sector tests from the number of persons tested in the public sector by applying the military ratio 8 of number of tests to number of persons tested (1.15), which was from a complete capture of testing. We assumed that the proportions of types of tests used was similar to that found in the private sector (86% by TST, 13% by QuantiFERON-TB Gold In-Tube [QFT-GIT (Qiagen, Germantown, MD)], and 1% by T-SPOT [Oxford Immunotec USA Inc, Marlborough, MA]). 7
We applied Medicare-allowable reimbursements in 2013 by test type to estimate LTBI testing costs: TST, $8.64 11 ; QFT-GIT, $85.20 12 ; T-SPOT, $102.99. 12 For total cost estimates of LTBI screening, we included 2-view chest radiographs ($30.96) 12 to rule out tuberculosis disease among persons who tested LTBI positive.
Results
We estimated that an average (low-high) 13.3 million (11.3-15.4 million) persons were tested for LTBI in 2013 in the private, public, and military sectors combined (Table 1). An estimated 13.2 million (11.1-15.3 million) TSTs and 2.1 million (1.8-2.4 million) IGRAs (QFT-GIT or T-SPOT) were conducted. Eighty percent of persons tested were in the public sector, 18% were in the private sector, and 2% were in the military. The estimated cost of LTBI testing was $295 million ($249-$341 million) in 2013 dollars (Table 2). Adding the costs of chest radiography raised estimates to $314 million ($265-$364 million).
Estimated number of persons tested for latent tuberculosis infection (LTBI), by type of health insurance, United States, 2013
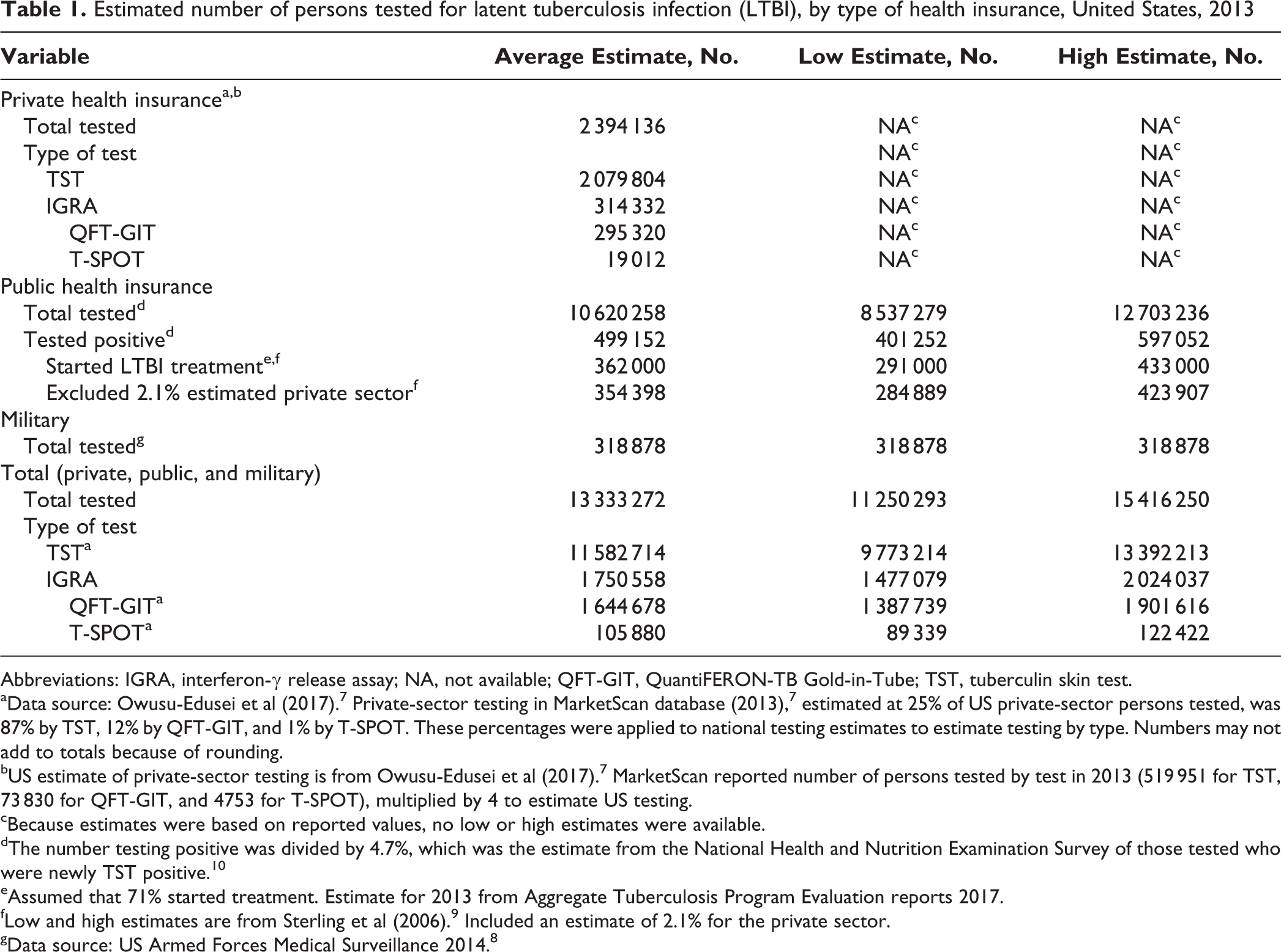
Abbreviations: IGRA, interferon-γ release assay; NA, not available; QFT-GIT, QuantiFERON-TB Gold-in-Tube; TST, tuberculin skin test.
aData source: Owusu-Edusei et al (2017). 7 Private-sector testing in MarketScan database (2013), 7 estimated at 25% of US private-sector persons tested, was 87% by TST, 12% by QFT-GIT, and 1% by T-SPOT. These percentages were applied to national testing estimates to estimate testing by type. Numbers may not add to totals because of rounding.
bUS estimate of private-sector testing is from Owusu-Edusei et al (2017). 7 MarketScan reported number of persons tested by test in 2013 (519 951 for TST, 73 830 for QFT-GIT, and 4753 for T-SPOT), multiplied by 4 to estimate US testing.
cBecause estimates were based on reported values, no low or high estimates were available.
dThe number testing positive was divided by 4.7%, which was the estimate from the National Health and Nutrition Examination Survey of those tested who were newly TST positive. 10
eAssumed that 71% started treatment. Estimate for 2013 from Aggregate Tuberculosis Program Evaluation reports 2017.
fLow and high estimates are from Sterling et al (2006). 9 Included an estimate of 2.1% for the private sector.
gData source: US Armed Forces Medical Surveillance 2014. 8
Estimated number of tests for latent tuberculosis infection (LTBI) conducted in the United States, by sector, 2013
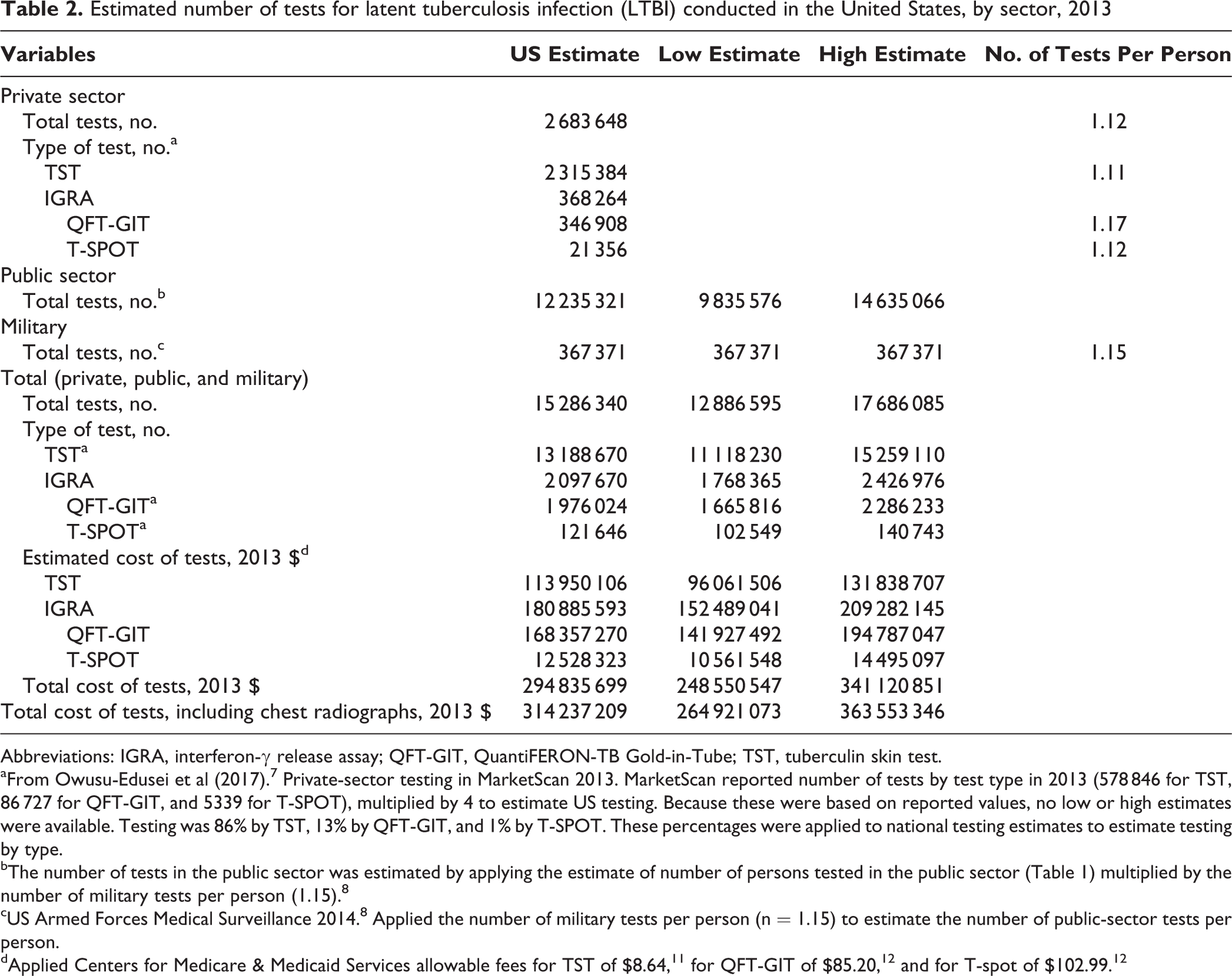
Abbreviations: IGRA, interferon-γ release assay; QFT-GIT, QuantiFERON-TB Gold-in-Tube; TST, tuberculin skin test.
aFrom Owusu-Edusei et al (2017). 7 Private-sector testing in MarketScan 2013. MarketScan reported number of tests by test type in 2013 (578 846 for TST, 86 727 for QFT-GIT, and 5339 for T-SPOT), multiplied by 4 to estimate US testing. Because these were based on reported values, no low or high estimates were available. Testing was 86% by TST, 13% by QFT-GIT, and 1% by T-SPOT. These percentages were applied to national testing estimates to estimate testing by type.
bThe number of tests in the public sector was estimated by applying the estimate of number of persons tested in the public sector (Table 1) multiplied by the number of military tests per person (1.15). 8
cUS Armed Forces Medical Surveillance 2014. 8 Applied the number of military tests per person (n = 1.15) to estimate the number of public-sector tests per person.
In a sensitivity analysis applying the lower and upper CIs of TST positivity, an average of 10.6 million to 17.4 million persons were tested, and an average of 12.2 million to 20.0 million tests were conducted. The average cost of LTBI tests ranged from $235 million to $385 million; including chest radiography, the average cost ranged from $256 million to $403 million.
Discussion
We estimated that health care providers engaged in a massive effort to test millions of persons for LTBI in 2013. Although we estimated that the public sector conducted 80% of the LTBI testing during that year, we expect that the proportion might change as private-sector primary care and congregate-setting (both public and private) providers test more populations at increased risk for tuberculosis disease, prompted by updated US Preventive Services Task Force (USPSTF) recommendations in 2016. 13 Increases in health insurance coverage resulting from Medicaid expansion through health care reform instituted in 2014 might also have expanded public-sector LTBI testing and treatment. Many persons with HIV or persons who are homeless and were previously uninsured now have health insurance, which provides access to LTBI testing and treatment. 14,15 Moreover, public-sector testing has likely increased since 2013, as state and local health departments fund additional LTBI testing and treatment efforts in an attempt to accelerate tuberculosis elimination. 16,17
Although we estimated LTBI testing by test type in 2013, health care providers are transitioning from using TSTs to using IGRAs, which are now recommended by CDC, the Infectious Diseases Society of America, and the American Thoracic Society as the preferred LTBI test for persons aged >5 years for whom testing is warranted and who have a low-to-intermediate risk of disease progression. 18 The proportion of LTBI testing by IGRA has likely increased since 2013. Evidence shows increased specificity (ie, fewer false-positive tests) and sensitivity (ie, more true-positive tests) of IGRAs compared with TSTs. 13 With an anticipated increase in the use of IGRA testing, LTBI testing costs may have increased because of the higher cost per test of IGRA, and the number of persons treated may have decreased because of the enhanced test specificity of IGRA. Tracking the volume, types, and costs of LTBI testing by sector and the resources spent on testing over time should help tuberculosis programs plan for and assess efforts toward tuberculosis elimination. We provided a baseline national estimate of LTBI testing and its costs for 2013 for comparison with estimates in later years.
In the absence of national reporting to CDC on LTBI testing by type of test and on LTBI diagnoses, we assembled data from various sources to arrive at our best estimate of annual LTBI testing in the United States for 2013, the year for which estimates were commonly available from military and private sectors. Although all US tuberculosis programs are required to report to CDC on tuberculosis testing of persons who have had recent contact with patients with tuberculosis disease, they are not required to report to CDC targeted LTBI testing data on other populations at high risk for tuberculosis, except for local area data from special studies. Sixteen US states have recently instituted LTBI reporting to the state (personal communication, Adam Langer, CDC, Division of Tuberculosis Elimination, March 2018), but it is a work in progress to ascertain the number and characteristics of those tested, the types of tests being used, and testing results. Because billing for the TST (at a reimbursement of $9 per test) is not routine in most public health tuberculosis clinics, counting the annual number of TSTs is not simple. As more providers use IGRAs to test for LTBI, the options to assess their use, including obtaining data from laboratories, increase. However, providers use both public laboratories and private laboratories, which are not compelled to share data with CDC. CDC researchers have contacted companies that produce IGRAs and TSTs to see if they might be willing to share sales data on LTBI tests; however, sales do not equate to actual use, and private companies are not required to share their data. Moreover, ownership of 1 of the 2 IGRA manufacturers recently changed, and data from years before the current ownership are not available for analysis of sales trends.
We compared our estimates of the number of persons tested in 2013 with the results derived from a published 3 mathematical model (Andrew Hill, unpublished data, July 2014) (n = 11.1 million), after applying our assumptions about the percentage of persons with positive LTBI tests who started treatment (71%) and the percentage of persons tested who had positive LTBI results (4.7%). That estimate is slightly higher than the low estimate of 10.6 million from our sensitivity analysis, which was based on 6.3% LTBI positivity, but is lower than our “high” sensitivity estimate of 17.4 million, which was based on 3.4% LTBI positivity. Newer models could be used for additional comparisons. 4
Limitations
Our analysis had several limitations. First, the data source for private-sector testing did not provide data on national-level testing, but it was a large claims database 7 that included more than 43 million persons; however, it was a convenience sample of primarily large employers in large cities with the South slightly underrepresented. As such, it might have been a biased source from which to extrapolate national testing data. 7 Second, our estimates of public-sector testing did not include estimates of patients who had public health insurance (eg, Medicare, Medicaid, state/local) and were tested in private clinic settings; thus, costs of testing in the public sector and the number of tests conducted in the private sector were likely underestimated. Third, we assumed that levels of LTBI testing in the public sector in 2013 would be similar to levels of LTBI testing in 2002, when data were available. 9 Those public-sector estimates came from a convenience sample of participating US sites (urban, rural, areas with low and high tuberculosis burden) that represented approximately 10% of the US population and of the tuberculosis case burden, which could also be subject to bias. This large study of public-sector testing was funded by CDC, involved a lot of staffing and labor, and is the only known source of such data; as such, it is not likely to be repeated soon. Fourth, publicly funded health departments provided the bulk of LTBI treatment in the United States and had not, until recently, routinely billed Medicare, Medicaid, or third-party health insurance for LTBI testing and treatment. From a survey of local health departments in 2014, only 36.5% billed both public insurers and private insurers for tuberculosis services, 8.2% billed only public insurers, and 1.4% billed only private insurers. 19 As such, health insurance claims data could not be used to comprehensively estimate public-sector testing. Fifth, by not excluding patients with private insurance from our public-sector cost estimates, we likely overestimated these costs. However, increased LTBI testing after the updated USPSTF recommendation might result in higher estimates of public-sector testing and costs since 2016. Sixth, testing among military reserves and in the Veterans’ Health Administration system was not included in military estimates; as a result, the total number of LTBI tests and their costs were underestimated. Seventh, our application of proportion of test types in the private sector to test types in the public sector and military sector may have resulted in bias. Eighth, we assumed a high rate of LTBI treatment initiation (71%) based on a study of management of US contacts to infectious tuberculosis cases (CDC, Aggregate Reports for Program Evaluation, December 18, 2017). 10 Using a lower treatment initiation rate would have increased estimates of LTBI testing. Finally, we estimated only direct medical costs of testing (inclusive of test antigen/assay, laboratory processing, and staff member time), which excluded the patient time spent traveling to and from the clinic and waiting at the clinic before testing.
Conclusions
Millions of persons were tested for LTBI in 2013, mostly by the public sector, at a minimum estimate of approximately $250 million. However, to achieve tuberculosis elimination, millions more will need to be tested by all sectors. By targeting testing to only those at high risk of tuberculosis and by using more specific IGRA tests, US tuberculosis incidence can be reduced and resources can be more efficiently used. Our estimates of LTBI testing in 2013 can be compared with future LTBI testing and cost estimates to help tuberculosis programs plan for and assess efforts toward tuberculosis elimination.
Footnotes
Authors’ Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
