Abstract
Objectives:
One strategy to increase the uptake of human papillomavirus (HPV) vaccine among adolescents is through the use of pharmacists. Our objectives were to (1) use a publicly available database to describe the statutory and regulatory authority of pharmacists to administer the HPV vaccine in the United States and (2) discuss how the current status of laws may influence achievement of the Healthy People 2020 goal of 80% HPV vaccination rate for teenagers aged 13-15.
Methods:
Using information from the Centers for Disease Control and Prevention’s (CDC’s) Public Health Law Program database, we identified state laws in effect as of January 1, 2016, giving pharmacists authority to administer vaccines. We used a standardized analysis algorithm to determine whether states’ laws (1) authorized pharmacists to administer HPV vaccine, (2) required third-party authorization for pharmacist administration, and (3) restricted HPV vaccine administration by pharmacists to certain patient age groups.
Results:
Of 50 states and the District of Columbia, 40 had laws expressly granting pharmacists authority to administer HPV vaccine to patients, but only 22 had laws that authorized pharmacists to vaccinate preadolescents aged 11 or 12 (ie, the CDC-recommended age group). Pharmacists were granted prescriptive authority by 5 states, and they were given authority pursuant to general (non–patient-specific) third-party authorization (eg, a licensed health care provider) by 32 states or patient-specific third-party authorization by 3 states.
Conclusions:
Most states permitted pharmacists to administer HPV vaccines only to boys and girls older than 11 or 12, which may hinder achievement of the Healthy People 2020 goal for HPV vaccination. Efforts should be made to strengthen the role of pharmacists in addressing this public health issue.
Keywords
Human papillomavirus (HPV), a sexually transmitted infection, causes many cervical and penile cancers and is 1 of 2 main causes of oropharyngeal cancer (the other is cigarette smoking). 1,2 The most common types of HPV can be prevented through immunization and screening; however, it is important that immunization occurs before patients become sexually active, because evidence suggests that the vaccine is substantially less effective after exposure to the virus. The Advisory Committee on Immunization Practices recommends beginning the 3-dose HPV vaccination series at age 11 or 12 for boys and girls. 3
Vaccination rates vary by state, and no state has achieved the Healthy People 2020 goal of an 80% HPV vaccination rate for teenagers aged 13-15. 4 In 2014, HPV vaccination coverage among states ranged from 20.1% to 56.9% for girls aged 13-17 and 9.0% to 42.9% for boys aged 13-17. 5 Possible reasons for low rates of HPV vaccine uptake include the reluctance of some parents and health care providers to discuss vaccination of preadolescents for sexually transmitted diseases, 6 misconceptions about susceptibility to HPV and associated cancers, 7,8 inadequate access to the vaccine, 9 and the fact that >1 vaccine dose is required. To help address the problems with access to HPV vaccination, the 2014 President’s Cancer Panel recommended that “states should enact laws and implement policies that allow pharmacists to administer vaccines to adolescents, including young adolescents.” 9
Expanding the authority of pharmacists to administer HPV vaccines could help improve HPV vaccination rates in several ways. It would substantially increase the number of vaccination providers, thereby enhancing availability and convenience for patients. 10,11 The extended operating hours of community and retail pharmacies would permit busy parents and patients to access HPV vaccinations on the way home from work, after school, or on weekends. 10,12,13 In addition, pharmacist-driven interventions, including press releases, flyers, and personalized letters, could be used to increase public awareness of the importance and availability of HPV vaccination. 14 –18
Some health care providers resist expanding the vaccination authority of pharmacists because of concerns that vaccinations at pharmacies might disrupt regular office visits, primary care providers might not be notified of vaccinations because of inadequate documentation and reporting by pharmacists, and some pharmacists may not have adequate vaccination-related training. 18 –20 Political resistance to HPV vaccination is another issue. Some interest groups are concerned that the health protection provided by the HPV vaccine will promote sexual promiscuity in adolescents. In 2007, opponents of HPV vaccination convinced the Texas state legislature to overturn an executive order to require HPV vaccination. 21 Yet, whereas some interest groups are opposed to mandating the vaccine, most have supported administration of the vaccine. Laws expanding the HPV vaccination authority of pharmacists have not faced the same resistance as other HPV-related laws, such as those requiring HPV vaccination for school enrollment. 22
The current literature is not sufficiently robust to rigorously assess whether and to what extent pharmacists are authorized by states to administer HPV vaccines. As a result, no studies have examined the impact of pharmacist vaccination laws on HPV vaccination rates, particularly among preadolescents. In 2012, Brewer et al assessed the authority of pharmacists to administer the HPV vaccine by surveying members of jurisdiction pharmacy associations, boards of pharmacy, and college of pharmacy faculty members. 23 In 2015, the American Pharmacists Association (APhA) and the National Alliance of State Pharmacy Associations (NASPA) reported on state laws granting pharmacists HPV vaccination authority, but their data, collection methods, and legal sources were not made available to the public. 24 The methods and reporting used by these 2 groups raised questions about the reliability and validity of their results. 25
To address the need for more information about the potential role of pharmacists in HPV vaccination, we sought to identify a robust, publicly available database containing information about state laws (statutes and regulations) pertaining to pharmacist vaccination authority (and specifically to pharmacist authority to administer HPV vaccine). Our objectives were to (1) use this database to determine the current status of pharmacist vaccination administration authority (including to what extent and under which conditions pharmacists are authorized to administer HPV vaccine), (2) ascertain how this authority is affected by vaccination age restrictions, and (3) discuss how the current status of these laws may influence achievement of the Healthy People 2020 HPV vaccination goals for adolescents.
Methods
We extracted the data for our study from CDC’s Public Health Law Program database, which contains state laws (statutes and regulations) from all 50 states and the District of Columbia and is hosted on the LawAtlas platform. 26 Researchers in CDC’s Public Health Law Program created and regularly update this database by detailing pharmacist HPV vaccination laws and using systematic and transparent data collection techniques, 26,27 as well as validated methods as set forth by Anderson et al. 25 When we accessed the database, it included state laws in effect on or before January 1, 2016. We found that it contained information about 219 unique legal attributes pertaining to pharmacist vaccination, including levels of and criteria for pharmacist vaccination authority and vaccination age restrictions. 26,27
In the database, we identified state laws that gave pharmacists authority to administer vaccines, establish third-party vaccination authorization requirements, stipulate vaccination age restrictions, and describe vaccination practice requirements (including training, reporting, record keeping, notification, malpractice insurance, and emergency exceptions). Although the database contained vaccination laws as far back as 1971, we excluded from our study any law that was not in effect on January 1, 2016.
We identified 41 unique legal attributes that were in some way related to the administration of HPV vaccine. Some attributes were directly related to HPV vaccination, whereas others were related to vaccinations in general but had applications to HPV vaccination. When possible, we placed the generally worded legal provisions into categories by general legal attributes. For example, we placed legal provisions that permitted pharmacists to administer CDC-recommended vaccines into a “CDC vaccines” category, rather than putting them into multiple categories for each type of vaccine, including HPV vaccine. Later, we combined the general and HPV-specific legal attributes in our data set to yield HPV-specific results.
We applied a coding methodology to these laws based on a series of questions that were designed to capture the scope of the authority of pharmacists to administer vaccines. For each question, we determined whether the provision met the coding criteria by using the following response options or codes: yes, no, ambiguous, not mentioned, or not applicable. 26 In the data set that we created from the CDC database, we used 3 variables to identify the types of pharmacist vaccination authority provided in laws, which we called “authorization types”: (1) prescriptive authorization (eg, pharmacist able to prescribe, no third-party authorization required), (2) general third-party written authorization (eg, a standing order that is not patient specific), and (3) patient-specific third-party written authorization (eg, a prescription by a licensed health care provider). In addition, we built the data set so that it contained legal attributes specifying various vaccination age restrictions. For example, some state laws permitted pharmacists to administer HPV vaccine but only to adult patients.
Next, using our data set, we evaluated legal attributes pertaining to pharmacist vaccination authorization types to determine the type of pharmacist HPV vaccination authority provided in each law (Figure 1). Using a standardized analysis algorithm, we considered pharmacists to be authorized to administer HPV vaccines when any of the following scenarios were expressly addressed in the law: (1) vaccination authority (prescriptive, general [non–patient specific], or patient specific) was not restricted by type of vaccine; (2) vaccination authority was restricted by type of vaccine, but HPV vaccine was expressly allowed (ie, HPV vaccine referenced by name); or (3) vaccination authority was restricted by type of vaccine, but vaccination authority was allowed for CDC-recommended vaccines. 28 If we found that a state did not have laws authorizing pharmacist-administered vaccinations and instead had laws allowing pharmacists to administer “drugs,” we did not consider these drugs to include vaccines, even if the states defined drugs as substances intended for the “prevention of disease” or to “affect the structure or any function of the body” and could be considered to include vaccines. 29 –32
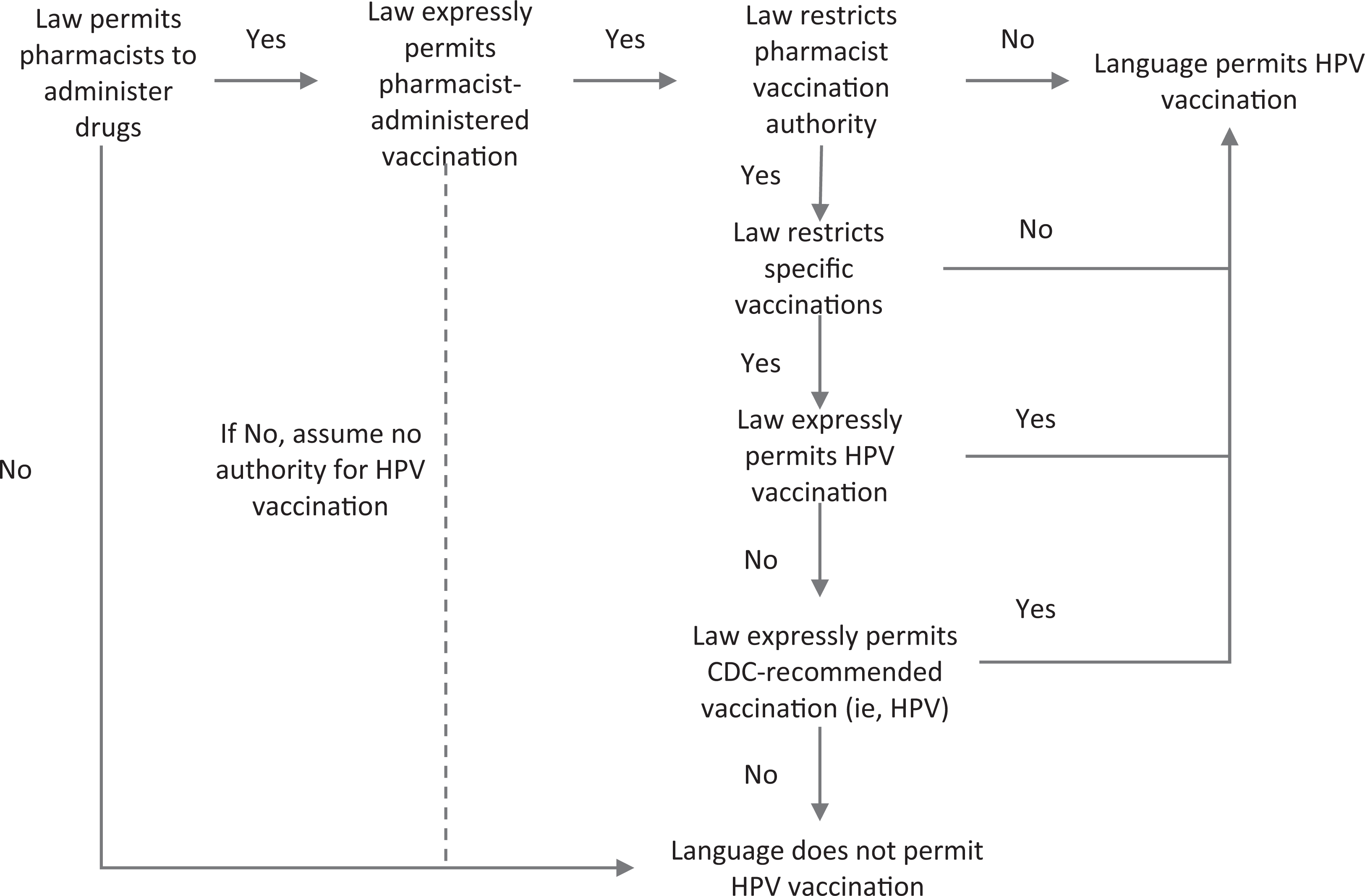
Standardized analysis algorithm used to determine whether a state law authorizes pharmacists to administer human papillomavirus (HPV) vaccines, applied to laws from all 50 states and the District of Columbia in the Centers for Disease Control and Prevention’s (CDC’s) Public Health Law Program database and in effect on January 1, 2016. Drugs are defined as substances intended for the “prevention of disease” or to “affect the structure or any function of the body.” 29 –32 Laws in Alabama, Mississippi, Tennessee, and Washington allowed pharmacists to do “drug administration.” Although this wording could reasonably be interpreted as authorizing pharmacist vaccination, 33 the CDC database consistently interpreted this wording narrowly, as not authorizing pharmacist vaccination.
Similarly, we evaluated legal attributes pertaining to patient vaccination age restrictions to determine patient age restrictions on pharmacist HPV vaccination authority in each law (Figure 2). We used the following standardized analysis algorithm in each pharmacist HPV vaccination authorization type (as described previously): (1) if pharmacist vaccination authority was expressly limited to a patient age, we used that minimum age; (2) if pharmacist vaccination was expressly allowed and patient age limits did not vary by the type of vaccine, we used that minimum age; (3) if patient age limits varied according to whether the vaccine was influenza or noninfluenza but did not mention HPV, we used the minimum age allowed for the noninfluenza vaccine; and (4) if the type of vaccine was restricted and HPV was not explicitly mentioned but CDC-recommended vaccines were allowed, we followed the logic described previously and used the minimum age allowed for CDC-recommended vaccines.
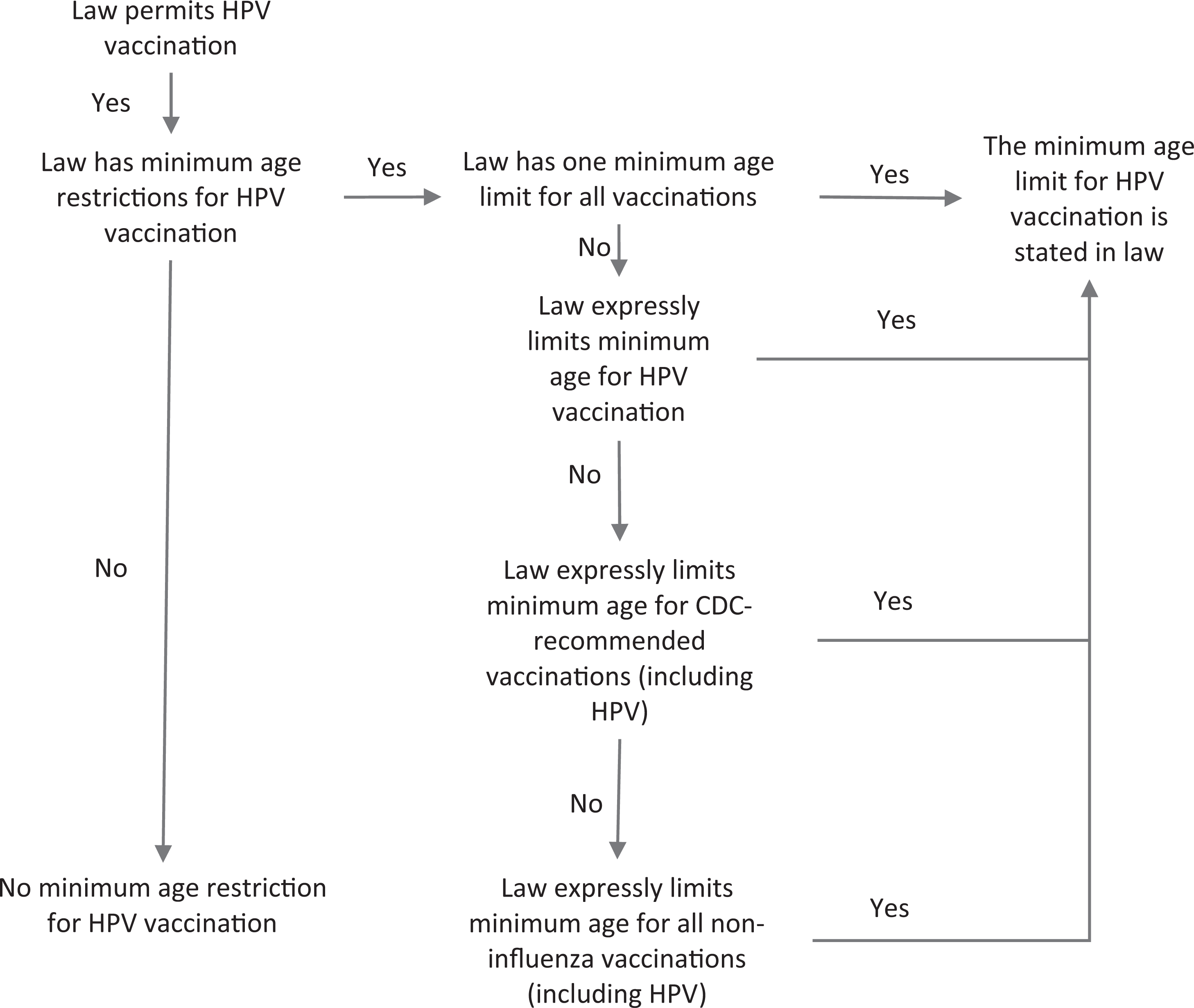
Standardized analysis algorithm used to determine minimum patient-age restrictions for human papillomavirus (HPV) vaccination, applied to laws from all 50 states and the District of Columbia in the Centers for Disease Control and Prevention’s (CDC’s) Public Health Law Program database and in effect on January 1, 2016.
Results
Pharmacist Authority to Vaccinate Against HPV
Of the 51 jurisdictions (50 states and District of Columbia), 47 had laws expressly authorizing pharmacists to administer vaccines (Table). The 4 states lacking laws authorizing pharmacist-administered vaccinations (Alabama, Mississippi, Tennessee, and Washington) instead had laws allowing pharmacists to administer “drugs.”
Types of authorizations allowing pharmacists to administer HPV vaccines, by state, based on laws from all 50 states and the District of Columbia in CDC’s public health law program database and in effect on January 1, 2016a
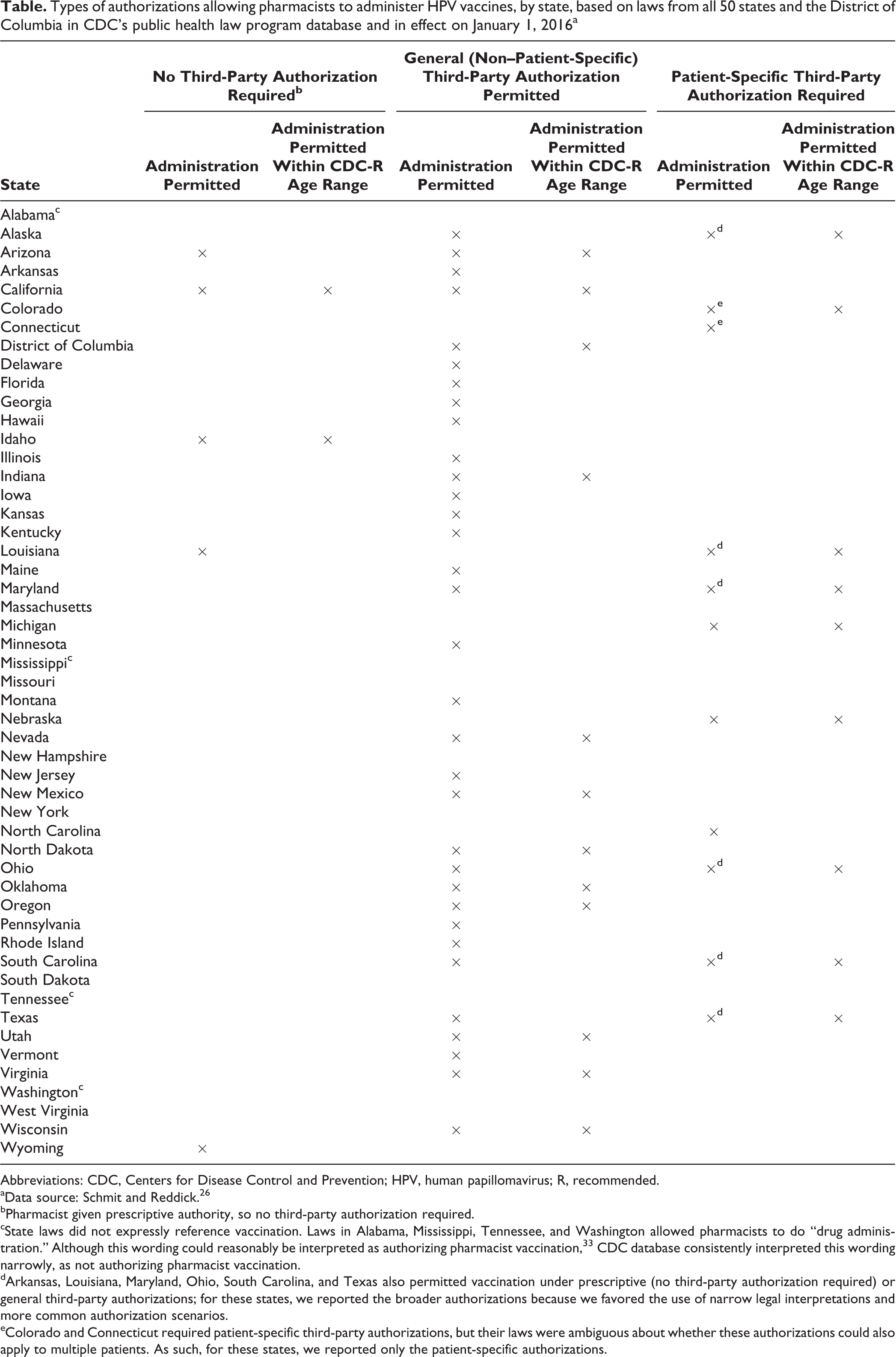
Abbreviations: CDC, Centers for Disease Control and Prevention; HPV, human papillomavirus; R, recommended.
aData source: Schmit and Reddick. 26
bPharmacist given prescriptive authority, so no third-party authorization required.
cState laws did not expressly reference vaccination. Laws in Alabama, Mississippi, Tennessee, and Washington allowed pharmacists to do “drug administration.” Although this wording could reasonably be interpreted as authorizing pharmacist vaccination, 33 CDC database consistently interpreted this wording narrowly, as not authorizing pharmacist vaccination.
dArkansas, Louisiana, Maryland, Ohio, South Carolina, and Texas also permitted vaccination under prescriptive (no third-party authorization required) or general third-party authorizations; for these states, we reported the broader authorizations because we favored the use of narrow legal interpretations and more common authorization scenarios.
eColorado and Connecticut required patient-specific third-party authorizations, but their laws were ambiguous about whether these authorizations could also apply to multiple patients. As such, for these states, we reported only the patient-specific authorizations.
Of the 51 jurisdictions, 40 had laws that expressly authorized pharmacists to administer HPV vaccinations. Of these 40 states, 5 (Arizona, California, Idaho, Louisiana, and Wyoming) granted pharmacists prescriptive authority to administer HPV vaccines (so no third-party authorization was required), 32 permitted pharmacists to administer HPV vaccines pursuant to general (non–patient-specific) third-party authorizations, and 3 (Michigan, Nebraska, and North Carolina) required that pharmacists have a patient-specific third-party authorization for HPV vaccinations (ie, prescriptive or general third-party authorizations were not permitted for other types of vaccinations). Colorado and Connecticut required patient-specific third-party authorization, but their laws were ambiguous about whether these authorizations could also apply to multiple patients.
Minimum Patient Age Restrictions for Pharmacist-Administered HPV Vaccines
Of the 35 states permitting pharmacists to vaccinate without patient-specific authorizations (eg, with prescriptive or general third-party authorizations), 16 (Alaska, Arkansas, Delaware, Florida, Hawaii, Iowa, Kansas, Maine, Maryland, Montana, New Jersey, Pennsylvania, Rhode Island, South Carolina, Vermont, and Wyoming) allowed HPV vaccination for only those aged ≥18. Louisiana authorized HPV vaccination for only those aged ≥17; Illinois, Kentucky, and Texas allowed HPV vaccination for only those aged ≥14; and Minnesota and Ohio authorized HPV vaccination for only those aged ≥13. Idaho allowed HPV vaccination for those aged ≥12 (under a pharmacist’s prescriptive authority), and the remaining 12 states authorized HPV vaccination of patients aged ≥11. These 12 states and Idaho authorized HPV vaccination of preadolescents aged 11 or 12 without patient-specific authorization. Only California and Idaho authorized HPV vaccination under a pharmacist’s prescriptive authority pursuant to CDC recommendations.
Discussion
Pharmacist Authority to Administer HPV Vaccine
Our review of laws in effect on January 1, 2016, identified 22 states that authorized pharmacists to administer HPV vaccines to patients who were the CDC-recommended age of 11 or 12 (under at least 1 of the 3 authorization types) and an additional 18 states that authorized pharmacists to administer HPV vaccines but only to those at older ages. States whose laws do not permit pharmacists to administer HPV vaccines at all or within the CDC-recommended age range create a potential barrier to meeting the Healthy People 2020 goal of an 80% HPV vaccination rate for those aged 13-15. 4 The focus on vaccinating within the CDC-recommended age of 11 or 12 makes sense, because more than half of all adolescents are sexually active by age 18 and >10% have their first sexual encounter before age 15. 34 If states place minimum age restrictions on who pharmacists may vaccinate, younger patients and their parents may have 1 fewer resource to access for HPV vaccination before beginning sexual activity. As a result, HPV vaccination rates may be less likely to rise; HPV may be more likely to spread; and the number of oral, cervical, and penile cancer cases may be more likely to grow. However, giving pharmacists authority to administer HPV vaccines, including to preadolescents, may help reverse these trends. It is for this reason that the President’s Cancer Panel, in a 2014 report, recommended that pharmacists be authorized to vaccinate younger adolescents. 9
In addition to age restrictions that may reduce patient access to pharmacists for HPV vaccination, limitations based on the type of authority granted to pharmacists may further reduce patient access. In our study, only 5 states allowed a trained and certified pharmacist to prescribe and administer HPV vaccination. Conversely, 30 states authorized HPV vaccination by a pharmacist only under a protocol or agreement with a third-party provider (usually a physician or other health care provider), who gives the pharmacist permission to administer HPV vaccine to a specified patient group. The terms of these protocols or agreements may deviate from CDC’s recommended HPV vaccination schedule or may even exclude the HPV vaccine altogether as approved for administration. Furthermore, 5 states required patients to obtain a prescription before receiving HPV vaccination from a pharmacist, which creates the need for patients to see a prescribing health care provider before receiving HPV vaccination from a pharmacist. In these states, some patients may find it more convenient to get the HPV vaccination directly from their health care provider (rather than obtaining the prescription and then going to a pharmacist); however, other patients may be less likely to pursue HPV vaccination if it requires a potentially more costly or inconvenient provider office visit. As such, laws requiring third-party or patient-specific authorization for pharmacist HPV vaccination services may create a barrier to HPV vaccination for some patients. In contrast, laws authorizing pharmacists to prescribe and administer the HPV vaccine may be one way to help improve HPV vaccination rates.
Another way that states might enhance access to pharmacist-provided HPV vaccination would be to tie pharmacist vaccination authority to CDC vaccination recommendations. In our study, 22 states already permitted pharmacist HPV vaccination based on more general laws allowing pharmacists to administer all CDC-recommended vaccines. One particular strength of this approach is that it ensures that pharmacist HPV vaccination practice is supported by the latest evidence on vaccine safety and efficacy according to CDC. Additionally, this approach could be combined with public education about the special training and reporting protocols required of pharmacists who can administer vaccines, with the intent of increasing patient trust and acceptance of pharmacist-prescribed and pharmacist-administered vaccination. The legislative strategy of linking pharmacist HPV vaccination authority to CDC recommendations is something that states not currently doing this could use, potentially resulting in an additional increase in patient access to HPV vaccination through pharmacists.
Contrast With Other Legal Analyses
At first glance, some of our findings related to pharmacist HPV vaccination authority were similar to those described by the authors of the 2012 Brewer survey 23 and the 2015 APhA/NASPA report. 24 For example, we found that 22 states allowed pharmacists to administer HPV vaccines to patients aged ≤12, and the APhA/NASPA report identified 23 states that did so. We found that all but 11 states had laws expressly allowing a pharmacist to provide HPV vaccines under some situations; the Brewer survey found this to be the case in all but 10 states; and the APhA/NASPA report noted that all but 5 states had such laws.
However, a closer look identified substantial differences between our findings and those of the 2012 Brewer survey and the 2015 APhA/NASPA report. For example, whereas the Brewer survey found that Georgia, Missouri, and South Dakota had laws allowing a pharmacist to provide HPV vaccines and the APhA/NASPA report found the same thing in Georgia, Massachusetts, Missouri, and South Dakota, we did not identify laws allowing pharmacists to administer HPV vaccine in any of these states.
Although some differences might be attributable to variations in researcher interpretation or to changes in laws among the dates of different investigations, these reasons do not explain all of the differences that we identified between our findings and those of others. For example, the Brewer survey found that pharmacists could administer HPV vaccines in Missouri, but we found a Missouri statute 35 in effect at the time of that survey that limited pharmacists to the administration of only “influenza, pneumonia, shingles and meningitis vaccines.” Furthermore, we found that that law was amended in 2014, 36 before the APhA/NASPA report, to authorize the administration of 5 additional vaccines (hepatitis A, hepatitis B, diphtheria, tetanus, and pertussis) but still did not authorize HPV vaccination. We reviewed the full text of the laws in effect at the times of the data collection by Brewer and APhA/NASPA, yet we were unable to find a basis for all of the discrepancies between our findings and theirs (full text for all laws is publicly available 26 ).
Some differences among the 3 studies may be attributable to the use of dissimilar methodologies. We used a regularly updated, publicly available, comprehensive CDC Public Health Law Program database, which contained state laws and used systematic and transparent data collection techniques, 26,27 as well as validated methods as set forth by Anderson et al. 25 Furthermore, Temple University’s Center for Public Health Law Research conducted independent evaluations and quality control investigations involving the CDC database, in which they compared samples from 2 secondary sources with random samples from a CDC data set. 37,38 They found only 2 coding differences between the CDC data set and secondary sources, and they determined that these differences were caused by variations in coding schemes, rather than by coding errors. 26
However, the Brewer survey determined the scope of pharmacist vaccination authority by interviewing 1 pharmacist spokesperson per jurisdiction. Although it is possible that some responses given by their interviewees were based on sources not included in our study (eg, informal agency guidance), they did not describe whether legal sources or authorities were used to support interviewee responses. Furthermore, the APhA/NASPA report did not describe their methodology, making it impossible to evaluate the reliability or validity of their findings. Specifically, we were unable to determine the following: (1) which types of laws or policies were analyzed (eg, statutes, regulations, pharmacy board policies), (2) which search terms were used to find the laws, (3) which criteria were used to evaluate the legal sources and authorities, or (4) whether the methods included checks on completeness, accuracy, reliability, and validity (as recommended by Anderson et al 25 ).
To assess whether pharmacist vaccination laws affect HPV vaccination rates, researchers need accurate legal data. 33 Unexplained differences in legal data obtained from various sources could confound future scientific evaluation efforts examining the public health impact of these pharmacist authority laws. The application of scientific methodologies to systematic legal research is a recent development, but it is likely to play a critical role in legal epidemiology and other fields that evaluate the impact of law on various issues. 33
Limitations
This study had several limitations. The CDC database uses a methodology that applies conservative, narrow interpretations to ambiguous or overlapping statutes and regulations. 27,28 For example, laws in Alabama, Mississippi, Tennessee, and Washington allowed pharmacists to do “drug administration.” Although this wording could reasonably be interpreted as authorizing a pharmacist to administer vaccines, 33 the CDC database consistently interpreted this wording narrowly, as not authorizing pharmacist vaccination.
We also used narrow interpretations when we evaluated CDC’s data for our study. Some state laws allowed pharmacists to vaccinate under some but not all 3 authorization types, and other state laws specified patient age limitations depending on the authorization type. For example, South Carolina allowed pharmacists to administer HPV vaccines to patients at age 12 if authorization was patient specific but otherwise only to adult patients in the case of general third-party authorization. We chose to narrowly interpret this type of authorization scenario. Because we believed pharmacists were much more likely to receive general third-party authorizations rather than patient-specific authorizations, for South Carolina we used the minimum patient age (ie, adult) associated with the general third-party authorization rather than the minimum age (ie, 12) associated with the patient-specific authority. Thus, whereas others may interpret similar legal data more broadly, we used narrow legal interpretations and focused on the most common authorization scenarios. Future research exploring the impact of laws related to vaccinations might investigate whether states with ambiguously worded laws have higher or lower vaccination rates than states with more specifically worded laws.
Conclusion
As of January 1, 2016, most states permitted pharmacists to administer HPV vaccines only to those older than the CDC-recommended age of 11 or 12, which may hinder achievement of the Healthy People 2020 goal of an 80% HPV vaccination rate for adolescents aged 13-15. Pharmacists could play an important role in addressing this public health issue, particularly if more states granted them authority to administer HPV vaccines to patients who are younger and not yet sexually active, according to CDC recommendations.
To determine how state laws on pharmacist vaccination authority affect vaccination rates, it will be necessary to use robust, publicly available databases and to collect legal data using transparent, reliable, and valid methodologies. We offer an example of the application of both. In the future, data sets similar to ours could be used to advance the understanding of the role that pharmacist vaccination laws have on health outcomes.
Footnotes
Authors’ Note
The CDC Public Health Law Program cannot provide legal advice on any issue and cannot represent any individual or entity in any matter. The Public Health Law Program recommends seeking the advice of an attorney or other qualified professional with questions about the application of law to a specific circumstance. The findings and conclusions in this article are those of the authors and do not necessarily represent the official views of CDC.
Acknowledgments
We thank Allison Reddick, Matthew Penn, and Tara Ramanathan for their contributions and insights.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant from the Robert Wood Johnson Foundation’s Public Health Law Research program and supported by the Oak Ridge Institute for Science and Education.
