Abstract
Objectives:
Drug-susceptibility testing (DST) of Mycobacterium tuberculosis is necessary for identifying drug-resistant tuberculosis, administering effective treatment regimens, and preventing the spread of drug-resistant tuberculosis. DST is recommended for all culture-confirmed cases of tuberculosis. We examined trends in delayed and unreported DST results in the Centers for Disease Control and Prevention’s National Tuberculosis Surveillance System.
Methods:
We analyzed culture-confirmed tuberculosis cases reported to the National Tuberculosis Surveillance System during 1993-2014 for annual trends in initial DST reporting for first-line antituberculosis drugs and trends in on-time, delayed, and unreported results. We defined on-time reporting as DST results received during the same calendar year in which the patient’s case was reported or ≤4 months after the calendar year ended and delayed reporting as DST results received after the calendar year. We compared cases with on-time, delayed, and unreported DST results by patient and tuberculosis program characteristics.
Results:
The proportion of cases with reported results for all first-line antituberculosis drugs increased during 1993-2011. Reporting of pyrazinamide results was lower than reporting of other drugs. However, during 2000-2012, of 134 787 tuberculosis cases reported to the National Tuberculosis Surveillance System, reporting was on time for 125 855 (93.4%) cases, delayed for 5332 (4.0%) cases, and unreported for 3600 (2.7%) cases.
Conclusions:
Despite increases in the proportion of cases with on-time DST results, delayed and unreported results persisted. Carefully assessing causes for delayed and unreported DST results should lead to more timely reporting of drug-resistant tuberculosis.
Tuberculosis (TB) is a substantial cause of morbidity and mortality within and outside the United States. 1,2 Multidrug-resistant TB, defined as TB resistant to at least isoniazid and rifampin, 2 is a global threat. In the United States, 1.0% to 1.5% of TB cases are multidrug-resistant TB. 3 Treatment regimens for drug-resistant TB have more side effects, are longer in duration, and can cost 100 times more than standard first-line TB treatment regimens. 3 –5 The Centers for Disease Control and Prevention (CDC) and the World Health Organization recommend making the prevention and control of drug-resistant TB a top priority for TB control programs. 6,7
Drug-susceptibility testing (DST) of Mycobacterium tuberculosis complex isolates is an essential tool in identifying drug-resistant cases, determining effective anti-TB drug regimens, and preventing the spread of drug-resistant TB. 4,5,8 In the United States, the American Thoracic Society and CDC recommend performing DST for first-line drugs (isoniazid, rifampin, ethambutol, and pyrazinamide) on the initial M tuberculosis isolate from each patient with culture-positive disease. 4 The National Tuberculosis Surveillance System (NTSS) is the main source of data on TB incidence rates and drug-resistance patterns in the United States and is an important source of data on clinical characteristics and risk factors among TB patients. NTSS helps guide national TB program priorities. Growth-based DST results have been included in NTSS since 1993, but culture-confirmed TB cases reported to CDC often have incomplete DST results. 1 To prevent and control drug-resistant TB, CDC recommends reducing the level of incomplete DST reporting and decreasing the time that it takes to report drug resistance to NTSS. 9 We examined DST reporting patterns and trends in NTSS to identify the factors relating to delayed and unreported results.
Methods
Study Population
We examined TB cases reported to NTSS during 1993-2014. TB cases are reported by the 50 state health departments and the District of Columbia via the Report of Verified Case of TB standard form. 1,10 The Report of Verified Case of TB form includes demographic, clinical, laboratory, and treatment outcome data. We restricted all analyses to cases with positive TB culture results because cultures are a prerequisite for performing DST on anti-TB medications. Of all reported TB cases, the proportion with a positive culture decreased from 81% in 1993 to 77% in 2014. 1 We analyzed DST results from the first cultured isolate for which DST was performed (ie, the initial DST). NTSS collects data only from conventional, growth-based DST and not from molecular DST.
Study Definitions
We defined complete DST reporting for a single anti-TB drug as being either resistant or susceptible. We considered missing or unknown results to be incomplete. We defined complete first-line DST reporting as having DST results for isoniazid, rifampin, and ethambutol. We excluded pyrazinamide from the definition of first-line DST for this study because it is difficult to perform pyrazinamide testing and because an estimated 11% of US laboratories do not routinely perform DST for pyrazinamide. 4,11 DST results are defined as those from growth-based assays on liquid or solid media. The guidelines for collecting DST results for NTSS did not change during the study period.
CDC asks state health departments to submit initial DST results on culture-confirmed cases within 2 months of submitting the initial Report of Verified Case of TB form to CDC or when DST results are available.
10
For our time-sensitive analyses, we categorized cases into 3 groups for first-line DST reporting: on time, delayed, or unreported. On time or within the expected time frame: DST results received during the same calendar year in which the patient’s case was reported or ≤4 months after the calendar year ended, when data for the previous year were frozen for analysis. For example, for all TB patients with confirmed cases in 1993, TB programs had until April 30, 1994, to report all initial DST results. Delayed: DST results received after the 4-month period after the year in which the patient’s case was reported. For example, for patients with confirmed TB cases in 1993, TB programs reported the initial DST results after April 30, 1994. Unreported: The initial DST results were never reported as of April 30, 2015, when data for all TB cases reported in 2014 and prior years were finalized by CDC. These data were the most recent available data at the time of our analysis.
We defined additional variables based on the standard Report of Verified Case of TB definitions. No health department involved in care meant that all care was provided by a non-health department provider (eg, private provider, hospital, correctional institution, or other health care provider that was not part of the state or local health department). We defined geographic regions by using US Census Bureau definitions. 12
NTSS data were collected as part of routine public health surveillance by local and state health departments. As such, approval by an institutional review board was not required, because we used existing surveillance data and the research did not involve human participants. The identities of people with TB are not reported to NTSS in accordance with section 308(d) of the Public Health Service Act (42 USC 242m). 13
Data Analysis
We examined 3 trends in DST reporting. We first examined the annual trend during 1993-2014 in the cumulative proportion of cases with DST reporting for each of the first-line anti-TB drugs by using the 2014 Report of Verified Case of TB data set. To determine how time since TB reporting affected DST result reporting, we grouped the cases by the year in which the case was reported, treated each year as a cohort, and examined the annual trend in the proportion of cases with complete combined first-line DST results (isoniazid, rifampin, and ethambutol combined). We used the start year of 2000 for this analysis because of the incompleteness of risk factor data before that time. Using the year cohorts, we examined the temporal trend during 2000-2014 in the proportion of cases with on-time, delayed, and unreported combined first-line DST results. We tested each trend for significance (P < .05) of the annual percentage change by using joinpoint regression analysis (Joinpoint Trend Analysis software version 4.2.0.2). 14
Additionally, for cases reported during 2000-2012, we compared those that had on-time DST reports with those that had delayed DST reports and those that had unreported DST results. We examined these 3 categories for differences in the quarter of year in which the case was reported as TB, the region where the case was reported, the state average annual TB case count during 2000-2012 for the state reporting the case, and patient demographic and clinical characteristics. We restricted these analyses to 2000-2012 because these were the most recent years with complete data for the patients’ providers. We set statistical significance at P < .01 because of the large sample size. We performed all analyses with the Cochran-Mantel-Haenszel χ2 test for odds ratios (ORs) and 99% confidence intervals (CI) with SAS version 9.3. 15
Results
During the early years of DST reporting, 1993-1996, initial DST result reporting increased significantly among culture-confirmed cases for isoniazid (annual percentage change, 3.0%) and rifampin (annual percentage change, 3.1%; Figure 1). However, during 2011-2014, reporting significantly decreased for isoniazid (annual percentage change, –0.7%) and rifampin (annual percentage change, –0.7%). Reporting DST results for ethambutol increased significantly in the early years (1993-1995, annual percentage change, 5.0%), plateaued during 1998-2011 (annual percentage change, 0.2%), and then declined significantly during 2011-2014 (annual percentage change, –0.6%). DST results for pyrazinamide significantly increased during 1993-2010 (1993-1997 annual percentage change, 14.6%; 1997-2010 annual percentage change, 2.3%) and plateaued during 2010-2014. Reported results for pyrazinamide had the largest absolute and proportionate increase during 1993-2014, compared with other anti-TB drugs; however, reported results for pyrazinamide DST were consistently lower than for the 3 first-line drugs. Reporting DST results ranged from a minimum of 87.5% for isoniazid, 87.3% for rifampin, and 84.5% for ethambutol, in 1993, to a maximum of 98.2% for isoniazid, 98.1% for rifampin, and 97.8% for ethambutol, in 2011; reporting DST results for pyrazinamide ranged from a minimum of 37.1% in 1993 to a maximum of 88.1% in 2011.
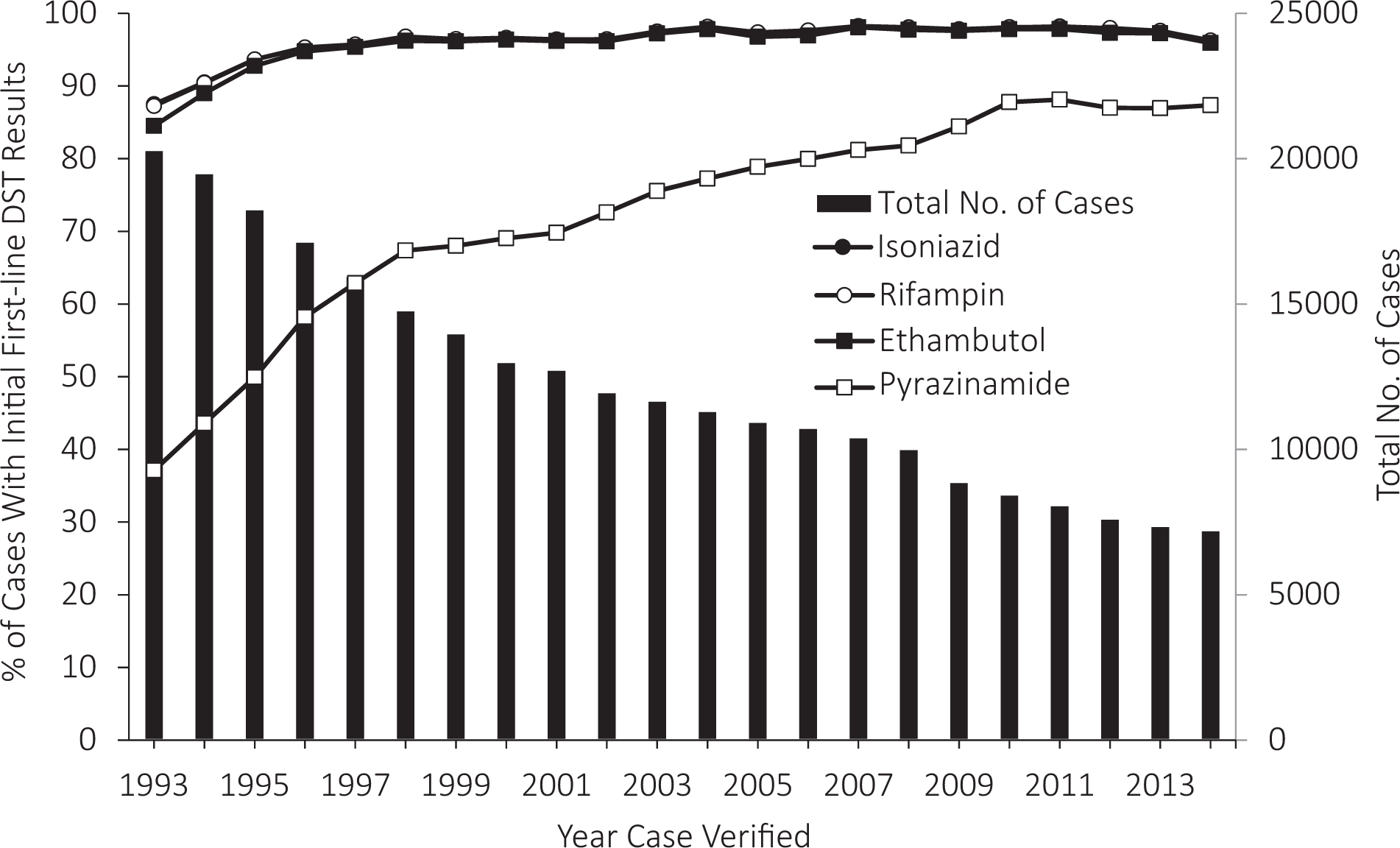
Trends in the proportion of cases with initial first-line drug-susceptibility testing (DST) results, by drug, among culture-confirmed cases of tuberculosis (N = 270 536), US National Tuberculosis Surveillance System, 1993-2014.
The proportion of cases with complete first-line DST results for 3 drugs (isoniazid, rifampin, and ethambutol) by cohort of year in which the case was reported increased after the first year for each cohort (Figure 2). Among all cohorts, the largest absolute increase in complete reporting occurred from the year in which the case was initially reported, or the first year, to the subsequent, or second year (range of increase, 1.0%-4.4%; mean, 2.7%); a smaller increase occurred from the second year to the third year (range, 0.1%-2.3%; mean, 0.7%). After the third year, annual increases were much smaller (range, 0.0%-0.2%), except for the cohort of cases reported in 2003, which increased by 1.0% during 2005-2006 and by 0.9% during 2009-2010. The proportion of cases with complete first-line DST results had a range of 90.1% to 96.3% for the first year reported and a range of 95.8% to 98.1% for the maximum proportion obtained for each cohort.
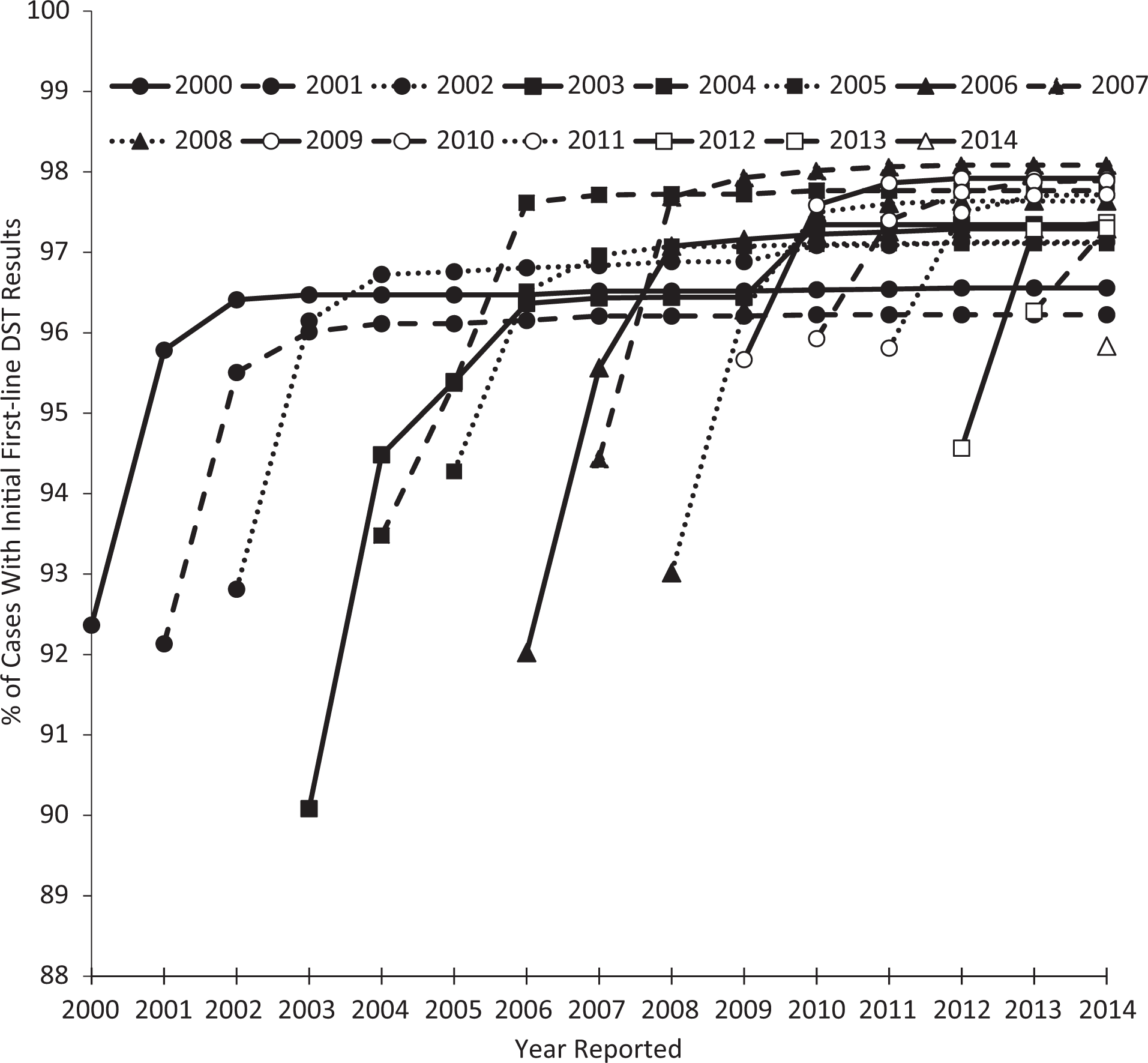
Trends in the proportion of cases with complete initial first-line drug-susceptibility testing (DST), by cohort of year reported, among culture-confirmed cases of tuberculosis, US National Tuberculosis Surveillance System, 2000-2014. Complete initial first-line DST is defined as cases with DST results for isoniazid, rifampin, and ethambutol.
The proportion of cases with on-time first-line DST results (isoniazid, rifampin, and ethambutol) increased significantly from 92.4% in 2000 to 95.8% in 2014 (annual percentage change, 0.4%), with delayed results decreased significantly from 4.2% in 2000 to 1.0% in 2013 (annual percentage change, –8.7%), and with unreported results decreased significantly from 3.4% in 2000 to 2.1% in 2010 (annual percentage change, –5.5%) but increased significantly from 2.1% in 2010 to 4.1% in 2014 (annual percentage change, 17.0%; Figure 3).
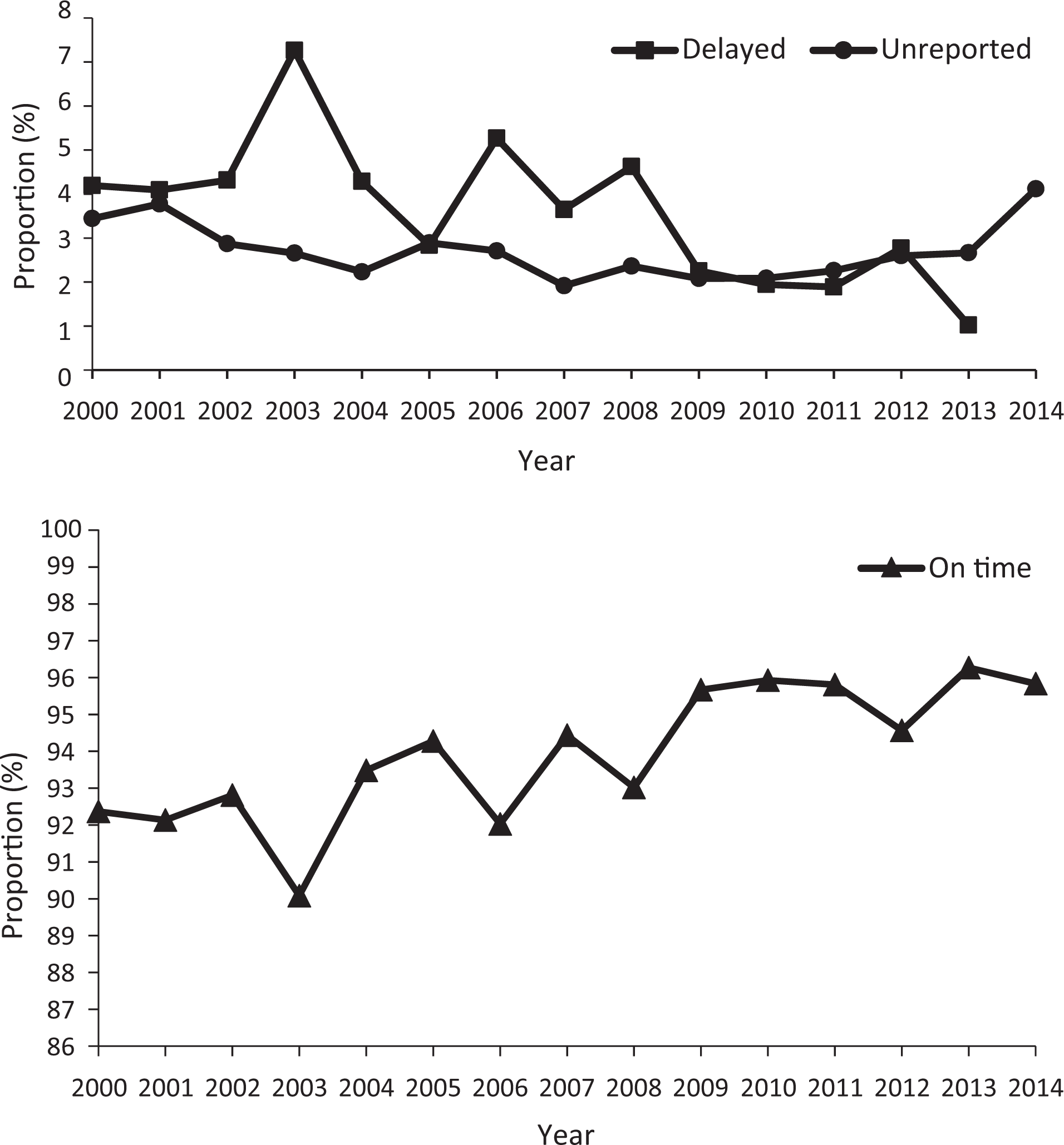
Proportion of cases with on-time, delayed, and unreported complete initial first-line drug-susceptibility testing (DST) among culture-confirmed cases of tuberculosis, US National Tuberculosis Surveillance System, 2000-2014. Any DST report received within the calendar year that the case was initially reported to the Centers for Disease Control and Prevention was considered on time. DST reports received after the case report year were classified as delayed. Unreported DST results were defined as cases without a complete DST report as of April 2015, when the data for all TB cases reported in 2014 and prior years were finalized by the Centers for Disease Control and Prevention. Complete initial first-line DST was defined as cases with DST results for isoniazid, rifampin, and ethambutol.
Of 134 787 culture-confirmed cases reported during 2000-2012, reporting of the initial first-line DST results to NTSS was on time for 125 855 (93.4%) cases, delayed for 5332 (4.0%) cases, and unreported for 3600 (2.7%) cases. The later in the year that cases were verified as TB, the lower the proportion of cases with on-time DST results and the greater the proportion of cases with delayed and unreported DST results (Table). The greatest odds of delayed reporting, compared with on-time reporting, were for cases reported in the fourth quarter of the year (OR = 3.2; 99% CI, 2.9-3.6). Compared with patients for whom the health department was involved in care, patients with no health department involvement had greater odds of unreported DST results (OR = 1.4; 99% CI, 1.2-1.5) yet the same odds of having delayed DST results (OR = 1.1; 99% CI, 1.0-1.2). States reporting an average of 202 to 2207 annual cases of TB, compared with states reporting an average of 93 to 201 annual cases of TB, had greater odds of delayed DST reporting (OR = 1.3; 99% CI, 1.1-1.4) but not unreported DST. Compared with the Northeast, all other regions had 1.2 to 2.4 greater odds of having delayed DST results. Compared with the West, other regions had 1.3 to 2.0 greater odds of having unreported DST results. The South had the highest odds of delayed (OR = 2.4; 99% CI, 2.1-2.7) and unreported (OR = 2.0; 99% CI, 1.8-2.2) DST results.
Characteristics of patients with on-time, delayed, and unreported first-line DST among culture-confirmed TB cases (N = 134 787), US National Tuberculosis Surveillance System, 2000-2012a
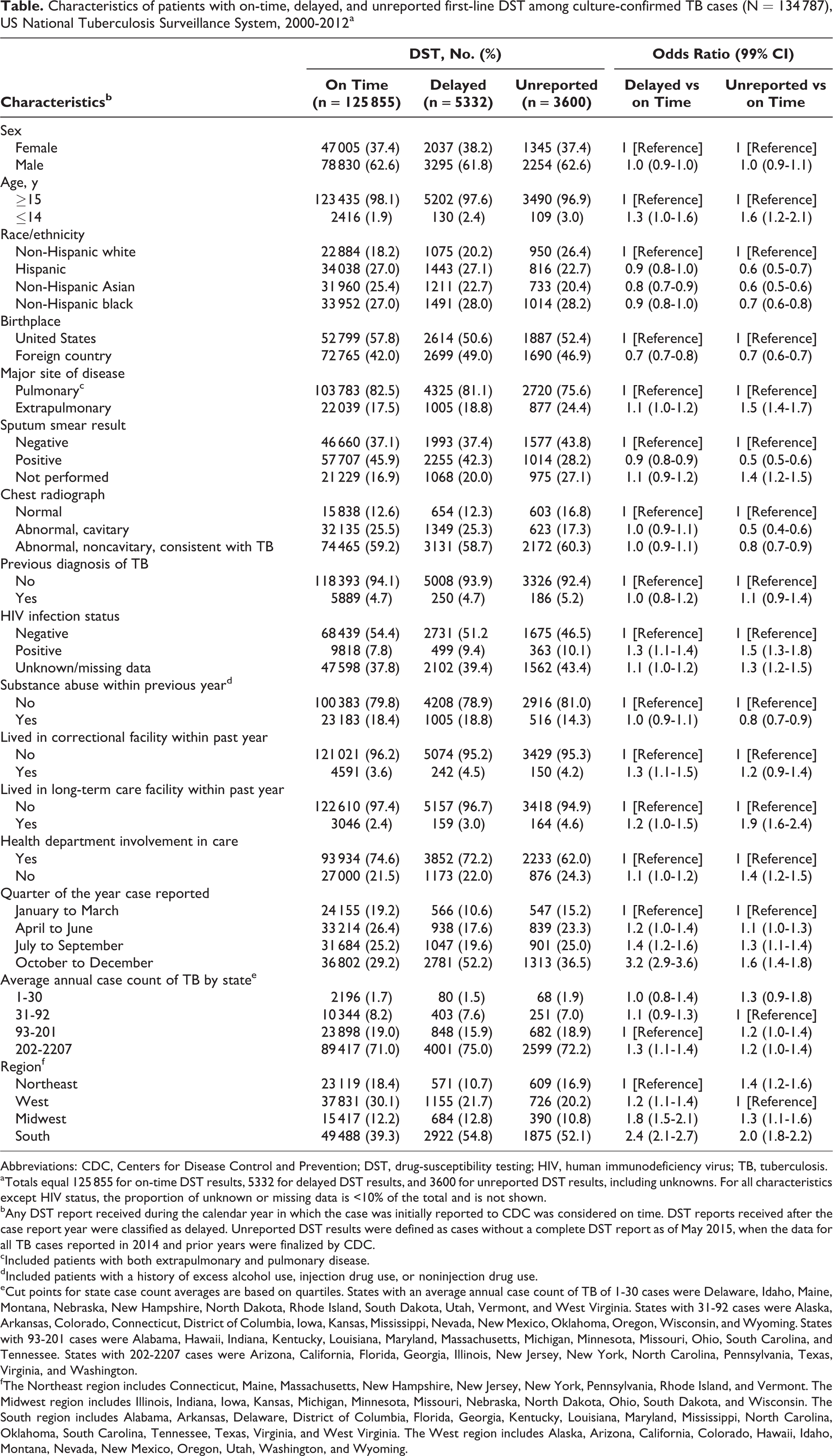
Abbreviations: CDC, Centers for Disease Control and Prevention; DST, drug-susceptibility testing; HIV, human immunodeficiency virus; TB, tuberculosis.
aTotals equal 125 855 for on-time DST results, 5332 for delayed DST results, and 3600 for unreported DST results, including unknowns. For all characteristics except HIV status, the proportion of unknown or missing data is <10% of the total and is not shown.
bAny DST report received during the calendar year in which the case was initially reported to CDC was considered on time. DST reports received after the case report year were classified as delayed. Unreported DST results were defined as cases without a complete DST report as of May 2015, when the data for all TB cases reported in 2014 and prior years were finalized by CDC.
cIncluded patients with both extrapulmonary and pulmonary disease.
dIncluded patients with a history of excess alcohol use, injection drug use, or noninjection drug use.
eCut points for state case count averages are based on quartiles. States with an average annual case count of TB of 1-30 cases were Delaware, Idaho, Maine, Montana, Nebraska, New Hampshire, North Dakota, Rhode Island, South Dakota, Utah, Vermont, and West Virginia. States with 31-92 cases were Alaska, Arkansas, Colorado, Connecticut, District of Columbia, Iowa, Kansas, Mississippi, Nevada, New Mexico, Oklahoma, Oregon, Wisconsin, and Wyoming. States with 93-201 cases were Alabama, Hawaii, Indiana, Kentucky, Louisiana, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Ohio, South Carolina, and Tennessee. States with 202-2207 cases were Arizona, California, Florida, Georgia, Illinois, New Jersey, New York, North Carolina, Pennsylvania, Texas, Virginia, and Washington.
fThe Northeast region includes Connecticut, Maine, Massachusetts, New Hampshire, New Jersey, New York, Pennsylvania, Rhode Island, and Vermont. The Midwest region includes Illinois, Indiana, Iowa, Kansas, Michigan, Minnesota, Missouri, Nebraska, North Dakota, Ohio, South Dakota, and Wisconsin. The South region includes Alabama, Arkansas, Delaware, District of Columbia, Florida, Georgia, Kentucky, Louisiana, Maryland, Mississippi, North Carolina, Oklahoma, South Carolina, Tennessee, Texas, Virginia, and West Virginia. The West region includes Alaska, Arizona, California, Colorado, Hawaii, Idaho, Montana, Nevada, New Mexico, Oregon, Utah, Washington, and Wyoming.
Patient characteristics significantly associated with delayed DST reporting included non-Hispanic white race/ethnicity, birth in the United States, sputum smear negativity, positive or unknown human immunodeficiency virus (HIV) status, and residence in a correctional facility (Table). Patient characteristics that were significantly associated with unreported DST results included age ≤14, non-Hispanic white race/ethnicity, birth in the United States, sputum smear negativity or sputum smear not performed, normal chest radiograph, positive or unknown HIV status, lack of substance abuse, residence in a long-term care facility, and lack of health department involvement in the care of TB.
Discussion
Reducing the level of incomplete DST reporting and decreasing the time to reporting of drug resistance to NTSS are recommended for improving the prevention and control of drug-resistant TB. 9 Among culture-confirmed TB cases in the United States, the proportion with initial DST results reported to NTSS improved for all first-line drugs during 1993-2008, and the proportion of cases with DST results reported on time significantly increased during 2000-2014. However, delayed and unreported DST results persisted. The delay or failure to report DST results might occur at multiple points along the reporting sequence: from the laboratory to the requestor of the DST, from the requestor to the health department (if requestor is outside the health department), and from the health department to CDC. Reporting dates for when DST results are sent to the requestor or to the health department are not collected in NTSS; therefore, we were unable to specify the type of delay in DST reporting in our analysis. The potential for delays or nonreporting to the health department is supported by the data on patients who are residents of correctional facilities or long-term care facilities or who receive care only from non-health department providers (TB program staff members might have difficulty obtaining complete patient records).
Delays might occur when health departments transmit data to CDC. The factor with the strongest association with delayed DST results in our analysis was the time of year during which the case was reported. Cases reported during October through December were more likely to have unreported DST results than cases reported earlier in the year. This reporting delay might be caused by the extended time that health departments have to report DST results for cases verified earlier in the year before they are considered delayed. Results from mycobacterial cultures can require 2 to 6 weeks to confirm the TB diagnosis, although acid-fast bacilli smear microscopy produces results in 24 hours and nucleic acid amplification tests produce results in 24 to 48 hours. 16 DST can take an additional month to produce results. 17 CDC requires all initial DST results to be reported during the calendar year in which the case is reported. However, even for cases verified in December, state health departments have until April of the next year to report DST results to CDC to be considered on time. Additionally, delayed DST results from cases reported in the fourth quarter might be related to the increased workload among health department staff members during the first and last few months of the calendar year because of disease reporting deadlines.
We based the definitions for on-time and delayed DST results used in this study on the timeliness of reporting these results to NTSS. Although bacteria in growth-based assays (eg, agar proportion or Mycobacteria growth indictor tube systems) can take weeks to grow and produce DST results, growth-based assays are commonly used for clinical management of drug-resistant TB cases. Surveillance reporting provides the public health and clinical community with data on the trends, patterns, and scope of drug-resistant TB in the United States and globally, which guide TB control measures.
We examined multiple factors in this analysis that could be related to any of the possible steps at which delays or nonreporting might occur. We found an association between geographic regions and both delayed and unreported DST results. This association might be attributable to differences in funding, number of staff members, method of DST reporting from local health departments and laboratories to the state health department, and method of DST reporting from the state health department to CDC (eg, type of software or electronic reporting). Further study of geographic differences might uncover causes of delayed and unreported DST results and lead to interventions for improving national DST reporting. States with the largest average annual TB case count were associated with delayed DST results, indicating that the number of staff members or funding in either the health department or the laboratory might be insufficient to handle the testing, management, and tracking of DST results.
We also found associations between patient characteristics and delayed or unreported DST results. This association indicates that providers or public health workers might be prioritizing reporting of DST results of selected patient populations (eg, patients with severe disease or those at risk for drug resistance). Positive sputum smears and cavitary disease are markers for severe disease 18 –21 and were both inversely associated with delayed or unreported DST results, possibly because of the culture isolates of patients with severe disease growing more rapidly 22 or because providers prioritized the reporting of DST results for patients with severe disease. Risk factors for drug resistance include foreign-born status or a previous diagnosis of TB 23 ; foreign-born status was inversely associated with delayed and unreported DST results, but previous diagnosis of TB was not associated with delayed or unreported DST results. Some researchers recommend that specimens from patients at risk for drug resistance, including foreign-born patients and patients with a previous TB diagnosis, should be fast-tracked to reference laboratories to avoid delays. 6,24 However, we found that HIV positivity was associated with unreported DST results. This finding is concerning because HIV-positive patients have a higher risk for poor outcomes than HIV-negative patients 25 and should be prioritized for obtaining and reporting DST results.
Despite the overall improvements in reporting of DST results to NTSS for 1993-2010, only 88.1% of all culture-confirmed TB cases verified during 2011 had DST results reported for pyrazinamide, the highest proportion of any year. The Association of Public Health Laboratories and CDC recommend that DST be performed for all first-line drugs, including pyrazinamide, yet not all laboratories perform DST for pyrazinamide. 6 Low rates of pyrazinamide susceptibility testing might be attributed to the following: (1) adding pyrazinamide to the standard first-line DST panel of test doubles the cost of first-line DST 6 and (2) pyrazinamide DST is technically more difficult to perform than other first-line anti-TB medications. 8
Limitations
This study had several limitations. Our definition of unreported DST results allowed more time for DST results to be reported for cases reported in earlier years, which might have led to an overestimation of unreported DST results for more recent years and might explain the increase in the proportion of cases with unreported DST results during 2011-2014. Additionally, for unreported DST results, we were unable to distinguish between DST not being performed and DST results not being reported to CDC.
Although our data cannot distinguish all the reasons for delay or nonreporting of DST results, we identified multiple concerns that should be addressed by health departments. Patients at high risk for poor clinical outcomes (eg, patients with diabetes or suspected of having drug-resistant TB) can be prioritized for obtaining and reporting DST results. Rapid molecular techniques are available for identifying mutations associated with drug resistance, and although rapid molecular test results will be collected in the future, NTSS captures only DST results through growth-based methods. CDC still recommends that all culture-confirmed TB patients have DST results by using growth-based methods. 25 States with the highest odds of delayed or unreported results might benefit from a systematic evaluation of their surveillance procedures and program capacity to identify the causes of delay and nonreporting of DST results. Improving communication efforts with corrections facilities, long-term care facilities, and non–health department providers caring for TB patients might also assist with identifying reporting gaps. State and local TB programs have taken steps toward electronic laboratory reporting, which could improve the timeliness of DST reporting in the future. Data sharing and linking among public health, providers, and laboratories could improve reporting timing and accuracy. 26
Conclusion
DST is recommended for all culture-confirmed cases of TB, and DST reporting is an important aspect of preventing and controlling drug-resistant TB. The proportion of cases with on-time reporting of DST results has improved; however, delayed and unreported DST results persist. Carefully assessing the causes for delayed and unreported DST results could lead to more timely and complete DST reporting to NTSS.
Footnotes
Authors’ Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of CDC.
Acknowledgments
We thank Beverly Metchock, PhD, and Angela Starks, PhD, for their help with data interpretation and editing, and C. Kay Smith, MEd, for editorial services.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by CDC.
