Abstract
This study explores the use of Fe-Mn-Cr-Si-C master alloy as a sustainable alternative to nickel in press and sinter powder metallurgy, with the goal to improve hardenability and mechanical performance. Two master alloy compositions were optimized for liquid phase sintering using thermodynamic software and then produced through gas atomization. The melting behavior of the master alloys was characterized and compared with thermodynamic predictions. Sintering experiments were performed on compacts from mixes with and without master alloy additions. Continuous cooling transformation (CCT) diagrams were generated to assess hardenability. The results demonstrated that the master alloys improve the hardenability, even at lower cooling rates compared to alloying with Ni. Mechanical testing showed notable improvements in yield strength and apparent hardness comparable to those achieved with Ni additions. These findings support the use of Fe-Mn-Cr-Si-C master alloy as a viable, more sustainable alternative to nickel in powder metallurgy steels.
Introduction
Powder metallurgy (PM) offers numerous advantages as a manufacturing process, such as producing near net shape components, enabling mass production, being cost effective and resulting in minimal material waste.1–3 For PM components made through press and sinter route to be used in highly demanding applications, the requirements for PM steels include high density and high hardenability.
Hardenability is the ability of steel to form martensite to a certain depth when quenching from austenitization temperature under specific cooling rates. The construction of continuous cooling transformation (CCT) diagrams using dilatometry data at different cooling rates is useful in understanding the hardenability of a steel. 4 Alloying elements in the steel influence the hardenability during the heat treatment process. 5
The main alloying elements conventionally used to boost the strength of PM steel base powders are Ni and Cu due to their suitability for industrial sintering atmospheres and the ease with which their oxides can be reduced during sintering.6,7 These elements are introduced into base powders through admixing or diffusion bonding methods. 8 However, Ni has a higher carbon footprint as well as rising cost, volatility, and health concerns and there is a growing focus on sustainability across industries. Cu reduces the recyclability of steel because the thermodynamics of steel refining do not favor the separation of Cu during steel making.9–11 Elements with a lower carbon footprint and higher recyclability, such as Cr, Mn and Si, which are commonly used in wrought steels, have been investigated as alternatives. They are inexpensive, and their prices are relatively stable.12,13 These elements also enhance the mechanical properties of steel, including tensile strength, hardness and yield strength.8,14 Microalloying with V and B as in wrought steel improves the hardenability. Boron particularly enhances the hardenability of steel at ppm levels with a low solubility in steel at room temperature.15,16 Using Cr, Mn, and Si as alloying elements in PM steel base powders poses a challenge. They are highly oxygen sensitive and form stable oxides that are difficult to reduce under conventional sintering conditions 17 which imply stricter requirements on the sintering temperature and atmosphere.
Chromium has been successfully incorporated into PM steels through pre-alloying method, up to a limit of 3 wt.%.6,18 The pre-alloying method ensures complete homogenization of elements within the matrix. However, it reduces compressibility due to the solid solution strengthening effect within the powder particles. 19 One method to overcome this is through the master alloy (MA) method. Master alloying involves combining the highly oxygen sensitive elements (such as Cr, Mn, and Si) with less oxygen sensitive elements, such as Fe, to form a master alloy. This master alloy is then mixed with base iron powder for alloying. The combination of these elements in MA lowers the activity of the oxygen sensitive elements and is attractive for PM steels.7,13,14,20–22 This is due to advantages such as the possibility of transient liquid phase sintering (LPS), limited loss in compressibility, flexibility to have a variety of final compositions using a single base powder and freedom to use more beneficial alloying elements.7,12,23 For the PM steels, B addition has been investigated for activating sintering by liquid phase formation leading to improved densities when B reacts with Fe to form a eutectic liquid at 1173 °C.1,24–26 There is limited work on the use of B to enhance the hardenability of low alloyed PM steels especially through the MA route.
The MA composition determines its behavior during the sintering process, interaction with base powder and final properties after sintering. Recent studies report that dimensional stability in master-alloyed steels depends not only on the onset of melting but also on the liquidus and the melting interval that govern the extent of the transient liquid in LPS. Distribution of Mn, Cr, Si from the master alloy into Fe-base powder improves homogenization and heat-treatment response. Moreover, carbon and boron tune these melting boundaries and transformation products, influencing shrinkage/distortion and the strength-ductility balance.27,28 LPS occurs when the MA melts at or below sintering temperature whereby industrial belt furnaces have typical working temperatures up to 1150 °C.18,29 To gain full understanding of the effect of individual elements, MAs with varying compositions should be analyzed. However, this process can be time-consuming and resource intensive. Therefore, thermodynamic simulation tools like Thermo-Calc software can be highly beneficial to find optimized compositions. 30
In the present work, a comprehensive investigation was conducted, covering the design of MA composition, powder production, characterization, and effects on final properties after sintering. A comparison was made between the effects of adding MA as a sustainable alternative versus adding Ni to a similar steel base powder. Thermodynamic simulations with Thermo-Calc software were used to optimize the master alloy, and the results were compared with experimental findings. The effect of MA on the hardenability of Fe-0.85 wt.%Mo base steel powder was also studied and compared with the results obtained using Ni addition.
Materials and methods
Optimization of master alloy powder composition and their production
The compositions of master alloys (MA) Fe-Mn-Cr-Si-C and Fe-Mn-Cr-Si-C-B were optimized using the CALPHAD method. 30 The relationship between the solidus temperature and the composition of individual elements in the MA, i.e., Cr, Mn, Si, C and B were determined through single equilibrium calculations using Thermo-Calc software with the TCFE14 database. The optimization criteria for achieving a low melting point MA were based on minimizing both the elemental content and the solidus temperature. The optimized compositions were used to produce two master alloys, MA1 (without boron) and MA2 (with boron) while the other elements (Mn, Cr, Si, C) held constant in both alloys, as shown in Table 1. The master alloy was optimized primarily for a low solidus to trigger transient liquid within the sintering window.
Master alloy identification and composition.
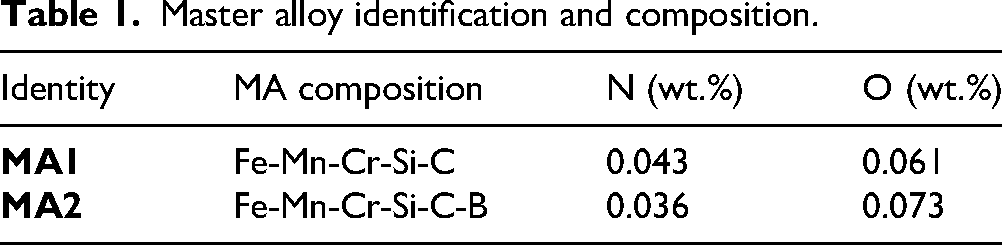
Both MA powders were produced through gas atomization using nitrogen and the powders were sieved to obtain a particle size below 45 µm. Both powders had a median size (D50) of 29 µm, selected to achieve better homogenization during mixing and sintering. Powder morphology was observed using a tabletop scanning electron microscope (SEM), Hitachi TM3030. Thermal analysis was performed to correlate the actual melting behavior of MA with the solidus temperature from thermodynamic simulations. Differential scanning calorimetry (DSC) measurements were performed using a Netzsch simultaneous thermal analyzer STA 449 F1 Jupiter by heating the samples up to 1300 °C with a heating and cooling rate of 10 °C/min in argon.
Powder mix preparation
Two powder mixes with 3 wt.% MA1 and MA2, were prepared by blending them with a water atomized iron powder prealloyed with 0.85 wt.% Mo (Astaloy 85Mo) along with UF-4 graphite (Kropfmühl GmbH) as the carbon source and 0.5 wt.% Lube E as the lubricant. Along with these mixes, two reference mixes with and without Ni addition were made, as given in Table 2. In all cases, graphite was added to achieve a total carbon content of 0.65 wt.% in the final composition.
Identities and compositions of powder mixes.

Compaction and sintering
For quenching studies in dilatometer, cylindrical samples measuring 6 mm in diameter and 10 mm in height were prepared. To evaluate mechanical properties, tensile and impact test specimens were prepared in accordance with ISO 2740:2023 and ISO 5754:2023, respectively. All samples were compacted at 700 MPa for all mixes. Sintering was performed in a belt furnace at 1150 °C for the dilatometer samples and 1120 °C for the mechanical test samples, with a holding time of 30 min in a 90%N2–10%H2 sintering atmosphere. The cooling rate was around 0.5 °C/s. Sintered tensile samples were tested according to EN 10002-1 in a Zwick 100 universal testing machine, while impact test specimens were tested according to ISO 148-1 using an Instron Motorized 450 J Pendulum system (Model: 450MP2J2). Vickers hardness was measured on the tensile test samples in accordance with ISO 6507-1:2023, using a 10 kg load using Struers Duramin-40 testing tester. Tensile and impact energy tests were each conducted on seven specimens while hardness was evaluated using eight indentations.
Quenching tests in dilatometer
Experimental CCT curves were constructed by performing quenching tests in a L78 RITA quench dilatometer using the samples sintered at 1150 °C. The samples were heated to 920 °C for austenitization with a heating rate of 10 °C/s, held for 3 min, and then cooled at 1, 5, 10, 50 and 100 °C/s in separate runs using individual samples. The carbon content of the samples was determined after quenching using LECO combustion method.
Microstructure characterization
The samples used for CCT evaluation and sintered tensile samples were sectioned, mounted, and polished to a 1 µm surface finish, then etched with 1% Nital. For metallographic analysis, DM6000 light optical microscope (LOM) was used. The samples used for constructing CCT curves were analyzed to estimate phase fraction using the point counting method with 100-point grids, in accordance with ISO 9042:2024. For each sample, three micrographs from different locations with a total analyzed area per specimen being 5.4 × 105 µm², were taken at x200 magnification.
Results
Thermodynamic optimization and characterization of master alloys
Figure 1 shows the effect of Mn, Cr, Si and C on the solidus of the Fe-Mn-Cr-Si-C(-B) master alloy as calculated using Thermo-Calc software. Calculations were performed in two steps: (i) an equilibrium calculation for the base composition, followed by (ii) one-axis step calculations in which either the composition of the element of interest was varied (to extract composition–solidus trends) or the temperature was stepped at fixed composition (to obtain the liquid phase fraction, fliq). The solidus as Tsol where fliq→0+ and the liquidus as fliq = 1, consistent with Gibbs-energy minimization. 30
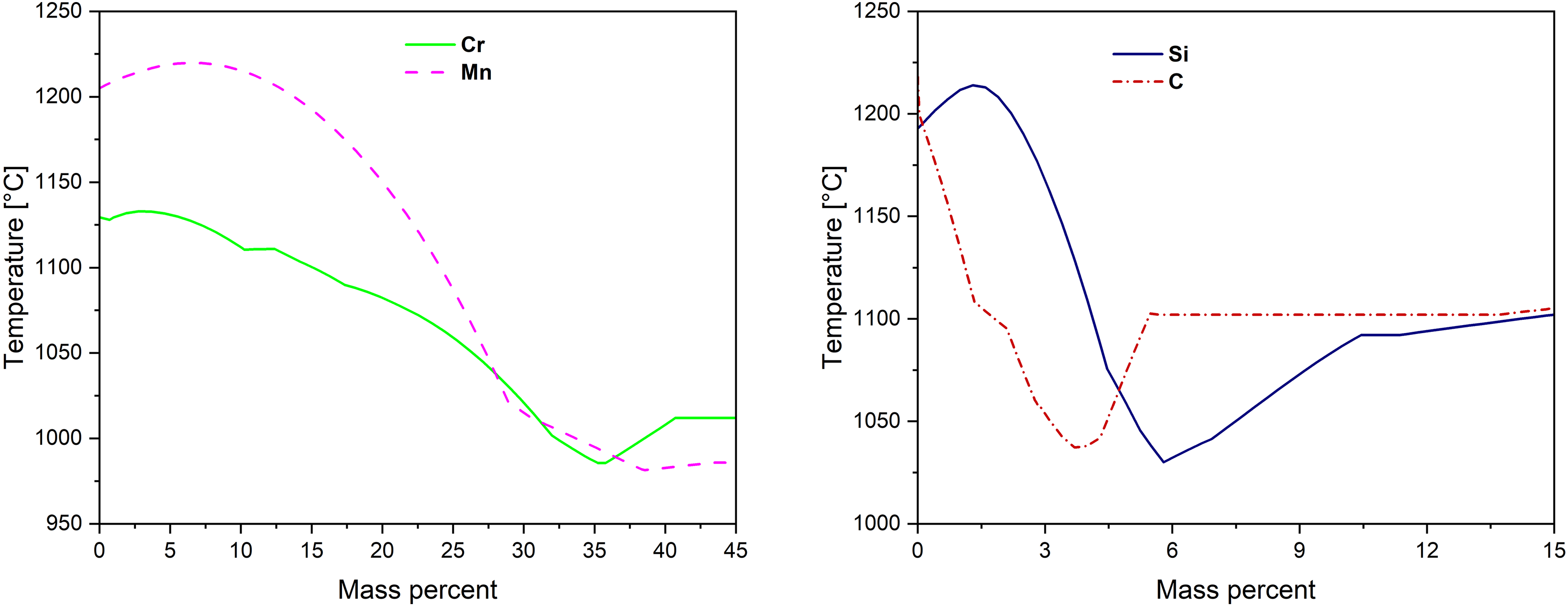
Effect of alloying elements on the solidus of the Fe–Mn–Cr–Si–C(–B) master alloy (Thermo-Calc).
Increasing Mn, Si and C lowers the solidus more strongly than Cr (steeper slopes in Figure 1). Higher content of Si (∼6.4 wt.%) and C (∼4 wt.%) show composition limits beyond which further additions raise the solidus temperatures. Adding small amounts of boron to MA1 (denoted as MA2) shifts the solidus to lower temperature (earlier liquid formation) and modifies the melting interval; this is reflected in the DSC curves and the liquid volume fraction plot from Thermo-Calc simulation (Figure 3).
SEM of the gas atomized powders MA1 and MA2 (Figure 2) shows predominantly spherical particles with satellites in both MA1 (optimized composition) and MA2 (boron-containing variant), with a higher satellite frequency in MA1. 31

SEM of gas-atomized powders (MA1, MA2). Spherical morphology with satellites.
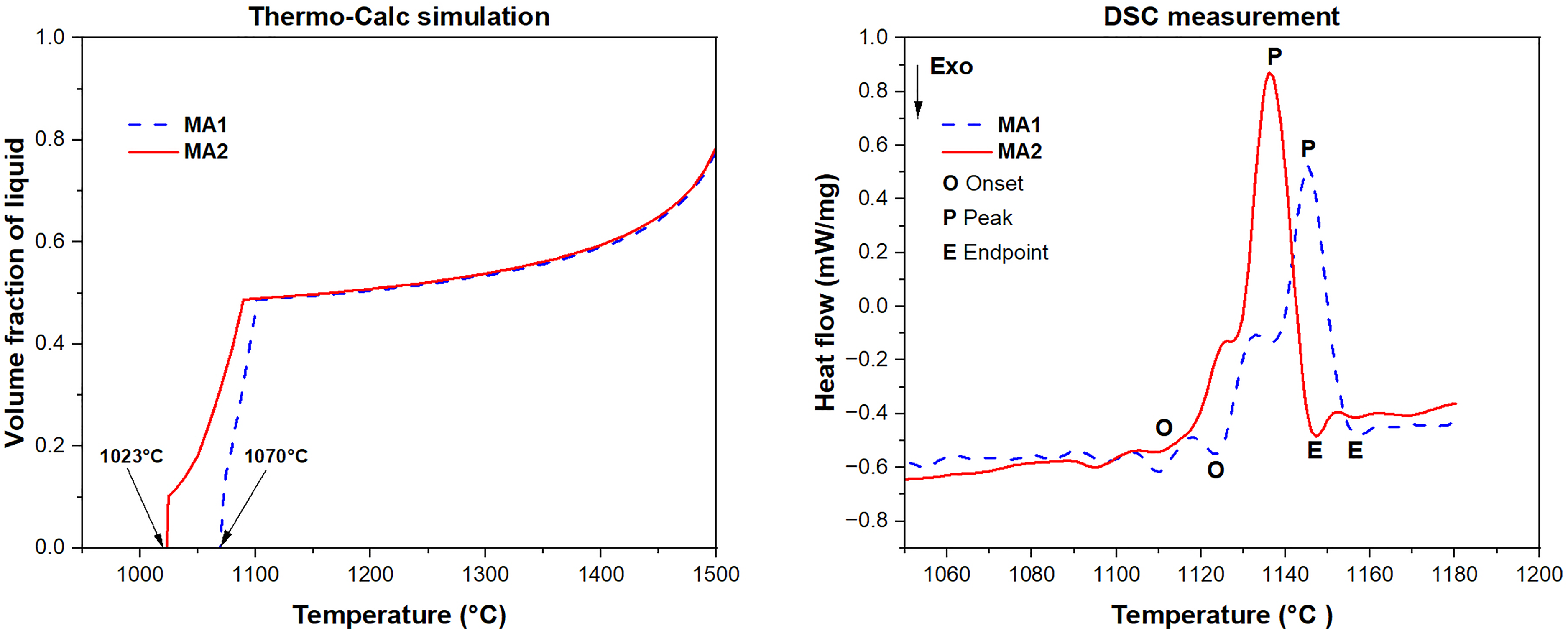
Liquid volume fraction vs. temperature from Thermo-Calc for MA1 and MA2 (Left); DSC curves with onset (O), peak (P), and endpoint (E); endpoints correspond to Tliq.
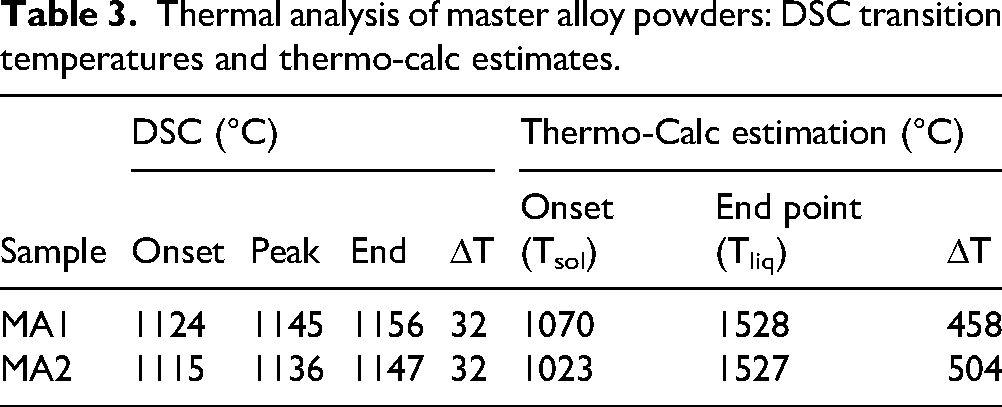
Figure 3 presents Thermo-Calc liquid volume fraction vs. temperature with DSC curves for MA1 and MA2. The onset of melting (DSC onset) occurs at 1070 °C for MA1 and 1023 °C for MA2. Endpoints on the DSC curves correspond to the liquidus, and the resulting melting intervals ΔT = Tliq-Tsol for each alloy are listed in Table 3 along with peak temperatures. The observed endothermic reactions correspond to melting. 26
Thermal analysis of master alloy powders: DSC transition temperatures and thermo-calc estimates.
Microstructural analysis of sintered alloys
Figure 4 shows LOM images of tensile samples sintered at 1120 °C. The 85Mo sample exhibits an upper bainite microstructure, while the 85Mo + 2Ni sample shows upper and lower bainite, along with Ni-rich austenite (highlighted in region marked A). The GA-R and GA-B samples display complex microstructures composed of upper bainite, martensite and retained austenite. Notably, the areas adjacent to the master alloy particles tend to develop martensitic microstructures. Coarser MA particles remain partially unmelted, while finer particles appear fully melted, leaving the characteristic secondary pores matching their original shape. 23 Compared with GA-R, GA-B shows a qualitatively more homogeneous redistribution of alloying elements from the MA into the matrix, evidenced by the more diffuse transformation zones/martensitic rims around MA2 in Figure 4, consistent with improved melting/wetting during sintering.
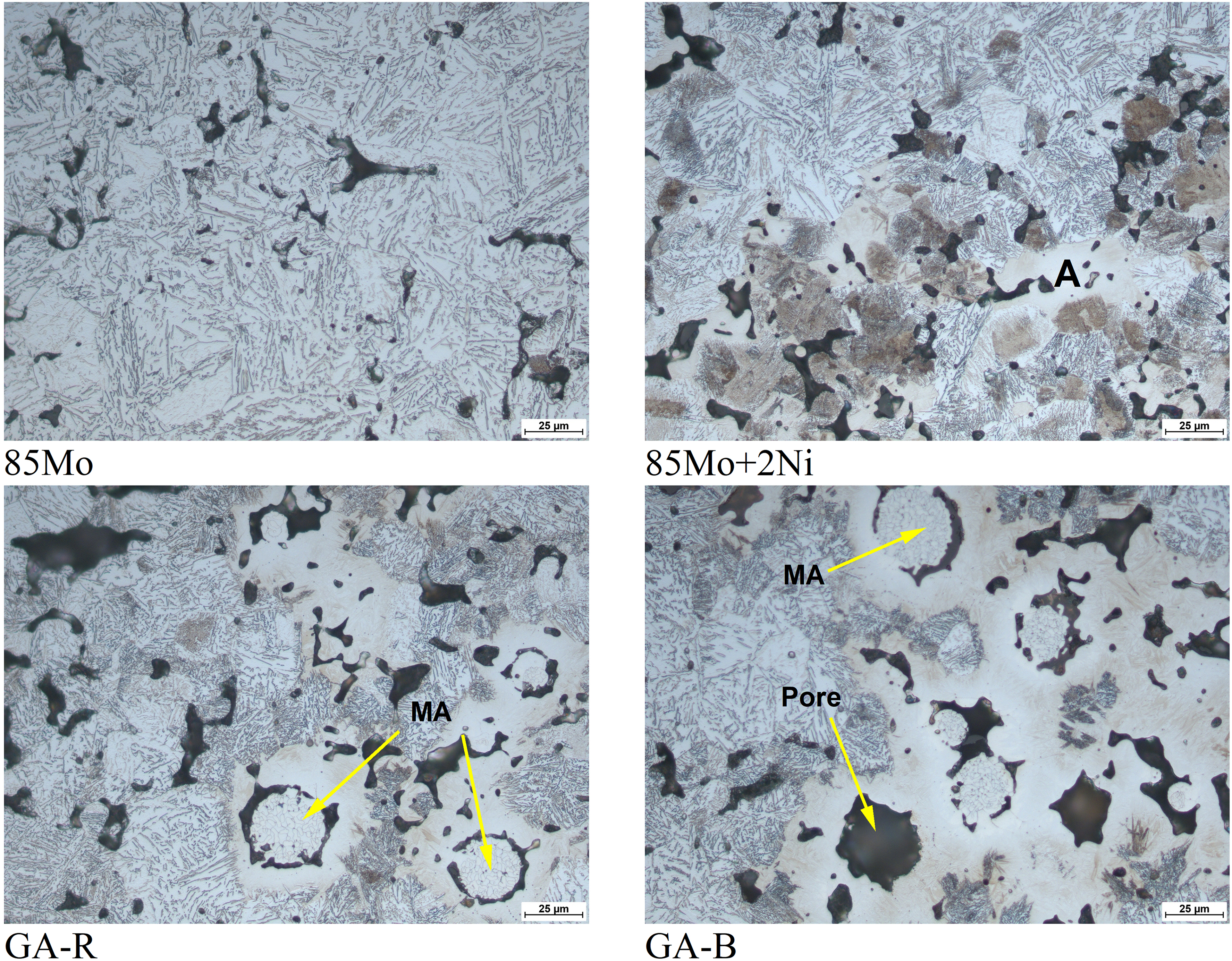
LOM images of sintered tensile test samples. Regions with master alloy (MA) particles and secondary pores are indicated.
Hardenability analysis
Figure 5 shows the CCT curves obtained from dilatometry quenching tests at different cooling rates for the samples sintered at 1150 °C. Ac1 and Ac3 are the start and end of austenitization process during heating. At a cooling rate of 1 °C/s, the 85Mo sample exhibits fully bainitic microstructure, while martensitic microstructure begins forming at 10 °C /s, with complete martensite formation at 50 and 100 °C/s. The martensite start (Ms) temperature drops from ∼300 °C at 10 °C/s to ∼250 °C at 100 °C/s. The 85Mo + 2Ni sample displays bainitic and martensitic transformation at 1 and 5 °C/s, and mainly martensitic transformation at higher cooling rates. Its Ms ranges from 250 to 290 °C. The GA-R sample shows martensitic transformation at all cooling rates. Bainite formation is observed at 1, 5 and 10 °C/s, while full martensite formation is observed at 50 and 100 °C/s (Ms:250–290 °C). The GA-B sample, which contains boron, exhibits both bainitic and martensitic transformation at 1 and 5 °C/s, while fully martensitic microstructure at 10, 50 and 100 °C/s.
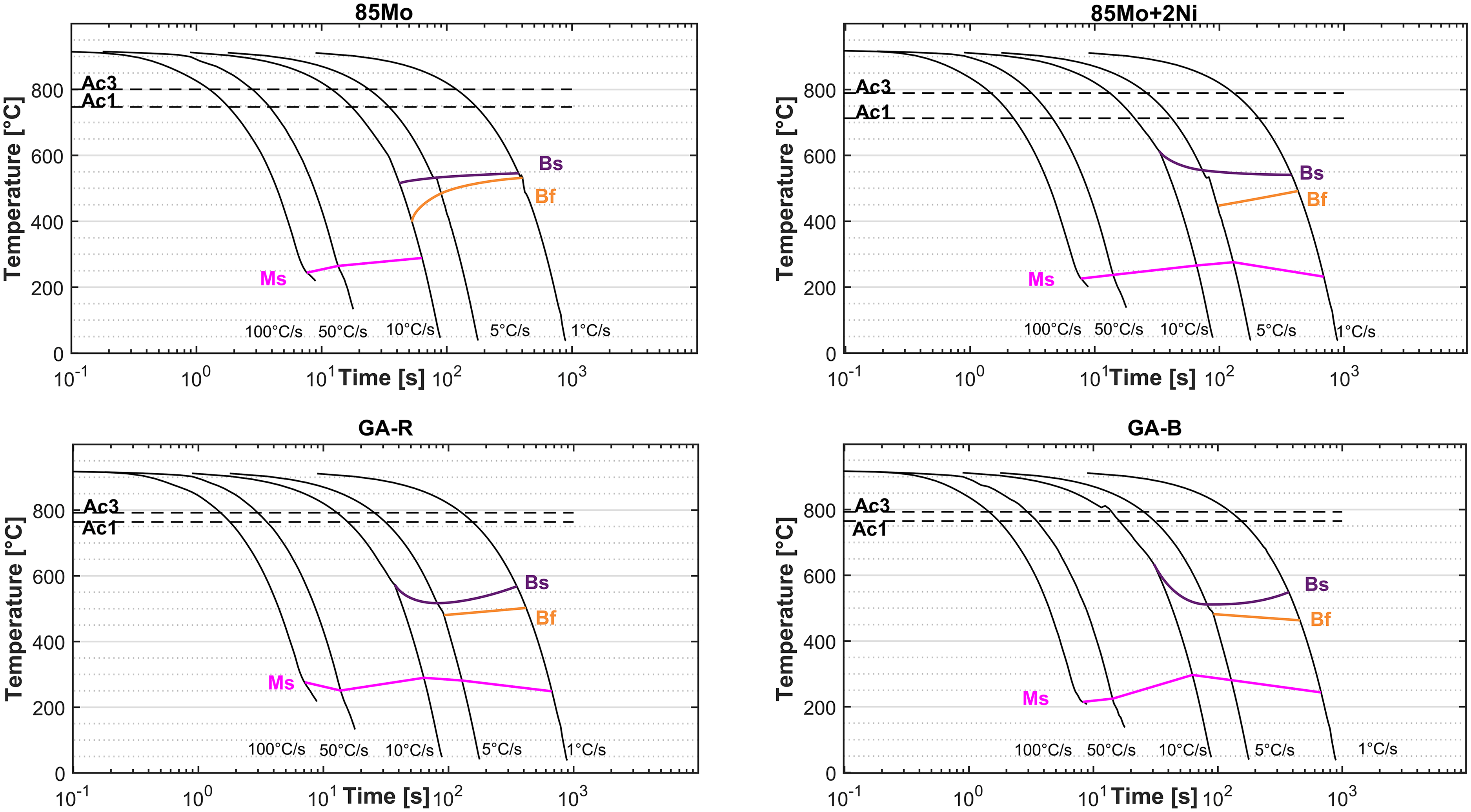
Continuous cooling transformation (CCT) curves of samples sintered at 1150 °C under different cooling rates.
Figure 6 shows phase fraction results by metallography, corresponding to the cooling rates in Figure 5. At 1 °C /s, both GA-B and GA-R samples contain more martensite than 85Mo + 2Ni sample, with GA-B having the higher amount. Figure 7 shows the carbon content of samples after quench tests. Although carbon content influences phase transformation temperatures, such as Bs and Bf (bainite start and end) temperatures, the measured values were almost similar across all samples, indicating negligible variation in this study.
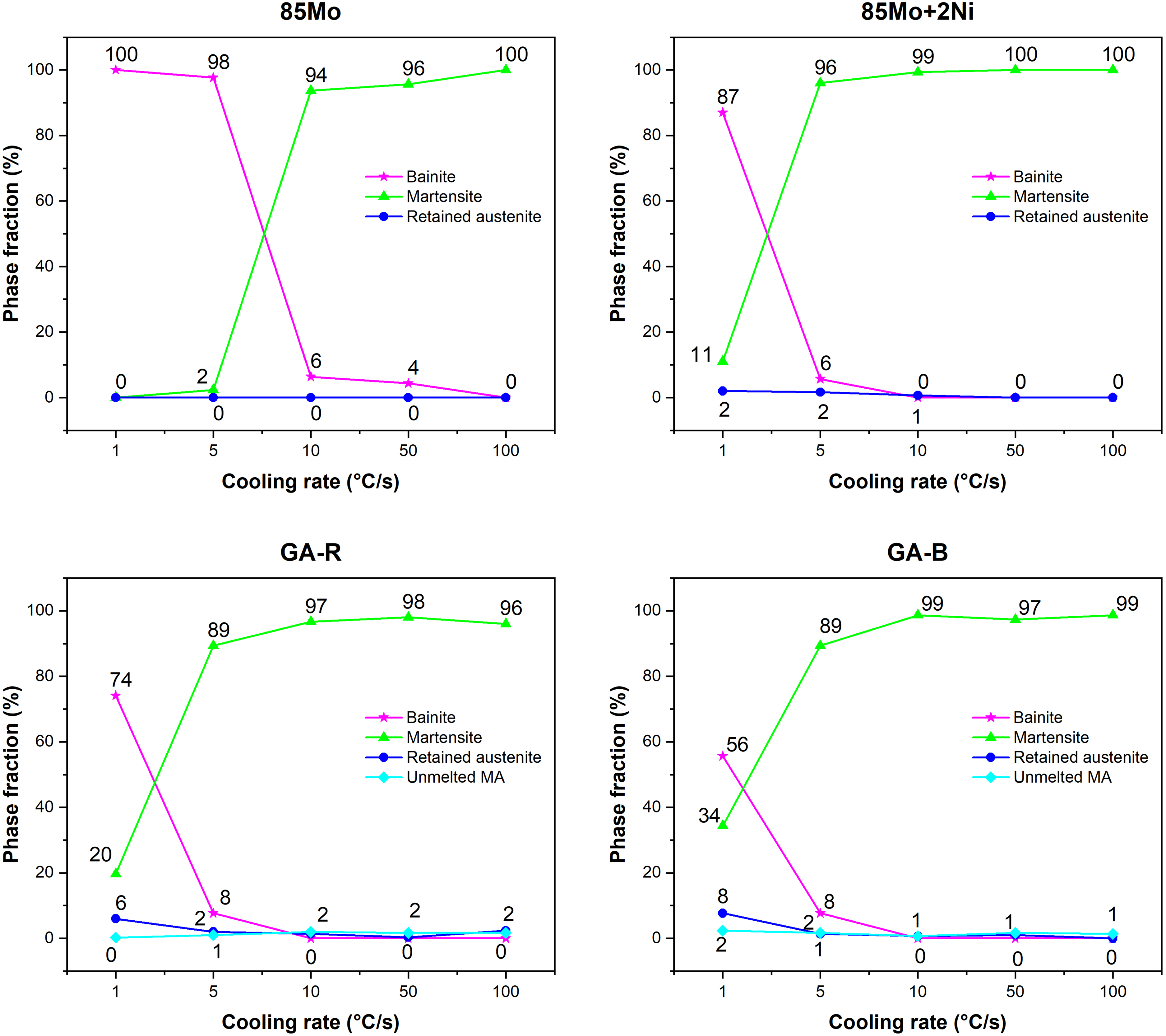
Phase fractions from metallographic images of samples subjected to different cooling rates.
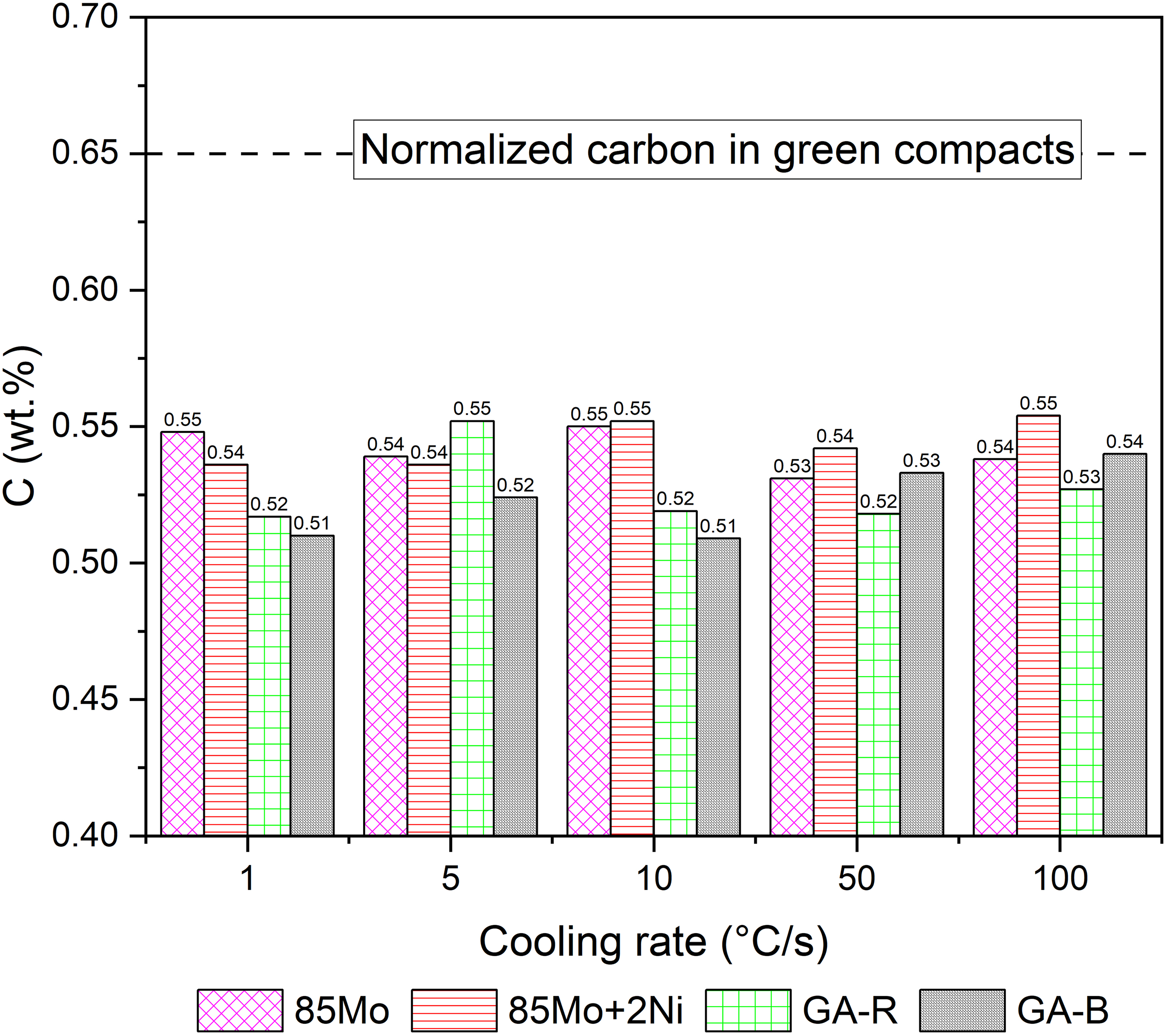
Carbon content (wt.%) of samples after quenching tests.
Evaluation of mechanical properties
Table 4 summarizes the green density (GD), sinter density (SD), dimensional change (DC), hardness (HV10), yield strength (YS), tensile strength (TS), elongation (A), impact energy (IE), carbon (C), oxygen (O) and nitrogen (N) of the samples at 1120 °C. Among the four samples, the 85Mo + 2Ni sample exhibits the highest sinter density, dimensional change, tensile strength, and impact energy. In contrast, the 85Mo sample shows the highest elongation while GA-B has the highest apparent hardness. Among the master alloy samples, GA-B also shows better elongation than GA-R.
Mechanical properties and C, O & N contents after 1120 °C sintering.

Discussion
Influence of Cr, Mn, Si, C and B on MA solidus
For transient-liquid-phase sintering, the master alloy must produce early but controlled liquid within the sintering schedule. The solidus of the master alloy is important especially for transient liquid phase sintering (LPS), making it a key factor in optimizing MA composition. For effective LPS, the MA should melt below sintering temperature.14,25 As shown in Figure 1, different elements in MA influence the melting behavior of the MA powder to varying degrees.
Thermodynamic simulations indicate that smaller additions of Si and C significantly lower the MA solidus, whereas Mn requires >25 wt.% for a notable effect on solidus temperature, and Cr should not exceed 35 wt.%. The composition is set considering producibility constraints, such as the reduced yield of Mn during melting due to its high vapor pressure. 6 Additionally, C is useful in preventing oxidation of the MA. 14
The final MA composition is determined by considering the possible limits for each alloying element. Boron has been studied and promotes liquid phase sintering through a eutectic reaction as B diffuses into Fe.25,32 This is reflected in Figure 3, where MA2 shows a lower solidus than MA1 consistent with B's eutectic-promoting effect under otherwise constant chemistry. Thermodynamic simulations predict a 57 °C reduction in solidus temperature with B addition, which is crucial in LPS and can help reduce furnace energy costs and sintering time.
As seen in Table 3, there is a variation between simulated and experimental solidus of both MA1 and MA2. The onset of liquid formation from Thermo-Calc simulation aligns with the DSC endothermic peak as shown in Figure 3. The difference for MA1 is 54 °C and 92 °C for MA2. This difference is due to the simulations being conducted under ideal equilibrium conditions, which is not the case in DSC 23 as well as the deviations that arise from the database used. Moreover, oxygen pickup during atomization can lead to the formation of oxides of Cr, Si and Mn, 14 shifting actual melting behavior. A previous study using high purity powders also reported deviations between calculated and measured melting points. 33
Effect of master alloy on hardenability
Steel hardenability is influenced by chemical composition, heat treatment and the austenite grain size.5,34 The size and part geometry also affect hardening depth for industrially produced PM components. Alloying elements impact the final microstructure and, consequently, mechanical properties. 35 From the quenching tests (Figure 5), the initial phase transformation is bainite. Higher cooling rates are required for the low alloyed Fe-base powder for martensite transformation. With the addition of Ni in 85Mo + 2Ni and Mn, Cr, Si in master alloyed samples (GA-R, GA-B), the martensite transformation occurs at lower cooling rates. Thus, alloying elements reduce the critical cooling rate for martensite formation. Additionally, higher cooling rates are needed to compensate for the absence of suitable alloying. Thus, cooling rates higher than 10 °C/s are required for martensite to form in 85Mo.
As seen in Figure 6, Mn, Cr and Si enhance hardenability more than Ni, where the GA-R and GA-B show a higher martensite fraction than 85Mo + 2Ni at 1 °C/s. This improvement is attributed to LPS, which enhances element homogenization in GA-R and GA-B compared to low-diffusing Ni.
The presence of boron in GA-B lowers the solidus of the master alloy, which enables earlier liquid formation during sintering, increasing the temperature window between melting and sintering (1115–1150 °C). Moreover, the presence of B can improve the hardenability of GA-B by retarding primary ferrite and pearlite formation, leaving more austenite available to transform into martensite on cooling. 36 The additional alloying causes notable differences since all samples contain nearly the same C content as shown in Figure 7. Boron further shifts the minimum required cooling rate to allow formation of martensite to even lower values.
Effect of master alloy on mechanical properties
The mechanical properties of steel are highly dependent on the resultant microstructure after sintering. Since most conventional sintering is carried out at 1120 °C, an evaluation at this temperature is relevant, especially for the master alloys (see Table 4). Sinter densities are closely similar among the materials (Table 4), indicating that porosity differences are not the primary cause of the mechanical trends. The 85Mo and 85Mo + 2Ni samples show negative dimensional change after sintering. GA-B has slightly higher sinter density than GA-R, likely due to better melting behavior from B addition. Samples with MA show higher hardness due to solid solution strengthening and higher martensite content, which also leads to increased yield strength. However, due to the sensitivity of high-carbon martensite to crack propagation, the elongation is lower with MA than with Ni, hence lower ultimate tensile strength and impact energy consistent with reduced toughness.4,37 While not all MA particles melt fully during sintering (Figure 4), the overall effect on mechanical properties remains significant. Oxygen is higher in GA-R/GA-B than in samples without MA (Table 4), and we observe a negative correlation between oxygen and ductility/impact energy, consistent with oxides acting as crack initiators. Due to lower cooling rates used in conventional sintering as in this study (∼0.5 °C/s), the effect of nitrogen on the mechanical properties is very low. In contrast, retained austenite may form at sinter-hardening rates (∼2–3 °C/s). Moreover, the measured nitrogen levels are very low. Overall, the addition of MA to 85Mo enhances heat treatment response, enabling the use of lower cooling rates to achieve desired properties even using conventional heat treatment techniques for PM components.
Conclusions
This study demonstrates that a master alloy for conventional sintering (1120–1150 °C) can be designed using thermodynamic simulation tools. The main conclusions are:
The solidus temperatures estimated via Thermo-Calc were 50 °C (MA1) and 90 °C (MA2) lower than the actual experimental values. This deviation is due to the assumption of equilibrium conditions and the database used during thermodynamic simulation. Nonetheless, the results effectively guide compositional design by illustrating the influence of individual elements on melting behavior. Master alloy addition enhanced the hardenability by promoting earlier martensite formation, even in belt furnace sintered samples, compared with the reference materials with and without Ni. CCT evaluation and phase quantification showed that boron-containing master alloy (MA2) resulted in a higher martensite fraction, up to 34% even at 1 °C/s, which is primarily due to its earlier melting during sintering. The mechanical properties of the base powder were improved by master alloy addition, particularly hardness and yield strength, confirming the alloy's effectiveness after standard sintering at 1120 °C. The master alloy offers to be an effective and potential replacement for Ni as a sustainable alternative, showing comparable or better hardness and yield strength but lower impact energy due to its enhanced hardenability.
Footnotes
Acknowledgements
This work was performed within the project “Sustainable Solution for high performance PM steels (SSPM) as a part of Wallenberg Initiative Materials Science for Sustainability (WISE) funded by the Knut and Alice Wallenberg Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Knut och Alice Wallenbergs Stiftelse.
