Abstract
In contrast to classical ingot metallurgy, in which case a chemically homogeneous liquid phase is the initial state, in powder metallurgy solid powders are the starting materials. This enables different alloying routes and thus widely differing degrees of chemical heterogeneity in the powder bulk or compact, according to the alloying variant chosen. This may range from homogeneous systems, as in the case of fully prealloyed powders, to fully heterogeneous ones, e.g. elemental powder mixes. Through appropriate sintering regimes, the process of homogenization to approach chemical equilibrium can be controlled and stopped at desired stages. In addition to metallurgical also chemical heterogeneity has to be considered, which is particularly relevant in reactions involving interstitials such as O, C and N. Transfer reactions, described as “internal gettering”, may have a profound effect on chemistry and properties of the resulting products. These effects have to be carefully assessed for selecting the optimum alloying route for a given material and application.
Introduction
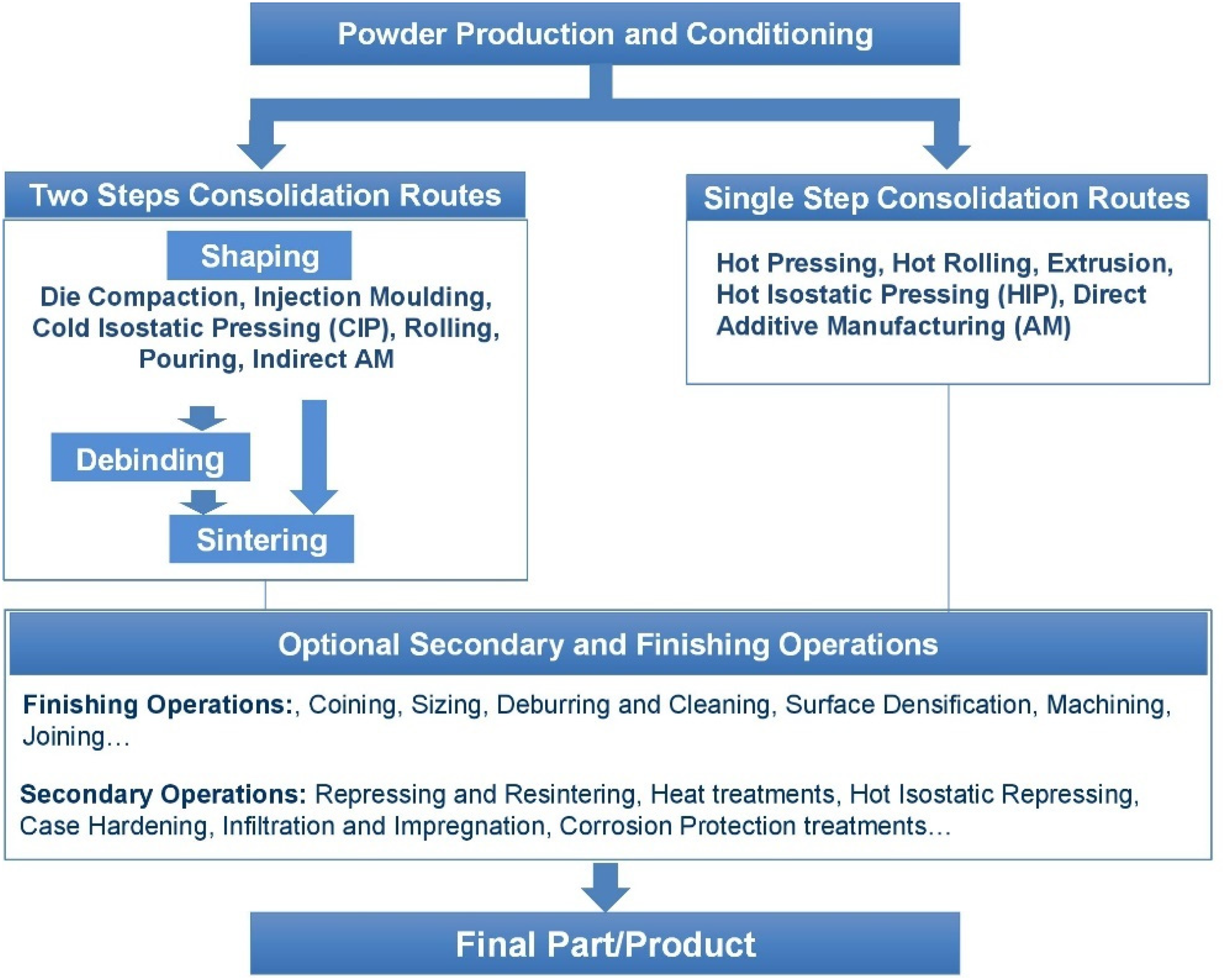
Powder metallurgy, the technique of manufacturing metal-based materials products from powders, is characterized by its enormous flexibility, which results in a wide range of product groups and applications, from precision parts for automotive applications to tool materials in cutting and drilling, lighting, electrical and electronic components to medical tools and devices.1–5 This flexibility includes the production routes, 6 which e.g. for consolidation may involve single stage processes that simultaneously apply pressure and temperature, or separate shaping and heat-treating stages (see Figure 1). This ranges from the classical press-and-sinter or HIP routes to direct (beam based) or indirect (sinter based) additive manufacturing. Even for the process of sintering, different options are available such as e.g. solid state sintering or liquid phase sintering either with persistent or transient liquid phase. Regarding the time axis, slow processes – as typically sintering - or fast reactions, as e.g. in beam-based AM, may be applied.
A second source of flexibility is the wide range of starting powders available, regarding composition, particle size and size distribution, particle shape, etc. Some examples are shown in Figure 2; the pronounced differences in particle size as well as particle shape are clearly visible. This wide range enables selection of the optimum powder(s) for a given manufacturing route and resulting product.

Typical starting powders for powder metallurgy manufacturing. (a) Cr-Mo steel powder, water atomized (prealloyed), (b) carbonyl nickel (elemental), (c) ultrafine WC (compound), (d) 316L, ultrasonically atomized (prealloyed).
A third, possibly less well universally known, feature is the variety of alloying routes available in powder metallurgy, as schematically depicted in Figure 3. Except for single-component systems such as e.g. plain Fe or Cu, there are different ways to combine the elements required for a given product. This is in stark contrast to ingot metallurgy in which a homogeneous melt, which is in chemical equilibrium, is the starting point for a metallic product. The equivalent route in powder metallurgy is prealloying, in which typically a melt is atomized by a gas or a liquid, 7 and the solidified droplets form powder particles which all have more or less the same composition. Powders from chemical compounds, such as WC or TiC1−x, can be treated as “prealloyed” in most respects (occasionally also as masteralloys). Alternatively, however, different elemental powders may be combined by mixing (“blended elemental” approach), or several alloy additives may be added to a base powder through a single “masteralloy” powder.8–13 To avoid segregation effects the alloy particles may be bonded to a base powder by a moderate sintering treatment named “diffusion bonding”, 14 or the alloy element may be added by coating a base powder. Finally, several alloying variants may be combined in the form of “hybrid alloying”, e.g. by admixing further elemental powder to a prealloyed base. Except for the prealloyed variant, the other alloying routes offer the option to start from chemical nonequilibrium and to control processing towards chemical equilibrium, stopping this process at a desired stage.
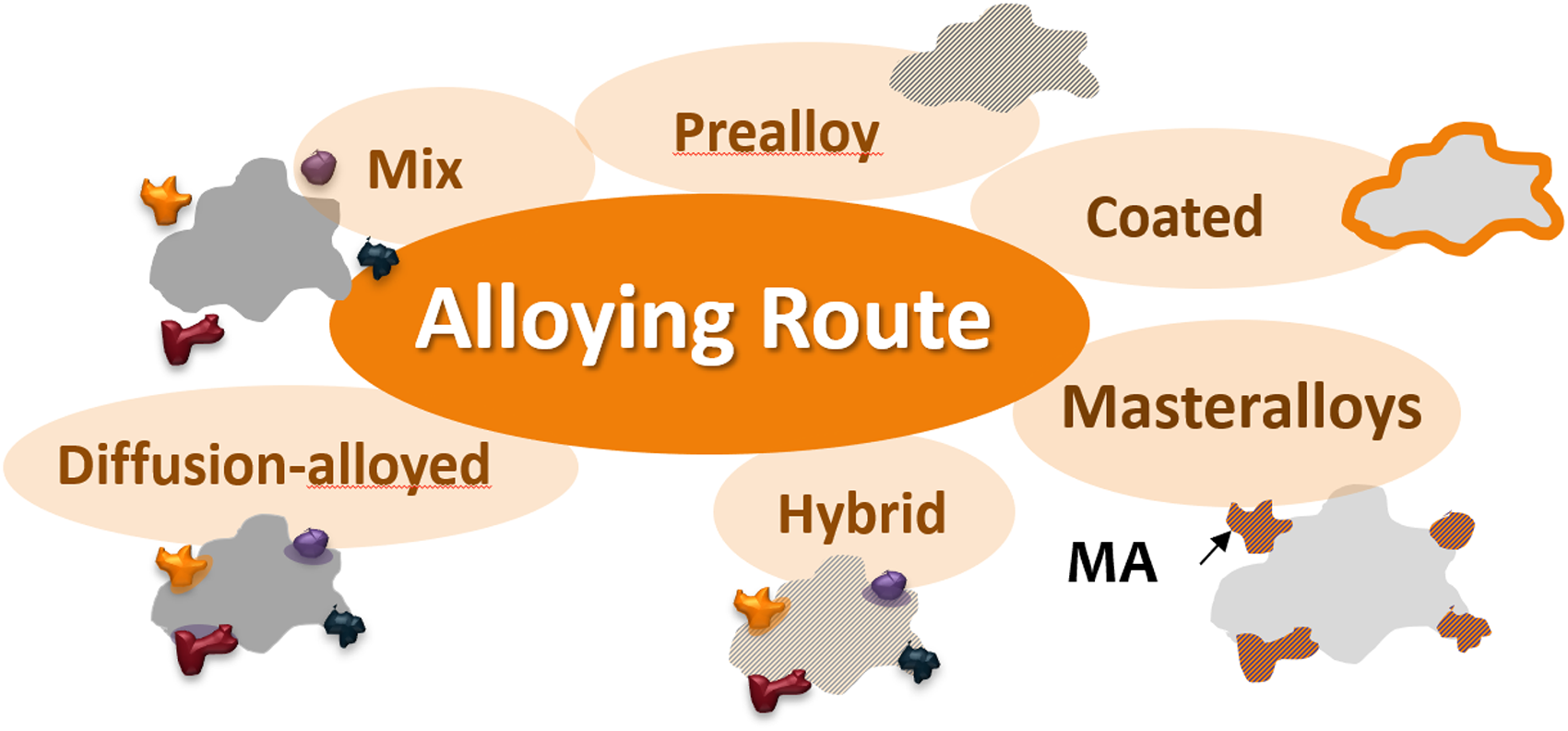
Alloying routes in powder metallurgy.
A comprehensive description of all aspects of alloying in powder metallurgy would require at least a book; in the present work, some selected examples are shown to give a preliminary insight into the potential of alloying in PM and how to select the best option.
Selection of the alloying route
The many available options for the alloying route in PM processing also involves the problem of selecting the best option for a given product. There are different aspects to be considered in this selection process, some of which are listed below:
Segregation risks
Desired microstructure
Sintering chemistry
Extreme variants
To better explain the differences between the alloying variants and their pros and cons, the extremes, prealloying and elemental powder mix, are compared in the following, and some typical examples are given.
Extreme 1: PM materials from prealloyed powders
Here, initially the metallic phase is chemically homogeneous.
Positive aspects
- Excellent homogeneity of the alloy elements
- Moderate heat treating / sintering temperatures possible
- Lower (and homogeneous) chemical activity of alloy elements = less reactivity e.g. with O2
- More straightforward sintering behaviour (also regarding the dimensions)
- No risk of secondary pores
- Better controllability esp. with beam-based AM
Negative aspects
- Available compositions restricted to those offered by the powder manufacturers (except for in-house production)
- Lower production volumes than for standard powders – mostly larger spread of properties
- Solid solution hardening = lower compressibility
- Only microstructures similar to cast / wrought materials possible
- Intraparticle internal getter effect may occur
- Impossible in many cases (W-Cu, WC-Co)
Figure 4 depicts the micrographs of materials produced from prealloyed powders. In Figure 4(a), the very regular and predictable solid state sintering process of prealloyed 316L is the major reason for using such a powder. Figure 4(b) shows a superalloy specimen produced by PBF-LB; here the very short time at temperature necessitates prealloyed powder grades since any powder mix would result in heterogeneous and probably unpredictable microstructures.

Systems for which prealloyed powders are preferable. (a) Stainless steel 316L, metal injection molded (photo courtesy Fraunhofer IFAM Bremen), (b) laser beam-powder bed fusion: as-built microstructure of superalloy 718L. 15
Figure 5 shows a further example for the benefit of prealloyed powders regarding control of the sintering process; the alternative, a powder mix, shows a very complex sintering behaviour, with first transient and subsequently persistent liquid phase. The secondary pores generated through transient liquid phase, as visible in Figure 5(a) (see16,17), act as nuclei for pore coalescence in the second stage of persistent liquid phase; the result is pore coarsening rather than pore elimination (=densification), as evident in Figure 5(b). In case of prealloyed base powder 410L, in contrast, only persistent liquid phase is formed, resulting in fully dense and regular microstructure, the process also being less sensitive to the temperature (Figure 5(c) and (d)).
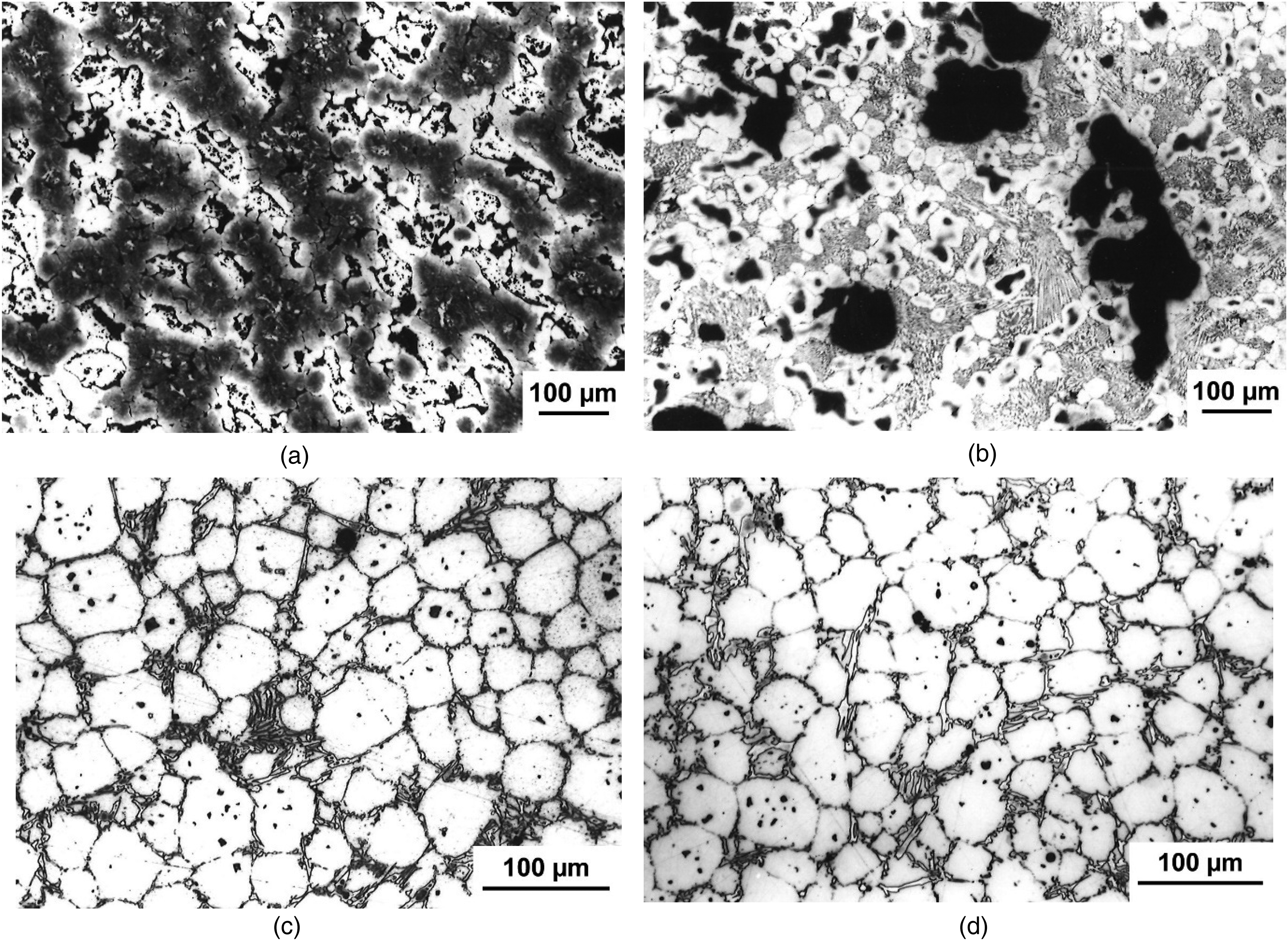
Elemental mix vs. prealloying: Wear resistant high Cr-high C steel sintered at different temperatures. (a) Elemental mix 1210 °C, (b) elemental mix 1240 °C, (c) prealloy 410L 1220 °C, (d) prealloy 410L 1240 °C.
However, prealloying is only feasible for systems in which a homogeneous melt can be produced that is subsequently atomized. For systems with limited miscibility other approaches are required, which will be discussed in the following.
Extreme 2: PM materials from mixed powders
Here, initially the metallic phases are chemically heterogeneous.
Positive aspects
- High compositional flexibility (unless diffusion bonded). Suited also for immiscible components (e.g. composites)
- Standard (e.g. elemental) powder grades available
- No solid solution hardening → good compressibility can be preserved
- Special microstructures available (Controlled heterogeneity)
- In part useful dimensional effects („copper swelling“)
Negative aspects
- Tendency to segregation and dusting (alleviated by diffusion bonding)
- Usually more complex sintering and homogenization behaviour.
- Intense heat treatment (sintering) necessary for full homogenization.
- Higher chemical activity of alloy elements = more danger of oxidation.
- Heterogeneity of oxygen affinity → interparticle internal getter effect
- In part secondary pores
Figure 6 shows examples of PM materials for which prealloying would not be possible. Tungsten heavy alloys are traditionally produced by mixing the elemental components, pressing and sintering with persistent liquid phase. In this case, the melting point of the main component W is above the boiling point of the binder component Ni, which precludes proper processing both through the ingot metallurgy route and through the manufacturing of prealloyed powders. In case of hardmetals, the manufacturing route is similar to heavy alloys, with the exception of much more intense mixing, typically by milling. Here, the peritectic decomposition of WC excludes producing of a homogeneous melt, as well as the boiling point of the binder element Co which is just slightly above the decomposition temperature of WC. Similar limitations hold e.g. for metal matrix composites such as Al-SiC; in these cases, the mixing route is the feasible option. Currently, hybrid variants are investigated for hardmetals, e.g. by combining WC with high entropy alloy binder powders produced by prealloying 18 ; time will show if these approaches will end up in technical products.

Systems for which classical prealloying is not feasible. (a) W-Ni-Fe heavy alloys: wide spread of melting and evaporation temperatures Fp(W): 3421 °C; Bp(Ni): 2730 °C, (b) WC-Co hardmetals: main component WC cannot be melted, decomposes at about 2800 °C.
There are also cases in which several options are attractive; one example is given in Figure 7. Low alloy Mo steels for PM precision parts may be produced from a powder mix or from an Fe-Mo prealloyed powder, if such a powder with desired composition is available. As evident from the micrographs, the prealloyed variant shows a very homogeneous microstructure (equivalent to that of a similar wrought steel) regardless of the sintering temperature, while the mixed type is rather heterogeneous, at least when sintered at moderate temperature. In this latter case, undissolved Mo particles are still present, due to a transformation to Mo-carbide. At higher temperature, transient liquid phase is formed which enhances Mo homogenization. The microstructure still seems to be slightly heterogeneous, but EPMA showed that the local differences of the Mo content are negligible. 19 This is also confirmed by the mechanical properties; as shown in Figure 8, the gigacycle fatigue behaviour of both high temperature sintered variants, elemental mix and prealloyed, are virtually the same. The mixed variant sintered at lower temperatures performs less in structural applications; however, it can afford attractive wear properties, e.g. for valve seat inserts, if the carbon content is properly adjusted to result in formation of the carbide Mo2C. 20 Such a carbide-reinforced microstructure is another example for a material that is accessible only through a mixing route, by stopping the Mo dissolution and homogenization at the desired stage.

Sintered steel Fe-1.5%Mo-0.7%C, manufactured through different alloying routes. 19 (a) Prealloyed, 1200 °C, (b) elemental mix, 1200 °C, (c) prealloyed, 1320 °C, (d) elemental mix, 1320 °C.
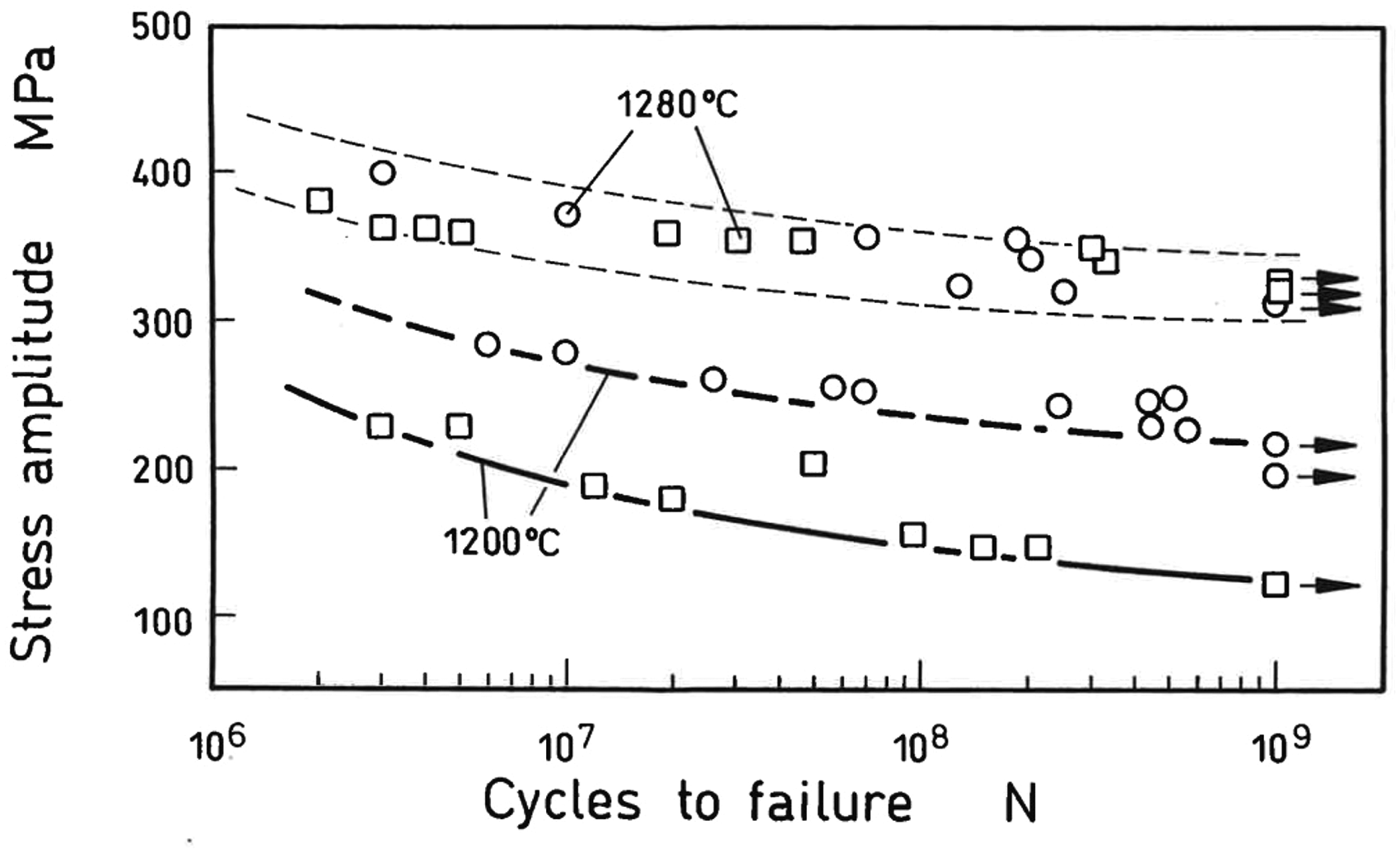
Gigacycle fatigue endurance strength of Mo steel prepared through different alloying techniques: elemental powder mix vs. prealloying. 20
Coated powders
In principle, coating a base powder with a thin layer of another metal is a very attractive alloying variants since it ensures regular distribution of the alloy element, avoids segregation – at least if the layer adheres well – and retains the compressibility of the base powders. The main difficulty is the coating process itself, which should be cost-effective and suited for upscaling.
There are several techniques for coating of metal powders with metals (see e.g. 3 ), some of which originate from treatment of cast or wrought metals. A simple and easily applicable method is the displacement reaction, i.e. depositing a more noble metal at the expense of the base metal, such as Cu on Fe. This just requires immersion of the powder in an aqueous Cu solution, stirring, and then collecting and drying the coated powder. This process has been applied e.g. to high speed steel powders to obtain materials with high wear resistance and good thermal conductivity, as required for valve seat inserts. 21 Compared to simple powder mixes, the coated powders result in finer and more regular microstructure, as shown in Figure 9.

High speed steel M2 (HS 6-5-2)-Cu sintered specimens; Cu coated by cementation vs. admixed. 22 (a) M2 powder, as delivered, (b) M2 powder, Cu coated, (c) M2-Cu, from powder mix, (d) M2-Cu, from coated powder.
For many other combinations, the displacement reaction is not feasible. Here, electroless plating may be an option, the reactions being easy to perform, and numerous commercial plating baths are available. However, the residues of the reducing agent, such as P in Ni layers, may offer problems. Electroplating, on the other hand, is rather flexible regarding the material combinations, but it requires sophisticated equipment to ensure regular contacting of each powder particle at the cathode to ensure homogeneous coating. A possible way of using a vibrating bed electroplating reactor has been described in 23 ; thus, regular plain Ni layers could be deposited on Fe powder particles. After admixing graphite, pressing and sintering, regular “microgradient” structures with pearlitic (after quench and temper treatment, martensitic) islands in a continuous austenitic network were obtained (Figure 10). This could be a logical extrapolation of the “Distaloy” microstructure which has been successfully used for PM precision parts for many decades. 14
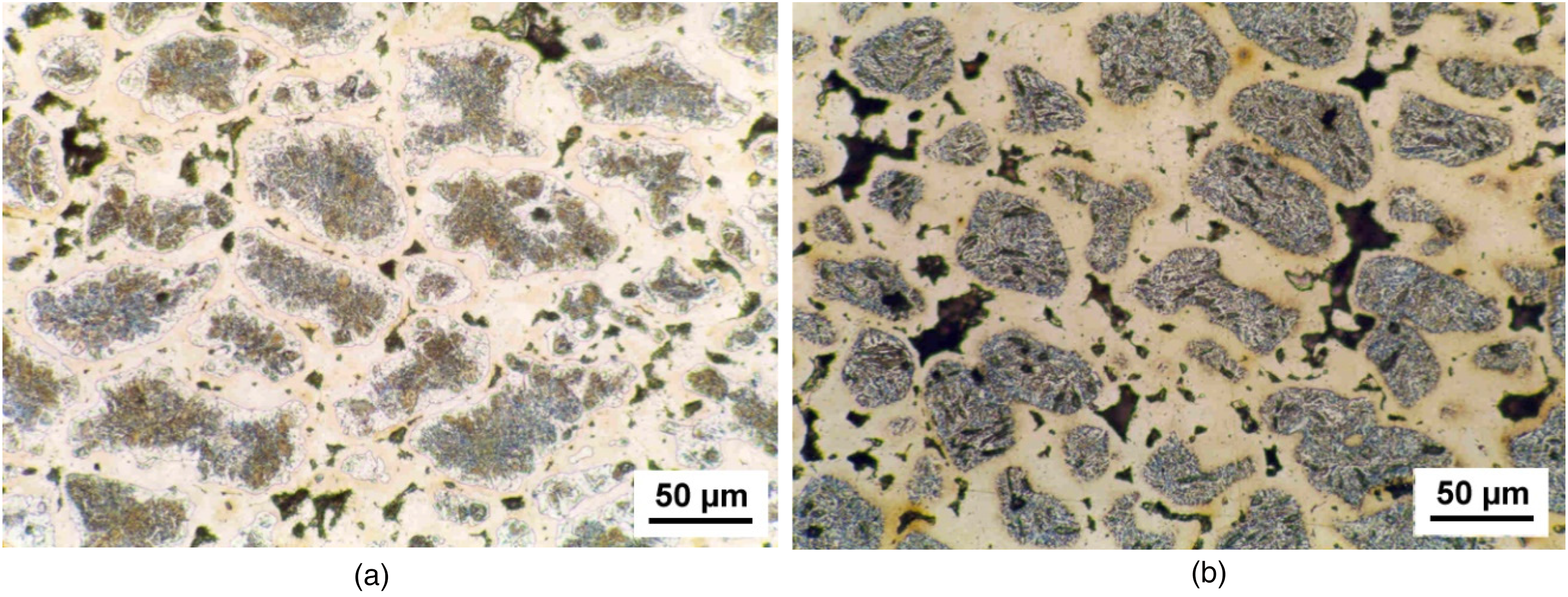
Fe-9%Ni-1.2%C, Ni electroplated on Fe; sintered 1 h 1000 °C. (a) As sintered, (b) quenched and tempered.
Deoxidation reactions
One characteristic feature – and frequently obstacle – is the oxygen content of all powders that have ever been exposed to air and/or humidity, which is mostly concentrated at the powder surfaces but in part is present also within the particles. In particular the surface oxides may pose problems unless removed in an early stage of consolidation, since such layers may adversely affect the interparticle cohesion and, because of the much lower surface energy compared to metal surfaces, also strongly decrease the sintering activity.
When removing the oxygen from a bulk powder or a compact, the interaction with densification is a crucial item. Typically, the powder systems have certain temperature intervals in which deoxidation occurs, mostly by reaction with H2 or C, and these intervals should be sufficiently below the temperature range at which densification results in closing of the pores and thus trapping of oxygen. This is an item that has to be considered e.g. in sintering of ultrafine hardmetals in which densification starts are at fairly low temperatures, but on the other hand the grain growth inhibitors necessary in these systems (usually V and/or Cr carbides) are carbothermally reduced at markedly higher temperatures than the main constituents W and Co. 24 Another example is shown in Figure 11: In this case the deoxidation interval is rather the same for all variants, but at higher C content the temperature of the densification interval decreases and finally intersects with the former interval, the CO generated being trapped within the compact and resulting in massive blistering.

Sintered steel (prealloy) Fe-Cr-Mo-Mn-Ni-Cu-P – x wt. %C, (x = 1.7%–2.0%–2.5%C), sintered at 1120 °C in N2-H2.
The deoxidation process also depends strongly on the alloy system chosen. For compositionally homogeneous systems - prealloyed powders and of course also single component systems - there is usually one well defined deoxidation interval, while for compositionally heterogeneous systems the reduction intervals of the individual components may vary considerably. In both cases, however, the element with the highest oxygen affinity (at least if present in significant amounts and thus with reasonably chemical activity) tends to dominate the temperature oxygen removal since oxygen transfer reactions, dubbed “internal gettering” may occur if the oxygen affinities of the elements involved differ significantly.
In prealloyed powders, this oxygen transfer occurs within the individual powder particles. Since the chemical activity of metallic elements present in oxides is much lower than in the metallic state, elements with high oxygen affinity tend to diffuse to the surface oxide layers, displacing less stable oxides there. This is schematically shown in Figure 12 which depicts a Cr-Mo prealloyed steel in which the surface initially consists of iron oxide with dispersed more stable oxides of Cr and Mn.25,26 As soon as Cr diffusion becomes possible – even at short distances – the iron oxide is transformed into a Cr-Mn-Fe oxides which requires markedly higher temperatures for reduction. Therefore, the main deoxidation peak is that for the mixed oxide and not that for iron oxide, as might be expected from the initial state. 27
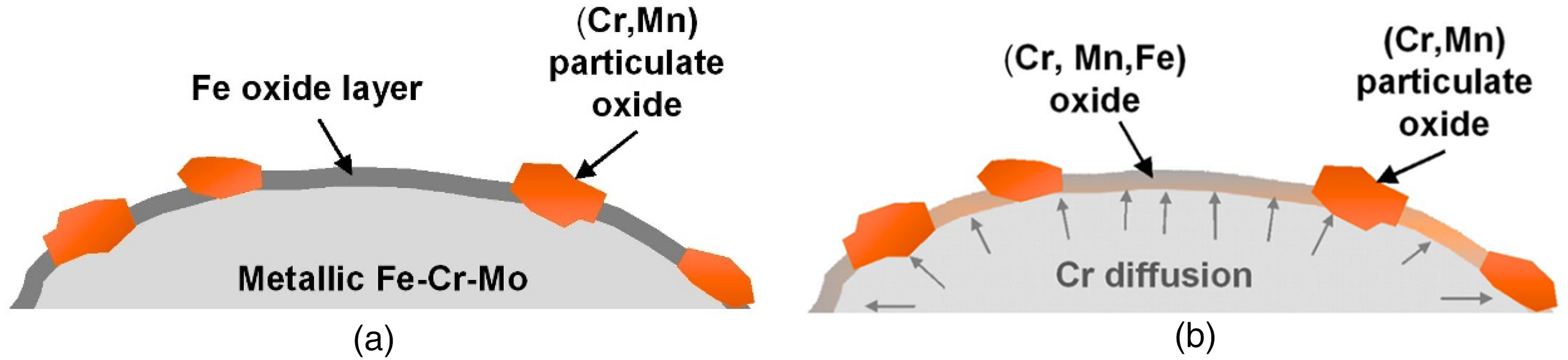
Schematic of „internal getter“ effect in Cr-Mo prealloyed steel powders: oxygen remains on site, Cr diffuses: „intraparticle“ effect. (a) Initial state (as delivered powder), (b) during annealing treatment / during heating.
In compositionally heterogeneous systems in which different powders are combined, as elemental or as masteralloys, interparticle oxygen transfer occurs, as schematically shown in Figure 13. This is particularly crucial if the oxygen affinity of the alloy element(s) is markedly higher than that of the base powder. In this case the surface oxides of the base powder are reduced, forming CO or H2O, but the atmosphere in the pore network thus formed is strongly oxidizing for the alloy element(s) which immediately getter the oxygen. In these conditions, hardly any oxygen is removed from the system until the conditions – which typically means the temperature – is such that also reduction of the most stable oxide in the system becomes possible. This is clearly visible when comparing the carbothermal reduction in mixed compacts Fe-C vs. Fe-Mn-C (see 27 ): while the surface oxides in Fe-C are removed in the temperature range 700 … 800 °C, addition of 4% elemental Mn shifts the deoxidation peak to 1200 °C, i.e. the reduction temperature for Mn oxides. This clearly illustrates that also here, oxygen removal is dominated by the most oxygen-affine constituent. This “internal getter” effect may be alleviated if it is possible to perform the deoxidation of the base powder at a temperature at which the reactivity of the alloy element(s) is still sufficiently low to prevent gettering. This may be the case when using the higher reducing power of H2 compared to C in the lower temperature range (see. e.g. 26 ).
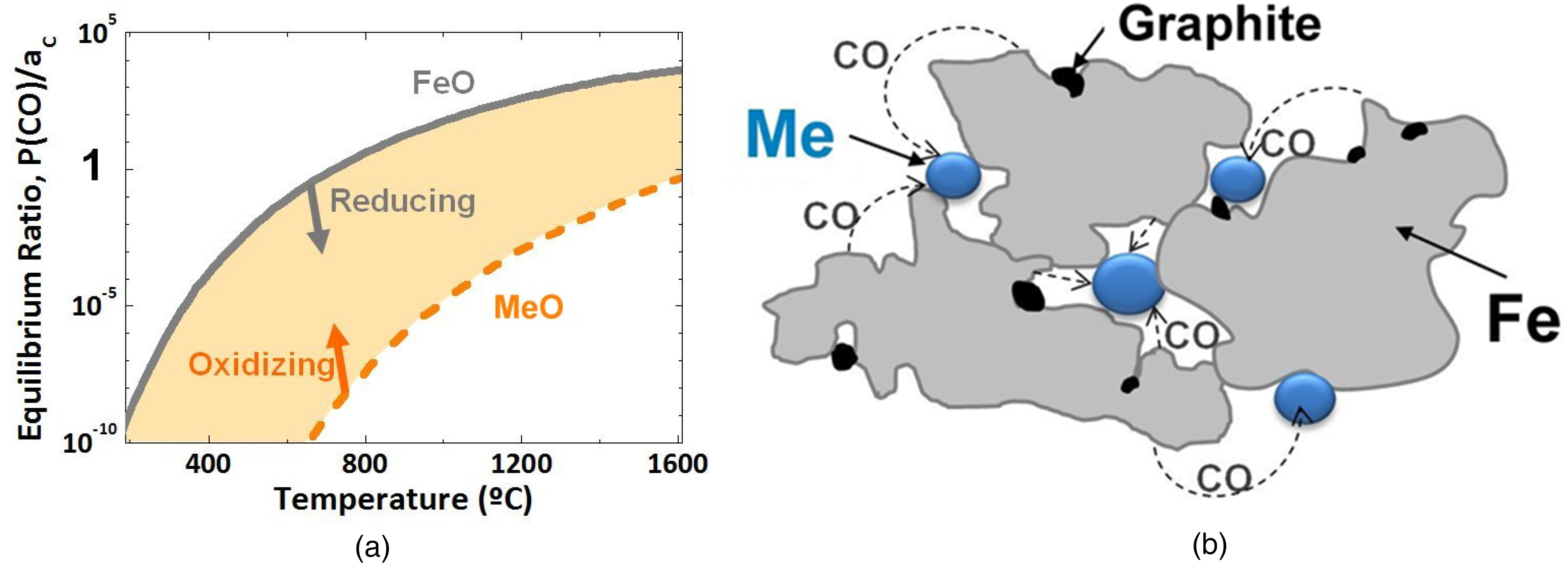
„Internal getter effect“ in ferrous powder mixes of components with different oxygen affinity: in fact metallothermic reduction. (a) Thermodynamics of carbothermal reduction, (b) schematic of interparticle internal getter effect in ferrous powder mix.
Of course the oxygen affinity of a given component is also affected by its concentration, strictly speaking its chemical activity. In elemental powders the activity is 1, and the oxygen affinity is therefore maximum for this element. In prealloyed systems, and to some degree also in masteralloys, concentration and activity are lower, and oxygen removal is somewhat easier; therefore, the masteralloy approach may be preferable to the elemental powder mix, in particular when element with high oxygen affinity are involved, as shown e.g. by Hryha et al. for Mn alloyed steels.28,29
Conclusions
- In addition to a wide range of materials, compositions and production variants, powder metallurgy also offers numerous alloying routes from which the best way can be chosen
- Selection of the optimum alloying variant has to be done carefully, balancing the pros and cons of each route and considering the production route, chemical aspects as well as the desired microstructure, esp. homogeneous vs. heterogeneous, and resulting properties
- Prealloyed powders are usually more easily processed if compressibility is not a major item and if preparation of the powders is technically possible
- Powder mixes offer higher compositional flexibility and the use of standard powder grades; however, there is risk of segregation and usually more complex sintering behaviour. If homogeneous microstructures are required, usually intense heat treatment (high temperatures and/or long times) are required. On the other hand, microstructures with defined heterogeneity are possible which would be inaccessible by ingot metallurgy.
- Powder coating is an interesting variant that enables e.g. insulating layers for SMCs or microstructures with desired and very regular heterogeneity, but is in part difficult to upscale
- Removal of the natural oxygen content can be retarded by “internal getter” effects, interparticle for mixes and intraparticle for prealloyed powders
List of abbreviations
AM: Additive Manufachturing; EPMA: European Powder Metallurgy Association; HIP: Hot Isostatic Pressing; MIM: Metal Injection Molding; PM: Powder Metallurgy; PBF-LB: Powder Bed Fusion – Laser Beam; SMCs: Soft Magnetic Composites.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
