Abstract
Binder jetting (BJT) utilises a sacrificial binder to shape green parts in a metal powder bed. The binder must be removed during debinding at elevated temperatures without contaminating the powder material. Efficient binder removal often requires oxygen in the atmosphere to aid the decomposition of the binder, but oxygen causes oxidation of the metal powders and hence negatively impacts the final material properties. Therefore, the use of tailored oxygen contents in the atmosphere during a standard debinding cycle performed at 300°C for 2 h was investigated for BJT printed 17-4 PH stainless steel. The change in bulk chemistry (carbon, oxygen, nitrogen, and hydrogen) from green parts to brown parts was thoroughly analysed and compared to the virgin powder subjected to the same conditions as used for debinding. While inert argon was beneficial for protecting the powder from oxidation, the removal of only ∼34% of binder was insufficient, resulting in 0.47 wt.% of residual carbon in the brown part. Debinding in atmospheres containing 3 vol.% O2 to 20 vol.% O2 led to almost complete binder removal, but also caused significant oxygen pickup by the powder between 1734 ppm and 1820 ppm. Furthermore, the brittleness of the brown parts increased, leading to undesirable powder losses during handling. The best debinding was obtained in Ar + 1 vol.% O2, which allowed the best combination of binder removal of ∼74% and lower oxygen pickup by the powder of 1617 ppm, together with sufficient brown part strength for handling.
Introduction
Binder jetting (BJT) is considered a promising additive manufacturing technology for the cost-efficient production of metal components with complex geometries at high volumes.1–3 The principle of BJT relies on the use of a binder to shape green parts in a powder bed. 3 The concept of the binder is sacrificial as its purpose is only to stabilise the geometry of the component until the sintering process begins. The binder needs to be completely and efficiently removed from the green part as organic binder residues are often detrimental to the material properties, limiting the adoption of BJT for high-performance materials and applications.
Thermal debinding is sufficient for binder removal, as the binder content in BJT green parts is relatively low compared to other binder-based metal manufacturing technologies such as metal injection moulding or material extrusion that might require a prior solvent debinding step. 2 During thermal debinding, the binder is burned out, pyrolysed or evaporated at elevated temperatures, typically below 600°C. The binder needs to be completely removed before the open porous structure is closed at higher temperatures by sintering densification. The thermal debinding process is mainly defined by the heating rate, holding temperatures, holding times and processing atmosphere. The choice of parameters depends mainly on the binder chemistry, part sizes and intended application. Most research on thermal debinding focused on heating rates, temperatures and holding times to optimise the debinding process. The effect of the debinding atmosphere on BJT parts has not been systematically studied despite its crucial impact on debinding efficiency.
The atmosphere during debinding can serve two purposes. On one hand, a constant stream of gas ensures that debinding products are carried away from the components. 4 On the other hand, the gas can provide reactive species such as oxygen (O2), water (H2O) or hydrogen (H2) aiding the decomposition of the organic binder. 5 Organic binders typically fragment into various vaporous products including hydrocarbons, water (H2O), carbon dioxide (CO2) and carbon monoxide (CO). 5 The composition of the process atmosphere determines the decomposition reactions and products occurring during debinding. The choice of atmosphere can vary from oxidising, reducing, wet (high dew point) to dry (low dew point) compositions. Widely adopted thermal debinding atmospheres include argon (Ar), nitrogen (N2), hydrogen (H2), vacuum or air.
Inert atmospheres, such as Ar or N2, are often not sufficient to remove the binder completely leaving behind excessive carbon contamination. 5 Debinding in vacuum, while viable, poses challenges such as clogging of vacuum pumps and high wear on sensitive furnace components due to condensing debinding products. Debinding and sintering in a pure hydrogen atmosphere is a common practice in powder metallurgy (PM). 6 The use of hydrogen might, however, be restricted due to safety concerns or local regulations regarding the risk of forming explosive mixtures with oxygen.7,8 Furthermore, debinding in hydrogen often requires high temperatures to efficiently remove the binder, which is not feasible for low-melting point metals.
A simple method to efficiently remove the organic binder, especially carbon, is debinding in air. 5 The oxygen in the air helps to decompose the binder into low-molecular weight products that are easier to remove. 6 Debinding under air is often performed in a separate and low-cost furnace to avoid the contamination of expensive and sensitive sintering furnace equipment with debinding products. The contamination of the high-purity sintering atmosphere is further avoided. The issue of debinding in air is, however, the oxidation of the metal powder. This limits the air debinding to low temperatures. Even at low temperatures, oxidation can have a detrimental impact on the subsequent sintering densification and final material properties. Oxides at the surface of powder particles hinder the formation of sinter necks resulting in a decrease in sintered strength and ductility. 9 While the removal of iron oxides is considered relatively simple, iron oxides can be transformed during heating into more stable oxides containing Cr.10–15
The weight fraction of organic binder in a BJT green part is comparable to the amount of organic lubricant in PM compacts. The importance of proper lubricant removal from the powder compacts was highlighted by several studies, especially for PM steels containing oxidation-sensitive elements such as Cr, Mn or Si.16–21 Hryha et al. 19 showed the importance of atmosphere control during delubrication and sintering to minimise powder oxidation during the heating stage. Karamchedu et al. 16 reported that delubrication in air at 450°C increased the thickness of the iron-oxide layer of Cr-alloyed powder. The oxidation led to higher carbon loss and higher oxygen content after sintering, which resulted in lower hardness and impact toughness compared to delubrication in dry N2 at 450°C. 16 Tan et al. 20 demonstrated the detrimental impact of oxidation of Cr-alloyed steels during delubrication in oxygen-containing atmospheres on as-sintered mechanical properties.
Dominant applications for stainless steel parts produced by binder-assisted powder processing technologies, including BJT, are structural components. 22 The required level of mechanical properties is sensitive to contamination by carbon, nitrogen and oxygen. 23 While oxygen, nitrogen and boron are usually not specified for stainless steels in contrast to carbon content, their concentrations are important for processing and the sintered properties. 23 Furthermore, corrosion resistance is negatively impacted by oxygen. One of the most popular stainless steels for BJT is 17-4 PH due to its unique combination of mechanical properties and corrosion resistance. 17-4 PH contains a high fraction of oxidation-sensitive elements such as Cr, Mn and Si. Hence, oxidation of the powder needs to be avoided. A potential approach to balance efficient binder removal with limited powder oxidation is to decrease the oxygen content in the debinding atmosphere.
In the first part of this study, the impact of decreasing oxygen contents in the debinding atmosphere was investigated for 17-4 PH stainless steel green parts manufactured via BJT. A thorough understanding of the impact of binder on the green part density and chemistry was established. The chemical evolution of carbon, oxygen, nitrogen and hydrogen was tracked from virgin powder to brown part and utilised to assess the binder residuals in the material. The investigated debinding atmospheres at 300°C for 2 h were Ar, Ar + 1 vol.% O2, Ar + 3 vol.% O2, Ar + 4 vol.% O2 + 5 vol.% CO2, Ar + 8 vol.% O2 and N2 + 20 vol.% O2 (air). The debinding efficiency was evaluated by weight loss, brown part chemistry and powder chemistry. In the second part of the study, the impact of the debinding atmosphere on sintering in inert Ar was examined. 24
Materials and methods
Materials and printing
Nitrogen-atomised 17-4 PH stainless steel powder (17-4 PH, Desktop Metal, USA) was used for the debinding study. The elemental composition of the virgin powder is listed in Table 1. The particle size distribution of the powder was D10 = 6.5 µm, D50 = 15.3 µm and D90 = 28.2 µm. The density of the 17-4 PH powder was 7.82 g/cm3 measured by helium pycnometry.
Elemental composition of the 17-4 PH stainless steel powder used for printing in wt.%.

A water-based binder comprising solvents and a thermoset polymer (SPJ-04, Desktop Metal, USA) were used for printing on a Production System™ P-1 (Desktop Metal, USA). The printer is based on Single Pass Jetting technology, where one layer is printed by powder spreading, powder compaction, powder bed steaming and binder deposition in one single pass of the carriage system.
Cuboid test specimens with nominal dimensions of 10 × 10 × 10 mm³ and cylinders with a height of 11.9 mm and a diameter of 2.9 mm were printed for debinding and sintering. The cuboid samples were used for density evaluation, whereas the cylinders were used for chemical analysis. The powder layer thickness was 65 µm. Printing was conducted in an inert nitrogen environment (<2.2 vol.% O2). A steamer in the printer was present to humidify each deposited powder layer to prevent powder ejection. The humidity in the print chamber was kept at a constant level of 40% relative humidity by purging with N2 during printing.
The used binder saturation values are given in Table 2. The slicing software defines the binder deposition based on the cross-sectional geometry at the specific layer height. The two-dimensional printing pattern of the part in each layer is subdivided into distinct regions with varying binder saturations: a shell, an inner shell and an inner area of the geometry. The shell thickness defines the contour thickness of the part. The shell and the inner region are separated by the inner shell with its own thickness and saturation. The inner saturation defines the binder saturation of the inner region. The first and last layers of a part are printed without shells. The bottom thickness and top thickness specify the number of first and last layers without shells and the corresponding saturation.
Binder saturation parameters used in this study.

The consumed binder volume for a cuboid sample was calculated to be 0.256 ml by the build job preparation software. To predict the binder content in a green sample, the weight change of the pure liquid binder was measured after drying at room temperature for 24 h, after curing and after debinding in ambient air.
The printed samples in the build box were subjected to curing to enable crosslinking of the binder. The curing process was performed in a furnace with forced air convection (TR 120 LS, Nabertherm GmbH, Germany) at 200°C for 4 h with a heating rate of 1.5°C/min and a natural furnace cooling. For depowdering of the green parts, brushes and a compressed air gun were used. To investigate the influence of curing on the binder in the green sample, a dedicated build job was left for drying for 24 h at room temperature. The dried green samples were removed from the powder bed for analysis.
The green densities of printed parts were determined after depowdering by weight and volume to ensure comparable starting conditions before debinding. The mass was determined with a precision scale (QUINTIX224-S1, Sartorius AG, Germany) with an accuracy of ±0.001 g. The dimensions of the cuboid specimens were measured in the three spatial directions to calculate the volume. The dimensions were measured using a micrometer screw gauge with an accuracy of ±1 μm.
Thermal debinding
After removing the green parts from the powder bed, the green parts were debinded in a tube furnace with a steel muffle at 300°C for 2 h. The heating rate for the debinding was 3°C/min. Natural cooling took place in the furnace for ∼12 h. A consistent temperature profile was ensured by a thermocouple placed near the samples. The investigated atmospheres for debinding with varied oxygen content are listed in Table 3. The oxygen content at the outlet of the tube furnace was monitored during the thermal treatment to ensure the purity of the atmosphere, especially during debinding in Ar. The weight of the cuboid brown parts after debinding was measured to determine the weight loss.
Investigated debinding atmospheres with varying oxygen content.

Furthermore, the 17-4 PH virgin powder was subjected to the same debinding cycle in the listed debinding atmospheres. The aim was to assess the impact on the isolated powder chemistry and compare it to the brown part chemistry. The experimental procedure for the green parts and powder is highlighted in Figure 1.
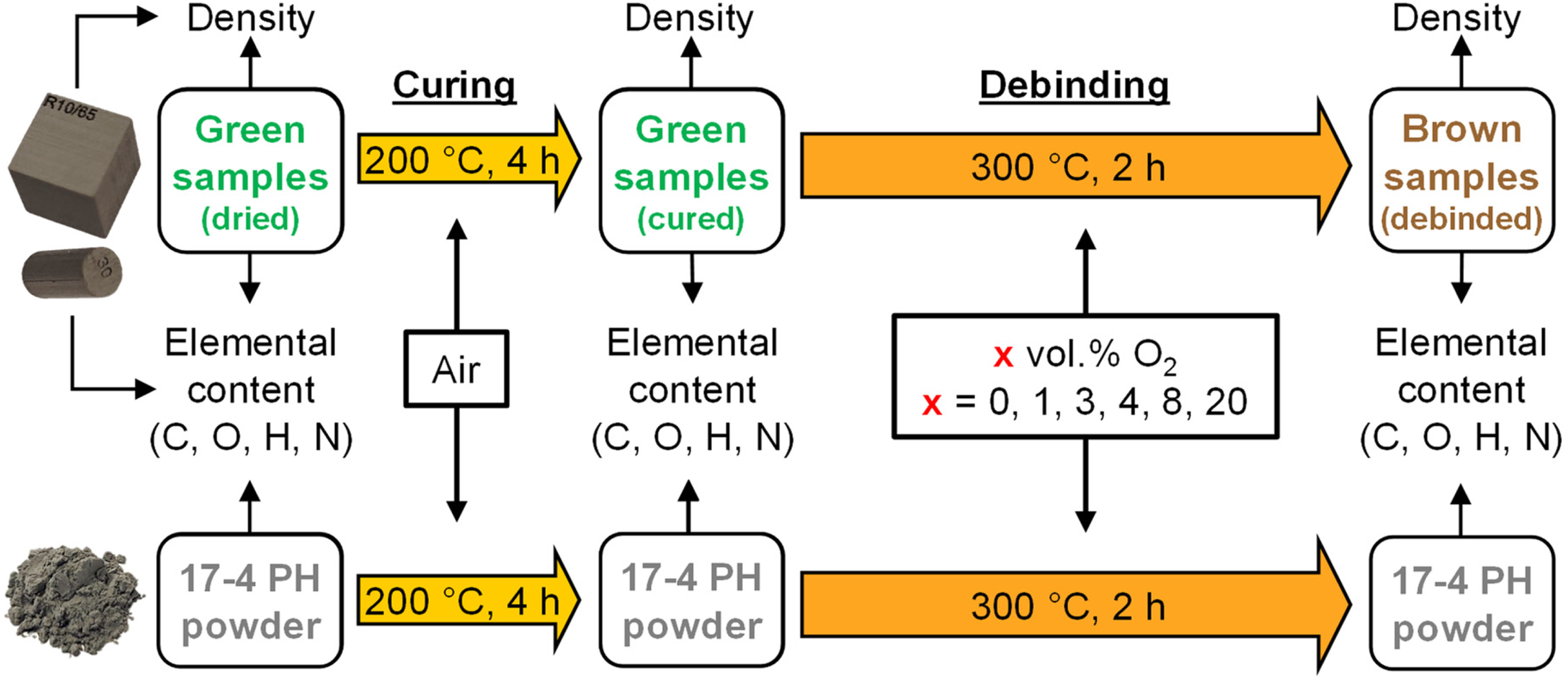
Experimental flow chart for the investigation of debinding in atmospheres with varying oxygen content.
Chemical analysis
The oxygen, nitrogen and hydrogen contents were measured through the inert gas fusion technique (ONH836, LECO Corporation, USA). Three cylindrical samples after printing and after debinding were analysed. The carbon content was also measured via the inert gas fusion method (CS 2000, ELTRA GmbH, Germany) using 3 cylindrical samples per debinding atmosphere. 17-4 PH powder samples were analysed by the same measurement techniques. The oxygen, nitrogen and hydrogen contents of the binder were also determined, but the measured sample mass was considerably reduced to ∼0.01 g to stay within the measuring range of the analytical equipment, as the maximum measuring range was designed for inorganic materials. The binder was put in uncured, cured and debinded condition into the measuring device.
Results and discussion
Analysis of binder and 17-4 PH powder
To estimate the binder content in a green part, the weight loss of the liquid binder after drying, curing and debinding was measured. The resulting weight of the binder is shown in Figure 2(a). The drying of the binder in ambient conditions at room temperature led to a loss of 71 wt.%. After curing at 200°C for 4 h, the mass of the dried binder decreased further by 11 wt.%. The binder residue after curing corresponded to 18 wt.% of the initial liquid binder mass. After debinding at 300°C for 2 h, 6.7 wt.% of the initial liquid binder remained. Debinding changed the visual appearance of the binder to a black and porous structure (see Figure 7(b)).
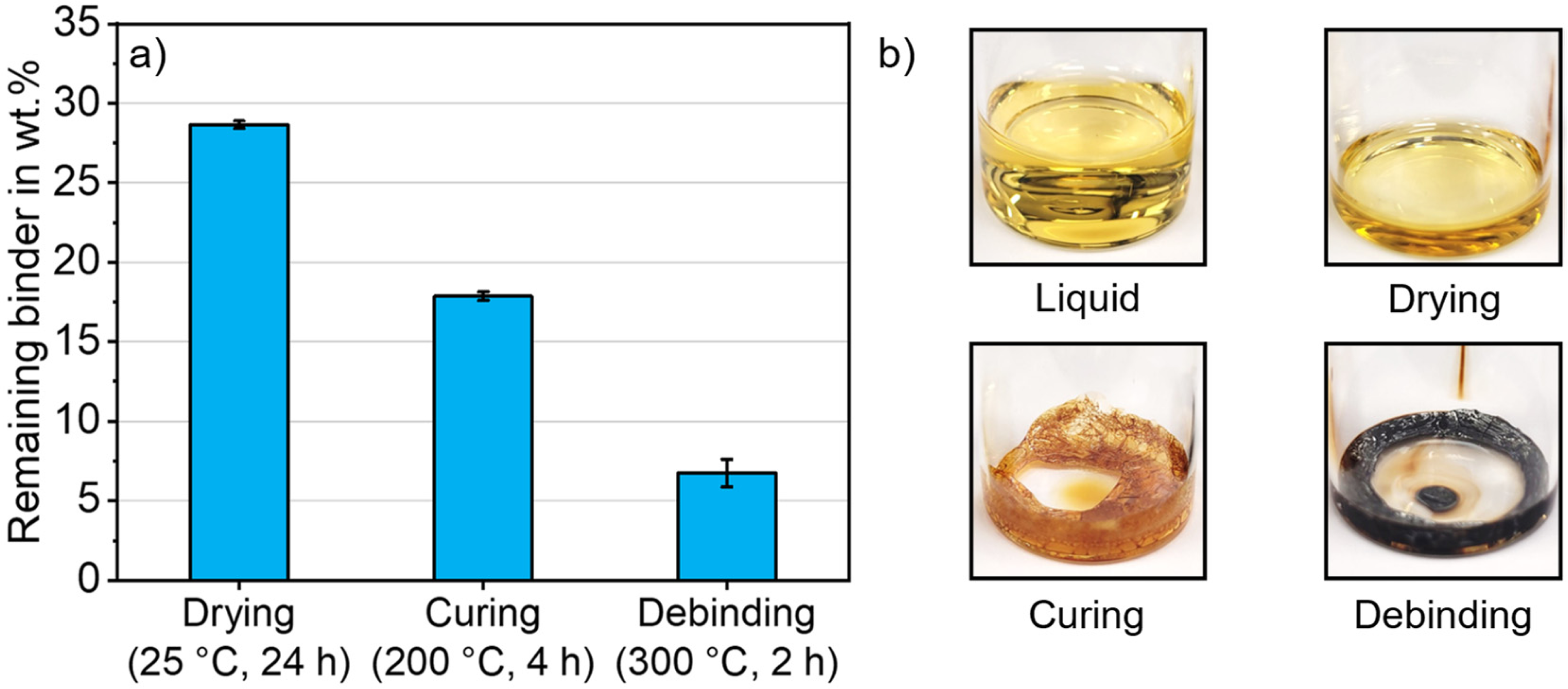
(a) Remaining binder weight after evaporation, curing and debinding in ambient air and (b) visual appearance of the binder in liquid state, after drying, after curing and after debinding.
The amount of binder used for printing a cuboid sample (1 cm³) was 0.256 ml, which was calculated by the build job preparation software applying the set saturation parameters. Considering the binder density of 1.04 g/cm³, the corresponding weight of the used liquid binder was roughly 0.246 g for a cuboid sample. Based on the measured weight losses, the binder was expected to add 0.062 g/cm³ to the green part density after drying and 0.044 g/cm³ after curing. The mass fraction of the binder in a green sample depends on the powder density and packing.
To assess how the binder affects the chemistry of a green part, the powder and binder were subjected to curing and debinding individually. The change in oxygen, nitrogen and hydrogen contents of the binder are presented in Figure 3(a). A significant loss of oxygen and hydrogen content was registered. This was mainly caused by the evaporation of water, which is the main constituent of the binder. After debinding at 300°C for 2 h, the oxygen content in the binder decreased further considerably, while the hydrogen fraction remained comparable. The remaining elemental content in the binder is expected to be mainly carbon.
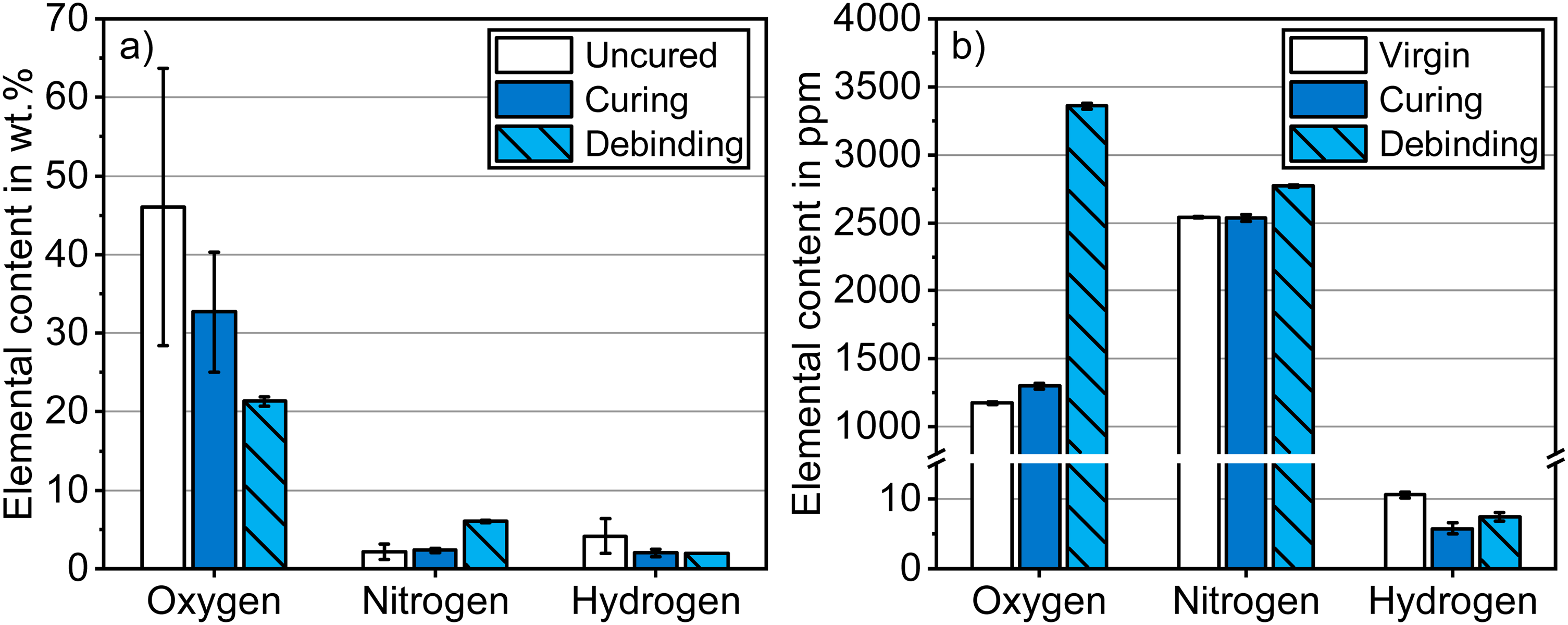
Oxygen, nitrogen and hydrogen contents of the (a) isolated binder and (b) 17-4 PH virgin powder before curing, after curing and after debinding in ambient air.
The oxygen, nitrogen and hydrogen contents of the 17-4 PH powder subjected to curing and debinding can be seen in Figure 3(b). The nitrogen and hydrogen contents of the powder remained comparable after thermal treatment. The nitrogen stems from the nitrogen atomisation process. The hydrogen content was negligible in the metal powder. A slight pickup of oxygen by ∼11% was measured after curing. A substantial increase in oxygen by 186% compared to the virgin powder was caused by oxidation of the powder during debinding at 300°C in ambient air. The carbon content of the 17-4 PH virgin powder was determined to be 245 ± 9 ppm.
Impact of binder on green part density and bulk chemistry
The impact of the binder on the green part chemistry is highlighted in Figure 4 displaying the bulk chemistry of the green part in comparison to the corresponding powder chemistry. The data confirmed the significant increase of carbon, oxygen, nitrogen and hydrogen content in a green part compared to the 17-4 PH virgin powder. Carbon and hydrogen were mostly independent indicators of the binder in the 17-4 PH green samples since their initial content in the 17-4 PH powder was comparatively low. In contrast, oxygen and nitrogen contents were more dependent on the powder composition.
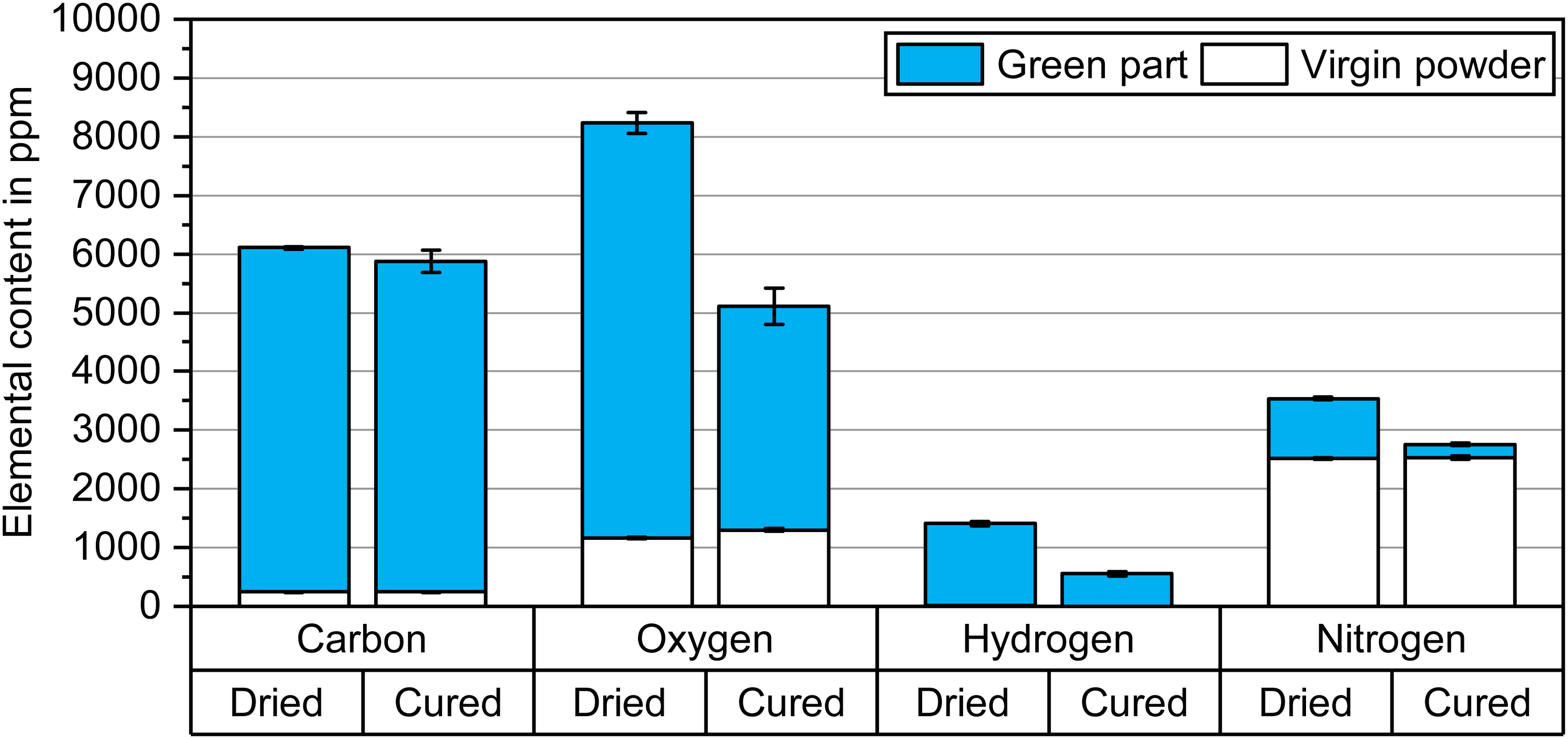
Carbon, oxygen, hydrogen and nitrogen contents of the 17-4 PH powder and green parts before and after the curing process at 200°C for 4 h.
The elemental analysis can be used to estimate the binder content by subtracting the 17-4 PH powder weight fractions of carbon, oxygen, hydrogen and nitrogen from the green sample weight fractions. This is a simplification, as the actual chemistry of the powder in the green part is unknown. It is assumed that the powder chemistry is comparable to the isolated powder subjected to the same thermal treatment. The sum of the additional weight fractions can, however, approximate the binder content in a green sample and hence provide a semi-quantitative estimation of the binder residue content. The results of the calculated binder contents are listed in Table 4.
Binder content calculations of the green parts after drying and curing based on the measured green part and powder chemistry.
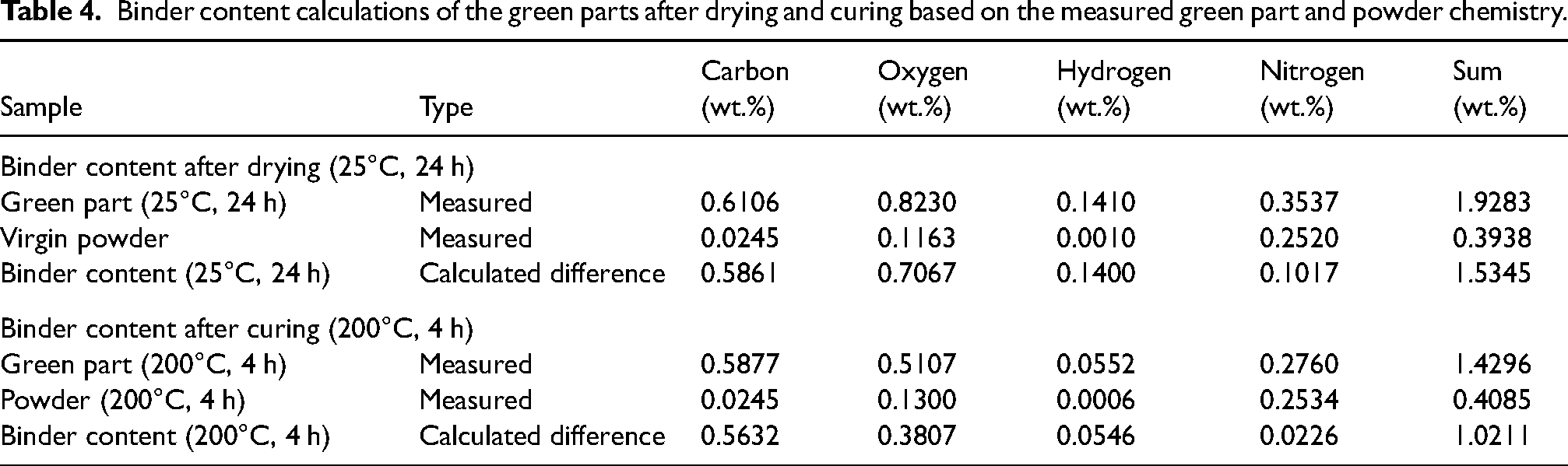
The average green density of the cuboid samples used in the study was 4.75 ± 0.04 g/cm³, which equals 60.7 ± 0.5% relative green density assuming a bulk density of 7.82 g/cm³. The estimated binder content in a dried green sample was 1.53 wt.%, which would amount to 0.072 g/cm³ of density added by the binder after drying. After curing, the remaining binder content was estimated to be 1.02 wt.%, which would correspond to a contribution of 0.048 g/cm³ to the green part density.
The calculated binder contents approximated by the green part and powder chemistry matched the previous calculations of binder content from the ‘Analysis of binder and 17-4 PH powder’ section, which were based on the printed binder volume from the printing parameters. Therefore, printing parameters and binder weight losses seem to provide a rather accurate estimation of the binder content in a green sample as confirmed by the bulk chemical analysis and calculations above. Catalytic effects of the powder might, however, change the behavior of the binder at elevated temperatures. The impact seemed to be negligible for the measured samples. In conclusion, the binder accounted for ∼0.6% of the relative density of the green part. Hence, the actual 17-4 PH powder packing density in the green part was around 4.70 g/cm³ or 60.1%, respectively.
Since the green samples were printed with the same parameters, especially considering binder saturation, the initial binder content and composition of all samples were assumed to be the same. The characterised carbon, oxygen, nitrogen and hydrogen contents of the cured green samples and 17-4 PH powder provided the baseline to assess the impact of the debinding atmosphere on the brown parts.
Elemental change of the binder after curing
Figure 5 visualises the binder weight composition inside of a green part after drying and after curing. In addition, the composition of the binder removed during the curing process is displayed. The main weight fractions of the dried binder (1.53 wt.%) inside the green part were oxygen at 46% and carbon at 38%. Hydrogen and nitrogen were present in a comparable weight fraction of 9% and 7% respectively. As the binder was already in a dried state, most of the water of the liquid binder had already been evaporated.
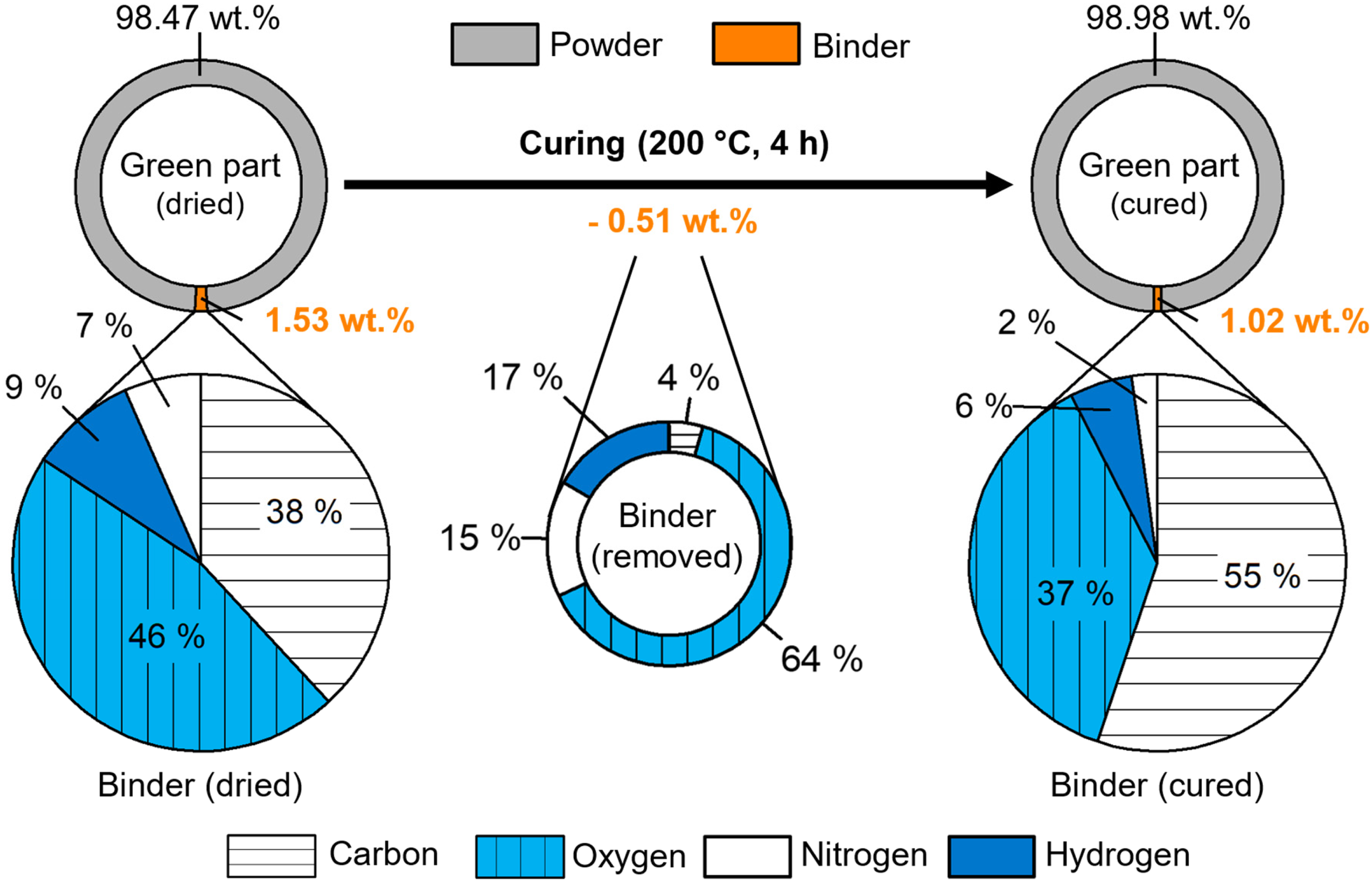
Change in the weight composition of the binder calculated by the chemistry of the green part and 17-4 PH powder.
The curing process reduced the binder content inside the green part by 0.51 wt.%, which is a decrease of 34% relative to the dried binder content. This was slightly less than the measured weight loss of the isolated binder by 38% after curing. The mass ratio of the removed elements during curing can provide insights into the evaporated binder constituents. The majority of the removed oxygen and hydrogen can be attributed to adsorbed water inside the porous powder compact. For water, the mass ratio of hydrogen to oxygen is 1:8 (2:16). The mass removed during curing comprised 17% of hydrogen and 63% of oxygen, which corresponded to a mass ratio of ∼1:3.7. Hence, the evaporated molecules could not be solely water. The removal of nitrogen and carbon indicated that low boiling point solvents or additives were evaporated, which can also contain hydrogen and oxygen. Binders for BJT usually consist of water, solvents and complex organic compounds. 3
The mass composition of the binder changed considerably from the dried to the cured state. The carbon mass fraction increased from 38% to 55%, while the oxygen mass fraction decreased from 46% to 37%. Nitrogen accounted for 6% and hydrogen for 2% of the binder mass after curing. As most humidity and solvents were removed after curing, the remaining carbon, oxygen, nitrogen and hydrogen belonged mainly to the crosslinked organic binder. The presence of these elements after curing can be understood by considering typical binder formulations in BJT.
Common polymers used in BJT contain carbon–carbon, carbon–oxygen and carbon–hydrogen bonds. 25 The carbon and hydrogen are part of carbon–hydrogen (C–H) and carbon––carbon (C–C) bonds, which are relatively unreactive. The oxygen in the binder is part of functional groups. Typical functional groups in binders include carbonyl groups (C = O), carboxyl groups (–C(=O)–OH), formyl groups (R–CH = O), ether groups (R–O–R′) and hydroxyl groups (–OH). Nitrogen is also part of functional amine groups. The mentioned functional groups are encountered in typical binders used for BJT such as polyvinylpyrrolidone, polyvinyl alcohol and polyacrylic acid. 26
Effect of decreasing oxygen content in the debinding atmosphere
Binder removal
A simple indicator of the debinding efficiency is the weight loss measured after the debinding process. Figure 6 depicts the measured weight loss after debinding at 300°C for 2 h and the percentage of binder removed relative to the previously determined green part (cured) binder content of 1.02 wt.%. The standard deviation was based on a minimum of six samples.
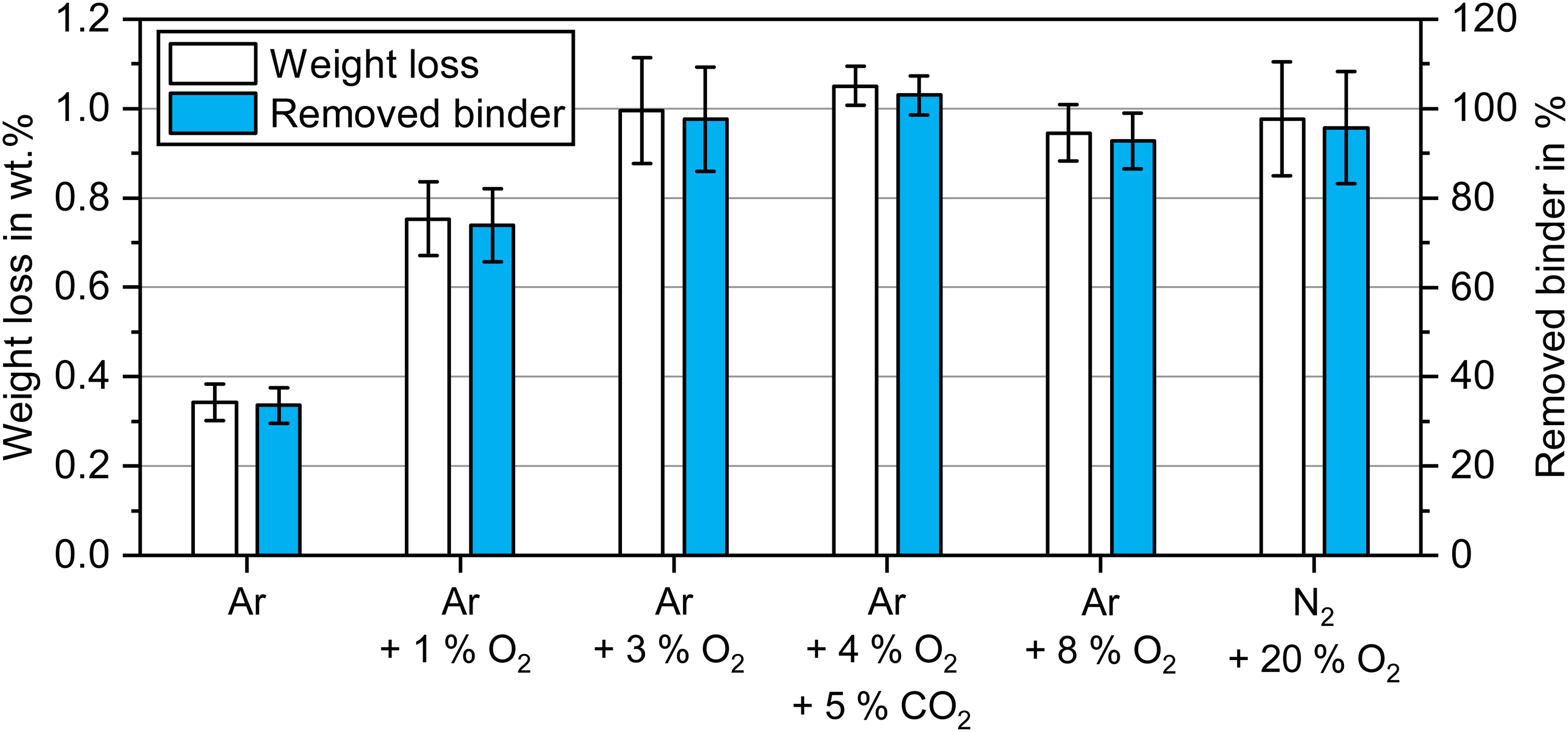
Total weight loss of green parts and corresponding percentage of removed binder after debinding at 300°C for 2 h in atmospheres with different oxygen content.
The lowest weight loss was seen after debinding in an inert Ar atmosphere at 0.34 wt.%, which corresponded to ∼34% of the initial binder mass removed. Debinding in Ar + 1 vol.% O2 resulted in a significantly higher weight loss of 0.75 wt.%, which equalled ∼74% of the initial binder mass. The highest binder removal was achieved in atmospheres from 3 vol.% O2 to 20 vol.% O2, which were comparable from 0.95 wt.% to 1.05 wt.%. Hence, 93%–100% of the initial binder was already removed at this stage. Since the binder was nearly completely removed, the powder packing density can be estimated to be ∼4.71 g/cm³ matching the previous calculations.
While almost complete binder removal in oxygen-rich atmospheres minimises the potential for carbon residuals after the sintering process, the brittleness of the brown parts increased. This makes the transport of brown parts challenging, especially risking the breaking of thin features. Powder loss was observed for debinding in 3 vol.% O2 to 20 vol.% O2 when brown parts were handled, which is demonstrated in Figure 7. The powder loss explains that in a few instances in Figure 6, the binder removal exceeded 100% when the brown parts lost small amounts of powder during handling. Debinding in Ar showed no noticeable change in visual appearance compared to the green part, while a color change for debinding in oxygen-containing atmospheres indicated powder oxidation. Powder loss for samples debinded in Ar and Ar + 1 vol.% O2 was not noticeable, since more residual binder was present in the brown parts.

Brown parts after debinding in different atmospheres with powder loss for oxygen-rich atmospheres.
Chemistry of the brown parts and 17-4 PH powder
While the weight loss after debinding indicates how much of the binder is removed, it does not reveal how the chemistry of the brown part or 17-4 PH powder was affected during debinding. Contamination of the stainless steel is still a possibility even though most of the binder seemed to be removed. The carbon and hydrogen contents of the brown parts debinded at 300°C in different atmospheres are compared in Figure 8.
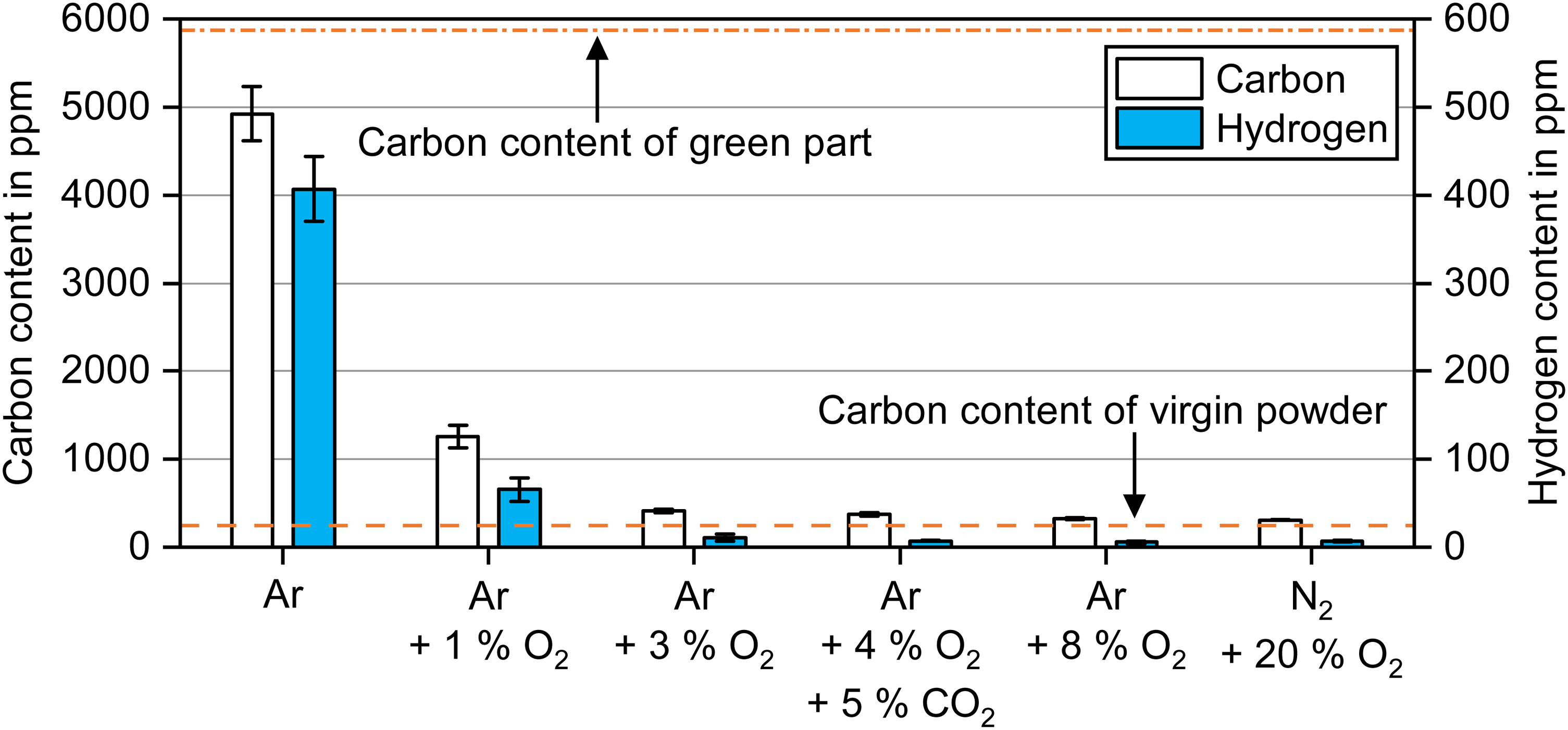
Carbon and hydrogen content of brown parts debinded in atmospheres with different oxygen content at 300°C for 2 h.
The carbon content after debinding in Ar dropped by ∼16% compared to the green state. For the addition of 1 vol.% O2, the carbon content decreased by ∼79% compared to the green state. For oxygen contents of 3 vol.% O2 and higher during debinding, the carbon content decreased by ∼93% to ∼94% in the brown part. A slightly decreasing trend in carbon content was found with a further increase in oxygen content in the debinding atmosphere from 3 vol.% O2 to 20 vol.% O2. Since the carbon contents stayed still above the virgin powder content of 245 ppm, some binder was still present in the brown part.
The highest residual hydrogen content was measured after debinding in Ar, whereas considerably lower hydrogen content was found after debinding in 1 vol.% O2. For debinding in 3 vol.% O2 up to 20 vol.% O2, the hydrogen content dropped below 10 ppm as in the 17-4 PH virgin powder. In general, a higher carbon removal was linked to a higher hydrogen removal.
It is noteworthy that the carbon contents of the brown samples after debinding at 300°C were already below the maximum value of 700 ppm (0.07 wt.%) specified for 17-4 PH by MPIF 35/ASTM B883-24 standards for metal injection moulding.27,28 These standards are frequently used as a benchmark due to the similarity of the green parts in terms of density and binder content before thermal debinding.
The oxygen and nitrogen contents of the brown parts debinded in different atmospheres are displayed in Figure 9 with the corresponding 17-4 PH powder, which was subjected separately to the same debinding conditions. The data demonstrated that the oxygen and nitrogen contents of the brown samples were highly dependent on the 17-4 PH powder chemistry as the difference was comparatively small for most debinding atmospheres.
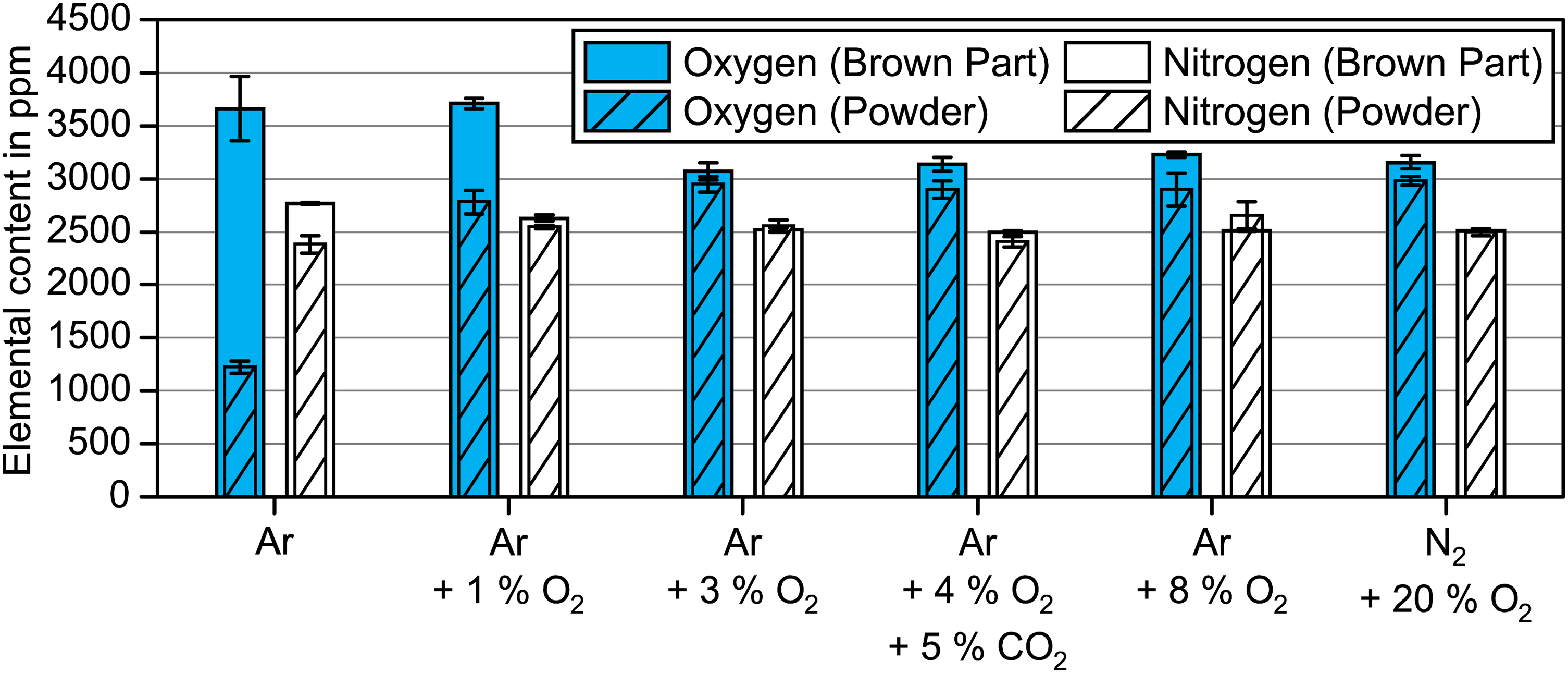
Oxygen and nitrogen contents of the 17-4 PH powder and brown parts subjected to debinding atmospheres with different oxygen contents at 300°C for 2 h.
The oxygen content of the 17-4 PH powder increased only slightly by 60 ppm compared to the virgin powder when subjected to 300°C for 2 h in an inert Ar environment. The oxygen pickup was highest for 3 vol.% O2 up to 20 vol.% O2 with 1734 to 1820 ppm. In contrast, 1 vol.% O2 resulted in a slightly lower oxygen pickup of 1617 ppm. The nitrogen contents of the 17-4 PH powders stayed unchanged.
The brown parts after debinding in Ar revealed a considerably higher oxygen level by 2440 ppm and a noticeably higher nitrogen content by 390 ppm in comparison to the corresponding 17-4 PH powder. Since nitrogen pickup was unlikely in an inert Ar environment, the added nitrogen must originate from the residual binder. Similarly, the high oxygen amount in the brown part belonged to the residual binder. The oxidation of the 17-4 PH powder in inert Ar cannot, however, be ruled out due to oxygen availability in the binder. The oxygen content in the brown parts decreased with increasing oxygen content in the processing atmosphere, which correlated with lower binder residue.
For debinding in Ar + 1 vol.% O2, excess oxygen of 33% was measured compared to the corresponding powder level, but the nitrogen content was only slightly higher than in the 17-4 PH powder. The lower nitrogen levels after debinding in Ar + 1 vol.% O2 implied a lower binder amount compared to debinding in Ar. Yet, debinding Ar + 1 vol.% O2 and debinding in Ar showed comparable oxygen levels in the brown sample. Since more oxygen must have originated from the binder after debinding in Ar due to the significantly higher amount of residual binder, a lower oxidation of the 17-4 PH can be confirmed after debinding in Ar.
When debinding was conducted in ≥3 vol.% O2, the oxygen content in the brown part was only slightly higher than in the corresponding powder. The nitrogen contents of brown parts and powder were comparable. The slightly increased oxygen levels suggest minor amounts of residual binder in the brown samples as indicated by the corresponding carbon contents shown in Figure 8.
The residual binder content can be estimated analogously to the calculation described in the ‘Impact of binder on green part density and bulk chemistry’ section and Table 4. The calculated binder contents in the brown parts depending on the debinding atmosphere at 300°C for 2 h are listed in Table 5. These calculations assume that the powder subjected to 300°C in the various atmospheres possessed the same chemistry as the powder in the brown part, which is a simplification. The oxidation of the powder might be affected by the binder covering the powder surface. The binder can protect the surface or provide oxidising species.
Calculation of residual binder contents in brown parts compared to the 17-4 PH powder after debinding in atmospheres with different oxygen contents.
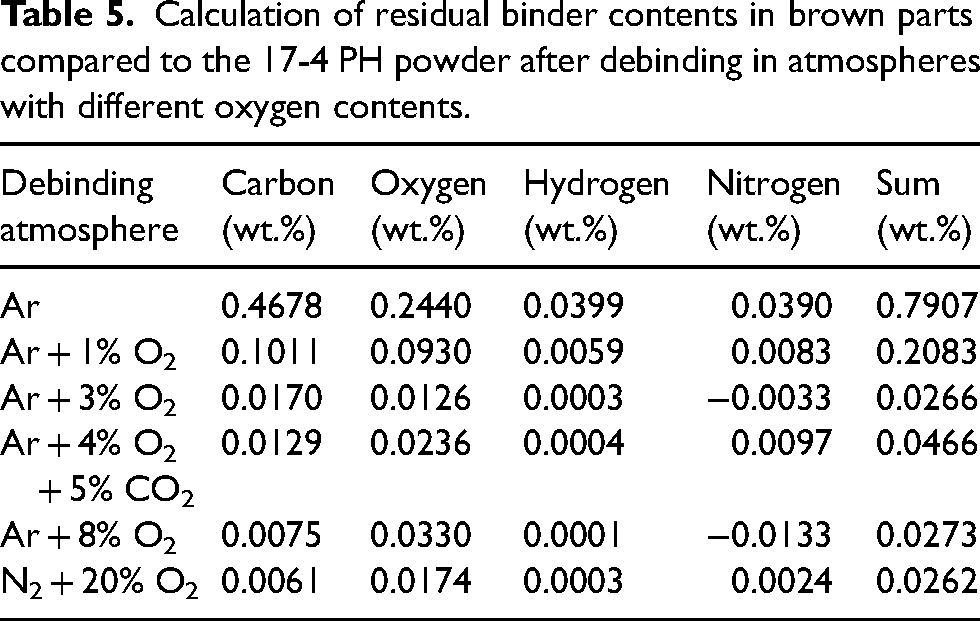
It is evident that the calculated residual binder contents in the brown parts matched the actual measured weight losses from green part to brown part in Figure 6 quite well except for debinding in Ar. The residual binder content of 0.79 wt.% after debinding in Ar would equal a weight loss of ∼0.23 wt.% after debinding. In contrast, the measured weight loss was higher at 0.34 wt.%. This difference could be explained by the oxygen content. As negligible oxidation of the 17-4 PH powder was measured after debinding in Ar, the 17-4 PH powder in the brown part was likely to be oxidised by binder constituents. Therefore, less oxygen would be attributed to the binder and more to the powder. Consequently, a lower binder content would be calculated overall. In this study, the influence of part geometry is assumed to be negligible due to the overall small green part sizes. This assumption is supported by the observation of similar weight losses in cuboid and cylindrical samples.
Lecis et al. compared debinding in inert Ar and air for BJT of 316L stainless steel with an aqueous-based binder. 7 The carbon content after debinding at 470°C in Ar was at 0.13 wt.% considerably higher than after debinding in air at 0.021 wt.%. 7 Even at a higher debinding temperature of 600°C, the carbon content for debinding in Ar was significantly higher at 0.15 wt.% compared to 0.020 wt.% for debinding in air. The oxygen values were not measured, but significant powder oxidation is expected in air, especially at 600°C. Even though the stainless steel powder, binder formulation and printing process were different, a similar impact of oxygen content during the debinding process was identified. Oxygen aids the decomposition of organic molecules and removal of carbon, which applies to most binder formulations.
It was shown that by tailoring the oxygen content in the debinding atmosphere, the debinding process can be adjusted. The debinding efficiency and powder oxidation for each atmosphere might, however, be further impacted by the green part size, binder formulation, binder saturation settings, particle size distribution and powder packing density.
Elemental change of the binder after debinding
To get a better insight into how the binder was removed during debinding depending on the oxygen content in the debinding atmosphere, the weight composition of the binder inside the brown parts was calculated as in the ‘Elemental change of the binder after curing’ section. The weight composition of the removed binder and residual binder in the brown part is highlighted in Figure 10 for debinding in Ar, Ar + 1 vol.% O2 and N2 + 20 vol.% O2. These debinding atmospheres were chosen for a representative comparison since the measured weight losses and chemistry of the brown parts were comparable for 3 vol.% O2 up to 20 vol.% O2. Note that while the weight compositions of the binder are compared, the mass of the removed and residual binder was different.
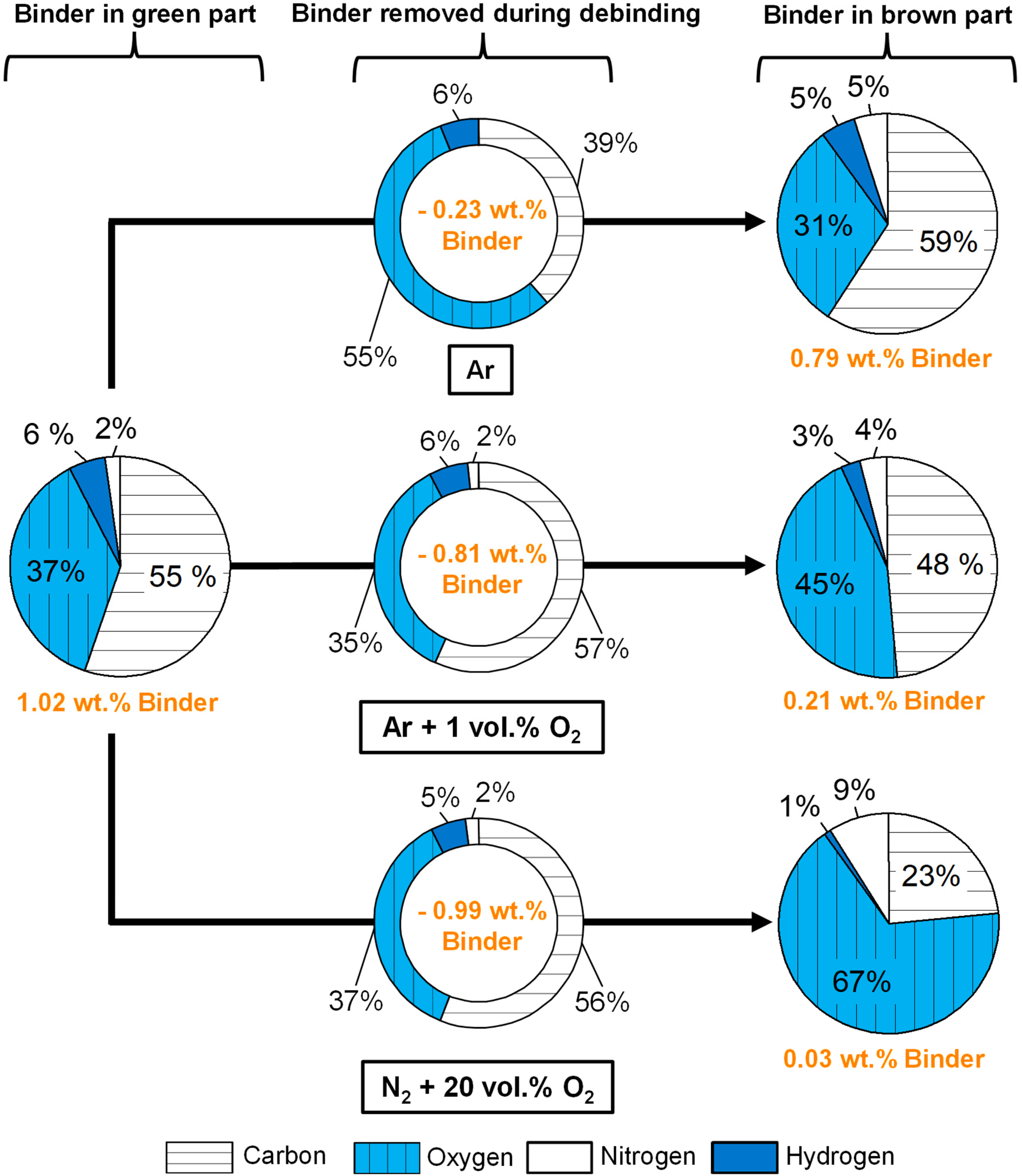
Elemental composition of the binder removed during debinding and in the brown part in wt.% for the debinding atmospheres of Ar, Ar + 1 vol.% O2 and N2 + 20 vol.% O2.
The residual binder after debinding in Ar revealed a slightly changed elemental distribution compared to the cured binder in a green part. A slightly higher fraction of nitrogen was seen in the residual binder compared to the cured binder, which is in accordance with the measurement in Figure 3. In contrast, debinding in 1 vol.% O2 resulted in more oxygen and less hydrogen in the residual binder composition of the brown parts. The carbon weight fraction of the binder in the brown part was reduced by 10% compared to debinding in Ar. Debinding in N2 + 20 vol.% O2 resulted in the highest oxygen fraction of 67%, whereas carbon decreased to 23%.
The combination of increasing oxygen, decreasing carbon and decreasing hydrogen content in the residual binder composition indicated that the binder was stronger pre-oxidised with higher oxygen content in the debinding atmosphere. More functional groups containing oxygen such as carbonyl groups, carboxyl groups, formyl groups, ether groups and hydroxyl groups might be present. A potential pre-oxidation of the binder results in lower molecular weight species that are easier to evaporate and remove, which likely caused higher weight loss with increasing oxygen content in the debinding atmosphere. It is important to note that increased powder oxidation is expected when debinding in atmospheres with higher oxygen content, which contributes to the estimated increase in oxygen content in the binder residues.
During debinding, interactions between the powder, the binder, the binder decomposition products and the respective process atmosphere can take place. 5 Complexity emerges from the different binders, which can depolymerise to release monomers, undergo random chain scissions or release side groups before degradation. 5 The mechanisms of debinding and decomposition products in BJT should in principle be comparable to delubrication of PM components due to the low organic binder content. The release of vapour species during the delubrication of PM steels has been thoroughly monitored in various studies.16,17,19 Hryha et al. 19 detected the formation of CO2 between 300°C and 330°C. This was attributed mostly to the decomposition of carbonates and hydrocarbonates present on the powder surface and also to lubricant decomposition. 19 Quadbeck et al. 29 measured the decomposition of ethylenbisstearamide for PM steel components at temperatures between 230°C and 480°C and detected mainly CH groups, CO, CO2, H2O and CH4 as decomposition products. Similar vapour products are expected for the debinding of BJT green parts.
When debinding was conducted in Ar at 300°C, the weight ratio of the removed carbon to oxygen was 1:1.4. The mass ratio of carbon to oxygen is close at 1:1.3 (12:16). Since no reactive species are present in the debinding atmosphere, the breakdown is mainly coming from the polymer itself. 5 In an inert atmosphere such as Ar, the decomposition of the binder is limited to vapour species formed by the binder. 5 Binders usually do not contain sufficient oxygen molecules to decompose into CO and CO2. This tends to lead to the formation of graphite or organic residues that can negatively impact sintered properties. In this case, the oxygen in the binder was not sufficient for the decomposition on its own at 300°C. Gilmer et al. 30 designed a binder formulation with excess oxygen groups enabling a cleaner binder burn-off and reduced carbon contamination.
For debinding in 1 vol.% O2, the removed debinding products contained more carbon at 57% and less oxygen at 35% corresponding to a weight ratio of nearly 1.6:1. Interestingly, the composition of the binder removed during debinding in 20 vol.% O2 was similar despite the significant higher binder weight lost compared to 1 vol.% O2. Furthermore, the weight distribution for the other oxygen-containing atmospheres was nearly the same. Therefore, the rate of thermal debinding was lower in 1 vol.% O2. The rate of thermal debinding is influenced by the exposed surface area and the diffusion of oxygen in the powder compact.31,32 The reaction temperatures might also be shifted by a varying oxygen content.
Exothermic oxidation reactions of the binder are expected in oxygen-containing atmospheres. The supply of oxygen increases the breakdown of hydrocarbon chains. Binder burnout in oxygen-containing atmospheres such as air results in the formation of CO, CO2 and water vapour (H2O). 5 Compared to debinding in Ar, less oxygen from the binder was removed. This indicated that the binder reacted with the oxygen from the atmosphere in contrast to debinding in Ar, where the removed oxygen fraction was higher as only the oxygen stemming from the binder could react.
Even though binder chemistry can vary for material and printer hardware, the introduced insight can be transferred since most binders rely on similar chemical bonds and decomposition mechanisms. The impact of the debinding atmosphere with varying oxygen contents on the density, microstructure and chemistry after sintering is discussed in part II of the study. 24
Conclusions
The influence of varying oxygen content in the debinding atmosphere was examined for 17-4 PH green parts manufactured via BJT. The debinding was conducted at 300°C for 2 h. Debinding atmosphere compositions with increasing oxygen content were studied including pure Ar (<15 ppm O2), Ar + 1 vol.% O2, Ar + 3 vol.% O2, Ar + 4 vol.% O2 + 5 vol.% CO2, Ar + 8 vol.% O2 and N2 + 20 vol.% O2. The focus of the study was on the binder content in the green parts, the binder residuals in the brown parts and powder oxidation after debinding.
The curing of green parts at 200°C for 2 h resulted in a decrease of hydrogen, oxygen and nitrogen content in the binder, which was attributed to the evaporation of water, low boiling point solvents and additives. After curing, the binder in the green part added 0.56 wt.% of carbon, 0.38 wt.% of oxygen, 0.05 wt.% of hydrogen and 0.02 wt.% of nitrogen compared to the 17-4 PH powder. This amounted to 1.02 wt.% of binder in total after curing adding ∼0.048 g/cm³ to the average green density of 4.75 g/cm³. The carbon and hydrogen contents in a green sample acted as good indicators for binder content due to their relatively low amounts in the 17-4 PH powder, while oxygen and nitrogen were convoluted with considerable amounts already present in the 17-4 PH powder.
Debinding in Ar was inefficient by removing only ∼34% of the initial binder mass leaving a high content of carbon- and oxygen-rich binder residuals in the brown part. The weight composition of the binder in the green part changed slightly in the brown part containing 59% carbon. High carbon residuals are unfavourable for sintering and material properties of 17-4 PH stainless steel. Debinding in Ar + 1 vol.% O2 achieved considerable binder removal of ∼74%, whereas debinding in 3 vol.% O2 to 20 vol.% O2 showed almost complete binder removal. The weight ratio of the elements removed during debinding was comparable for all the oxygen-containing atmospheres despite higher residual binder content for 1 vol.% O2. The almost complete binder removal for oxygen-rich atmospheres (3 vol% O2 to 20 vol.% O2) resulted, however, in increased brittleness of the brown parts, causing minor powder loss during handling. This is problematic in practice, especially for intricate components.
Subjecting the 17-4 PH virgin powder to the debinding conditions at 300°C for 2 h showed that inert Ar allowed to minimise powder oxidation, but partial oxidation in the brown part was still expected due to oxygen originating from the binder residuals. For 3 vol.% O2 to 20 vol.% O2, the oxygen pick-up by the powder was between 1734 ppm and 1820 ppm. In 1 vol.% O2, a slightly lower oxidation of 1617 ppm by the powder was measured. The findings demonstrated that Ar + 1 vol.% O2 provided the most favourable balance of high binder removal and low powder oxidation combined with sufficient brown part strength and stability. The study highlighted the potential of tailoring the oxygen content in the atmosphere to improve the debinding process for BJT components.
Footnotes
Acknowledgments
This work was conducted in the framework of the Centre for Additive Manufacturing – Metal (CAM2), supported by the Swedish Governmental Agency of Innovation Systems (Vinnova). Linde GmbH acknowledges the funding by the Federal Ministry of Research and Education (BMBF, Germany) for the project SINEWAVE (FKZ 03HY123A) within the Hydrogen Flagship Project H2Giga. The authors acknowledge the contribution of Mumtahena Shahabuddin Jim for carrying out parts of the experimental work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swedish Governmental Agency of Innovation Systems (Vinnova) with grant number 2016-05175 and the Federal Ministry of Research and Education (BMBF) (under SINEWAVE (FKZ 03HY123A).
