Abstract
This paper reviews the original reasons of the organic farming movement for excluding mineral (inorganic) fertilizers. In this paper, their theories and decision criteria for excluding use of inorganic fertilizers in crop production were revisited. Original reasons for banning inorganic fertilizers were subjected to scientific scrutiny, which was not possible when they were formulated 50–100 years ago due to limited knowledge of the soil-crop system. The original reasons were as follows: Rudolf Steiner, the founder of biodynamic farming, played down the physical role of plant nutrients and pointed out “flow of forces” as being most important for soils and crops. Eve Balfour and Albert Howard, founders of the Soil Association in England, claimed that inorganic fertilizer increases the breakdown of humus in soil, leading to a decline in soil fertility. Hans-Peter Rusch, the founder of biological organic farming, considered inorganic fertilizers to be imbalanced products not matching crop composition and not in synchrony with crop demand. When testing these historical statements as scientific hypotheses, older and modern scientific literature was used for validation. Steiner’s belief about the “flow of forces” has not be verified using current methodologies. The claim by Balfour and Howard that inorganic fertilizers accelerate soil organic matter decomposition is not substantiated by data from long-term field experiments on carbon and nitrogen cycling in soil-plant systems. The statement by Rusch that inorganic fertilizers supply crops inappropriately is difficult to uphold, as the composition, time, and rate of application and the placement of fertilizer in soil or on foliage can be fully adapted to crop requirements. In light of accumulated scientific evidence, the original arguments lack validity. The decision to ban inorganic fertilizers in organic farming is inconsistent with our current scientific understanding. Scientific stringency requires principles found to be erroneous to be abandoned.
Introduction
Exclusion of inorganic fertilizers is often perceived as a quality-improving principle that makes organic farming superior to conventional agriculture. The ban on the use of inorganic fertilizers in organic farming was introduced almost a century ago, when scientific knowledge about interactions between fertilizers and the soil-crop system was limited. This paper analyzes the original arguments in the different schools of organic agriculture on prohibiting the use of inorganic fertilizers and re-evaluates these reasons based on accumulated science and evidence. As inorganic fertilizers are the most powerful management input affecting the performance and quality of crops, it is highly important to gain a correct scientific understanding and go beyond popular biases against the use of inorganic fertilizers.
The analysis consisted of the following steps: First, original statements by the initiators of biodynamic farming (Steiner, 1924), the Soil Association (Balfour, 1943; Howard, 1940), and biological organic farming (Rusch, 1978) were identified and cited. The reasons cited for not using mineral fertilizers were then treated as scientific hypotheses, following the principles of the philosophy of science (Popper, 1959), and tested for their validity. The principal question addressed was whether there is evidence supporting the decision to ban the use of synthetic fertilizers in agriculture, i.e. whether the doctrine is corroborated by science.
At the time when exclusion of inorganic fertilizers was proposed, it was difficult to corroborate or reject the specific reasons put forward. There was a lack of knowledge about soils and about nutrient turnover in the soil-plant system. Furthermore, scientific methodologies were limited or had not been developed, e.g. isotope techniques were not available, measurements of nutrient leaching and gaseous emissions were scarce, and an understanding of soil biological processes was only in its infancy. Thus it was difficult to prove the validity of the original statements when they were first made.
Founders of organic farming were concerned about maltreatment of nature and deteriorating food quality
Two initiators of organic farming, the Austrian philosopher Dr. R. Steiner (1861–1925), who founded biodynamic farming, and the British agronomist and farmer Lady E. Balfour (1899–1990), who co-founded the Soil Association, were convinced that food quality had declined through the use of inorganic fertilizers. Their explicit response was to exclude inorganic fertilizers, in order to produce food of high quality. Steiner believed that food products would degenerate to such an extent that they would not be suitable as food for humans within a century (Steiner, 1924: 12). Balfour believed that if the fertility of soils is built up with an adequate supply of humus, crops do not suffer from diseases and animals fed on these crops develop high resistance to diseases. She also believed that humans fed with such plants and animals could achieve a high standard of health and the power to resist diseases and infections, from whatever cause (Balfour, 1943: 91). She was convinced that there must be something lacking in the foods of the day that was not lacking in the foods of the nation’s more robust forefathers (Balfour, 1943: 35). Her view was shared by the British agronomist Sir Albert Howard (1873–1947), her co-founder in the Soil Association, who stated that perfectly healthy soils are the basis for health on earth and that undernourishment of the soil is at the root of all problems (Howard, 1947: 12).
The German medical doctor H.-P. Rusch (1906–1977), the initiator of biological organic farming, believed that nature must be treated as a biological wholeness (Rusch, 1978: 15). He taught that each unnatural intervention in biological element cycling will cause damage (Rusch, 1978: 235); that each nitrogen fertilization prevents biological cycling and decreases biological quality (Rusch, 1978: 236); and that the quality of food is dependent on the biological functioning of soils (Rusch, 1978: 26), through proper humus management (Rusch, 1978: 234).
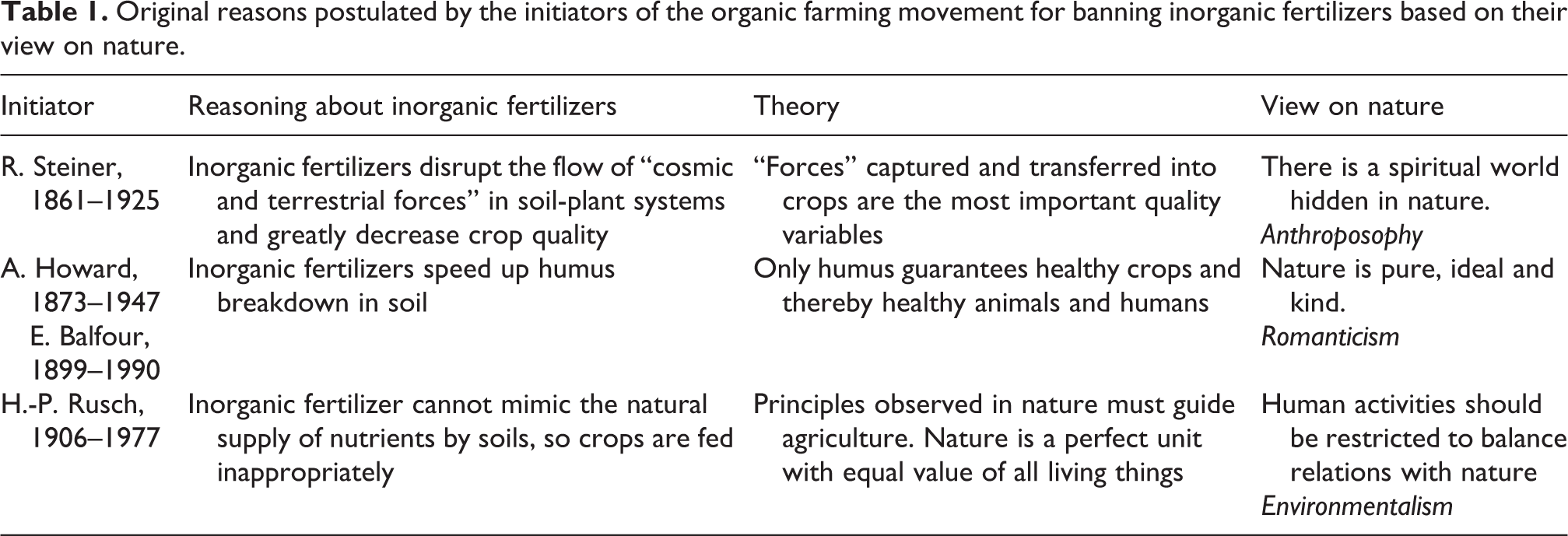
The different views on nature held by the initiators of the organic farming movement are summarized in Table 1. Note that the initiators did not argue against the use of inorganic fertilizers due to concerns about resource shortage, energy demand for nitrogen fertilizer production, or environmental impacts. It is only more recently that followers of the organic movement have declared that inorganic fertilizers are the root cause of eutrophication (e.g., Granstedt, 2000; Granstedt et al., 2008; Koepf, 1973) and that exclusion of inorganic fertilizers decreases greenhouse gas emissions (e.g., Muller et al., 2017; Smith et al., 2019).
Original reasons postulated by the initiators of the organic farming movement for banning inorganic fertilizers based on their view on nature.
Testing Steiner’s statements about inorganic fertilizers
Steiner’s context of thinking was non-scientific. He described “auras” and “forces” around organisms, phenomena that are not known to science. His perspective was spiritual teaching, mysticism, and esoteric wisdom. He applied his spiritual perception to transform society, e.g. arts and architecture, medicine, religion, pedagogics, and also agriculture and bee-keeping. He created a wide-ranging spiritual system called “anthroposophy.”
Biodynamic agriculture builds upon a series of lectures by Steiner during a 1-week course on agriculture in Koberwitz (now Wroclaw), Poland, in 1924. There, he taught a group of followers on consideration of spiritual matters in agriculture, in a lecture entitled “Geisteswissenschaftliche Grundlagen zum Gedeihen der Landwirtschaft” (Spiritual foundations for the renewal of agriculture), with instructions on how to produce organic food supplying “forces” to mankind (Steiner, 1924). This was the birth of the first distinct form of organic agriculture. Key statements from his book are as follows: “Es weiss zum Beispiel kein Mensch heute, dass alle die mineralischen Dungarten gerade diejeningen sind, die zu dieser Degenerierung, von der ich gesprochen habe, zu diesem Schlechterwereden der landwirtschaftlichen Produkte das Wesentliche beitragen” (Nobody knows today that all sorts of mineral fertilizers are leading to the degeneration, of which I have talked, to the significant quality decline in agricultural products). (Steiner, 1924: 20) “Man hört heute sehr oft die Phrase: der Dünger enhalte die Futterstoffe für die Pflanzen. Da muss sie (Wissenschaft) sich korrigieren, weil eben sie eben von einer ganz falschen Anschauung ausgeht in Bezug auf die Ernährung irgeneines Wesens” (You often hear the phrase: Fertilizer contains the nutrients for plants. In this case, science must correct itself, because this is a completely wrong view about nutrition of any organism). (Steiner, 1924: 87) “Man muss wissen, dass das Düngen zu einer Verlebendigung der Erde bestehen muss…” (You need to know that fertilization means making the soil more alive). (Steiner, 1924: 91) “Für die Pflanze sind viel wichtiger lebendige Kräfte als bloss die substanziellen Kräfte, als bloss die Substanzen” (For the plant, living forces are much more important than only substances). (Steiner, 1924: 124) “Daher werden Pflanzen, welche unter dem Einfluss irgendwelchen mineralischen Düngern stehen, ein solches Wachstum zeigen, das verrät, wie wie es nur unterstützt wird von angeregter Wässrigkeit, nicht von lebendiger Erdigkeit” (Therefore plants treated with mineral fertilizers will show growth affected by waterishness, not living soil). (Steiner, 1924: 94) “Man muss die Erde direkt beleben, und das kann man nicht, wenn man mineralisierend vorgeht…” (One needs to revive the soil, and this cannot be done using minerals…). (Steiner, 1924: 122) “Denn jeder mineralsiche Dünger bewirkt, dass nach einiger Zeit dasjeninge, was auf den Feldern erzeugt wird, die mit ihm gedüngt werden, an Nährwert verliert. Das ist ein allgemeines Gesetz” (Hence, all mineral fertilizers affect, after some time, what is grown on fields, and those (crops) lose nutritional value. This is a general law). (Steiner, 1924: 176)
The auras and forces mentioned by Steiner are not known to science, but nevertheless there have been several attempts to visualize them. Two attempts in particular are described below, a photographic technique (Kirlian photography) to visualize the phenomenon of an aura (Kirlian, 1949) and a crystallization technique to visualize formative forces in a plant (Engqvist, 1970).
The Russian researcher Semyon Kirlian discovered in 1939 that when a plant part, e.g. a leaf, was placed on a photographic plate with a metal plate underneath and connected to an electricity supply (high voltage, high frequency), an image was produced on the photographic plate. The image showed color fields around the electrified object that resembled an aura. Such plant images were described as revealing “the secret life of plants.” However, when the method of using electricity to reveal auras around objects was subjected to further research, it was found that when high voltage enters an object electric discharge occurs, leading to ionization of water and gaseous molecules around surfaces of the object. In fact, it was found that variations in color, length, density, and curvature of the field around plants could be explained by variations in the water content on the surface and in the tissue, causing different discharge conditions (Pehek et al., 1976). Forms and colors of the electrical discharge zone around a plant are thus not an intrinsic plant quality, but a random effect of the water content, and do not represent etheric auras.
The method devised by Engqvist (1970) to illustrate “forces” in plants was based on crystallization. It involved mixing freshly pressed solution (juice) from a plant with copper chloride, placing drops of the mixture on a glass plate, and evaporating the liquid off under controlled temperature and moisture conditions. The crystallization pattern formed during evaporation was interpreted as showing the plant’s organized and formative “forces.” The more regular the pattern the better, with irregularity taken to indicate less organized plant “forces.” From a scientific perspective, crystallization of plant solution with copper chloride during evaporation is primarily affected by the concentration of compounds in the plant juice and their reactions with copper chloride. Even if strictly controlled conditions are applied, i.e. constant temperature, moisture and rate of addition, uniform surface properties of plates, plants of similar age, etc. crystallization patterns are governed by plant solution concentrations and composition. Interpretation of crystallization patterns as being intrinsic formative “forces” of the plant, is a pseudoscientific approach.
Despite impressive scientific and technical advances, it has not been possible to identify living “forces” or “auras” surrounding plants or animals. Steiner’s descriptions of such phenomena were based on his spiritual experience (Kirchmann, 1994). Note that Steiner never addressed known crop quality characteristics such as protein, carbohydrate, or fat content, or mineral or vitamin concentrations.
The statement by Steiner that our view about the function of minerals as nutrients for crops is completely wrong is not substantiated by science. Soil and plant scientists have identified 14 elements in the periodic table as being essential plant nutrients (Marschner, 2012). These nutrients are taken up as inorganic ions by roots and are involved in photosynthesis, amino acid and carbohydrate synthesis, formation of cell walls and structural components, and enzyme reactions regulating growth and reproduction of plants (Mengel et al., 2001). In stark contradiction of the statement by Steiner, supplying inorganic nutrients to crops contributes significantly to food production (Smil, 2002; Stewart et al., 2005), and has become a critical tool in improving crop yields and quality (e.g., Cassman, 1999). Inorganic fertilizers allow plant protein content to be regulated through precision agriculture (Raun et al., 2002), increase soil fertility (e.g., Carlgren and Mattsson, 2001), and enable crops to be fortified with essential trace elements, e.g. selenium (Hartikainen, 2005).
There is no evidence to support Steiner’s statement that science must “correct itself” about the nutritional role of minerals for plants. His statement (hypothesis) of “living forces” affecting crops cannot be tested, and is thus not falsifiable. However, when a hypothesis is not falsifiable, this is a sign of pseudoscience (Hines, 2003).
Testing Balfour’s and Howard’s statements about mineral fertilizers
The 1940s brought the next wave of organic pioneers, with Balfour and Howard as prominent figures in the United Kingdom. In her highly influential book “The Living Soil,” Balfour (1943) pointed out the importance of a healthy soil and the nutritional superiority of organically grown food. The central hypothesis in writings by Balfour and Howard was that there is a close relationship between soil fertility, soil humus, and human health.
According to Howard, perfectly healthy soils are the basis for health on earth: “The undernourishment of the soil is at the root of all” and health is a “birthright of life” (Howard, 1947: 12) and “Humus is the most significant of all nature´s reserves” (Howard, 1947: 26). Thus, the aim of Balfour and Howard was to increase and maintain organic matter content in soils, which was regarded as a guarantee of soil health and human health. A decline in soil humus was interpreted as deterioration of soil quality and inorganic fertilizers were considered to lead to humus decreases.
Balfour stated that: “Artificial fertilisers speed up the rate at which soil organic matter is exhausted” (Balfour, 1943: 53). She postulated that inorganic fertilizers accelerate decomposition of organic matter in soil, leading to a decline over time. Her statement that inorganic fertilizers increase the breakdown of soil organic matter and, as a result, reduce stocks of soil organic matter has been a topic of debate since the 1940s.
Understanding of the interactions between inorganic fertilizers and soil has greatly increased since isotopic research was introduced as a tool in soil and crop science. Early nitrogen isotope studies revealed no significant impact of added mineral nitrogen fertilizer on decomposition, as microbiological activity was not stimulated and no additional carbon dioxide was released (Jansson, 1958). Studies on mineral nitrogen fertilizer and soil nitrogen interactions further corroborated that humus breakdown is not accelerated by mineral fertilizer application (Jenkinson et al., 1985).
Nevertheless, the hypothesis by Balfour that inorganic nitrogen fertilizers cause soil organic matter breakdown and reduce soil organic matter stocks is still being put forward, e.g. by Mulvaney et al. (2009). A later study concluded the interpretation by Mulvaney et al. (2009) “is false and not supported by their data or data from numerous studies worldwide” (Powlson et al., 2010). In fact, inorganic fertilizers enhance soil humus formation. With increasing doses of nitrogen fertilizer, more roots and above-ground crop residues are produced, forming the raw material for creation of more soil organic matter (e.g., Kätterer et al., 2012; Poffenbarger et al., 2017; Powlson et al., 2011). Long-term field experiments have revealed that the level of soil organic matter is the result of the production level of an agroecosystem (Johnston et al., 2009, 2017).
Recently, nutrient supply to microorganisms has been identified as influencing soil organic matter formation. It has been reported that a sufficient supply of inorganic nitrogen to microbes when decomposing organic matter consistently increases microbial growth (Spohn et al., 2016) and reduces soil organic matter decomposition (Mahal et al., 2019). Decomposition appears to be linked to the stoichiometry of nutrients available to microbes (Kirkby et al., 2013, 2014). If an optimal stoichiometric nutrient ratio for microbes is provided, for example by inorganic fertilizers, humus formation in soil can be improved by 30% (Kirkby et al., 2016). In summary, in stark contradiction to Balfour’s statement, inorganic fertilizers increase organic matter in soil.
Howard (1940) considered supplying nutrients to plants through soluble inorganic fertilizers to be a “fatal error”: Artificial fertilisers were born out of the abuse of Liebig’s discoveries of the chemical properties of soil. The effects of the physical properties of the soil were by-passed: its physiological life ignored, even denied, the latter a most fatal error. The essential co-partnership between the soil and the life of the creatures, which inhabit it, to which Darwin´s genius had early drawn attention, is wholly forgotten. (Howard, 1947: 71–72)
Two pathways of beneficial plant-microbe interactions that resemble the living bridges referred to by Howard are symbiotic nitrogen fixation in legume roots by Rhizobium bacteria and symbiotic associations between plants and arbuscular mycorrhizal fungi (AMF). However, these biological symbioses can supply legumes with nitrogen and plants with phosphorus, and some micronutrients, so the entire nutrient requirement of crops is not covered. Furthermore, the contribution of AMF varies with soil conditions and systems, and the association can be both parasitic and symbiotic for crops (Ryan and Tibbett, 2008). In the present understanding, crop requirements for essential plant nutrients are met by root uptake of dissolved inorganic ions (Marschner, 2012; Mengel et al., 2001), dissolved chelated metal ions (e.g., Chen et al., 2001; Ullah and Gerzabek, 1991), and dissolved amino acids (e.g., Jones and Darrah, 1994; Näsholm et al., 2000). In summary, the statement by Howard about living bridges by which plants are fed is incorrect.
Testing Rusch’s statements about inorganic fertilizers
Rusch aimed to apply principles that he observed nature in agricultural production, to keep farming in harmony with nature. He defined this as analogical, biological thinking, which he defined in a book about biological organic farming (Rusch, 1978). His focus was on soil microbes and their role in organic matter turnover in soil. He developed a microbial test as a tool to determine soil quality. He stated that disturbing and mixing of soil must be avoided, in order to mimic nature (Rusch, 1978: 80, 215). One major consequence of his approach is that plowing, which involves mixing soil and disturbing the formation of layers in soil, is prohibited. As a consequence, in biological organic farming organic manures may not be incorporated into the topsoil, and may only be used as a surface cover (Rusch, 1978: 158). Moreover, organic manures are considered unsuitable for the root zone, composting is considered not to be in line with nature, and decomposition of organic materials should only take place on the soil surface (Rusch, 1978: 166).
The statements by Rusch (1978) about inorganic fertilizers were as follows: “Artificial fertilization is not a normal, physiological and natural form of plant nutrition” (Rusch, 1978: 17) and “It is completely impossible to mimic the natural supply of minerals between soil and crop and this is the unavoidable error of artificial fertilization” (Rusch, 1978: 73). Rusch postulated that substances added with inorganic fertilizers are not a natural form of plant nutrition and that this type of supply is not in accordance with the physiological needs of crops. These statements question the basic understanding of plant nutrition that has prevailed since the findings by Liebig (1847) to modern times (Marschner, 2012), and require scrutiny.
A limited number of elements in the periodic table (14) have been identified and classified as being essential plant nutrients (Mengel et al., 2001). Common compounds of these elements present in soil and water can be taken up by roots as inorganic ions in dissolved form (Marschner, 2012). Organic molecules larger than single amino acids cannot penetrate root cells. Instead, organic compounds are synthesized by plants from inorganic ions, water, and carbon dioxide. Plant roots do not discriminate between sources from which inorganic ions originate. Identical ions act physiologically alike whether added to soil with urine, slurry, manures, or compost, deposited with rainfall, supplied with irrigation water, released by soil minerals and soil organic matter, or applied with inorganic fertilizers. Furthermore, all nutrients are involved in the same processes in the soil-crop system, whether added with organic or inorganic fertilizers. For example, identical molecules such as an ammonium ion in urine undergoes the same reactions in soil as an ammonium ion added as ammonium fertilizer (Kirchmann and Pettersson, 1995).
Rusch claimed that inorganic fertilizers cannot “mimic the natural supply of minerals between soil and crop,” where a “natural supply” means that the amount and type of plant-available nutrients are in synchrony with crop demand. This claim is not corroborated by science. Soils vary in their composition (Brady and Weil, 1996) and in their native state can be rich or deficient in nutrients and trace elements, depending on geological conditions, i.e. the bedrock from which the soils derive (Kabata-Pendia, 2000). Soils can also contain undesirable elements such as cadmium, arsenic, etc. that can affect crop composition and human health (Selinus et al., 2005). For example, there is a low content of selenium in Scandinavian soils (Eriksson et al., 2010), which can be overcome by fertilization with selenium (Hartikainen, 2005). Similarly, the zinc content is naturally very low in Turkish soils, but the problem can be overcome by adding zinc to fertilizers (Cakmak et al., 1999). Thus it is wrong to believe that soils in their natural state have a perfect nutrient composition for crop growth.
Rusch postulated that there is synchrony between nutrient release in soil and crop demand, and that the natural nutrient release in soil cannot be mimicked by inorganic fertilizer. This is an erroneous belief as such conditions rarely exist. Release of nutrients in soil is mainly controlled by temperature and moisture conditions (Kätterer and Andrén, 2001), whereas demand for nutrients is driven by crop growth (Hunt, 1982). For example, under temperate climate conditions, soils can have low temperature in spring and cannot provide sufficient nutrients when crop demand is high. In late summer, when soils are warm and moist, nutrient supply from soil can be large, but there is little or no demand by crops (Kirchmann and Bergström, 2008).
The underlying assumption by Rusch that inorganic fertilizers have an imbalanced composition and are an inferior nutrient source is also not corroborated by science. In fact, it is organic manures that are often not well adapted to crop demand. Manures produced by grazing cows exemplify this. Grazing cows deposit urine and feces on the ground and return nutrients to the site where they originated. However, nitrogen added with a urine or dung patch amounts to about 1000 kg N ha−1, which is far more than surrounding plants can take up during the grazing period (Haynes and Williams, 1993; Wachendorf et al., 2005). Nitrogen in urine patches consist of around 10% organic compounds (hippuric acid, allantoin, and creatine) and 90% inorganic compounds (urea plus ammonium), causing high ammonia losses of up to 60% within a few hours (Whitehead et al., 1989). The nitrogen to phosphorus ratio, a measure of how well nutrient composition is adapted to crop need, is highly imbalanced in urine, which contains much nitrogen, but no phosphorus (Haynes and Williams, 1993). A nitrogen to phosphorus ratio of 5:1 is required to be balanced with crop demand (Sadras, 2006). Fecal material in dung patches contains mainly organically bound nitrogen (Floate and Torrance, 1970), which must undergo microbial mineralization before becoming plant available. Dung also has an imbalanced nitrogen to phosphorus ratio (1:0.25–0.4) (Haynes and Williams, 1993), not matching crop needs. While the composition of animal manures is controlled by their metabolism, inorganic fertilizers can be formulated to meet specific demands of different crops even over a growing period (through foliar or split application).
In summary, agricultural management can be inspired by nature, but mimicry of processes in natural ecosystems can be misguided. According to Rusch (1978: 90), nature is a perfect wholeness in itself. However, nature’s wisdom cannot be found at the ecosystem level, but in the adaptation of individual plant and animal species (Denison, 2012).
State-of-the-art of inorganic fertilizers
Production of fertilizers is based on the nutritional demand of higher plants (Marschner, 2012). Composition and use of inorganic fertilizers can be adapted to match different crops. The objective is to supply crops with plant nutrients in water-soluble/plant-available form instead of applying untreated minerals with low water solubility. The qualifier “artificial” or “synthetic” fertilizer gives the impression that these materials are unnatural and analogous to synthetic biocides. This is a misunderstanding as inorganic fertilizers provide essential plant nutrients comparable to organic sources. Inorganic fertilizers feed crops first of all and increase the soil nutrient pool. Organic fertilizers supply organic matter to soil (Kirchmann and Witter, 1992; Sommer et al., 2013) and to a lesser degree provide nutrients to crops (Figure 1).
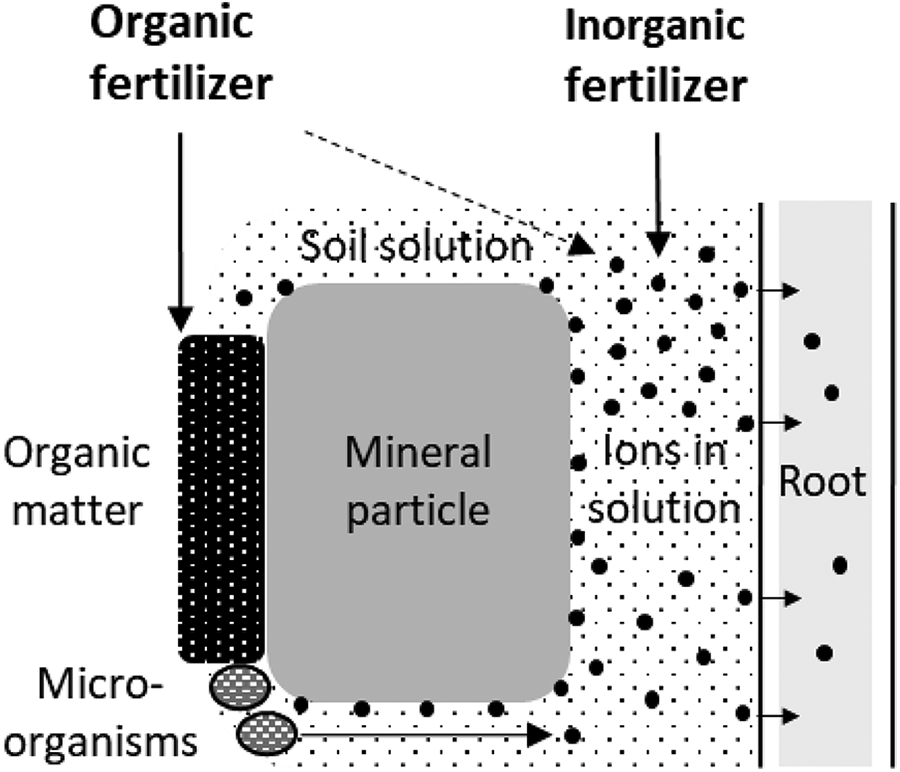
Schematic illustration of the functions of organic and inorganic fertilizers in agricultural soils. Inorganic and organic fertilizers provide water-soluble nutrients in the form of ions dissolved in soil solution and organic fertilizers also add organic matter.
Soils can be natively depleted in nutrients or have an imbalanced nutrient composition. These conditions can be overcome through the use of fertilizers, restoring and building up the depleted nutrient pool with plant-available nutrients. Using untreated minerals instead, e.g. replenishing phosphorus-depleted soil with apatite, would mean wasting a resource, as most apatite forms have no significant impact on crop production. Phosphorus content in soil would increase, but no plant-available phosphorus would be provided and the deficiency problem would remain.
Nutrients are removed from soil through harvested products and through losses by leaching and gaseous emissions. Exclusive recycling of harvested nutrients would not compensate for losses, the balance between outtake and input would become negative and, over time, soils would become depleted lowering soil fertility. Inorganic fertilizers can compensate for losses and/or non-recycled nutrients, while organic fertilizers recycle harvested nutrients only even when redistributed.
Some key points about inorganic fertilizers: Use of inorganic fertilizers means reactivation of accumulated inorganic deposits from earlier geological periods (Jansson, 1971). Fertilizer production means that the chemical structure of mineral deposits is modified to become water-soluble whereby unwanted elements can be removed (Finck, 1982). When nitrogen gas from the atmosphere is chemically bound to hydrogen to form ammonia in the Haber-Bosch process (Smil, 2004), the same inorganic compound is produced as in biological nitrogen fixation. Inorganic N fertilizers can be produced in sustainable manner using renewable energy in the Haber-Bosch process (Ahlgren et al., 2010; Bertilsson and Kirchmann, 2021). Appropriate agricultural use of inorganic N does not cause more greenhouse gas emissions than using organic manures exclusively per unit crop yield (Kirchmann et al., 2016). Leaching losses of inorganic N are lower than when using organic N sources per unit crop yield (Aronsson et al., 2007; Stenberg et al., 2012; Torstensson et al., 2006). The composition of inorganic fertilizers can be adapted to match the need of crops with the right type and amount of nutrients at the right time.
Example of environmental benefit using inorganic fertilizer
Some Swedish literature references from similar organic and conventional rotations were compiled (Table 1) to illustrate how N leaching losses are affected by the exclusive use of inorganic or organic N. Supply of N in the organic rotations was exclusively through legumes (Aronsson et al., 2007; Stenberg et al., 2012; Torstensson et al., 2006). Losses of N leaching were expressed per hectare and unit yield. Yield differences between systems were similar to those reported in Swedish statistics (Kirchmann, 2019). In two studies, leaching of N per hectare was somewhat higher from conventional rotations (38 vs 34 and 13 vs 11 kg N ha−1 yr−1) but lower in the third study (5 vs 19 kg N ha−1 yr−1). However, the amount of N leached per product revealed the true environmental impact. Expressing leaching losses per unit product (Table 2), showed that organic rotations resulted in consistently higher leaching losses ranging from 3.6 to 11.6 kg N Mg−1 organic yield as compared to 0.8 to 6.6 kg N Mg−1 conventional yield. In fact, leaching losses per unit product were 78–69% lower in the conventional rotations. These measures indicate that environmental leaching loads of N are increased by organic farming when the boundary condition to produce similar amount of crops is applied.
Leaching losses of N from comparative crop rotations without livestock using inorganic N (conventional) and exclusively legume N (organic).
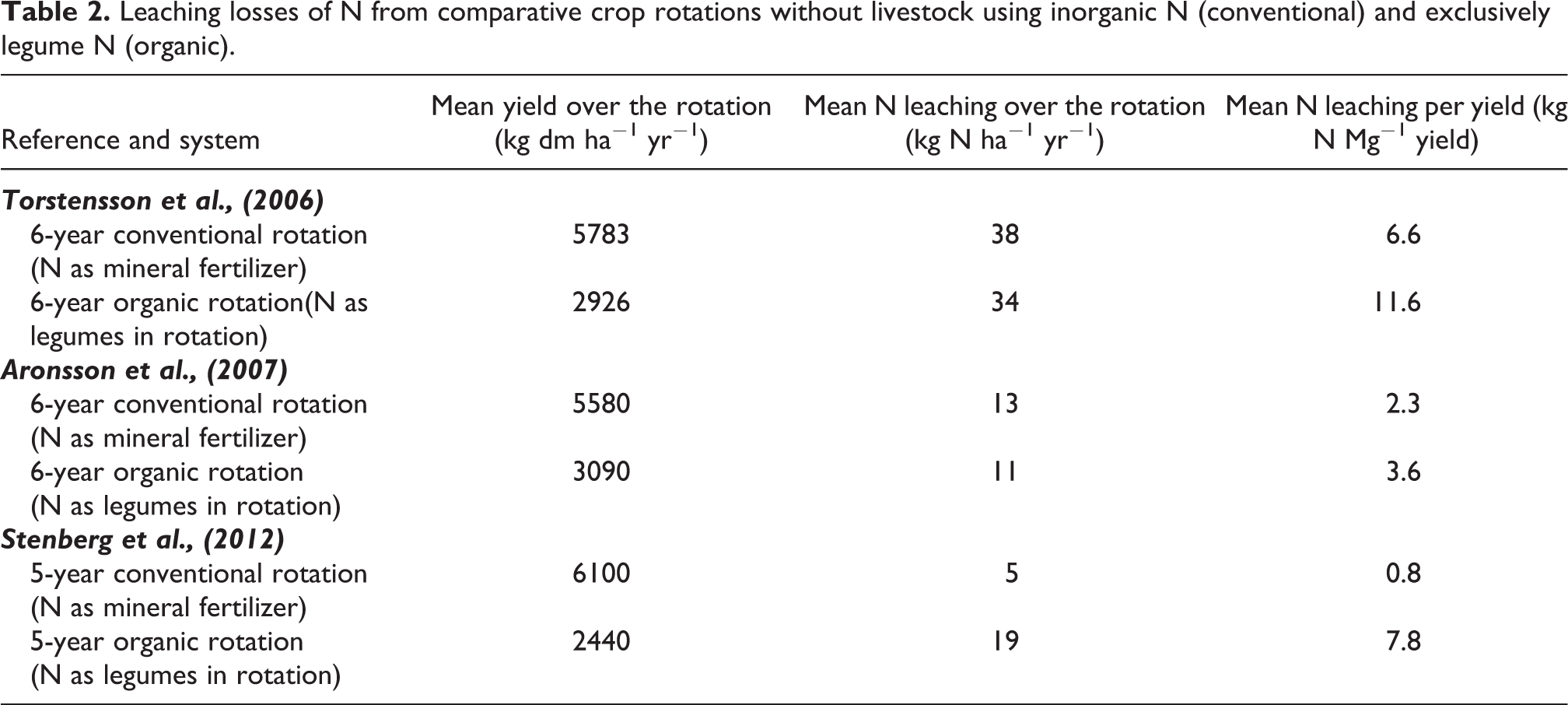
Concluding remarks
Organic farming was founded on the doctrine the inorganic fertilizers should not be used, in the belief that they are harmful for soil, food quality, and human health. Statements about inorganic fertilizers made by the initiators of organic farming reflect their understanding of nature. The reasons they gave for banning inorganic fertilizers could not be verified at the time, due to poor understanding of nutrient turnover in soil-plant systems, insufficient experience, and lack of scientific methodology. Understanding of soil-plant systems has greatly improved since the 1950s, however, so stringent analysis of the original arguments is now possible.
This review and analysis of core statements by the initiators of the organic farming movement showed that their stated reasons for prohibiting inorganic fertilizers are not corroborated by scientific facts. Their decision criteria were found to have no scientific legitimacy, and thus exclusion of inorganic fertilizers cannot be categorized as a principle based on science. Modern science requires invalid hypotheses to be rejected and false concepts to be corrected, so if theories and principles are maintained despite lack of evidence, science is ignored. If non-valid theories/concepts are not rejected upon falsification, they become articles of faith and make matters of science into matters of doctrine. Once a concept has become a doctrine, revision is often regarded as irrelevant.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
