Abstract
Juveniles and infants are often believed to decompose faster than adults due to smaller body masses, yet research on juvenile decomposition rates remains limited. Studies examining body size and decomposition yield contradictory results and typically rely on human analogues of adult-like proportions. Thus, whether smaller or larger cadavers decompose more rapidly remains unclear. Limited research exists on juvenile decomposition rates, despite their relevance to forensic investigations. In the UK, 46% of homicides involving victims under 16 remained unsolved in the year ending March 2022, compared to 30% across all homicides. Moreover, from 2012 to 2022, children under 1 year old had the highest homicide rate. Increased forensic understanding of juvenile decomposition could improve case resolution and investigative accuracy. This study investigated the relationship between cadaver size and decomposition rates using juvenile-sized domestic pigs in the UK. Over 18 weeks, total body score and percentage mass change were recorded. Given the subjective nature of visual scoring, three independent observers assessed the total body score to evaluate inter-rater reliability. Larger cadavers (12.3–16.1 kg) decomposed faster in early stages, while smaller cadavers (6.6–7.1 kg) showed an accelerated rate between weeks 4 and 12 based on total body score. Inter-rater analysis confirmed strong reliability in the scoring system. Both total body score and percentage mass change showed strong positive correlations with time, indicating that each measure reliably tracks the progression of decomposition.
Introduction
Many factors are known to influence the rate of decay. Some of the most widely accepted influences on decomposition include temperature, moisture, pH, partial pressure of oxygen, and time. 1 Many other extrinsic factors, such as clothing and coverings, affect the rate of decomposition. However, this is primarily due to their effect on the ambient temperature, 2 though it is also important to note that clothing and coverings may also influence decomposition as they are likely to retain moisture and restrict access to carrion. The effect body size has on the decomposition rate is an essential subject for investigation as it is a conspicuously under-researched area of taphonomic study. Two key articles3,4 investigated the impact of body size on cadaver decomposition using pigs as human proxies. Interestingly, the results of both studies contradict one another.
Sutherland et al. 4 observed a relationship between the initial carcass mass and the rate of decomposition in a study based in South Africa. Their findings indicated that smaller cadavers (<35 kg) achieved skeletonisation at twice the speed of larger cadavers (60–90 kg) when measured in accumulated degree days. In a similar study conducted in Hawaii, USA, Hewadikaram and Goff 3 found an increased rate of decomposition in larger cadavers during the earlier stages of decomposition. The largest variation was seen during the intermediate stages of decomposition. However, their findings revealed a less noticeable difference in the decomposition rate during the latter stages. It was suggested that this might have been due to the lack of samples used in their study as they based their experiment on one larger cadaver (15.1 kg) and one smaller cadaver (8.4 kg). Though, it must also be noted that in Hewadikaram and Goff's 3 study the larger cadaver was considerably smaller than almost half of Sutherland et al.’s 4 ‘small’ samples. Other researchers have reported body size potentially influencing decomposition,5–8 but none of these studies adopted body size as their primary focus. For example, in research conducted in the UK by Simmons et al., 9 it was observed that body size is significant when cadavers are assessed by insect colonisation. However, when insects are omitted, such as when bodies are buried or submerged, decomposition occurs at a constant rate, regardless of body size. Inconsistencies in results and methodological approaches justify the expansion of decomposition studies examining the impact body size has on the rate of decomposition. Moreover, as most of the above studies were conducted outside of the UK in vastly different climates, this study will also develop an understanding of how body size impacts decomposition rates in a more temperate climate.
In addition to the value of expanding knowledge in this area of study, progressing a practical understanding of the processes relating to decomposition will lead to more accurate reconstructions of events leading up to suspicious deaths, including vital information leading to more accurate post-mortem interval estimations.
This is particularly important in cases involving juvenile remains since there is minimal research 10 on whether juvenile remains would decompose at differing rates from adults. This issue is directly reflected in modern UK crime statistics as according to the Office for National Statistics, 11 no suspect had been charged in 46% of homicides involving victims under 16 years of age in the year ending March 2022 compared with 30% of cases with no charge when looking at all homicide victims. In addition, children under 1 year old were victim to the highest rate of homicide in the year ending March 2020, a fact that had been consistent since the year ending March 2012. 12 This further exemplifies the need for additional research in this area of taphonomic study with an added necessity for research conducted in the UK within a juvenile-focused context. In further regards to the motivation of this study, in 2018 Ross and Hale stated the importance of investigating and understanding juvenile and infant decomposition as being pertinent and essential due to such remains being more often concealed intentionally after death. This holds true for both accidental and intentional (including homicide) deaths. 10 As such, the rates of homicide and post-mortem actions relating to juvenile and infant remains are intrinsically tied to associated decomposition, which in turn is a crucial field of study.
This study aims to investigate the relationship between body size and decomposition rate in juvenile-sized remains under temperate UK conditions, using both total body score (TBS) and percentage mass change (PMC) as complementary measures, while also assessing the inter-observer reliability of TBS in real-time, in situ scoring. However, it is critical to acknowledge the anatomical and physiological differences between pig cadavers and human juveniles, particularly infants, which limit the direct translatability of decomposition findings. While pigs are widely used as proxies in decomposition research,13–22 human infants are born neurologically and physiologically under-developed compared to most mammals, including pigs. 23 Compared to pigs, which are born with mature organ systems and functional motor ability, human infants exhibit delayed brain development and immature organ systems. 23 Furthermore, structural differences in connective tissues may influence decay rates and subsequent ecological interactions with carrion, potentially altering the rate and pattern of decomposition. 24 Variations in skin thickness, tissue composition, and moisture content between pigs and human infants add further complexity. Another particularly relevant anatomical parameter is the head-to-body mass ratio, which varies considerably between species and developmental stages. While adult pigs show similar head-to-body mass ratios to adult humans, juvenile pigs differ notably from human infants. 25 This distinction has potential implications for decomposition progression, particularly in the cranial region. Therefore, anatomical properties and decomposition dynamics may not translate directly and must be interpreted with caution when pig models are used for estimating post-mortem intervals in forensic contexts.15,26
Measuring decomposition
Numerous visual methods can be used to measure the extent of terrestrial cadaver decomposition. These methods include accumulated decomposition score, 27 degree of decomposition index, 28 TBS, 29 and total decomposition score. 30 TBS is one of the most used approaches in taphonomic and entomological studies and has been used in a wide variety of decomposition studies.16,18,31–37 Descriptions of five main decomposition stages were devised by Galloway et al., 38 later adopted by Megyesi et al., 29 and used as a foundation for the point-based scoring system we now refer to as TBS. It remains one of the regularly selected methods of measuring cadaver decomposition. Several researchers have adapted this point-based system intending to make the scale more precise when used in combination with different human analogues.39–42 Keough et al. 42 adapted the scale for pig cadavers, although this system has not yet been exposed to the meticulous scientific scrutiny and experimentation of its predecessor. One study conducted by Indra et al. 43 supports the general applicability of the TBS system adapted by Keough et al. to pig cadavers, however the paper highlights the need for further refinement and validation to ensure its reliability under varied taphonomic conditions and across different stages of decomposition. The system developed by Megyesi et al. 29 visually inspects the stage of decomposition across three different regions of the body: the head and neck, the trunk, and the limbs. The scoring system consists of descriptions of differing post-mortem changes seen at varying stages of decomposition. To implement the scoring system, a cadaver is observed, and a description is assigned to each of the three regions of the cadaver. Each description corresponds to a numerical score. When each area has been scored, the composite score can be determined by adding together the three assigned values.
While TBS is commonly used in taphonomic and entomological studies, the scale's subjectivity has been criticised. Particularly subjective indicators associated with cadaver decomposition include tissue colour, intensity of maggot activity, and percentage of bone exposure. There is also some uncertainty on the reliability of these visual scoring methods in different research contexts. Owing to the brief nature of the descriptions provided by Megyesi et al. 29 and the nature of decomposition studies, it is assumed that individuals will likely understand and apply these subjective descriptions differently 44 ; although research by Dabbs et al. 45 demonstrated high inter-observer agreement among 16 participants using the TBS system, their assessments were based on digital observation packets comprising photographs and written descriptions, which may have introduced bias into the scoring process. Although photographic analysis can yield high inter-observer agreement, Ribéreau-Gayon et al. 46 found that scoring decomposition from photographs can produce significantly different results compared to real-time, in situ assessments, suggesting that photographs may not capture the full range of taphonomic changes necessary for accurate evaluation. Therefore, the inter-observer reliability of these subjective methods should be assessed in different geographical locations, owing to the huge variations in climate and other influences on the decomposition process. Due to the subjective nature of TBS, additional quantitative analyses, such as measuring the change in percentage mass of a cadaver, would be valuable to include in decomposition studies. 47
Methods
Cadavers
Six pig (Pigs were used as human analogues due to their likeness to humans including their skin and omnivorous diets. They attract much of the same entomology 14 and share similar soil electroconductivity. 48 ) cadavers were utilised in the study, three smaller (between 6.6 and 7.1 kg) and three larger (between 12.3 and 16.1 kg). For comparison, a 1-year-old human child's weight is expected to be between 6.9 and 13.3 kg with a median of 10.6 kg, and a 5-year-old human child's weight is expected to be between 12.4 and 27.9 kg with a median of 18.4 kg. 49 The cadavers used in this study were by-products of the food industry, sourced from a local abattoir and euthanised with a captive bolt pistol. Their skin and internal organs remained in place to ensure that the cadavers represented whole intact bodies. Autolysis begins in the abdomen because of bacteria and enzyme activity in the gut. Thus, without organs in situ, the typical sequence of decomposition would be disrupted. 50 The cadavers were transported a short distance and placed in an outdoor taphonomic research facility registered by the Department for Environment, Food, and Rural Affairs located in Wrexham, UK, within an hour of being euthanised. Ethical approval for this study was gained in accordance with the University's Code of Practice on Ethical Standards for Research.
Experimental procedure
At the facility, each cadaver was individually marked by an identification tag on a rear limb. Identification tags were made of plastic to withstand all environmental conditions. Each cadaver was placed in the centre of a piece of tarpaulin (50 cm apart) which acted as a barrier to minimise contact between the cadavers and the soil. Because the facility has been used for repeated decomposition experiments over time, it is likely to have become proliferated by microbes which could increase the rate of decomposition in this study.51,52 The tarpaulin was also essential in allowing the cadavers to be easily weighed at regular intervals. Each corner of the tarpaulin was attached to an Odowalker digital scale hook, and the cadavers’ mass was then measured in kilograms to three decimal places with an error margin of <5%. This method also prevented small bones and decomposed tissue from being lost when the cadavers were moved. The percentage of mass change from the initial mass was calculated at each interval to better compare samples of different sizes. This value is preferred because it accounts for the proportional aspect of the study, that is, that a loss of 1 kg from a cadaver weighing 7 kg is more significant than a loss of 1 kg from a cadaver of 15 kg.
The project ran for 18 weeks between August and December 2019. Data collection intervals occurred once every 7 days for the first 8 weeks. Once the advanced decay stage was observed, decomposition slowed, so data was collected once every 14 days for 4 weeks and then once every 21 days for 6 weeks.
Photographs of each cadaver were taken using a 20-megapixel Huawei P20 camera to record their appearance at the start of the study. Wire mesh cages were placed on top of the cadavers and secured to the ground with pegs to protect them from scavengers.53–55 The cages were constructed into triangular prisms using planks of wood and chicken wire with hexagonal openings of approximately 5 cm. The cages measured 60 × 30 × 26 cm3 for the smaller cadavers and 60 × 60 × 52 cm3 for the larger cadavers. These cages were chosen as they replicate those used in other taphonomic studies53–59 as they do not impede insect colonisation or affect the cadavers’ temperature while also protecting samples from larger scavengers, which could significantly disrupt or eliminate samples.
From week 1 to 10, the on-site temperatures ranged between 10 °C and 20 °C. From week 10 until the end of the study, temperatures observed ranged between 1 °C and 12 °C. Trees heavily shade the facility; however, it must be noted that all the samples were placed in the shade. Shaded areas slow the rate of decomposition because the temperature tends to be lower and fluctuates less than the temperature in non-shaded areas. 60 Three rain gauges were situated at various locations within the research facility to monitor if the amount of rainfall each cadaver was subjected to was equivalent (see Figure 1).
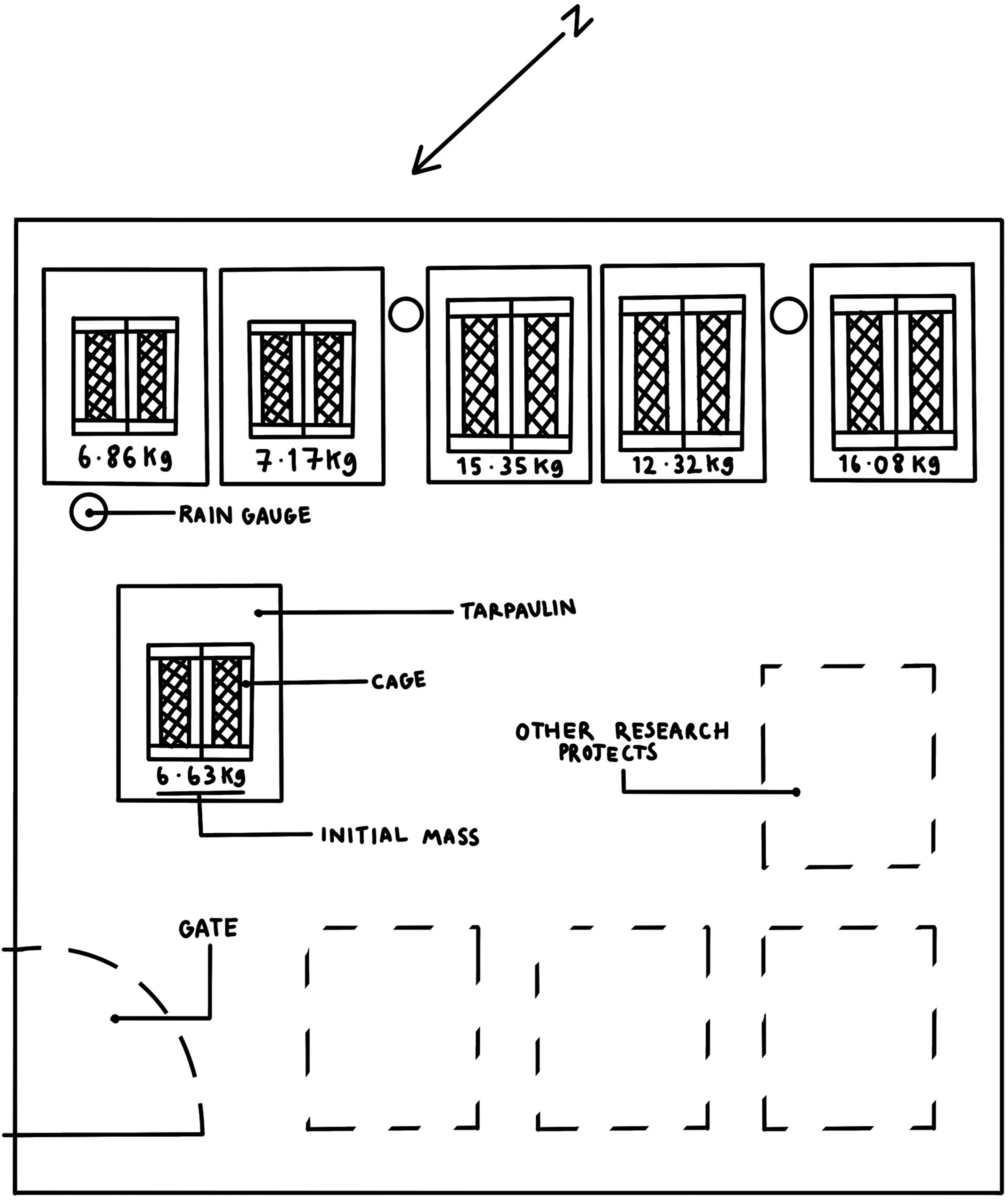
The layout of the Department for Environment, Food and Rural Affairs licenced taphonomic research facility at Wrexham. Illustration by Madeleine Thomson.
Data collection
At each interval, the TBS of each cadaver was documented by three independent observers using the system proposed by Megyesi et al. 29 (The system proposed by Megyesi et al. 29 was chosen as it is generally the most favoured TBS method and is used most frequently.) to establish whether inter-rater reliability of the scale used was present. Scores were assigned to three pre-agreed regions of the body: the head and neck, the trunk, and the limbs as defined by Megyesi et al. 29 The mass of the cadavers was recorded using the method above using an Odowalker digital scale. Temperature data was collected using the closest weather station located 14 km away. Rainfall was collected using rain gauges. The precipitation was transferred from the rain gauge and measured using a 100 cL measuring cylinder (error margin ≤1%). Photographs of each cadaver were also taken using a 20-megapixel Huawei P20 camera.
Statistical analyses
Two Spearman correlations were conducted to test whether there was a relationship in the changes seen in TBS and PMC between the smaller and larger cadavers throughout the study. In addition, a series of Mann–Whitney U tests were carried out to test for changes in TBS and PMC between the smaller and larger cadaver groups at different points in time throughout the 18 weeks. Specifically, tests were run using data collected from weeks 2, 4, 6, 8, 10, 12, 15, and 18. These periods were selected to allow for further analysis of changes over time and to account for any differences between the groups at varying points within any overall trend. Non-parametric tests were exclusively used due to sample sizes and to make the statistical tests as robust as possible. In addition, Dunn–Bonferroni corrections were applied to all significant values to account for multiple tests being run simultaneously. Krippendorff's alpha statistic was used to assess the inter-rater reliability of TBS between participants. All statistical tests were performed on SPSS v.28.
Results
Total body score
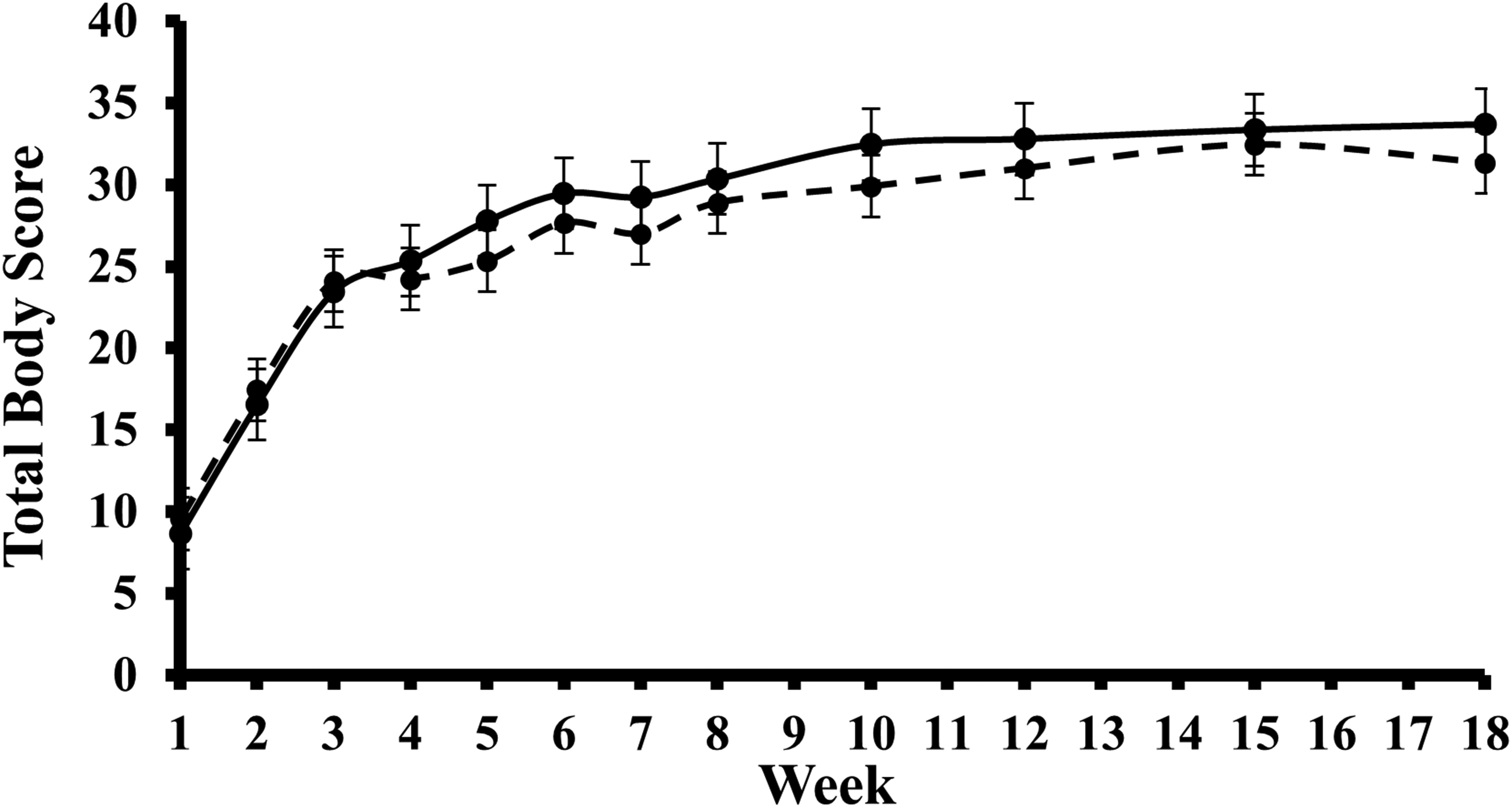
Table 1 shows the difference in TBS between the smaller and larger cadavers at selected project intervals. At week 2, it was noted that the mean TBS for the larger cadavers was greater than the smaller cadavers. However, the mean TBS for the smaller cadavers was greater at every other interval (see Figure 2). The mean TBS of the smaller cadavers was significantly greater than that of the larger cadavers from weeks 4 to 12. The greatest mean difference was recorded in week 10, where the smaller cadavers’ mean TBS was 2.55 greater. Krippendorff's alpha statistic returned an α = 0.9353. An α value of 0.9–1 demonstrates high inter-rater reliability between participants. 61
A box plot displaying the changes over time in total body scores assigned to the smaller (—) and larger (- -) cadavers during the experimental period. Dots represent mean scores and the vertical bars ±1 SD around the mean.
Results of Mann–Whitney U tests comparing TBS between the smaller and larger cadavers for select weeks over the 18-week period.
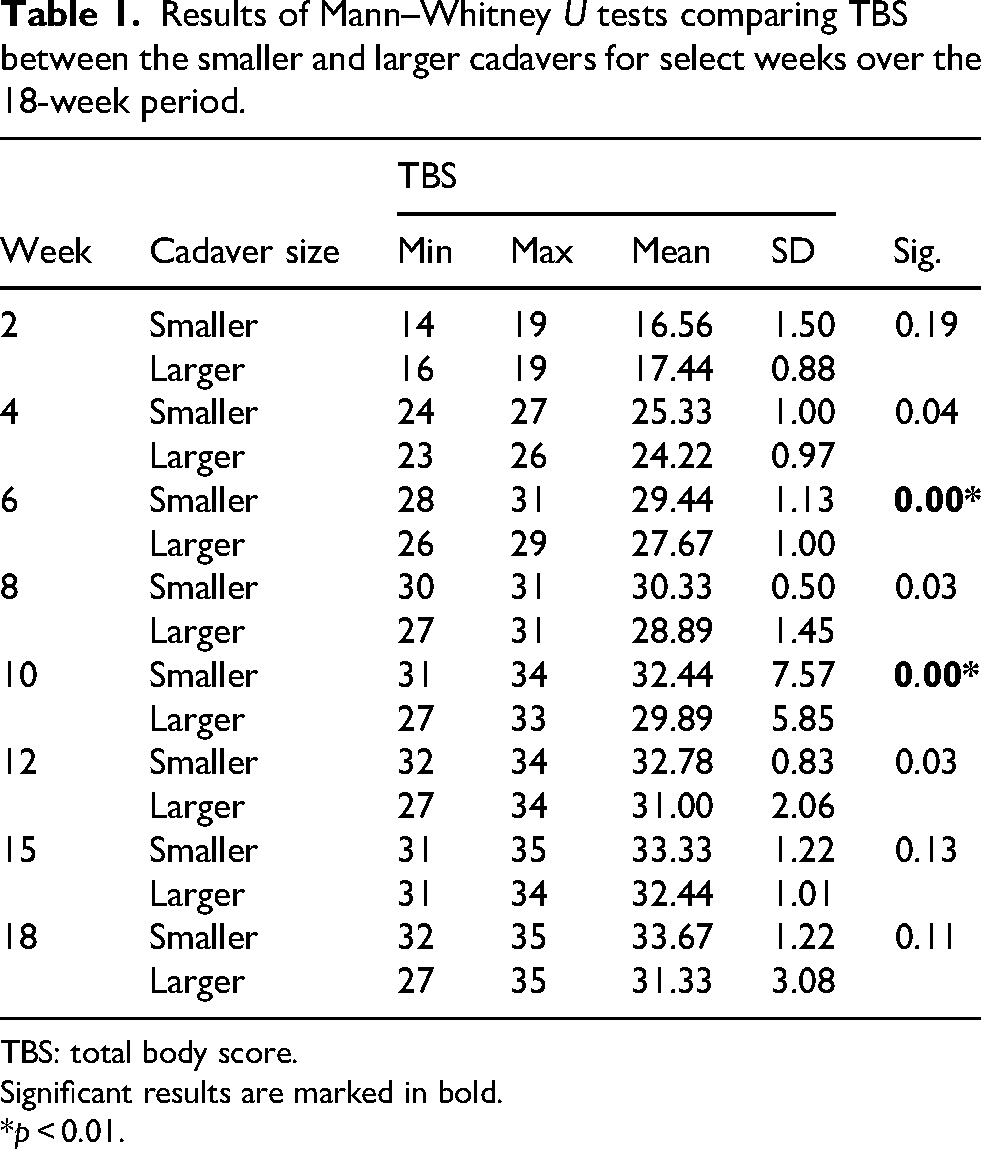
TBS: total body score.
Significant results are marked in bold.
*p < 0.01.
Percentage mass change
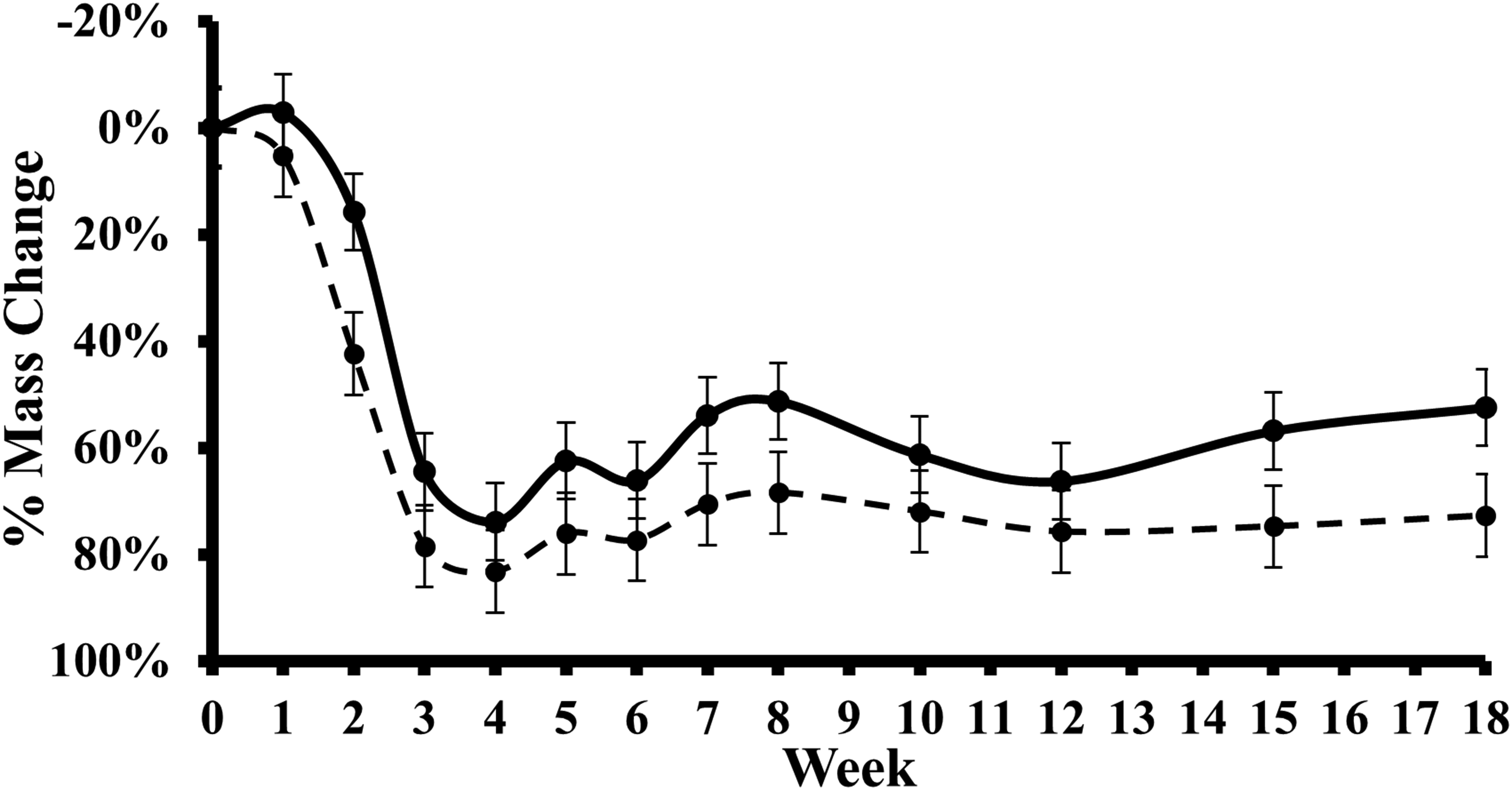
Table 2 shows the difference in PMC between the smaller and larger cadavers at pre-selected intervals during the study. Mean PMC was consistently greater in the larger cadavers compared to smaller cadavers throughout the study (see Figure 3). This was the greatest in week 2, where the difference in mean PMC was 25.66%, whereas the second highest difference recorded was only 18.00% in week 8. However, no statistical significance was identified by any test for any week.
A box plot displaying the changes over time in a comparison of the mean PMC recorded for the smaller (—) and larger (- -) cadavers during the experimental period. Dots represent mean scores and the vertical bars ±1 SD around the mean. PMC: percentage mass change.
Results of Mann–Whitney U tests comparing PMC between the smaller and larger cadavers for select weeks over the 18-week period.
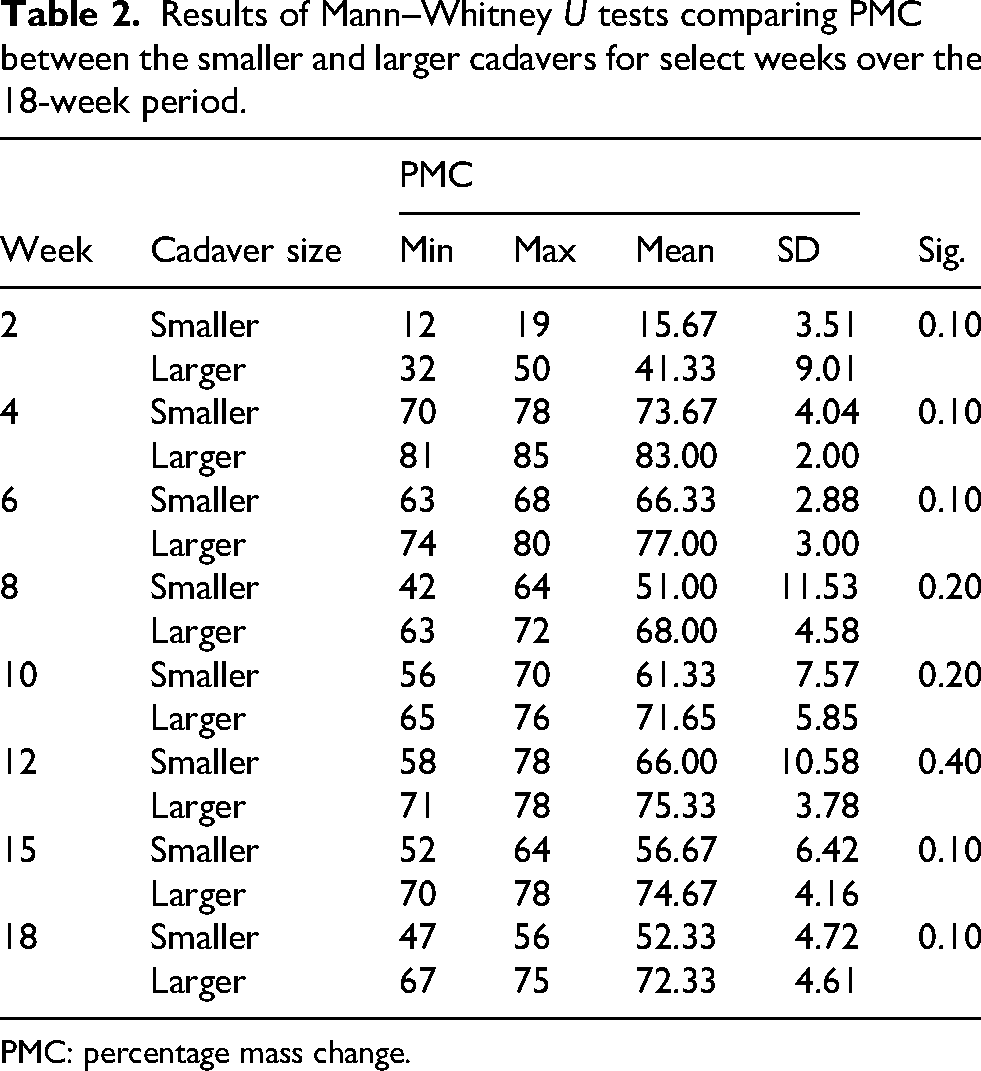
PMC: percentage mass change.
Correlation of TBS and PMC
Table 3 shows the correlation between the smaller and larger cadavers, for both TBS and PMC, over the 18-week research period. Results showed that both TBS and PMC significantly correlated between the two cadaver size groups over the course of the study.
Results of Spearman's rho tests for correlation between the smaller and larger cadavers for both TBS and PMC over the 18-week period. Significant results are marked in bold.

TBS: total body score; PMC: percentage mass change.
Significant results are marked in bold.
*p < 0.01.
Discussion
Total body score
Statistical analysis indicated a faster decomposition rate among the larger cadavers during the earlier stages of decomposition when looking at the TBS. This difference can be attributed to the understanding that larger cadavers lose heat at a slower rate than smaller cadavers due to a reduced surface area-to-volume ratio. As decomposition begins internally, initiated by the temperature-dependent bacteria and enzymes of the gut, larger cadavers would have more energy available for the autolytic process, meaning that it would occur faster.62,63 In contrast, between weeks 4 and 12, the smaller cadavers were consistently assigned a higher TBS than the larger ones.
The variation between the smaller and larger cadavers between weeks 4 and 12 highlights a need for a better understanding of the progression of decomposition happening during this period. The difference in the rate of decomposition seen during this period could be attributed to the fact that smaller cadavers have a larger surface area-to-volume ratio, meaning there is a vast surface area available for insects to lay eggs. However, once hatched, there is less food available due to the smaller volume of the pig. Hewadikaram and Goff 3 presented similar findings in their study, which used similar-sized cadavers. They highlight that the largest difference in decay rates is seen during the intermittent stages of decay and that fewer notable differences were identified during the earlier and later stages of decomposition.
Percentage mass change
Although no statistical significance was found, which may have been a result of the small sample size utilised, observations saw that the larger cadavers decomposed at an accelerated rate during the earlier stages of the study (see Figure 3). However, there was a less considerable difference seen during the later stages. In the first week of study, minor changes in mass were recorded. Measurements showed that the smaller cadavers gained weight during this period which could have been due to water saturation from rainfall which was particularly heavy across the site. Similar weight gain is likely to have occurred among the larger cadavers, but this could have been obscured by the overall mass loss caused by the faster decomposition seen in the early stages. However, Figure 3 shows clear deviations in PMC between the smaller and larger cadavers, particularly after weeks 2 and 6.
This finding is consistent with the delayed onset of the active decay stage in smaller cadavers seen in Hewadikaram and Goff's study. 3 Similarly, Matuszewski et al. 17 noted that bloating started earlier in larger cadavers. Although, they remained in this stage for longer than the smaller cadavers, explaining why larger cadavers would have lost a greater percentage of their mass at this stage. Denno and Cothran 64 observed a relationship between carcass size and fly population, which could also explain why the larger cadavers would have lost more biomass during the earlier stages of decay. However, the findings presented here differ from those of Spicka et al., 65 who found that smaller cadavers decomposed faster than their larger counterparts in the first 6 days. Although, it should be noted that in Spicka et al.’s 65 study, the smaller cadavers were categorised as <20 kg and would therefore include both the sample groups in this study. Once the active decay stage was reached, and entomological activity became the driving factor, the smaller cadavers decomposed at an accelerated rate. This acceleration could be due to the increased mass of maggots colonising the larger cadavers and impacting the overall mass. 3
Additionally, Matuszewski et al. 17 found that active decay was more efficient in smaller cadavers resulting in a quicker mass loss and a shorter period of active decay overall. Spicka et al. 65 suggested that it is easier for larvae to access smaller cadavers’ body cavities, gaining access to internal tissues for consumption faster, which they suggest is likely due to smaller cadavers having reduced tissue volume and less structural resistance. This study observed that larger cadavers decomposed at an accelerated rate during the fresh and bloat stages, whereas during the intermittent stages of decay, the smaller cadavers decomposed at an accelerated rate. Therefore, it is possible that juvenile remains could decompose faster than those of infants in the first few weeks after death. However, once the cadavers reach advanced decay, it is possible infants could decompose at an accelerated rate. However, further research is needed in this area to establish whether this would be the case in humans. In addition, it must be noted that in humans, infants have a larger body fat content than juveniles. 66 On the other hand, smaller pigs have a lower body fat content than larger pigs. 67 Therefore, care must be taken when interpreting these findings to cases involving human remains. The results of this study extend our understanding of the effects body size has on the rate of decomposition while also contributing to our understanding of decomposition processes in cooler climates. In addition, the study used cadavers more representative of infant and juvenile-sized remains, as opposed to most similar studies that used much larger cadavers.
Limitations
While this study provides novel insights into the decomposition of juvenile-sized remains under temperate UK conditions, several limitations should be acknowledged.
The use of pig cadavers, although widely accepted as human analogues in forensic taphonomy,68–71 may not fully replicate the anatomical, microbial, or physiological properties of human remains, especially human juveniles.14,15 This limits the generalisability of the findings to actual forensic cases involving humans. To enhance the forensic applicability of animal-based decomposition research, future studies should incorporate retrospective analysis of forensic case data involving actual human children and infants, where ethically and legally permissible, to evaluate whether anatomical differences result in measurable difference in decomposition patterns.
The study was conducted in a single location and under specific environmental conditions. Factors such as soil composition, humidity, rainfall, and regional insect fauna likely influenced decomposition rates and patterns and may differ considerably in other geographic or climatic contexts. 72
While decomposition was measured using both TBS and PMC, each method presents limitations. TBS relies on visual assessments, which, despite demonstrating inter-observer reliability, 45 these systems are inherently subjective and may vary with assessor experience. PMC, on the other hand, assumes all mass loss is attributable to decomposition alone. However, the use of a tarpaulin to measure mass may result in over-estimation of soft tissue mass by including decomposed fluids, rainwater, and other by-products, particularly during the active decay stage. Future research may benefit from exploring alternative weighing strategies that distinguish between intact tissue and decomposition by-products.
Additionally, internal temperature loggers were not used in this study. While ambient temperatures were recorded periodically, they do not reflect internal body temperatures, particularly during periods of intense maggot activity. Future studies should consider the use of internal data loggers or thermal imaging to monitor internal temperature profiles more precisely and help assess the thermal impact of body size on decomposition.
Furthermore, larvae and insect activity were not quantitatively measured throughout the decomposition process. Given the critical role of insect colonisation in soft tissue breakdown, 8 future work should incorporate detailed entomological sampling to better understand how body size influences insect access, larval development, and overall decomposition rate.
Conclusion
This study aimed to investigate whether there is a relationship between body size and the rate of decomposition in juvenile-sized remains. The TBS and PMC of two groups of pig cadavers (representative of two sizes of humans) were recorded at regular intervals over 18 weeks. Both TBS and PMC showed strong positive correlations with time, indicating that each measure reliably tracks the progression of decomposition. Based on the interpretation of the findings, the larger cadavers decomposed at an accelerated rate during the earlier stages of decomposition. However, during the later stages, the smaller cadavers decomposed at an accelerated rate. The inter-rater reliability of the TBS system was statistically evaluated and showed a high rate of inter-rater reliability.
These results highlight the need for continued decomposition research to improve our understanding of the relationship between the initial size of a cadaver and the decomposition rate. It is recommended that future studies include experimental groups with a greater range of initial body mass to better capture size-related variability. Investigations conducted over extended post-mortem intervals are also encouraged to identify long-term trends in decomposition between cadavers of different sizes. In addition, future research using human remains is essential to determine whether these findings translate to human decomposition.
Footnotes
Acknowledgements
The authors would like to thank Madeleine Thomson for providing the artwork for this paper.
Ethical considerations
Ethical approval for this study was gained in accordance with the University's Code of Practice on Ethical Standards for Research.
Consent for publication
Consent to publish has been provided in writing by the designer of the figures in written format.
Author contributions
PT contributed to the conceptualisation, data curation, formal analysis, investigation, methodology, and writing original draft and review and editing. AER contributed to the conceptualisation, data curation, investigation, methodology, supervision, and writing – review and editing. CA contributed to the formal analysis and writing – review and editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Christopher Aris is an consulting editor of this journal. The other authors declared no potential conflicts of interest.
