Abstract
The safety and efficacy of mifepristone antagonization with progesterone to avert medication abortion, also known as abortion pill rescue, is a subject of vigorous debate. Two prominent medical associations have taken positions that either entirely reject or fully support its use. This scoping review aimed to gain insight into the safety and efficacy of its use. Analysis of 16 studies showed that the continuing pregnancy rate after ingesting mifepristone alone is ≦25 percent for gestational ages ≦49 days. Analysis of four studies showed that two-thirds of the women who changed their minds and received progesterone after initiating their medication abortion with mifepristone could safely continue their pregnancies. There is no increased maternal or fetal risk from using bioidentical progesterone in early pregnancy. If a woman has already taken mifepristone for her medication abortion and then changes her mind, timely supplementation with progesterone may allow her pregnancy to continue. The conclusion that mifepristone antagonization with progesterone is a safe and effective treatment has implications for medication abortion informed consent.
Keywords
Introduction
Medication abortion is a two-drug regimen that uses the drugs mifepristone (aka Mifeprex and RU-486) and misoprostol (FDA 2016) (FDA 2017). The regimen begins with mifepristone ingestion, followed 24–48 h later with 800 mcg of misoprostol taken buccally (in the cheek pouch) (ACOG 2020). The FDA manages the regimen under the Risk Evaluation and Mitigation Strategy (REMS). In 2023, the FDA permanently removed the REMS in-person dispensing requirement and added a process to certify pharmacies but retained other REMS requirements, such as the need for prescriber certification (FDA 2023, 1).
Mifepristone contraindications include confirmed/suspected ectopic pregnancy, chronic adrenal failure, concurrent long-term corticosteroid therapy, concurrent anticoagulant therapy, and the presence of an intrauterine device in place (FDA 2016). The American College of Obstetricians and Gynecologists (ACOG) recommends that healthcare providers administer Rh D immune globulin to Rh D-negative women who have a medication abortion (ACOG 2017, e63). Recent studies propose a “no-test” protocol that eliminates pre-abortion ultrasound, pelvic examination, and Rh D-negative testing (Raymond et al. 2020, 362).
In 2016, the FDA modified the regimen by expanding use from 49 to 70 days gestation and by reducing mifepristone dosage from 600 to 200 mg. The FDA also removed the requirement that prescribers submit adverse event reports (AER) but retained the requirement to report deaths (Aultman et al. 2021, 7). In 2021, the FDA removed the requirement to dispense mifepristone in clinics, medical offices, and hospitals (Cavazzoni 2021). This decision created the opportunity for women to receive mifepristone without being examined by a medical professional.
Medication abortion accounts for 50 percent to 60 percent of all US abortions (Guttmacher Institute 2022). Dozens of websites such as PlanCPills, HeyJane, and AidAccess sell medication abortion kits for $200—$400 per kit (Mosbergen and Vibhuti 2022). Many of the websites provide incomplete information that undermines informed consent and increases the risk of severe illness and possible death. For instance, AbortionRx does not mention mifepristone contraindications such as ectopic pregnancy, concurrent long-term corticosteroid therapy, or concurrent anticoagulant therapy. Nor does it mention the need for Rh D immune globulin for Rh D-negative women (Abortionrx 2023, 1).
Progesterone plays a critical role in the normal functioning of the human female reproductive system. It promotes placental development, prepares the uterus for embryo implantation (Coomarasamy et al. 2015, 2142), and inhibits uterine contractions (Scarpin et al. 2009, 1). Mifepristone is a progesterone receptor antagonist that binds more aggressively to the progesterone receptors in the uterus than progesterone. The primary effect of mifepristone is to cause a separation of the decidua basalis from the trophoblast, which results in embryo demise (AAPLOG 2022, 1). Misoprostol triggers uterine contractions, expelling the embryo.
Some women change their mind about their medication abortion after ingesting mifepristone but before taking misoprostol. For example, a study by Aultman et al. analyzed 19 years of mifepristone AER. They found 102 of the 452 patients (22.6 percent) with a continuing pregnancy after mifepristone alone changed their mind and chose to continue their pregnancy (Aultman et al. 2021, 4). Thus, even after initiating their medication abortion, some women change their mind.
The concept of mifepristone antagonization is to use high doses of progesterone to halt the effects of mifepristone and increase the continuing pregnancy rate. A patient must receive progesterone within 72 h after taking mifepristone but before taking misoprostol (AAPLOG 2022, 3). The increase in progesterone concentration displaces mifepristone binding at the progesterone receptors to reestablish regular progesterone binding and encourage healthy embryo development (AAPLOG 2022, 2). Proponents claim that there have been thousands of documented cases of mifepristone antagonization (aka “abortion pill rescue”) that resulted in live births (Heartbeat International 2023).
At least twelve states require physicians to inform women seeking a medication abortion of mifepristone antagonization using progesterone (Forsythe and Harrison 2022, 406). Two prominent medical associations have taken positions that either entirely reject or fully support mifepristone antagonization with progesterone to avert medication abortion. The American Association of Pro-life Obstetricians and Gynecologists (AAPLOG) states that the action of mifepristone “can be slowed or arrested with progesterone based on biological plausibility and limited cohort data” and is based upon “limited and inconsistent scientific evidence (Level B)” (AAPLOG 2022, 4). They fully support informing women of the option of mifepristone antagonization as part of informed consent prior to a medication abortion (AAPLOG 2019). The ACOG states that claims of medication abortion reversal are “not supported by science” and “do not meet clinical standards” (ACOG 2022). They entirely reject any attempts to mandate disclosing mifepristone antagonization as part of informed consent prior to medication abortion. This paper examines the scientific evidence to answer the question, “Is mifepristone antagonization with progesterone a safe and effective treatment to avert medication abortion?”
Methods
Since 2015 three journal articles have been published that reviewed mifepristone research studies to determine the continuing pregnancy rate after mifepristone alone (Grossman et al. 2015; Davenport et al. 2017; Creinin and Chen 2019). In total, the three articles reviewed 16 studies. However, each article reviewed a different subset of the studies and reported different results. This scoping review began by analyzing the 16 studies cited by the three articles to determine the continuing pregnancy rate after mifepristone alone.
Since 2012 four journal articles have been published that documented the continuing pregnancy rate after ingesting mifepristone followed by progesterone (Delgado and Davenport 2012); Garratt and Turner 2017; Delgado et al. 2018; Creinin et al. 2020). This scoping review next analyzed the four articles to determine the continuing pregnancy rate after ingesting mifepristone followed by progesterone.
Lastly, this scoping review analyzed safety considerations, including the safety of using progesterone during pregnancy and the safety of using mifepristone alone without misoprostol.
Results
The following section organizes results into three groups: continuing pregnancy rate after mifepristone alone, continuing pregnancy rate after mifepristone and progesterone, and safety considerations.
Continuing Pregnancy Rate after Mifepristone Alone
It is essential to establish the continuing pregnancy rate after ingesting mifepristone alone because it provides a baseline against which to compare the efficacy of progesterone as a mifepristone antagonist. ACOG states that “as many as half of women who take only mifepristone continue their pregnancies” (ACOG 2022). ACOG's only citation for this statement relies upon a systematic review that analyzed continuing pregnancy after mifepristone alone (Grossman et al. 2015). However, since 2015 there have been two additional publications (Davenport et al. 2017; Creinin and Chen 2019) that analyzed continuing pregnancy after mifepristone alone.
There are three possible results after ingesting mifepristone alone; (1) embryo demise with the complete evacuation of the uterus (complete abortion); (2) embryo demise with incomplete uterine evacuation or no evacuation at all (incomplete abortion); (3) embryo survival (continuing pregnancy) (Davenport et al. 2017, 9). The 16 studies aimed to determine the efficacy of mifepristone as an alternative to surgical abortion (complete abortion). As a result, four of the 16 studies did not clearly distinguish between an incomplete abortion and continuing pregnancy when reporting mifepristone treatment failure. The remaining 12 studies used ultrasound to determine the presence of a living embryo to differentiate between an incomplete abortion and a continuing pregnancy.
In the Grossman article previously mentioned, the authors reviewed 11 studies (1092 women). They concluded that the “proportion of pregnancies continuing 1–2 weeks after mifepristone alone varied from 8 percent (95 percent CI 3–22 percent) to 46 percent (95 percent CI 37–56 percent)” and that “continuing pregnancy was more common with lower mifepristone doses and advanced gestational age” (Grossman et al. 2015, 206). However, the review included four studies (Birgerson and Odlind 1988; Grimes et al. 1988; Swahn et al. 1989; Zheng 1989) that did not use ultrasound to verify the presence of a living embryo when they reported mifepristone treatment failure.
Birgerson relied upon changes in hCG levels to determine “continuing pregnancy” (Birgerson and Odlind 1988). Grimes defined therapeutic success (complete abortion) as vaginal bleeding and declining beta-human chorionic gonadotropin (ß-hCG) titer. Otherwise, the patient was classified as “failed to abort” and underwent suction curettage (Grimes et al. 1988). Swahn classified women with “intact amniotic sac” as a “treatment failure” but did not verify the existence of a living embryo (Swahn et al. 1989). Zheng define “persisting pregnancy” as “no expulsion of the conceptus” and “gradual” increase of serum or urine hCG. The authors did not define “gradual” (Zheng 1989).
Thus, the reported continuing pregnancy rates for these four studies (N = 516) may have overstated the actual continuing pregnancy rate. The remaining seven studies used ultrasound to verify embryo survival and reported continuing pregnancy rates ranging from 0 percent to 25 percent (N = 576).
In 2017, Davenport published a systematic review of 12 studies (675 women). All studies cited by Davenport used ultrasound verification to document living embryos were present at follow-up. The continuing pregnancy rate using “total doses of 200–300 mg ranged from 10–23.3 percent” and “regimens with total doses ≥400 mg had embryo survivals ranging from 0–18.1 percent when followed ≥14 days post mifepristone, and 0–50 percent when followed for 6–8 days after mifepristone.” The continuing pregnancy rate for “studies including gestations up to 70 days was ≤25 percent in three of four studies” (Davenport et al. 2017, 3–4). Eleven of the 12 studies reported continuing pregnancy rates ranging from 0 percent to 25 percent (N = 657). One study had a continuing pregnancy rate of 50 percent (Elia 1985, N = 18).
In 2019, Creinin published a review of seven studies (550 women). All studies cited by Creinin used ultrasound verification to document living embryos were present at follow-up. This review reported that “the continuing pregnancy rate was higher with 200 mg (7/30 [23 percent, 95 percent confidence interval 8–38 percent]) than 600 mg (29/420 [7 percent, 95 percent confidence interval 4–9 percent])” (Creinin and Chen 2019, 428). The authors concluded that the data was inadequate to determine the continuing pregnancy rate after mifepristone alone because there was not enough data from studies using the FDA recommended 200 mg dosage (N = 30) (Creinin and Chen 2019, 428). These seven studies reported continuing pregnancy rates ranging from 0 percent to 25 percent (N = 550).
In total, the three articles analyzed 16 studies (1191 women). Each article analyzed a different subset of the 16 studies. Table 1 summarizes the findings.
Studies Reporting Continuing Pregnancy Rate Following Mifepristone.
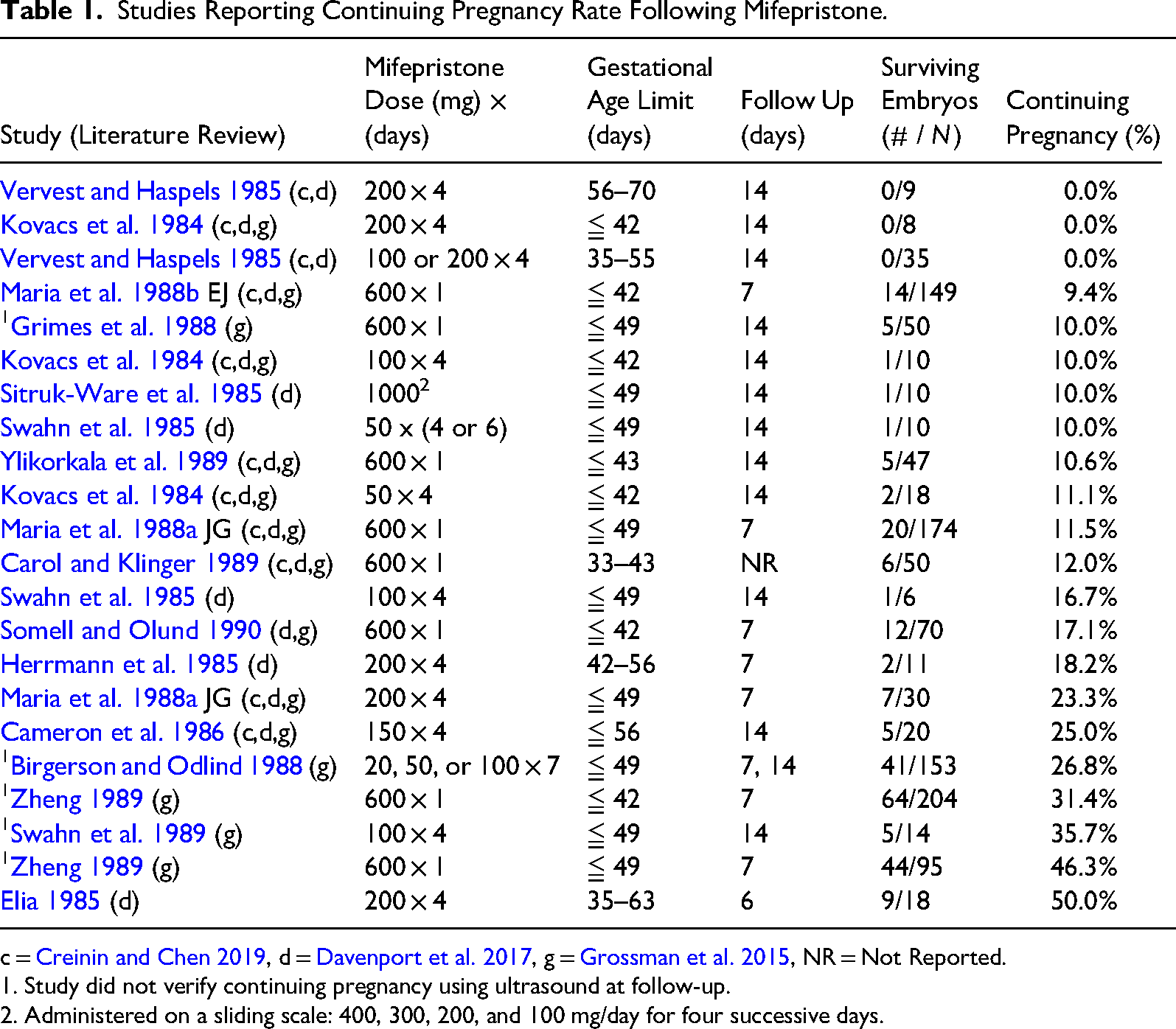
c = Creinin and Chen 2019, d = Davenport et al. 2017, g = Grossman et al. 2015, NR = Not Reported.
1. Study did not verify continuing pregnancy using ultrasound at follow-up.
2. Administered on a sliding scale: 400, 300, 200, and 100 mg/day for four successive days.
Combining data to perform a meta-analysis across the 16 studies is not possible because of the varied dose regimens. However, the continuing pregnancy rate for each of the 12 studies that verified embryo survival never exceeded 25 percent, except for Elia (1985), N = 18, regardless of mifepristone dose, gestational age, or follow-up period. Studies with a shorter follow-up period after the last mifepristone dose, such as Elia (1985) (2 days after last dose), tended to have higher embryo survival rates, and vice-versa.
Mifepristone doses varied from 200 to 1000 mg across the 16 studies. Some studies used multiple treatment groups resulting in 22 treatment groups across the 16 studies. Six treatment groups used mifepristone doses of 200–400 mg (N = 88). Ten treatment groups used mifepristone doses of 600–700 mg (N = 1012). Six treatment groups used mifepristone doses of 800–1000 mg (N = 91).
The gestational age limit ranged from ≤42 days to ≤70 days across the 22 treatment groups. Seventeen treatment groups reported a gestational age limit of ≤ 49 days (N = 1098). Five treatment groups reported a gestational age limit of 50–70 days (N = 93).
Four studies (Elia 1985; Grimes et al. 1988; Herrmann et al. 1985; Zheng 1989) found that the continuing pregnancy rate was more common with advanced gestational age. Davenport et al. also reported data from a 1986 Elia report showing that the continuing pregnancy rate increased as the gestational age limit increased (Davenport et al. 2017, 12). Table 2 summarizes the five studies (754 women) that reported continuing pregnancy rates by gestational age.
Studies Reporting Continuing Pregnancy Rate by Gestational Age.

1. Study did not verify continuing pregnancy using ultrasound at follow-up.
2. Elia had the shortest follow-up period of all 16 studies (6 days). Studies with shorter follow-up periods tended to have higher embryo survival rates.
In summary, Tables 1 and 2 show that the continuing pregnancy rate after ingesting mifepristone alone is generally ≤25 percent for gestational ages ≦49 days. There is insufficient evidence to establish the continuing pregnancy rate after mifepristone alone for gestational age ≥50 days. The evidence also shows that continuing pregnancy is more common with lower mifepristone doses and greater gestational ages.
Continuing Pregnancy Rate after Mifepristone and Progesterone
ACOG states there is “no credible scientific evidence” that progesterone will halt mifepristone binding from uterine progesterone receptors (ACOG 2022). However, researchers at French pharmaceutical company Roussel Uclaf, developer of mifepristone, published research in 1984 demonstrating the dissociation rate of mifepristone binding from rat uterus progesterone receptors when exposed to progesterone (Baulieu and Segal 1985, 91, Figure 3). Their research demonstrated that high progesterone concentrations can trigger mifepristone disassociation from uterine progesterone receptors.
Research published by Yamabe et al. studied how mifepristone and progesterone affect the luteal function during pregnancy in rats (Yamabe et al. 1989). The researchers gave one group of pregnant rats mifepristone and a second group mifepristone and progesterone. The study found that 33 percent of the mifepristone only group delivered live offspring but 100 percent of the mifepristone plus progesterone group delivered live offspring (AAPLOG 2022, 2).
Two case series documented mifepristone antagonization in humans. In 2012, Delgado documented a series of six case reports. Four of six women (67 percent) who received progesterone after ingesting mifepristone alone delivered a viable infant without birth defects (Delgado and Davenport 2012, e36). The other two cases resulted in a complete abortion. In 2017, Garratt documented a series of three case reports. Two of three women (67 percent) who received progesterone vaginally after ingesting mifepristone alone delivered a viable infant without birth defects (Garratt and Turner 2017, 473). The other case resulted in a complete abortion.
In 2018, Delgado published a case series of 547 patients who attempted mifepristone antagonization with progesterone to avert medication abortion (Delgado et al. 2018, 25). The patients received progesterone using six delivery regimens; intramuscular injection of progesterone in oil, oral administration of micronized progesterone, vaginal use of oral micronized progesterone capsules, compounded micronized progesterone vaginal suppositories, progesterone vaginal gel, and progesterone vaginal suppositories (Delgado et al. 2018, 24). Patients who received intramuscular progesterone had a 64 percent continuing pregnancy rate (N = 125), and those who received high-dose oral progesterone had a 68 percent continuing pregnancy rate (N = 31) (Delgado et al. 2018, 26). Delgado reported that the gestational age at the time of the mifepristone ingestion was directly related to the embryo survival rate: 35 days (25 percent), 42 days (46 percent), 49 days (49 percent), 56 days (61 percent), 63 days (77 percent). 71 percent of the patients (N = 291) in the Delgado case series report had a gestational age of 49 days or less.
Delgado reported seven birth defects (2.7 percent) for the 257 patients that had live births and post-delivery follow-up. The reported birth defects were port wine stain (1), bilateral absent toe (1), unilateral two absent fingers (1), choroid plexus cyst (1), cystic kidney (1), unilateral failed hearing test (1), heart murmur (1). This birth defect rate is comparable to the 3 percent birth defect rate in the general population (Delgado et al. 2018, 26).
In 2020, Creinin published a randomized controlled trial designed to study the safety and efficacy of mifepristone antagonization using high-dose oral progesterone (Creinin et al. 2020). Two of the 12 patients voluntarily exited the study. Of the ten remaining patients, five received 200 mg of oral progesterone, and five received a placebo. Gestational cardiac activity continued for two weeks for four of five patients (80 percent) receiving progesterone and for two of five patients (40 percent) receiving a placebo (Creinin et al. 2020, 158). Three patients experienced hemorrhage and were transported by ambulance to the hospital. Two of three patients received a placebo, the other received progesterone. Both patients who received a placebo required aspiration, and one placebo patient also required transfusion. The patient who received progesterone did not require any medical treatment. The authors halted the study after the third hemorrhage, though all hemorrhages requiring medical treatment occurred in the placebo group.
Table 3 summarizes the four human studies, broken down by delivery regimen and progesterone dose. The continuing pregnancy rates for all doses and delivery regimens ranged from 32 percent to 100 percent (N = 359). The continuing pregnancy rate for the delivery regimens intramuscular (all groups) and high-dose oral was 65 percent (N = 130) and 69 percent (N = 36), respectively. The continuing pregnancy rate for the vaginal delivery regimen was 38 percent (N = 193).
Studies Reporting Continuing Pregnancy Rate Following Mifepristone and Progesterone.
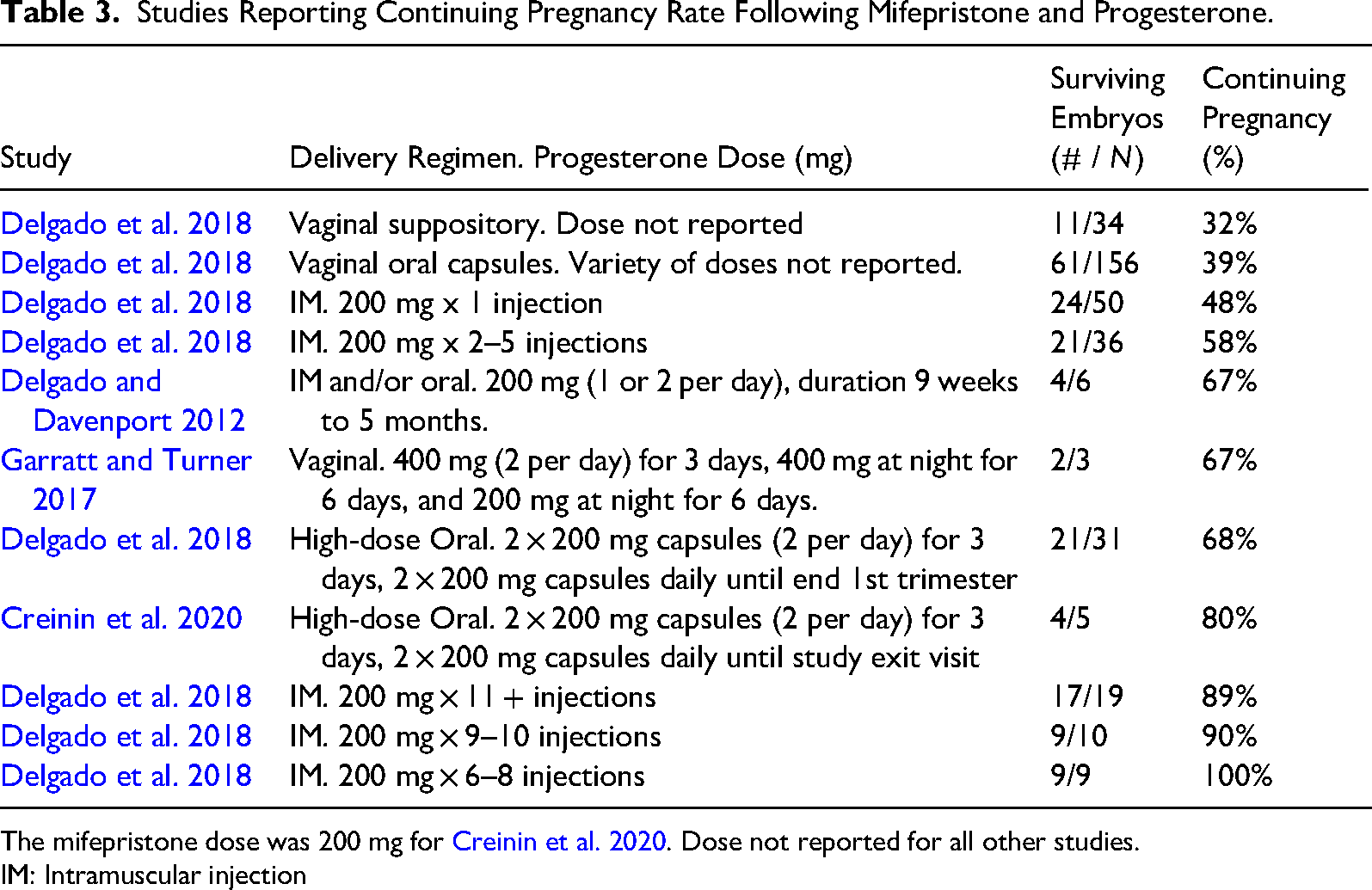
The mifepristone dose was 200 mg for Creinin et al. 2020. Dose not reported for all other studies.
IM: Intramuscular injection
The Delgado case series articles have been criticized because they lacked the rigor of double-blind, placebo-controlled randomized trials (Grossman and White 2018, 1493). The case series articles did not use a randomly assigned, mifepristone-only placebo control group. This limits the generalization of the case series data because it is not possible to compare the effectiveness of the progesterone treatment against a control group that did not receive the treatment.
However, placebo usage opposes the bioethical principle of autonomy (“self-rule”) because it rejects the patient's decision to continue her pregnancy (Beauchamp and Childress 2019, 99). Placebo usage also opposes the bioethical principle of beneficence (“to do good”) because it withholds a treatment that increases the probability of embryo survival (Beauchamp and Childress 2019, 157). Placebo usage in this case lacks equipoise and is therefore unethical.
AAPLOG recommends the delivery regimens high-dose oral and intramuscular injection (7 or more) for women who change their mind after taking mifepristone (AAPLOG 2022, 3). They also recommend an immediate sonogram to determine “intrauterine location, viability, and gestational age” and advise delaying treatment if there is suspicion of “an ectopic pregnancy, septic abortion or other complication that requires immediate gynecologic attention in a hospital or similar setting” (AAPLOG 2022, 4).
In summary, the evidence shows that the continuing pregnancy rate using the progesterone delivery regimens intramuscular injection and high-dose oral are 65 percent and 69 percent, respectively. The vaginal delivery regimen is less effective, having a continuing pregnancy rate of 38 percent.
Safety Considerations
Physicians have used bioidentical progesterone in reproductive medicine for over fifty years (Di Renzo et al. 2020). The term bioidentical progesterone refers to progesterone that is biologically identical to that produced by the human body (Di Renzo et al. 2020, 3). Progesterone is “a critical regulator” of normal human female reproduction (Scarpin et al. 2009, 1) and reduces the risk of premature birth (Di Renzo et al. 2021, 1).
The American Society for Reproductive Medicine reports no increased risk from using bioidentical progesterone in early pregnancy (ASRM 2008, 791) (The case series study by Delgado et al. found that there was no increase in birth defects after receiving progesterone treatment [2.7 percent] as compared to the birth defect rate in the general population [3 percent] [Delgado et al. 2018, 26]).
The AGOG has questioned the safety of using mifepristone alone without misoprostol. They state that “limited available evidence suggests that use of mifepristone alone without subsequent administration of misoprostol may be associated with an increased risk of hemorrhage” (ACOG 2020, e33). The only evidence cited by ACOG is the previously discussed randomized controlled trial in which there was bleeding after mifepristone without misoprostol (Creinin et al. 2020). Three of the ten patients visited the hospital because of bleeding, two in the placebo group and one in the progesterone group. Both patients in the placebo group experienced an incomplete abortion and required dilation and curettage. One patient in the placebo group also required a transfusion. The patient in the progesterone group experienced a complete abortion and required no treatment.
Each of the previously discussed 16 studies reported the level of uterine bleeding experienced by research participants after mifepristone alone. There was a total of 19 hemorrhage events among the 1191 research participants (1.6 percent). Table 4 lists the number of hemorrhage events for each study and the reported hemorrhage event description.
Studies Reporting Hemorrhage Events Following Mifepristone Alone.
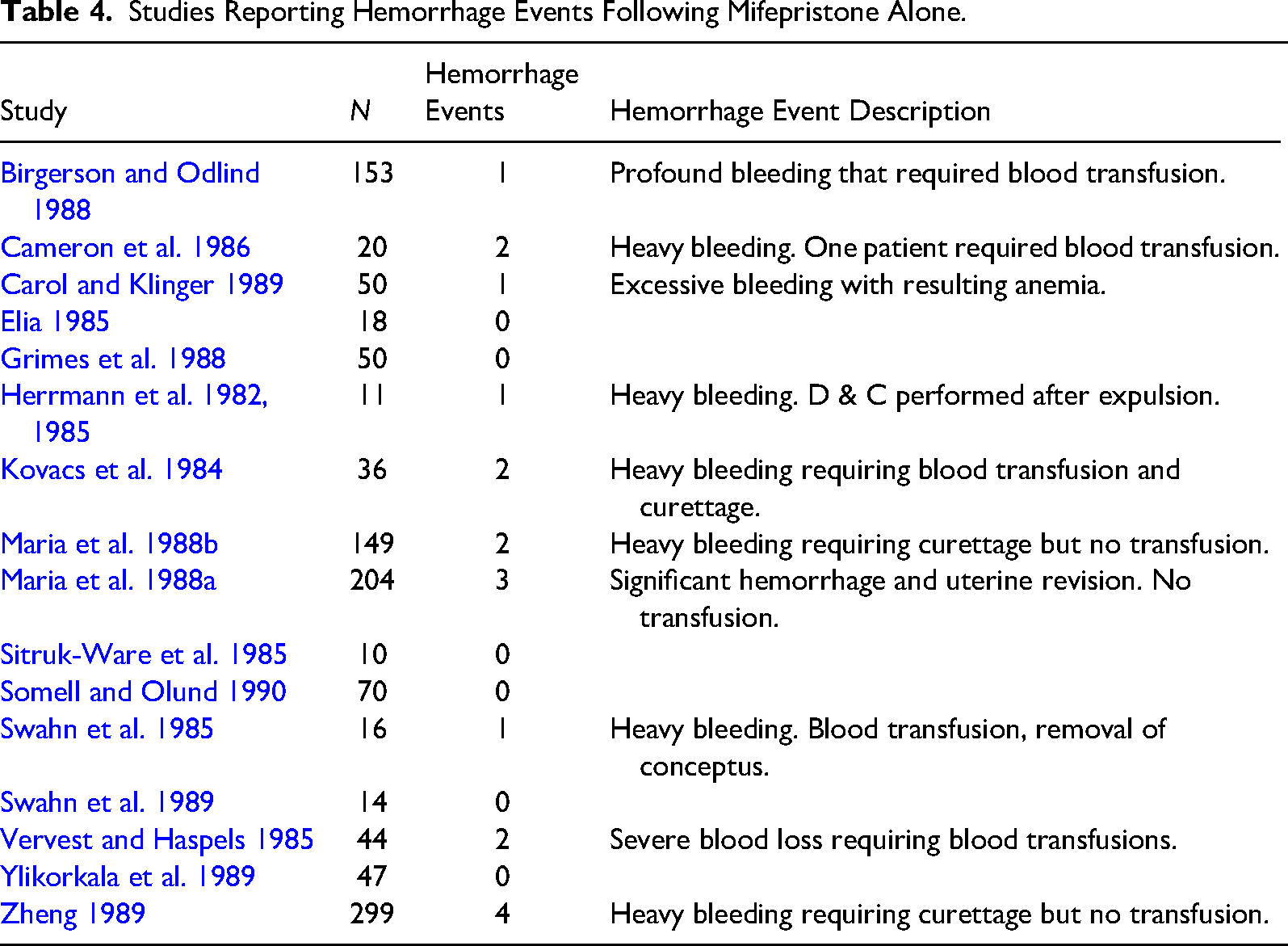
In summary, healthcare professionals have safely used progesterone in reproductive medicine for over fifty years. There is insufficient evidence to establish that mifepristone, followed by progesterone, has a higher risk of hemorrhage than mifepristone, followed by misoprostol.
Conclusion
Mifepristone antagonization with progesterone to avert medication abortion is a safe and effective treatment. The continuing pregnancy rate after ingesting mifepristone alone is ≤25 percent for gestational age ≦49 days. The continuing pregnancy rate after ingesting mifepristone, followed by progesterone, is 65 percent and 69 percent using the delivery regimens intramuscular injection and high-dose oral, respectively. There is no increased maternal or fetal risk from using bioidentical progesterone in early pregnancy.
The conclusion that mifepristone antagonization with progesterone is a safe and effective treatment has implications for medication abortion informed consent. The primary bioethical principle underlying informed consent is patient autonomy. Physicians must safeguard patient autonomy by disclosing medical risks and benefits (AMA 2023). This obligation includes the legal responsibility to disclose safe and effective treatment options that a reasonable person in the patient's position would find important (Nixdorf v. Hicken 1980). Before this treatment was studied, many women changed their minds about their medication abortion (Aultman et al. 2021, 4). It is, therefore, reasonable to assume that women seeking a medication abortion would find the knowledge of this treatment important.
Mifepristone antagonization with progesterone is a time-sensitive treatment. The patient must receive progesterone no later than 72 h after taking mifepristone but before taking misoprostol. Therefore, physicians should not only disclose this treatment to their patients but should do so at the time of informed consent. Failure to inform the patient prior to mifepristone ingestion could cause a delay that leads to fetal demise as the patient searches for a treatment to avert medication abortion.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
