Abstract
Minipigs are used as in vivo endovascular models, particularly in stroke and aneurysm research. However, detailed knowledge of the diameters of forelimb arteries that are commonly used as surrogates for human brain–supplying arteries are lacking. This study aimed to determine the diameters of forelimb and neck arteries in Aachen minipigs and to compare those to the diameters of human cerebral brain–supplying arteries in order to assess the validity of the Aachen minipig as a human intracranial in vivo model. We measured the diameters in the external carotid artery and eight different branches of the subclavian artery in 12 Aachen minipigs using angiographic imaging. Analysed arteries comprised the external carotid artery, axillary artery, brachial artery, subscapular artery first segment, subscapular artery second segment, external thoracic artery, caudal circumflex humeral artery, suprascapular artery and thoracodorsal artery. We compared these diameters to diameters of the following human brain–supplying arteries: terminal internal carotid artery (carotid-T and petrous segment), M1 segment of the middle cerebral artery, M2 segments of the middle cerebral artery, anterior cerebral artery, vertebral artery and basilar artery. Median diameters of porcine forelimb arteries ranged from 1.8 to 4.9 mm, and human brain supplying arteries ranged in diameter from 1.4 to 4.3 mm. Depending on the intended use, this allows porcine forelimb arteries to be selected which are statistically comparable to human brain–supplying vessels. In conclusion, we identified several equivalent arteries of the porcine subclavian branches that are comparable to human brain–supplying arteries. This may help to validate the minipig as a suitable in vivo model for neurovascular experiments.
Introduction
Stroke is the second leading cause of death worldwide.1,2 Europe alone registered around 1.1 million strokes with 440,000 deaths in 2017. 3 Therefore, endovascular treatment techniques for stroke have been continuously developed and are still being improved. In endovascular stroke research, pigs are commonly used as in vivo models for training purposes and development of new therapeutic devices.4 –10 Pigs are gaining more popularity as an animal model because conventional endovascular devices from human medicine can be used due to the similarity of porcine body size and anatomy, as well as haemodynamic and haemostatic parameters.11,12 This results in a high transferability of research results to humans.13 –16 In pigs, branches of the subclavian artery are used because direct endovascular access to the cerebral vessels is not possible due to the rete mirabile – a network of small arteries between the cervical and intracranial cerebral arteries.5,8,17
German Landrace pigs can be used for acute neuroradiological experiments in a weight class of 45–60 kg at five to six months of age.8,13,17 –19 Full-grown German Landrace pigs, however, are too large for the use of standard endovascular material. Moreover, handling these animals can also be difficult and dangerous. 20 This limits the suitability of German Landrace pigs for long-term experiments. Minipigs, on the other hand. represent a suitable animal model for chronic experiments due to their smaller adult body size and stable vessel diameters.21,22
Even though minipigs are considered a reliable in vivo model, the literature is lacking precise data on vessel diameters and the anatomy of minipigs. 23 Siefert et al. assessed the diameters of the main branches of the thoracic and aortic aorta, including the subclavian artery and proximal carotid artery, in juvenile minipigs aged 8–12 weeks, but not in adult animals and without addressing the branches of the subclavian artery. 23 Furthermore, to the best of our knowledge, there are no studies that compare the subclavian and carotid artery diameters in minipigs with arterial diameters of central brain–supplying arteries in humans. Therefore, our goals were to assess diameters of the main branches of the subclavian arteries in minipigs and to compare these diameters to those of human brain–supplying arteries.
Methods
Experimental design and animal groups
Twelve female Aachen minipigs (Gerd Heinrichs, Heinsberg-Karken, Deutschland; mean weight 45.95 ± 5.07 kg; age 17–21 months) were included in this retrospective study.
All animals were part of another endovascular study, with three minipigs receiving a single angiography and nine minipigs receiving two angiographies. The animal experiments were performed in accordance with the German Animal Welfare Law and the EU Directive 2010/63/EU. The experimental protocol was approved by the governmental animal care and use committee (Landesamt für Natur, Umwelt und Verbraucherschutz (LANUV) Nordrhein Westfalen, Recklinghausen, Germany; approval number AZ-81-02.04.2019.A412). The housing conditions corresponded to the requirements of Appendix III of EU Directive 2010/63/EU and the Appendix of the European Agreement of 18 March 1986. Institutional guidelines for animal welfare and experimental implementation were followed.
Animal preparation and procedures
Animal housing, handling, and anaesthesia were performed as previously described. 18 Animals were weighed before angiographies. Subsequent euthanasia at termination of experiments was also carried out as previously described. 18
Image acquisition and measurement of vessel diameters
For angiographies, a short 6-French sheath was placed in the femoral artery and flushed with heparinized saline after induction of anaesthesia. Standard 5- or 6-French catheters and 0.035″ guidewires were used to gain access to the carotid and subclavian arteries. Digital subtraction angiography of the forelimbs and neck was carried out using Iopamidol (Solutrast 300, 300 mg/mL; Bracco Imaging Deutschland GmbH, Konstanz, Germany) as contrast agent. Therefore, a maximum dose of 5 mL per kilogram of bodyweight Iopamidol was administered via the femoral artery.
Images were acquired using either a Siemens (Arcadis Avantic/Arcadis ORBIC; Siemens Healthcare GmbH, Erlangen, Germany) or a Ziehm (Ziehm Vision; Ziehm Imaging GmbH, Nürnberg, Germany) C-arm angiography unit. A standard image management system (IntelliSpace PACS 4.4 Enterprise; Philips GmbH, Eindhoven, The Netherlands) was used to determine the diameters of the arterial vasculature of both forelimbs. Imaging of the external carotid artery (ECA) was limited to one side due to the original experimental set-up. We measured diameters at 1 cm after the respective vessel origin in the following predefined vessel segments: axillary artery (AA), brachial artery (BrA), subscapular artery first segment (SSA), external thoracic artery (ETA), caudal circumflex humeral artery (CHA), suprascapular artery (SPA), subscapular artery second segment (SSA2) and thoracodorsal artery (TA; Figure 1). A careful calibration process of the angiographic images, based on the size of the guide catheter, was performed to increase the accuracy of the measurements.
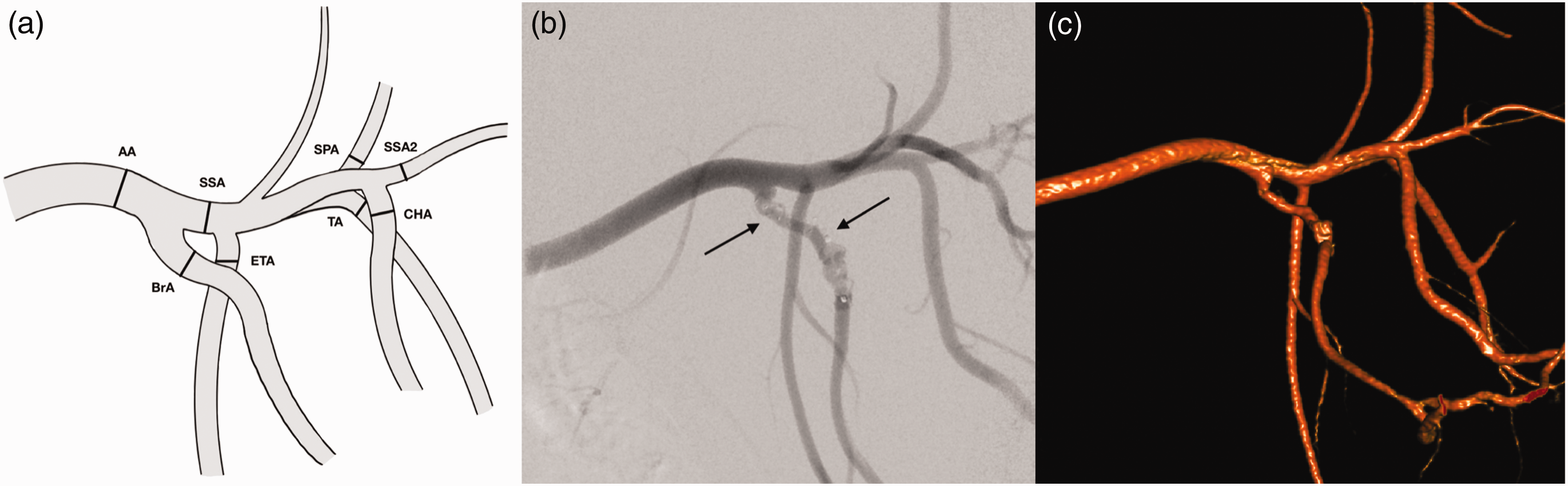
Anatomy of the main branches of the left porcine subclavian artery. (a) Schematic drawing of the main branches of the subclavian artery. Diameters were determined at the measuring points (black lines) in the axillary artery (AA), brachial artery (BrA), subscapular artery first segment (SSA), external thoracic artery (ETA), caudal circumflex humeral artery (CHA), suprascapular artery (SPA), subscapular artery second segment (SSA2) and thoracodorsal artery (TA) and (b) Digital subtraction angiography of the left axillar artery with endovascular devices (black arrows) of the original experiment. (c) Angiography-based three-dimensional reconstruction of the left subclavian artery.
Human brain–supplying artery diameters were assessed using computed tomography angiography and digital subtraction angiography of 100 acute stroke patients. The consecutive stroke collective includes 45 female patients and 55 male patients aged 3–105 years, with an average age of 71 years. This retrospective analysis was approved by the ethics board of our institution. We measured at the following predefined vessel segments: internal carotid artery (carotid-T and petrous segment), middle cerebral artery (MCA, M1 segment), M2 segments of the MCA (M2 superior trunk, M2 inferior trunk), anterior cerebral artery (ACA, A1 segment), vertebral artery (VA intracranial, terminal part of V4 segment) and basilar artery (BA, proximal segment, below basilar top).
Statistical analysis
The mean and standard deviation as well as median and interquartile range (IQR) were calculated for each diameter. Data were tested for normality with a Shapiro–Wilk test.
Mann–Whitney U-tests were performed for analysing statistical differences between right and left subclavian arteries in minipigs and statistical differences between porcine and human arterial diameters. Bonferroni correction was used to address multiple comparisons. A corrected p-value (p < 0.0006) was calculated as significant. SPSS Statistics for Windows v28 was used for statistical analyses (IBM Corp., Armonk, NY).
Results
The diameter of eight different arterial branches of each subclavian artery as well as the ECA diameter were measured in 12 minipigs, resulting in 17 measurements per minipig. In our sample of 100 stroke patients, we measured 10 segments of brain-supplying vessels each.
The diameters of the measured artery sections are presented in Table 1 and Figure 2. The largest measured section in the minipig was the AA (4.9 mm), and the smallest was the SSA2 (1.8 mm). There were no significant differences between the subclavian branches of the left and right side in minipigs. In humans, the petrous segment of the ICA had the largest diameter (4.3 mm), and the M2 segment of the MCA had the smallest diameter (1.4 mm). All but the ICA, MCA, M2 segments, ACA, VA and BrA diameters were normally distributed.
Arterial diameters of human intracerebral arteries and porcine subclavian arterial branches according to quantitative vascular angiography.
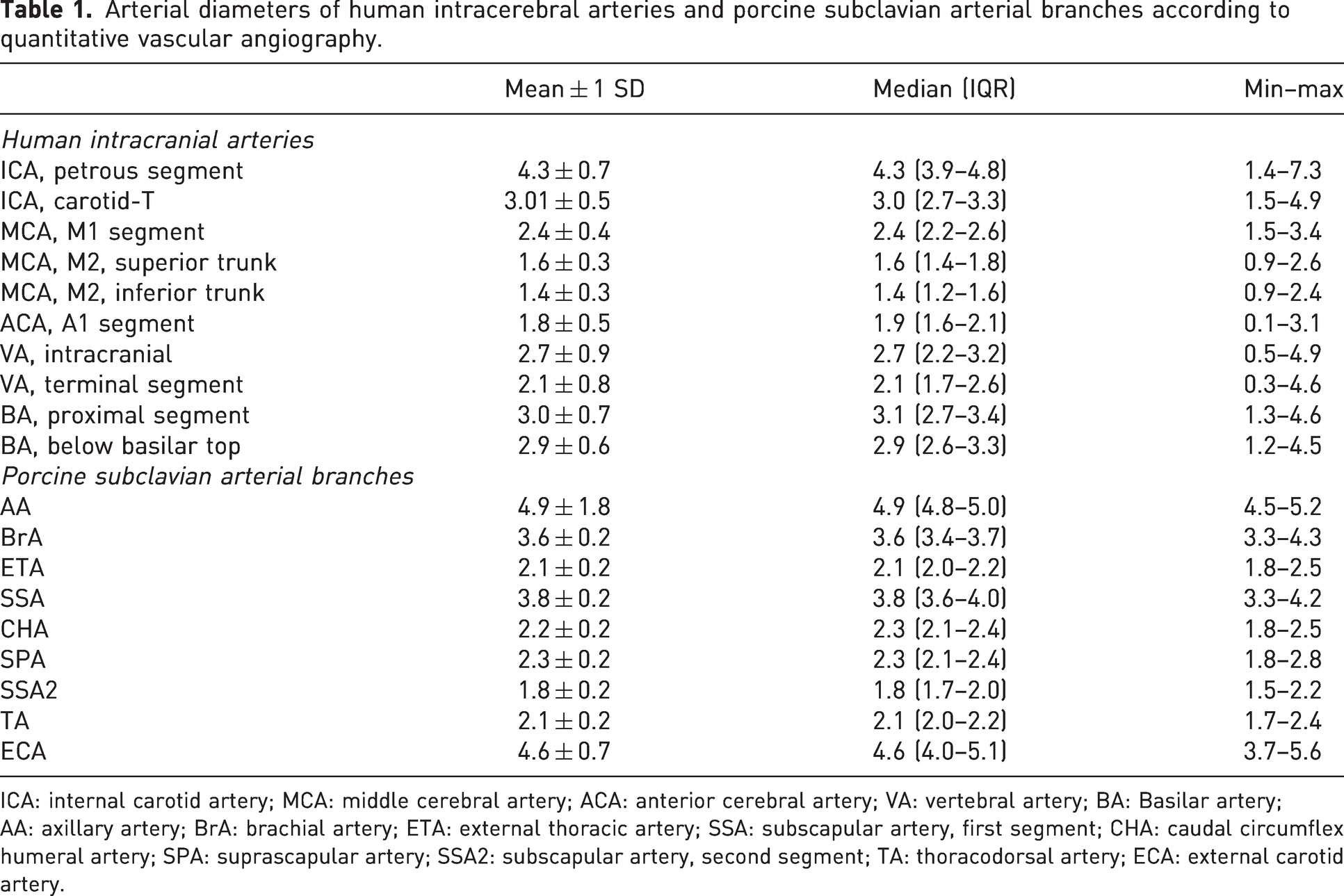
ICA: internal carotid artery; MCA: middle cerebral artery; ACA: anterior cerebral artery; VA: vertebral artery; BA: Basilar artery; AA: axillary artery; BrA: brachial artery; ETA: external thoracic artery; SSA: subscapular artery, first segment; CHA: caudal circumflex humeral artery; SPA: suprascapular artery; SSA2: subscapular artery, second segment; TA: thoracodorsal artery; ECA: external carotid artery.
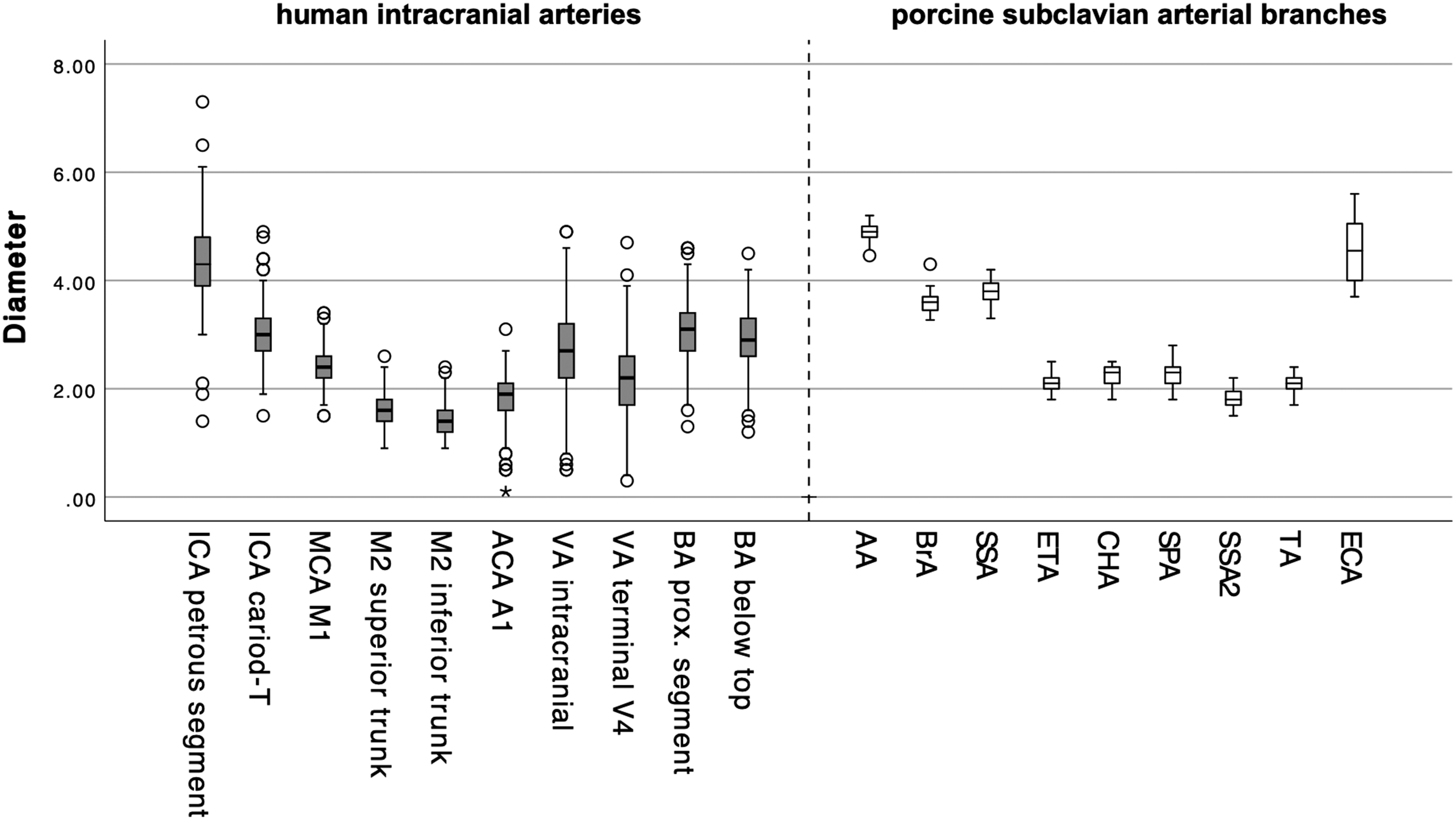
Box-and-whisker plot of human intracranial (grey) and porcine subclavian (white) arterial diameters (in millimetres): internal carotid artery (ICA, carotid-T and petrous segment), middle cerebral artery (MCA, M1 segment), M2 segments of the middle cerebral artery (M2 superior trunk, M2 inferior trunk), anterior cerebral artery (ACA, A1 segment), vertebral artery (VA, intracranial, terminal V4 segment of VA), basilar artery (BA, proximal segment, below basilar top), AA, BrA, SSA, ETA, CHA, SPA, SSA2, TA and external carotid artery (ECA).
The diameter of the terminal VA was comparable to five arteries in minipigs: the ETA (p = 0.553), SPA (p = 0.438), TA (p = 0.427), CHA (p = 0.613) and SSA2 (p = 0.002; Table 2).
Presentation of the Mann–Whitney U-test results (p-values) for differences of artery diameters between porcine subclavian arterial branches and one of the selected human intracerebral arteries.
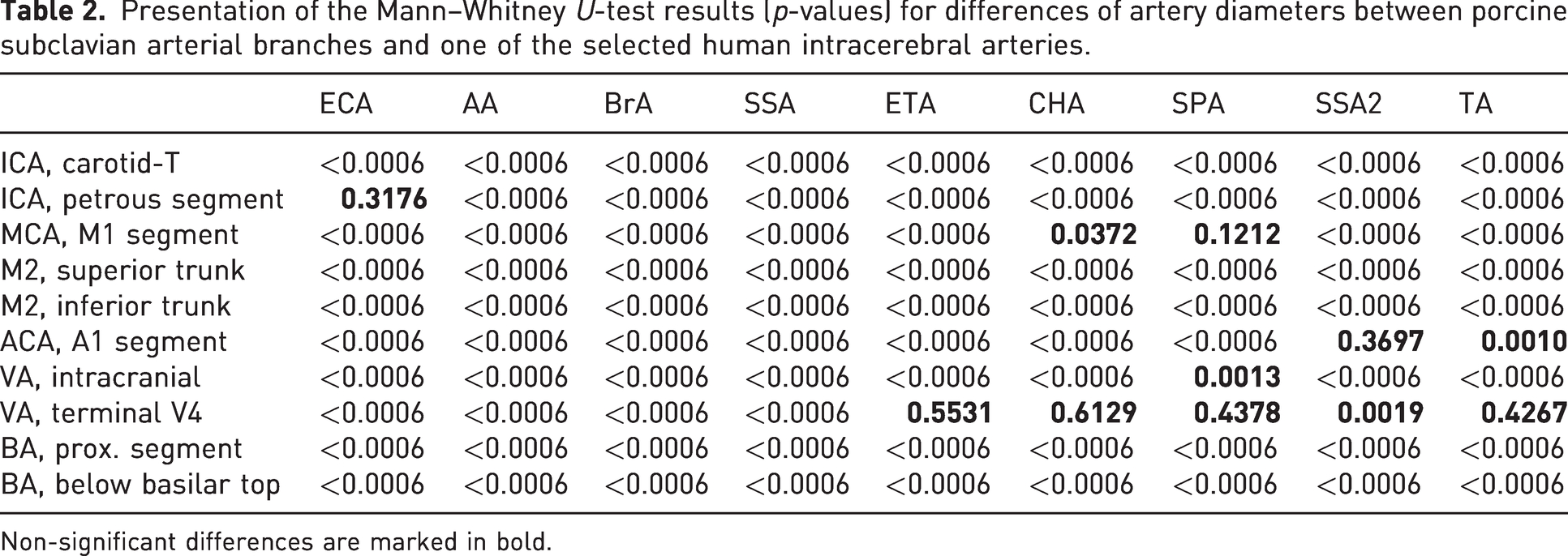
Non-significant differences are marked in bold.
The diameter of the A1 segment of the ACA was comparable to the SSA2 (p = 0.370) and TA (p = 0.001). The diameter of the M1 segment of the MCA was also comparable to the SPA (p = 0.121) and CHA (p = 0.037; Table 2). Furthermore, the intracranial segment of the VA did not differ significantly in diameter from the SPA (p = 0.0013), and likewise the petrous segment of the human ICA from the ECA in minipigs (p = 0.3177; Table 2).
Discussion
One finding of our study is that vessel diameters of the left and right forelimb did not differ significantly. This implies that both limbs can be used equally in studies and that both limbs can be used for comparisons of interventions on one side and control on the other.
As was to be expected, the diameters of the porcine forelimb arteries cover a wide range from 1.8 to 4.9 mm. This compares well to human brain–supplying arteries, which range from 1.4 to 4.3 mm. Consequently, it is possible to select a porcine forelimb artery which matches the human vessel that is to be used as the in vivo model. For instance, the AA and ECA are not suitable as vascular models for small intracerebral vessels, but they serve well as a model for the cervical or petrous segment of the human ICA. In addition, it is possible to select more distal or more proximal vessel segments in certain arteries that we have included in our analysis to optimise comparability further.
Comparison of our data with the literature would be interesting, this but is not yet possible, as other authors have not measured the diameters of the subclavian arterial branches of Aachen minipigs. Mathern et al. investigated vessel diameters of German Landrace pigs and compared those with human brain–supplying arteries. 19 Another study dealing with porcine and human-vessel diameters was based on data from three Landrace pigs. 14 A particular characteristic of Landrace pigs is that they grow rapidly, reaching bodyweights of >300 kg after approximately one year, whereas full-grown minipigs have a stable weight of approximately 40–60 kg. Landrace pigs are suitable for neurointerventional endovascular research when they are juvenile, at a weight range of approximately 50–65 kg. Their rapid weight gain, however, limits their suitability for long-term experiments. 19 As outlined above, diameters of subclavian branches have been reported for Landrace pigs with bodyweights comparable to the minipigs we included in our study. However, since these measures were obtained from juvenile Landrace pigs, they cannot be extrapolated to full-grown minipigs, since the growth of vessels and increases in bodyweight do not necessarily correlate linearly. Therefore, our data provide basis for future use of minipigs in endovascular research.
In conclusion, we identified several equivalent arteries of the subclavian branches of Aachen minipigs that are comparable to the human brain–supplying arteries. This may help to validate the minipig as a suitable in vivo model for neurovascular experiments.
Limitations
A limitation of our study was the provision of the angiographic images by a superordinate aneurysm project. Images were not always taken from the same angle, and the ECA was not recorded on both sides. However, in terms of the 3Rs (Replacement, Reduction and Refinement) according to Russel and Burch, 24 preliminary results could be drawn regarding the comparability of vessel diameters between humans and minipigs, which is why this limitation seems acceptable. A degree of measurement inaccuracy is another limitation of our study, especially since it deals with small millimetric arteries.
Footnotes
Declaration of conflicting interests
Martin Wiesmann has the following disclosures: consultancy for Stryker; payment for lectures from Bracco, Medtronic, Siemens and Stryker; educational presentations for Bracco, Codman, Medtronic, Phenox and Siemen; and grants for research projects or educational exhibits from Ab Medica, Acandis, Bracco Imaging, Cerenovus, Kaneka Pharmaceuticals, Medtronic, Mentice AB, Microvention, Phenox, Siemens Healthcare and Stryker Neurovascular. Omid Nikoubashman has the following disclosures: grants from Stryker and payment for lectures from Stryker and Phenox. Hani Ridwan has the following disclosures: consultancy for ThrombX Medical, Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was funded by EFRE NRW 2014-2020 (Leitmarkt.Agentur NRW) through the project IANIS – Intraaneurysmales Implantat mit flussmodulierenden Eigenschaften (funding ID: EFRE-0801324, LS-2-01-012e).
