Abstract
Rat models employing cranial implants are increasingly employed to facilitate neural stimulation and recording in freely moving animals. Due to possible damage to wound, implant or attached devices, rats with cranial implants are traditionally housed singly, and little information is available on group- or pair-housing. Here we describe a protocol for pair-housing rats following cranial implant surgery and describe our experience with pair-housing during post-surgical recovery and up to 16 weeks following surgery.
Thirty-six adult Wistar rats of both sexes were implanted with deep brain stimulation electrodes. Ten rats were equipped with an additional wireless headstage. Rats were housed in stable pairs before surgery and re-introduced 0–18 h post-surgery. Rat grimace scores did not indicate pain after conclusion of the analgesia protocol, physiological parameters were in the normal range three days post-surgery and weight loss did not exceed 10%. Rats with a cement cap only were pair-housed continuously without damage to the headcap. Rats carrying an additional fragile headstage had to be separated during lights-off periods to prevent headstage damage but could be pair-housed during lights-on periods.
Pair-housing is a feasible and effective method to facilitate the rats’ need for social companionship following cranial implant surgery.
Introduction
Wireless telemetry systems and cranial implants in rodents for neural recording, stimulation, optogenetics, intra-cranial catheterisation and combinations thereof are increasingly used in experimental protocols in neuroscience. These systems typically consist of a cranial implant embedded in dental acrylic cement, held in place by bone screws, with a neurostimulator or amplifier in the form of a headstage, 1 rucksack 2 or implant under the skin, 3 temporarily or continuously attached.
In these types of experiments, individual housing of animals remains standard practice due to the risk of damage to the device or wound during interactions between animals.4,5 However, social isolation is long known to reduce animal welfare,6,7 affecting stress hormone levels, 8 anxiety, 9 social interaction, 10 brain metabolism and structure, 11 blood pressure and heart rate 12 and food intake. 13 Group-housing has been shown to prevent these detrimental physiological and behavioural effects of social isolation and increase animal welfare.
Although research studies often do not report housing conditions in rodents, pair-housing is possible in non-human primates.14–16 In addition, two groups have reported pair-housing of rodents with cranial implants. Pinnell et al. presented a headstage purposefully covered with a protective cap to allow pair-housing and behavioural assessment in the water.17,18 Koletar et al. reported co-housing of rats after creation of a cranial window for two-photon fluorescence microscopy 19 . Neither reported damage to the device or wound. In addition, Koletar et al. reported improved handling in group-housed rats compared with individually housed rats. Norecopa, Norway’s National Consensus Platform, have published limited descriptions of experiences with pair-housing of rodents with cranial implants while highlighting the lack of literature on the topic. 20
The aim of this article is to present our protocol for pair-housing of rats with cranial implants and report our experience with the protocol in the period immediately post-surgery and up to 16 weeks after surgery. The housing protocol was developed in the context of a project to examine the effect of chronic deep brain stimulation on the electrode tissue interface and surrounding brain parenchyma. Two different types of head-mounted devices were used: (1) a basic cement cap and (2) a larger headstage continuously connected to the top of the cement cap for deep brain stimulation of the basal ganglia (S-series, Triangle Biosystems).
Methods
Animals
Thirty-six adult male (n = 34) and female (n = 2) naïve Wistar rats (weight: 404 g ± 70 g (SD); age: 9.8 weeks ± 2.6 weeks, bred in the local facility using Charles River breeders) that had undergone successful cranial implant surgery for deep brain stimulation in the time period 2017–2021. The animals were divided into two groups: 26 rats had a cranial implant secured by a cement cap and 10 had an additional headstage attached throughout the protocol. No control group was included because pair-housing was required by the licensing body. Welfare assessments were conducted twice daily for the first four days and three times per week thereafter by the same researcher at the same time in no set order. The rat grimace score, 21 ranging from 0 (no pain) to 2 (severe pain) depending on four facial features, was rated by one observer in real-time. A facility specific welfare score including general appearance, behaviour, respiration, weight, state of the surgical site and adverse effects was also filled, scoring each point from 0 (normal) to 3 (see Supplemental material online). Training was provided by the facility beforehand. Rats were assessed in no set order in the home cage. After eight or 16 weeks rats were perfused with formalin and brains harvested for histological assessment. All experiments were approved by the UCD Animal Research Ethics Committee (AREC 17-22/20-02) and licensed by the Health Product Regulatory Authority Ireland (AE18982-P122/P190).
The primary welfare measures reported for each animal here are adverse effects, welfare scores, rat grimace scores and weight development.
Cranial implant surgery
Stereotaxic surgery was performed under inhalation anaesthesia (induction: 4.5% isoflurane (Iso-Vet, Chanelle Pharma, Loughrea, Ireland) in 4 l/min oxygen; maintenance: 1.2–1.8% isoflurane in 1 l/min oxygen) in a procedure room. In addition, local anaesthesia at the skin incision (maximum 0.05 ml 0.5% lidocaine, diluted from Lidocaine 1%, Hameln Pharmaceuticals Ltd, Gloucester, UK) and anaesthetic cream at the non-rupture ear bars (Emla 5% cream, AstraZeneca, Cambridge, UK) were used. The depth of anaesthesia was monitored continuously using the pedal withdrawal reflex and corneal reflex. Surgical practice included temperature control using a heating blanket, aseptically prepared surgical field, subcutaneous (s.c.) antibiosis given pre- and four days post-op (metronidazole (20 mg/kg, B. Braun Medical Ltd, Dublin, Ireland) and gentamicin (6 mg/kg, diluted from Gentaject 10%, Alpha Med Ltd, Fermoy, Ireland) q.d.), tear replacement ointment (Vidisic, Dr. Gerhard Mann, Chem.-pharm. Fabrik GmbH, Berlin, Germany) and intra-operative fluid. Buprenorphine (0.05 mg/kg s.c., Buprenodale, Dechra, Shrewsbury, UK) was given as analgesia 1 h before surgery and post-operatively two to four times per day for three to four days.
All rats underwent craniotomy, electrode insertion into the basal ganglia area and formation of a cement cap (Dentalon plus, Heraeus Kulzer GmbH, Hanau, Germany) held by four bone screws (1.59 mm, Stoelting). The skin was re-aligned cranially and caudally using an intra-dermal suture (Vycyl 4-0, Ethicon Inc., Somerville, USA). Ten rats had a wireless and inductively powered headstage for deep brain stimulation (S-series, Triangle BioSystems Inc., Durham, USA) continuously attached. In the other 26 rats the connector in the cement cap was accessed for stimulation or recording for short time periods.
Animal husbandry
The general housing conditions in a specific pathogen free facility following the FELASA recommendations 22 included a 12/12 h light/dark cycle in a temperature (21.8°C ± 0.3°C) and humidity (49% ± 4.1%) controlled environment and access to water and standard rodent diet (209/18 Tekland irradiated diet, Envigo, Indianapolis, USA) ad libitum. Individually ventilated rat cages (size: 34 cm × 46 cm × 29 cm) were adapted for housing. The metal food dispenser and water bottle holder were removed to increase headspace and remove possible sites for entanglement or headstage damage. For the same reason, initially no other enrichment was supplied. Hammocks were supplied for the last 10 rats to undergo surgery. Cages contained woodchip bedding (Gold Chip Original, LBS, London, UK), paper shreds (Enviro-Dri, LBS) as nesting material and enrichment in the form of wooden balls or sticks. Rats were fed on the ground and water bottles fed through the ceiling. Cages were cleaned two or three times per week.
Rats were housed in stable pairs for a minimum of one week before recovery surgery. If surgeries took place on the same day, rats were placed with their former cage-mate immediately after recovery. If surgeries were on consecutive days, rats were housed singly for up to 18 h to recover from the procedure and then re-introduced to their former cage-mate. A heating blanket was provided under half the cage for 18 h post-surgery. While continuous pair-housing of rats with a cement cap caused little to no damage to the cement cap, gnaw marks on the headstage penetrating the protective cover of one rat that had a headstage attached were found after the lights-off period. Therefore, to prevent damage to the headstage, rats in the headstage group (five cages) were separated in the same cage during the lights-off period by a grid partition in the middle of the cage. 23 This allowed olfactory, visual and auditory cues. Rats were housed together during the lights-on period.
Statistics
Descriptive statistics and graph creation were performed in Prism 5 (GraphPad, San Diego, USA). The mean and standard deviation for both groups were calculated based on individual rats.
Results and discussion
Examples of the pair-housing with separation during the lights-off period and enrichment are presented in Figure 1(d) to (g). The rat grimace score was under 0.5 after three days post-surgery and the welfare score was under 2 from three days post-surgery (Figure 1(a) and (b)), indicating mild pain and good recovery post-operatively with little to no physiological changes. Rats’ weight loss after surgery was 5.8% ± 3.9% seven days post-surgery and returned to the weight at surgery 12–14 days post-surgery, continuing to increase thereafter (Figure 1(c)). When comparing between male and female rats, welfare scores developed similarly post-operatively, but weight loss was less in female rats (–0.7%, –2.8% at seven days post-operative) while weight gain was minimal at eight weeks (+13.6%, +6.8%). While it is not possible to draw conclusions regarding outcomes compared with singly-housed animals due to the lack of a control group, Pinnell et al. reported lower weight gain in pair-housed rats compared with singly-housed rats in a related cranial study with headstages. 17 It is widely accepted that group-housing provides animal welfare benefits, especially in regard to behaviour and stress6,7,11–13 and is therefore recommended by Norecopa. 20 The addition of hammocks to the environmental enrichment did not result in headstage loss in any of the rats housed in those cages (Figure 1(g)).
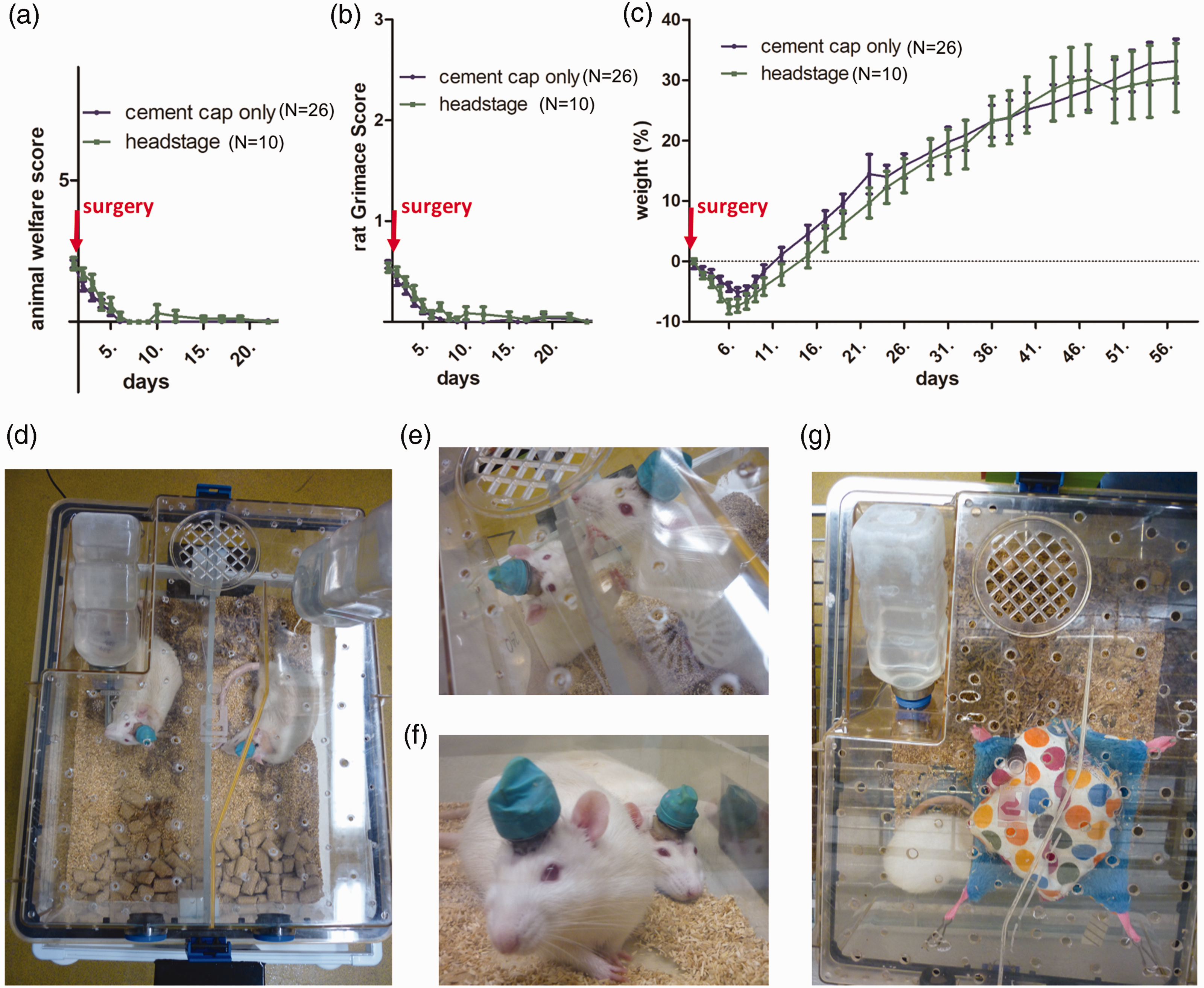
Mean ± SEM of rat welfare score, rat grimace score and weight gain/loss in relation to weight at surgery post cranial surgery; cement cap only (n = 26); headstage (n = 10) ((a) to (c)). Separation (d). Interaction during temporary separation (e). Pair of rats during unseparated period (f) and Housing with enrichment and hammock for two rats (only one visible in picture) (g).
Only mild adverse events occurred as a result of pair-housing (Table 1). These included minor headstage damage and two incidents of biting that resolved within a maximum of two days occurring in <10% of rats (Table 1). No animals were excluded as a result of pair-housing, which meets the principle of reduction. In all three animals that lost the headstage, softening of the bone around at least one screw was observed post-mortem, linking the loss to local inflammation but no cracked bones that would indicate mechanical dislodgement. The number of animals (4/36) that lost the headstage in this cohort was in line with previous outcomes in the facility (1/5–1/8 with single-housing). In summary, observed side effects were mild and headstage loss was not attributed to pair-housing and thus pair-housing represents a refinement in the husbandry of rats with cranial implants.
Summary of adverse events in pair-housed rats with cranial implants.

In the case of fragile headstages, the protocol was further refined to allow temporary pair-housing while protecting the equipment from detrimental damage. For long-term continuous housing of animals in pairs, however, sturdy headstages or devices that can withstand gnawing are needed.17,18
Finally, with pair- or group-housing, behavioural data from rats that are housed together may not be statistically independent as behaviour is directly impacted by the presence of a conspecific. This can be controlled for through the use of mixed models or similar methods to model the effect of cage or group.
Conclusions
We report our experience with a protocol for pair-housing of rats with cranial implants including modifications for fragile devices. Rats showed good recovery after surgery and no additional moderate or severe adverse events were observed that could be attributed to pair-housing. In conclusion, to increase animal welfare, rats with cranial implants can be housed in pairs and safe enrichment can be provided which would counteract behavioural and physiological changes associated with individual housing.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772221122099 - Supplemental material for Feasibility of pair-housing of rats after cranial implant surgery
Supplemental material, sj-pdf-1-lan-10.1177_00236772221122099 for Feasibility of pair-housing of rats after cranial implant surgery by Judith Evers, Karthik Sridhar and Madeleine Lowery in Laboratory Animals
Footnotes
Acknowledgements
We would like to acknowledge Emer Conroy for her input on the drafting of the manuscript.
Data availability
Data presented in this short report is available on reasonable request from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science Foundation Ireland (SFI) and the European Regional Development Fund (grant number 13/RC/2073) and the European Research Council (grant number ERC-2014-CoG-646923).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
