Abstract
The objective of this study was to describe prolonged surgical anaesthesia and recovery in fire salamanders (Salamandra salamandra) using tricaine methanesulfonate (MS-222). A total of 14 salamanders were anaesthetised for electromyography wire implantation. Sodium bicarbonate buffered solutions (0.5–4 g l–1) of MS-222 were prepared (adjusted to pH 7.0). Anaesthesia was induced by partial immersion in pre-oxygenated 3 g l–1 solution for 20 min. Buprenorphine (0.5 mg kg–1) was administered subcutaneously. During microsurgery, heart rate (HR), solution pH and temperature were recorded. Reflectance pulse oximeter (SpO2) (Masimo Rad-57) was recorded in two salamanders. Anaesthetic plane and MS-222 pH stability (pH 7.6) were maintained by renewing administration of oxygenated MS-222 solution (0.5–3 g l–1) onto swabs that partially covered the body. Recovery started at the end of surgery (MS-222 0 g l–1). Postoperatively, salamanders were given oral meloxicam (0.2 mg kg–1). Mean time for loss of righting reflex during induction was 13.7 ± 2.2 min. Duration of anaesthesia and time to recovery were 111 ± 24.2 and 31 ± 10.3 min, respectively. Due to complications, two salamanders did not recover. Baseline HR was 67.4 ± 34.5 beats/min, and it decreased significantly until recovery (p ≤ 0.0001). In two salamanders, baseline SpO2 was 85.5% ± 14.5, SpO2 during surgery was 61% ± 6.4, improving to 80.5% ± 2.1 on recovery.
In conclusion, prolonged recovery anaesthesia is achievable with MS-222 dilutions in salamander. Reflectance SpO2 could prove valuable during immersion anaesthesia.
Introduction
Amphibians are maintained in captivity as pets, in research setting and in zoos. Due to their unique anatomical and physiological characteristics they are increasingly subjects of research. 1
Fire salamanders, being terrestrial use a two-stroke buccal pump and breath through their nares. 2 During anaesthesia though, the pulmonary respiration ceases and they switch to transdermal respiration. 3 This is sufficient to prevent clinical hypoxia during anaesthesia. 4 however, the skin needs to remain moist to allow this process. In order to maintain an appropriate saturation level, oxygen is bubbled in the solutions used for immersion anaesthesia. 5 Due to the cessation of ventilation, heart rate (HR) is the most useful variable that can be easily monitored. 6
A number of anaesthetic options exist for amphibians, with differing success rates and ranges in physiological effects. The three main categories are injectable, volatile and immersion anaesthesia. Injectable drugs have narrow margins of safety and prolonged recovery times. White’s tree frogs (Ranoidea caerulea) anaesthetized with propofol can take up to16 hours to completely recover from a deep anaesthetic plane 7 whilst another study achieved light but non-surgical anaesthesia in leopard frogs (Rana pipiens). 8 An alternative is ketamine; induction and recovery times and anaesthetic depth vary considerably between species 3 and its use has been reported in salamander species. Alfaxalone, a neurosteroid that acts on the γ-aminobutiric acid (GABAA) receptors, has been trialled in frogs, resulting in immobilisation after intramuscular injection. However, a surgical plane of anaesthesia was not achieved. 9 With regards to volatile anaesthesia, isoflurane can be considered as a viable option, taking care to avoid desiccation from high oxygen flow rates through a face mask. 1 Intubation can be used with species with lungs, but the trachea is very short in most 10 , which can cause difficulty for surgical positioning.
The third category of immersion includes, but is not limited to, clove oil, benzocaine or tricaine mesylate (MS-222). Lafortune et al. 8 used clove oil to induce leopard frogs in 15 minutes but concluded anaesthetic duration was widely variable, whilst the extent of analgesia clove oil provides is unknown 11 and its use in surgical procedures remains questionable. Benzocaine has been shown to cause rapid induction but has narrower margins of safety. 12 Generally, the preferred method for amphibian anaesthesia is immersion with MS-222.
MS-222, as used in this case series, is the most commonly used agent in frogs. 13 It is a water-soluble benzocaine derivative which blocks sodium channels. 6 When used for immersion, it depresses the central nervous system, producing anaesthesia. It is formulated as a white powder and the preparation is crucial for effective use as it forms an acidic solution when made, resulting in long recovery times and irritation to amphibian skin. 12 It must therefore be buffered with sodium bicarbonate to minimise these effects. It is a favoured choice of anaesthetic for many due to its ease of maintenance and administration. 1 Guenette and Lair 13 concluded MS-222 to be the most effective agent to induce a surgical plane of anaesthesia when compared to benzocaine, clove oil and Emla® (lidocaine and prilocaine). It concluded MS-222 had, compared to the other agents, more reliable anaesthesia and that was corroborated with by Wojick et al. 14 .
MS-222 has been reported to vary between species, individual traits and life history, 15 making it difficult to establish a generalised protocol, especially for long and invasive procedures.
This study demonstrates the feasibility of using MS-222 at different concentrations for prolonged anaesthesia and recovery in fire salamanders. As there is limited literature on prolonged surgical anaesthesia in salamanders, we are hoping that this study could be of benefit for amphibian anaesthetic management.
Animals
A group of 14 fire salamanders (Salamandra salamandra) weighing between 15–30 g were anaesthetised for implantation of electromyography wires around muscles of the thoracic or pelvic limbs for a biochemical study (Pierce et al., 2020). The number of salamanders anaesthetised was decided by the statistical needs of the biomechanical study. One of the salamanders was used as a pilot to trial the anaesthetic technique and was not included in the original biochemical study. Animals were housed separately in moist and cool environments. Food and water were offered ad libitum. Water was regularly changed and dechlorinated, while animals’ skin was inspected daily for abrasions and any sign of parasites or infection.
All procedures in this study were approved by the **** Animal Welfare and Ethics Review Board (approval number ****) and conducted under a Home Office project license following the Animals (Scientific Procedures) Act of 1986 (******).
Materials and methods
Anaesthesia was induced with tricaine methanesulfonate (Tricaine-S MS-222, Western Chemical. Inc., WA, USA). A trial anaesthesia was attempted with a 1 g L−1 but failed to achieve surgical anaesthesia, with the salamander recovering uneventfully. Subsequently, infusions of different concentrations were prepared by adding equivalent amounts of MS-222 in dechlorinated water to achieve concentrations of 0.5 g L−1, 1 g L−1, 2 g L−1, 3 g L−1 and 4 g L−1 respectively. Amounts of MS-222 added were measured with high accuracy laboratory scales. Infusions were further homogenised with a magnetic stirrer. The pHs of the solutions were measured with a laboratory pH meter (RS Component Ltd, Watford, UK). A two-point calibration of the pH meter was performed before starting the measurements.
MS-222 anaesthetic solutions were buffered for each reconstitution, as the pH can be significantly acidotic, varying with time, depending on the water hardness. Sodium bicarbonate (BHD Laboratory Supplies, UK) was added as the buffer agent. Amounts needed to buffer each of the MS-222 concentrations to a target pH of 7.0 are reported in Table 1 in the appendix. The sodium bicarbonate added to make each solution strength is represented in Figure 1.
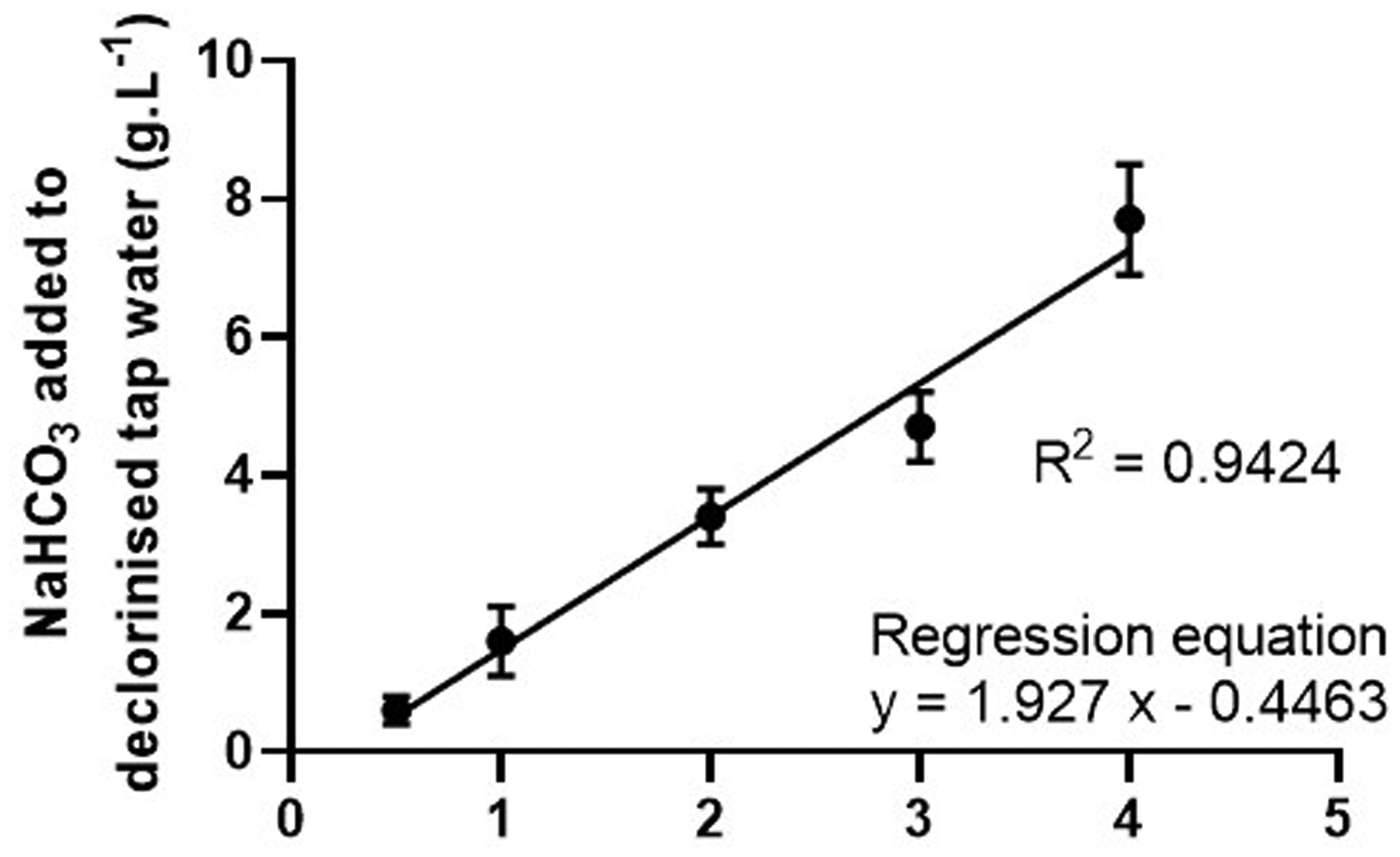
Mean amounts (g/L) of sodium bicarbonate (+/− standard deviation) used to buffer dechlorinated tap water with MS-222 to yield concentrations 0.5–4 g/L with a target pH of 7.0 (n = 14 occasion days).
Before each anaesthetic, respiratory rate was taken by observation and when feasible baseline HRs were acquired. For induction, each of the 13 salamanders were placed in a transparent plastic bag containing 100–150 mL of 3 g L−1 MS-222 solution. Their head was kept above the solution, and the solution was supplemented with oxygen bubbled through a metal tip attached to a T-piece breathing system and an anaesthetic machine at a flow of 2 L min−1. Once loss of righting reflex occurred, salamanders were placed in a petri dish (15 cm diameter) containing a shallow layer of the same concentration of MS-222 solution (3 g L−1). They were placed in left lateral recumbency and their bodies were partially covered with two moist gauze squares (5 × 5 cm). Oxygen was supplemented in the MS-222 by immersing a metal tip, during the entire procedure at the same flow rate. Animals were constantly kept moist by slowly pouring, with a 20 mL syringe, a few millilitres of the petri dish solution across their body with special care to keep the entire length of the body moist including the tail. The surgical procedures undergone are reported elsewhere; 16 briefly electrodes were placed in either thoracic or pelvic limb muscles. Regarding analgesia, buprenorphine 0.5 mg kg−1 (Vetergesic, Alstoe, UK) was administered intramuscularly (proximal pelvic limb) during induction. A volume of 0.1 mL of lidocaine 2% was used as a splash block in the area of the surgical incision before the beginning of the surgery.
HR was monitored every 5 minutes by placing the Doppler probe (model 811-B, Parks Medical Electronics, NE, USA) ventrally on the precordial area. Measurement of the solution’s pH was also performed at the same time intervals. If the pH rose above 7.6 (likely due to the buffering capacity of the skin and mucous), the solution was renewed with the same or lower concentration, depending on the phase of the surgery. This was normally necessary at 10-minute intervals. Temperature was monitored with a temperature probe placed in the petri dish and maintained between 20°C to 24°C. In the last two salamanders of the series, a Masimo pulse oximeter using a LNOP TF-1 Transflectance Forehead Sensor (RAD-57, Masimo, UK) was used to monitor oxygen saturation. The probe was placed on the precordial area alternating with Doppler measurements so a concordance of the acquired HR with the methods could be possible.
The concentration of MS-222 solution was decreased gradually from 3 g L−1 to 0.5 g L−1 during the procedure to maintain an appropriate plane of anaesthesia and optimal immobility conditions for surgical completion.
Once surgery was completed, salamanders were placed in a petri dish, rinsed with dechlorinated water (0 g L−1 MS-222) applied with a 20 mL syringe and monitored until their full recovery. Ocular reflex was the first sign of recovery followed by resumption of buccal cavity expansion and head movement. Once mobile and alert, they were moved to their normal housing environment.
Salamanders were administered 0.2 mg kg−1 of meloxicam orally postoperatively (Metacam, Boehringer Ingelheim Animal Health UK Ltd, UK).
Salamanders were euthanised 48 hours after completion of the locomotion and treadmill studies, according to the research protocol, by using 8 g L−1 buffered MS-222 solution and were pithed through the base of their skull.
All data and anaesthesia records are available through the corresponding author and are kept and registered in the relevant research facility.
Statistical Analyses
Change in HR from the baseline taken prior to induction was statistically analysed using a mixed effect model. Due to the heterogeneity of variance (Levene’s test, p < 0.0001), HR data was logarithmically transformed first. Dunnett's multiple comparisons test was used for post-hoc HR comparison at different times. Additionally, correlation analyses were performed to investigate relationships between length of anaesthesia and time to recovery as well as between solution pH and HR. Significance was set to p < 0.05 for all statistical tests in GraphPad Prism 8 (GraphPad Software, CA, USA).
Results
The anaesthetic depth was described as adequate once the righting reflex and the ocular reflex were absent. In deeper anaesthetic planes, respiration occurred cutaneously and the withdrawal reflex elicited by toe pinch was abolished.
Eleven salamanders recovered from the surgical procedure for the EMG wires implantation in either the thoracic or pelvic limb. They underwent treadmill trials in the following 48 hours in order to evaluate their locomotor function for research purposes. Data collection was deemed successful which could suggest the adequacy of the analgesia level provided. Two salamanders did not recover, yielding an overall survival rate of 85%. One salamander that did not recover experienced significant haemorrhage during surgery. The circulating blood volume of the salamander, weighing 30 g was 3 ml. The estimated blood loss was more than 50% of the total blood volume. However, due to the presence of saline flush in the surgical field, it was not possible to calculate accurately. The second one experienced a less profound haemorrhage, however still significant and maintained a heartbeat 24 h after anaesthesia but never started moving again and was euthanised. There were no significant findings in the histopathology report that could explain the inability of these animals to recover, and profound haemorrhage was described as the most likely cause.
Mean time for loss of righting reflex during induction was 13.7 ± 2.25 minutes (range 8–20). Mean duration of anaesthesia was 111.4 ± 24.2 minutes (range 75–160). Average duration of exposure to each solution was 25.0, 61.8, 15.0 and 10.4 min for the 3, 2, 1, and 0.5 g L−1 solution, respectively. The average time weighted MS-222 concentration during anaesthesia was 1.93 ± 0.29 g L−1.
Mean time for recovery from cessation of the MS-222 to ocular reflex was 31.2 ± 10.28 minutes (range 20–75). One of the surviving salamanders had a prolonged recovery (75 minutes). There was no statistically significant correlation (p = 0.7198) between duration of anaesthesia and time for recovery (Figure 2).
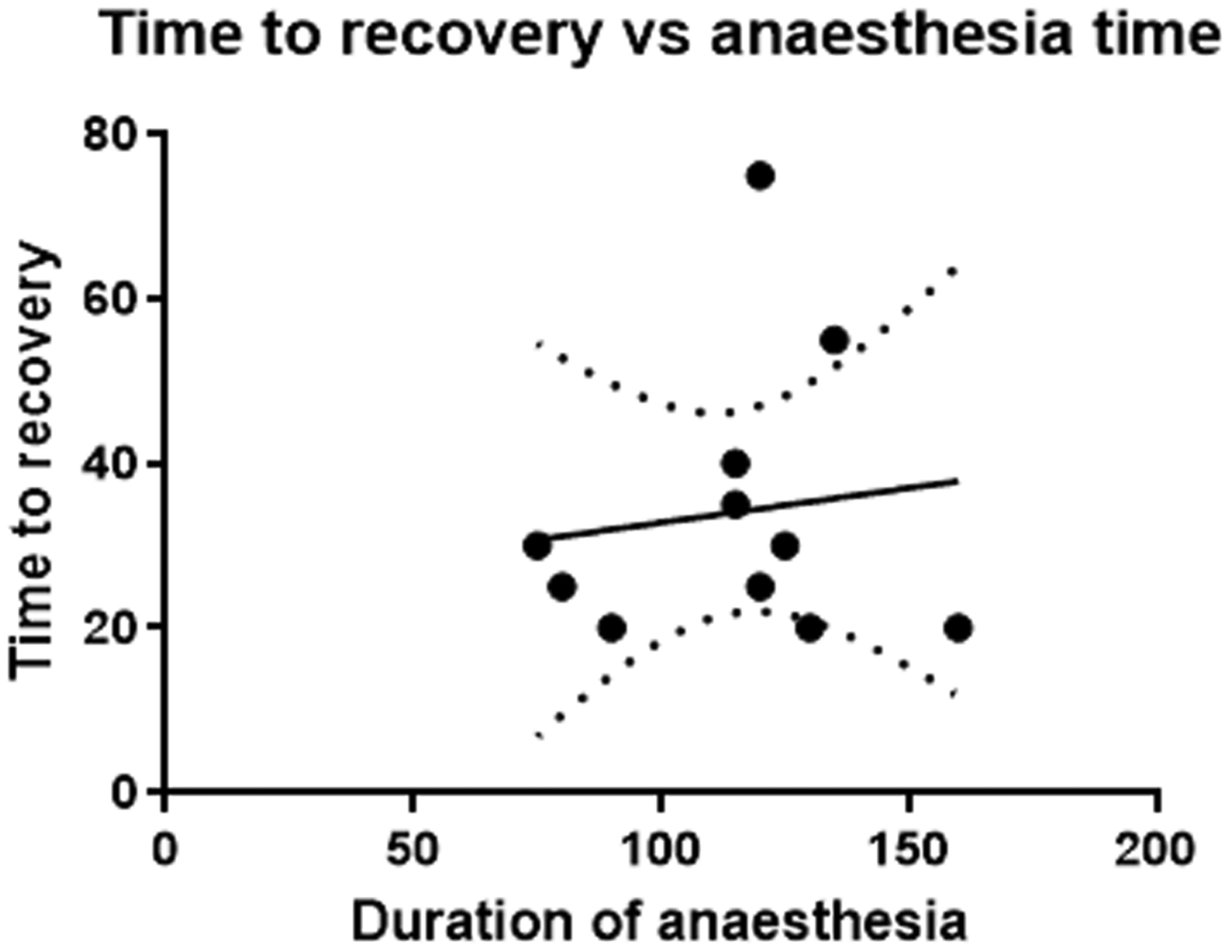
Relationship between duration of anaesthesia (minutes) and time to recovery (minutes). No statistically significant relationship was observed (p = 0.7198).
It was not always possible to measure HR just prior to induction because of the fractious nature of the salamanders. In these cases, the HR obtained previously during the physical examination for the specific animal, was used as a baseline. Baseline was found to be 67.6 ± 9.2 beats per minute. Mean HR at time 0 after loss of righting reflex was 67.4 ± 34.5 beats per minute (range 55–100). The HRs, taken at 5-minute intervals, are displayed in Figure 3. There was a decrease in HR over time with a significant difference from baseline between 35 minutes (p = 0.026) and 170 minutes (p = 0.048).
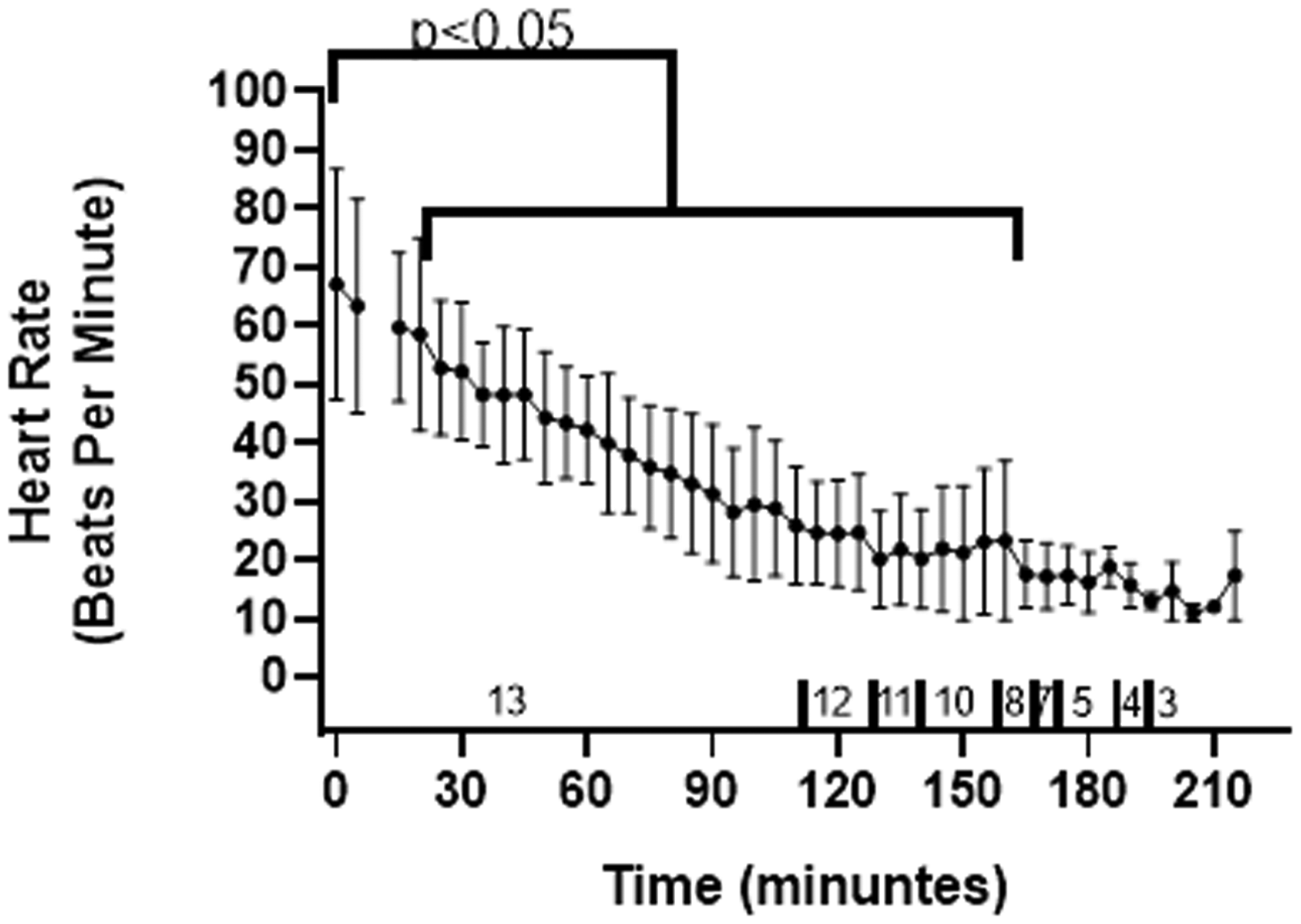
The heart rates (including standard deviation) every 5 minutes over time for the 13 salamanders that underwent surgical anaesthesia. Heart rate was significantly lower than baseline between 35 minutes (p = 0.026) and 170 minutes (p = 0.048), as denoted by the brackets. Numbers over the x-axis represent the number of salamanders included in the dataset for these timepoints. Data points after 110 minutes relate to 12 salamanders or less (the number of remaining salamanders is indicated over the x-axis further declines as time progresses and each of the surgeries are completed).
Changes in pH over the course of the anaesthesia for each salamander are displayed in Figure 4. There was no statistically significant correlation between HR and pH (p = 0.7937) (Figure 5). Temperature was kept constant between 20.5 to 24.0°C for the entire length of the procedure.
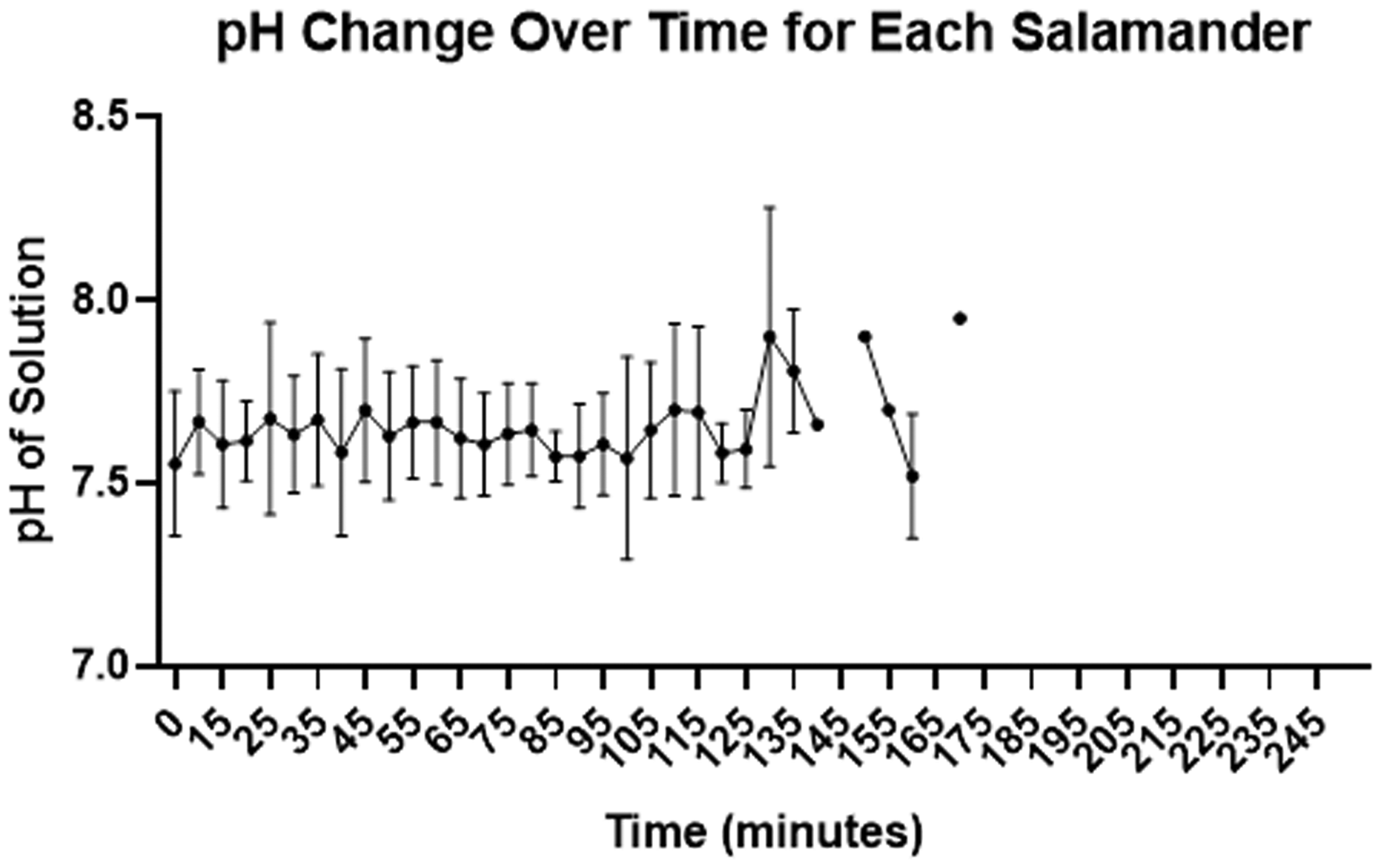
MS-222 pH changes over time for the 13 salamanders. Data is presented as mean ± SD. Data points before 90 minutes represent all 13 salamanders.
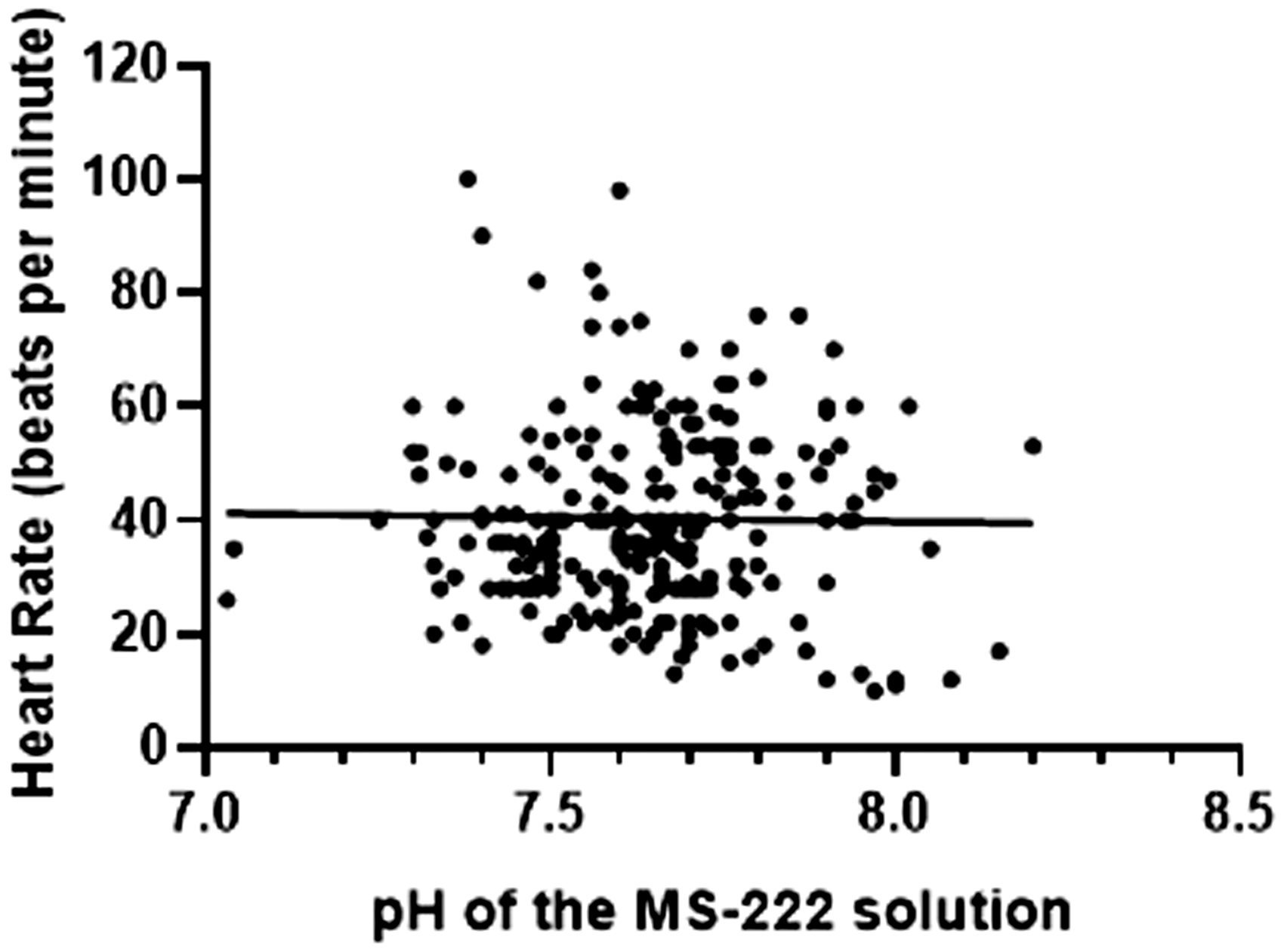
Correlation between pH and heart rate (beats per minute) in 13 salamanders. No statistically significant relationship was observed (p = 0.7937).
A Masimo pulse oximeter was used as part of the monitoring in two of the salamanders. Mean baseline pulse oximetry measurement in these two animals was 85.5% ± 14.5. There was a large decrease during surgery with mean 61% ± 6.4 and clear improvement at recovery after removing the moist gauze square with mean 80.5% ± 2.1.
DISCUSSION
The results of this case series highlight the anaesthetic effects of MS 222 when used for prolonged anaesthesia on fire salamanders and may provide a framework other species can be compared to.
The mean duration of anaesthesia of 111.4 ± 12.1 minutes is considerably longer than the typical lengths of anaesthesia in amphibians reported in literature. While authors mention MS-222 can be used for prolonged immersion anaesthesia, 1 limited studies demonstrate this. Most of the current literature describes MS 222 use for short procedures up to 20 minutes, despite its wide anecdotal clinical use for longer procedures. 17
Survival rate presents as lower than in other studies (85%). Studies in leopard frogs (Guenette and Lair 13 mean deep anaesthetic time of 64.5 minutes) and desert toads (Bufo alvarius) (Wojick et al. 14 mean anaesthetic time of 23.9 minutes) recorded a survival rate of 100% when using MS-222. However, in the present case series, long anaesthesia (mean of 111.4 ± 12.1 minutes) and invasive surgery was carried out that could have affected mortality. One fatality can be accounted for due to significant haemorrhage. The second died due to unknown causes. Unfortunately, no histopathology was performed to confirm or rule out underlying pathology. Even though the blood loss was less significant than in the first salamander, it is difficult to estimate accurately the percentage of the total blood volume lost and the effect that might have to the specific animal. However, it is recommended that an overall blood volume for sampling should constitute less than 10% of their body weight. 18 Unlike more common veterinary patients, there are no established expected peri-anaesthetic mortality rates for any amphibians in which comparisons can be made.
Time to loss of righting reflex was 13.7 ± 2.25 minutes. In literature, induction time using MS-222 is variable, both between and within species. In one study by Kinkead et al. 19 both dusky and seal salamanders (Desmognathus fuscus and Desmognathus monticola respectively) were induced within 5–10 minutes and similarly leopard frogs were induced within 5 minutes. 13 In contrast, cane toads (Rhinella marina) had a mean time to induction of 40 minutes at a dose of 3 g/L whilst Argentine horned frogs (Ceratophrys ornata) have ranges from a few to approximately 30 minutes. 6 No previous study was identified describing the loss of righting reflex in fire salamanders, therefore this approach using 3 g/L resulting in a loss in 13.7 ± 2.25 minutes is the first recorded. This author hypothesises that the range obtained in this study (8–20 minutes) may be due to differences in mass between individuals (15–30 grams). Tail length and mass of Eastern red-backed salamanders (Plethodon cinereus) were both found to significantly affect induction time 15 as was mass in a study with Chinese giant salamanders (Andrias davidianus). 20 Large species of frogs seem to exhibit longer induction times, but this relationship is less clear in distantly related species. Also, the available data for bufonid toads shows a gradual increase of induction time with body mass. 21 Further studies into this with fire salamanders would be needed to confirm this.
Alternating between MS-222 bath solutions of decreasing concentrations is a technique used in the present study. The closest variation described involves “dripping maintenance concentration onto the animal or periodically re-immersing them in a maintenance bath”. 1 Alternating between the concentrations of MS-222 (0.5–4 g/L) rather than dripping maintenance MS-222 or re-immersing periodically allowed more accurate control over anaesthetic depth and immobility. The plane was more consistent and reliable with less alternation over time.
Time to recovery is less varied in associated literature despite different species. This case series result (31.2 ± 10.28 minutes) largely falls within the generally stated range of 30–90 minutes21,22 despite the longer anaesthetic time than other studies. No relevant literature was identified that looked for a correlation between length of anaesthesia and duration of recovery time. This author hypothesises that MS-222 is cleared by first order kinetics in amphibians, so despite increased total exposure to MS-222 for the longer anaesthesia performed, the same proportion is cleared as with the lower exposures found in more common shorter anaesthetics. This would explain similar recovery times despite differences in anaesthetic length. A series of repeat drug-plasma concentration tests would be needed to determine the elimination kinetics behind MS-222 in amphibians.
HR is the easiest measurable parameter given the cessation in pulmonary respiration under anaesthesia. Salamanders have three chambered hearts: two atria and one shared ventricle. The use of Doppler allowed identification of the HR and due to the anatomy described, three beats are heard. Notably, normal values for HRs have not been published 23 so the results obtained cannot be further compared. However, they demonstrate a statistically significant decrease from the baseline value. This may be due to MS-222 inducing bradycardia in fire salamanders given the length of anaesthesia but the large range in baseline HRs at time 0 after inductions (55–100 beats per minute) suggests some salamanders experienced stress tachycardia at the start of the procedure. Studies using MS-222 have yielded mixed results for different species. Leopard frogs have been recorded in multiple studies with decreased HRs suggesting bradycardia is to be expected. A decrease of 18% was found in one study. 13 Two Rio Cauca caecilians (Typhlonectes natans) 24 have also experienced bradycardia in an anaesthetic study. Desert toads however have not shown statistically significant changes from baseline 14 and nor have African clawed frogs (Xenopus leavis) in two studies. 6,17 In contrast, Zullian et al 21 found an increase in heart rate in Mexican axolotls (Ambystoma mexicanum) anaesthetised with MS 222. This suggests variable species-specific responses to MS-222. Cellular hypoxia in mammals results in cell membrane depolarisation in the sinoatrial node, followed by bradycardia. 25 However, ectothermic species are more tolerant to hypoxia compared to endothermic species. There are several studies where hypoxia is followed by increase in heart rate in frogs 26 while in others there was no significant change in heart rate. 27 According to Fritsche 28 the cardiovascular response of the amphibians to acute hypoxia depends on the development stage as well. Young larvae of Xenopus laevis show no change in the heart rate, while older ones can develop bradycardia. Tiger salamanders (Ambystoma tigrinum) demonstrate increase cardiac output, decrease heart rate and decrease of the oxygen consumption, that is in agreement with the findings of our study. 29 Atropine was administered to one salamander with profound bradycardia and there was no increase in HR. Bradycardia as a result of cardiac cellular hypoxia is unresponsive to antimuscarinics (atropine). 30 Also, the amphibians show a different degree of cholinergic and adrenergic innervation depending on the different species and development stage. 28 One more explanation for bradycardia, could be related with the effects of a local anaesthetic on sodium channels in the heart that results in depolarisation reduction. 31
Despite only using the Masimo Rad 57 pulse oximeter for two salamanders, results show a decrease in oxygen saturation from baseline during surgery and a rise during recovery, suggesting pulse oximetry may have a valid place in monitoring. The baseline value of 85.5% ± 14.5 appears low compared to typical veterinary species 5 but salamanders have a shared ventricle, meaning mixing of deoxygenated (from the right atrium) and oxygenated (from the left atrium) blood occurs. Cessation of pulmonary respiration in salamanders under anaesthetic and reliance on cutaneous respiration is notable. A compromise is required between maximising surface area for cutaneous respiration and ensuring anaesthetic absorption/keeping the skin moist using gauge squares. No conclusions can be drawn from the use of pulse oximetry in only two animals. However, it remains of interest that once gauge squares were removed from the body surface, the rise in saturation was evident. Potential for pulse oximeters to provide valuable information such as this may allow individuals to adjust their techniques based on readings obtained. In one study African clawed frog’s oxygen saturation changed significantly 27 suggesting pulse oximetry may be used to assess response to MS-222 and the development of hypoxia, whilst another showed no change in saturation despite marked respiratory depression, including apnea. 6 This presents a difficulty in using pulse oximetry reliably but in combination with monitoring HR it may be of benefit to include in anaesthetic protocols for amphibians. Performing blood gas analyses, that could also validate the pulse oximetry readings, was not possible due to the amount of blood required in relation to the size of the animals.
Analgesia has a pivotal role in every balanced anaesthetic. Amphibians possess the neurological components for transmitting pain and antinociceptive mechanisms to modulate pain.32,33 Machin 33 concludes pain perception in amphibians is likely analogous to that in mammals and invasive, painful procedures should be tempered with analgesics. In addition to this, MS-222 has not been proven to provide analgesia in amphibians. 22 Buprenorphine, a partial mu agonist has been recorded providing significant analgesia. 34 Typically, higher doses have been used; 14 mg/kg in leopard frogs 1 as opposed to 0.5 mg/kg used here. Notably, a multimodal approach to analgesia was used for surgery in this case series and reduced doses were deemed appropriate. Lidocaine (2%), as used in this case series as the second agent has been reported to generally provide suitable analgesia when infused at the site of surgical incision for minor surgeries. A dose of 0.5 mL of 2% lidocaine has also been reported to provide surgical analgesia in leopard frogs. 35 Salamanders in this case series received 0.1 mL as a splash block, well within these ranges but it does constitute a very high dose. Similar doses within this range have been reported by Stevens et al. 36 It cannot be excluded that this dose might have caused a degree of immobilisation as well. It is also unknown the amount of lidocaine that was absorbed through and whether it could have reached the systemic toxicity level. Meloxicam administered post-operatively was given at a dose of 0.2 mg kg−1. The same dose provided adequate analgesia to African clawed frogs. 37 The fire salamanders were able to perform the treadmill work set out in the original study which suggests the adequacy of the analgesic protocol used. The ability to detect and quantify severity of pain in amphibians is an area that would benefit greatly from further studies. Development of pain scores would allow greater precision recognising pain and choosing appropriate multimodal analgesia approach.
Limitations
Most studies on amphibian anaesthesia revolve around the use of research species, with focus on African clawed frogs and leopard frogs. Due to the issue with vastly differing species-specific reactions to MS-222, it is difficult to draw comparisons with the results of this case series.
Further prospective studies are warranted to draw relevant conclusions for prolonged anaesthesia and analgesia in salamander. It was not possible to use standardised doses of MS-222 in all animals and as a result no conclusions can be drawn for the recovery times.
The use of pulse oximetry was limited in only two salamanders and no conclusions can be derived for its use and accuracy. Ideally it would have been utilised in all animals to draw further conclusions on validity and reproducibility, but that was not possible due to equipment issues.
Buprenorphine was injected intramuscularly in the pelvic limb. There is evidence for the existence of the hepatic portal system in amphibians and the fore limb would have been the preferred injection site. 38 However, the hind limb was chosen from the surgeon for consistency with previous similar studies and that could have lowered bioavailable fraction of buprenorphine. In absence of pharmacokinetic data that is difficult to demonstrate.
In conclusion, prolonged recovery anaesthesia is achievable with MS-222 dilutions in salamander in a safe and predicted manner. Also, reflectance SpO2 could be used as part of monitoring. However, no further conclusions could be made from this study for its accuracy and sensitivity.
Footnotes
Acknowledgement
The authors are grateful to John Hutchinson for securing the funding for the biomechanical study. This work was supported by a UK’s Natural Environment Research Council grant [NE/K004751/1 awarded to John Hutchison and S.E. Pierce]. We thank the staff of the RVC’s Biological Services Unit for animal husbandry.
Author’s contributions
T.S.: anaesthesia provision, data collection and interpretation, preparation of the manuscript; J.L.: data interpretation, statistical analysis, preparation of the manuscript; L.L.: study design, surgery provision; S.E.P.: study design, surgery assistance, project management, L.P.: study design, anaesthesia provision and data collection, data interpretation, statistical analysis, preparation of the manuscript. All authors provided comments on the manuscript and approved the final version.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The original data for this study can be accessed by the interested parties after contacting authors via the email address:
Ethical statement
All procedures in this study were approved by the Royal Veterinary College’s Animal Welfare and Ethics Review Board (approval number AWERB-A-2013-5064) and conducted under a Home Office project license following the Animals (Scientific Procedures) Act of 1986 (United Kingdom).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
