Abstract
Immunotoxicity assessment is an important part of non-clinical safety evaluation of biotechnology-derived pharmaceuticals. The reference ranges of evaluated parameters, which depend on the sex, age and geographical origin of animals, play a significant role in interpreting the study results. The aim of this study was to determine the reference ranges of parameters commonly used for non-clinical immunotoxicity studies in cynomolgus monkeys (Macaca fascicularis) of different ages. The percentages of CD3+, CD4+, CD8+, CD20+, CD16/56+ lymphocytes, and the serum levels of immunoglobulins A, M, G and E in clinically healthy cynomolgus monkeys of both sexes and different ages (37–104 months) were analysed. The lymphocyte subsets were assessed by flow cytometry and serum immunoglobulin levels were determined by enzyme-linked immunosorbent assay. Statistical analysis showed a weak rank correlation of CD3+, CD20+ lymphocyte levels with the ages of female and male cynomolgus monkeys, and no significant correlation with age for the combined data of both sexes. The serum levels of immunoglobulins A, M, G and E did not have a significant rank correlation with age for the combined data of both sexes, whereas IgA levels in females and IgG levels in males were weakly correlated with age. Overall, these results justify the use of animals aged three to eight years for non-clinical immunotoxicity evaluation of biotechnology-derived pharmaceuticals. It is important to avoid the formation of animal groups with extreme ages (three years or eight years) in one group during randomization.
Introduction
Studying the effect of biologicals (monoclonal antibodies) on the nature and severity of immune responses is crucial for non-clinical safety studies of certain classes of drug products. Lymphocyte subsets and immunoglobulin levels in peripheral blood are some of the most significant parameters characterizing cellular and humoral immunity. Given the species specificity of pharmacological targets, cynomolgus monkeys are commonly used in non-clinical studies. In this regard, a clear understanding of the reference ranges of the studied parameters is essential for interpreting the obtained results. According to published data, the percentage of peripheral blood lymphocyte subsets in healthy monkeys depends on sex, age and geographical origin,1–5 and immunoglobulin levels can vary significantly depending on the analytical procedure,6–10 which casts a doubt on the possibility of using these study results to evaluate the historical ranges variability. Considering the practical significance of the existing problems, we aimed to analyse the baseline values of parameters in clinically healthy monkeys (cynomolgus monkeys) in this study to determine the reference ranges of the studied parameters in animals (of different sex and age groups) maintained in the laboratory. Determining the optimal age range for monkey studies is of essential practical importance for understanding the variability of the reference ranges of lymphocyte subpopulations and immunoglobulin levels in the peripheral blood of cynomolgus monkeys, since this is the only monkey nursery located in the territory of the Eurasian Economic Union.
We aimed to determine the optimal age range of monkeys in the nursery population of the Research Institute of Medical Primatology (RIMP) in Sochi for a preclinical immunotoxicity study.
Methods
Animals
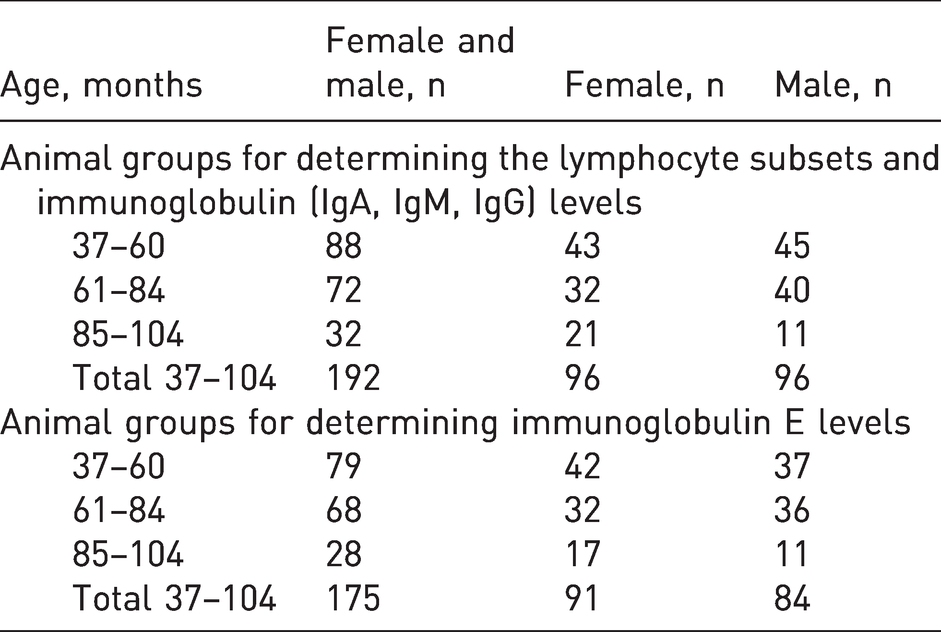
The studies were carried out on non-human primates, cynomolgus monkeys (Macaca fascicularis), based on the Federal State Budgetary Scientific Institution RIMP. Each study (eight studies were performed in total) was considered at a meeting of the Bioethics Commission of CJSC ‘BIOCAD, Russia’ (protocols numbers 1/2015, 22/2015, 28/2016, 34/2016, 1/2017, 7/2017, 11/2017, 13/2017) and the need for the study was confirmed. For data processing, only background values of parameters concerning lymphocyte subsets and serum immunoglobulin levels were used in non-clinical studies. The animals were not previously subjected to any experimental intervention and were obtained from the RIMP. RIMP originally obtained these animals from Vietnam. The monkeys used in the study were the offspring of animals born in the nursery (generation F2 or later). In total, 192 animals (96 females and 96 males) were assigned to three groups based on their age (Table 1), with an interval of approximately two years between the groups; these were the most commonly used age ranges of animals in preclinical studies. IgE levels were analysed in 175 primates (91 females and 84 males). The age and sex details are presented in Table 1.
Distribution of animals by sex and age.
The monkeys were housed in outdoor enclosures connected to indoor-shelter housing units. The monkeys were housed in metal cages, each containing a colony of 16 individuals (15 females and one male) and were exposed to natural conditions throughout the year. All primates were fed grain-based pellets (LLC ‘TL UNION’, Russia) supplemented with two apples or seasonal vegetables per day; water was supplied from the central water pipeline using an automatic watering system (ad libitum). All animals were monitored once a year for tuberculosis using the tuberculin skin test and were subjected to prophylactic antiparasitic treatment every six months. Each animal was subjected to haematological blood testing. Bacteriological examination of rectal smears was performed for all monkeys and faecal samples were examined for the causative agents of protozoanosis (giardiasis, coccidiosis, balantidiosis and blastocystosis) and helminthiasis (trichocephalus and strongyloids).
Animal husbandry and the studies were carried out in compliance with the Guidelines.11–13
During the quarantine period (30 days before testing) and investigation, the monkeys were housed in individual metal cages (66 cm × 70 cm × 90 cm). The following environmental conditions were maintained in the animal husbandry facilities: air temperature 27 ± 2°C, relative humidity 65 ± 5% and natural daylight. The monkeys received additional enhanced nutrition in the form of various vegetables, fruits (orange, apple or banana), dried fruits and nuts. Each macaque was observed once daily for general well-being. Feeding behaviour was assessed daily. Bacteriological examination of faeces was carried out at the beginning and end of the quarantine period. The animals were injected with an antimicrobial agent Metronid 50 (Metronidazole; LLC NITA-PHARM, Russia; 0.2 ml/kg) and an anthelmintic agent Ivermek (Ivermectin, vitamin E; LLC NITA-PHARM, Russia; 0.02 ml/kg). The weight of the animals ranged from 2.5 kg to 7.0 kg. None of the females were pregnant during the study period. All monkeys used in the study were considered clinically healthy based on haematological and biochemical blood test findings, microbiological and parasitological analysis of faecal samples, general conditions, gastrointestinal tract functions and body temperature.
Sampling
Blood samples of each subject were collected under aseptic conditions from the inguinal vein using tubes, and without anaesthesia. Primate containment cages with sliding partition were used to immobilize monkeys. To study a subset of lymphocytes, blood samples (1 ml) were collected in heparin tubes and analysed on the day of blood collection. To study the immunoglobulin levels, blood samples (1.5 ml) were collected in tubes with a coagulation activator to obtain serum, which was stored at –20°C until measurement.
Materials and methods
Flow cytometry
The monkey peripheral blood lymphocyte subsets were evaluated using fluorescent-conjugated monoclonal antibodies, anti-CD3-PerCP-Cy5.5 (Clone SP34-2), anti-CD4-FITC (Clone L200), anti-CD8-PE (Clone RPA-T8), anti-CD20-FITC (Clone 2H7), anti-CD16-PE (Clone 3G8) and anti-CD56-PE (Clone MY31) (BD Biosciences, USA). Red blood cells were lysed using BD FACS® lysis solution (Becton Dickinson, USA). The sample analysis was performed using the Guava® easyCyte flow cytometer (Merck Millipore, USA) and 5000 events were collected. The data were processed using InCyte™ guavaSoft™ software. The following cell subpopulations were determined (percentage of the total lymphocyte count): B lymphocytes (CD20+), T lymphocytes (CD3+), T helper cells (CD3+CD4+), killer T cells (CD3+CD8+) and NK cells (CD3–CD16+/CD56+).
Determination of immunoglobulin levels in the blood
Determination of the immunoglobulin A, E, G and M levels in blood was performed by enzyme-linked immunosorbent assay (ELISA) using Monkey Ig ELISA kits (Life Diagnostics Inc., USA) according to the manufacturer’s protocol.
Statistical analysis
Emission analysis was performed using the double median method. Emissions were replaced by cutoff threshold values. The normality of the distribution was evaluated using the Kolmogorov–Smirnov test. The distribution in the subgroups differed from normal distribution; therefore, the comparisons of two groups on quantitative scales were based on the non-parametric Mann–Whitney test and comparisons of three or more groups were based on the non-parametric Kruskal–Wallis test. The mean values and standard deviations were used to describe quantitative parameters. Statistical significance was set at p < 0.05.
Comparisons between age groups were performed using the Kruskal–Wallis test with further post hoc analysis using the Nemenyi test. Significance differences between males and females were estimated based on the non-parametric Mann–Whitney test. Statistical significance was set at p < 0.05.
The statistical significance of various values for binary and baseline parameters was determined using Pearson’s chi-squared test. Correlation analysis was performed based on the non-parametric Spearman's rank-order correlation coefficients.
Multiple regression analysis was used to determine the possibility of predicting a target quantitative variable based on several independent variables (factors).
Statistical data processing was performed using Statistica 10 and SAS JMP 11 packages.
Results
For statistical analysis, the animals were divided into groups according to Table 1.
Changes in the percentages of peripheral blood lymphocytes in animals of different age groups
Comparative evaluation showed that the relative counts of T-lymphocytes (CD3+) and cytotoxic lymphocytes (CD8+) were lower: (63.67% ± 9.57% (CD3+) and 36.33% ± 7.13% (CD8+), respectively) in females aged 37–60 months versus those in females aged 85–104 months (71.86% ± 9.31% and 42.38% ± 9.13%, respectively); in contrast, in the same age group of males (37–60 months), the values of this parameter were higher (69.49% ± 6.78% (CD3+) and 41.42% ± 6.87% (CD8+)) than those in males aged 61–84 months (64.28% ± 8.05% and 37.15% ± 7.82%, respectively); the differences were statistically significant.
The relative count of CD20+ lymphocytes (12.95% ± 7.41%) was decreased in the female group (85–104 months), whereas it (27.55 ± 11.33%) was increased in males relative to the values in the age control groups (37–60 and 61–84 months); the differences were statistically significant. Upon combining these data of females and males, the relative counts of CD20+ cells in the group aged 85–104 months were lower (17.97% ± 11.24%) than those in animals aged 61–84 months (24.08% ± 11.83%).
There were no statistically significant differences in the relative counts of CD4+ and NK cells in groups of females and males of different ages, as well as those of CD3+, CD4+, CD8+ lymphocytes, and NK cells in the combined groups, including data from animals of both sexes (Table 2).
Percentages of lymphocyte subsets in the peripheral blood of cynomolgus monkeys.
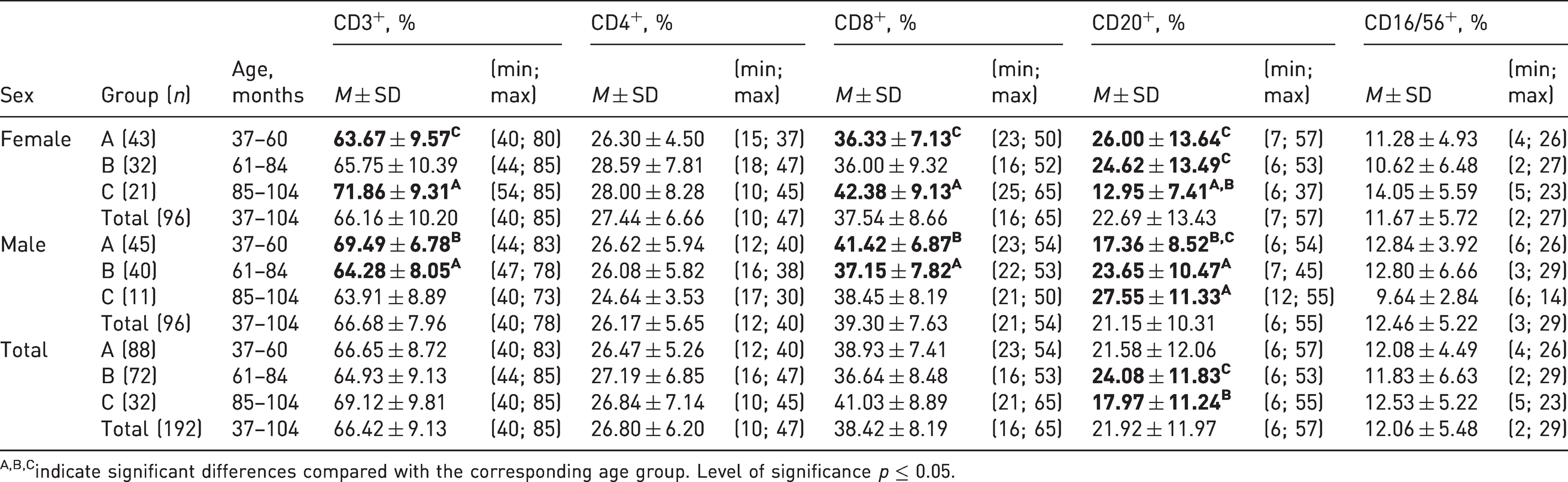
A,B,Cindicate significant differences compared with the corresponding age group. Level of significance p ≤ 0.05.
Changes in the relative counts of peripheral blood lymphocytes in animals of different sexes
According to the study results, the relative counts of CD3+ lymphocytes in males aged 85–104 months were lower (63.91% ± 8.89%) than those in females of the same age (71.86% ± 9.31%), and the relative counts of CD3+ and CD8+ lymphocytes in males aged 37–60 months were higher than those in the females (69.49% ± 6.78% and 41.42% ± 6.87%, respectively). The relative numbers of B-lymphocytes in the blood of cynomolgus monkeys aged 85–104 months were 27.55% ± 11.33% and 12.95% ± 7.41% in males and females, respectively, whereas at the age of 37–60 months, the values in the group of females were higher (26.00% ± 13.64%) than those in males (17.36% ± 8.52%); the differences were statistically significant (Figure 1(a), (c) and (d)).
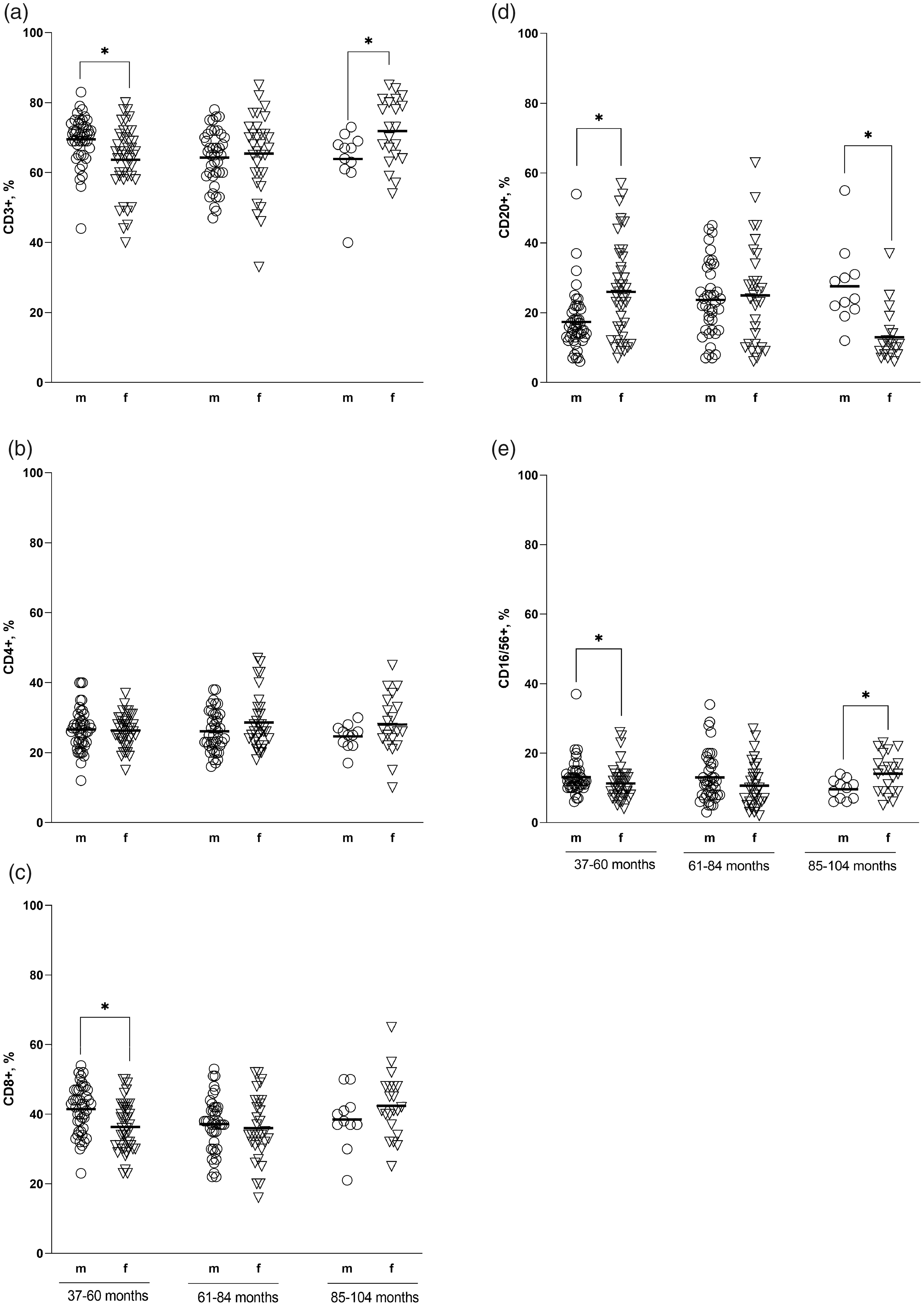
Phenotyping of peripheral blood lymphocytes in male and female cynomolgus macaques divided into three age groups. The relative count of each subset was assessed in the peripheral blood samples of animals aged 37–60, 61–84 and 85–104 months (x-axis) and was expressed as the percentage of the total lymphocyte count (y-axis). Individual data points are represented by sex-specific shapes. Coordinated horizontal bars represent the group mean values. All results were considered statistically significant at p < 0.05.
Significant differences in the percentage of CD16/56+ lymphocytes were noted in the age groups of 37–60 and 85–104 months; the relative counts of NK cells in males were 12.84% ± 3.92% and 9.64% ± 2.84%, and those in females were 11.28% ± 4.93% and 14.05% ± 5.59%, respectively (Figure 1(e)).
Statistical analysis showed a weak correlation between the relative counts of CD3+ and CD20+ lymphocytes and the age of animals, divided by sex. Spearman’s rank-order correlation coefficient in terms of the relative counts of CD3+ lymphocytes in males and females was –0.31 and 0.33, respectively, and the percentages of B-lymphocytes were 0.32 in males and –0.38 in females (p < 0.01). However, no significant rank correlation was noted when the data of the females and males were combined.
Changes in the level of serum immunoglobulins IgA, IgM, IgG, IgE in animals of different age groups
According to the results, the IgA level in female cynomolgus monkeys aged 85–104 months was higher (12.07 ± 5.78 mg/ml) compared with those in the groups of younger animals (4.75 ± 2.31 mg/ml and 4.87 ± 3.36 mg/ml in age groups 37–60 and 61–84 months, respectively); the difference was statistically significant. Similar changes over time were observed in combined groups of females and males, where the IgA contents were 9.82 ± 5.84 mg/ml, 6.85 ± 5.33 mg/ml and 6.31 ± 5.82 mg/ml at the ages of 85–104, 37–60 and 61–84 months, respectively.
The IgM level in females aged 85–104 months was significantly higher than those in the groups aged 37–60 and 61–84 months and amounted to 7.53 ± 1.99 mg/ml, 5.63 ± 1.79 mg/ml, and 4.81 ± 1.71 mg/ml, respectively. In the combined groups of females and males aged 85–104 months, the IgM level was also significantly higher than those in the compared groups (37–60 and 61–84 months) and amounted to 6.8 ± 1.97 mg/ml.
An increase in IgG levels were observed in males aged 61–84 months (36.77 ± 11.46 mg/ml) relative to the values in the group of younger (37–60 months) animals (30.92 ± 10.43 mg/ml). When comparing the summarized data in groups of both sexes, there were no significant differences in the values of the studied parameters.
There were no significant differences in the IgA and IgM levels in the male groups, IgG levels in the female groups and IgE levels in females and males of all age groups (Table 3).
Serum immunoglobulin levels in the peripheral blood of cynomolgus monkeys.
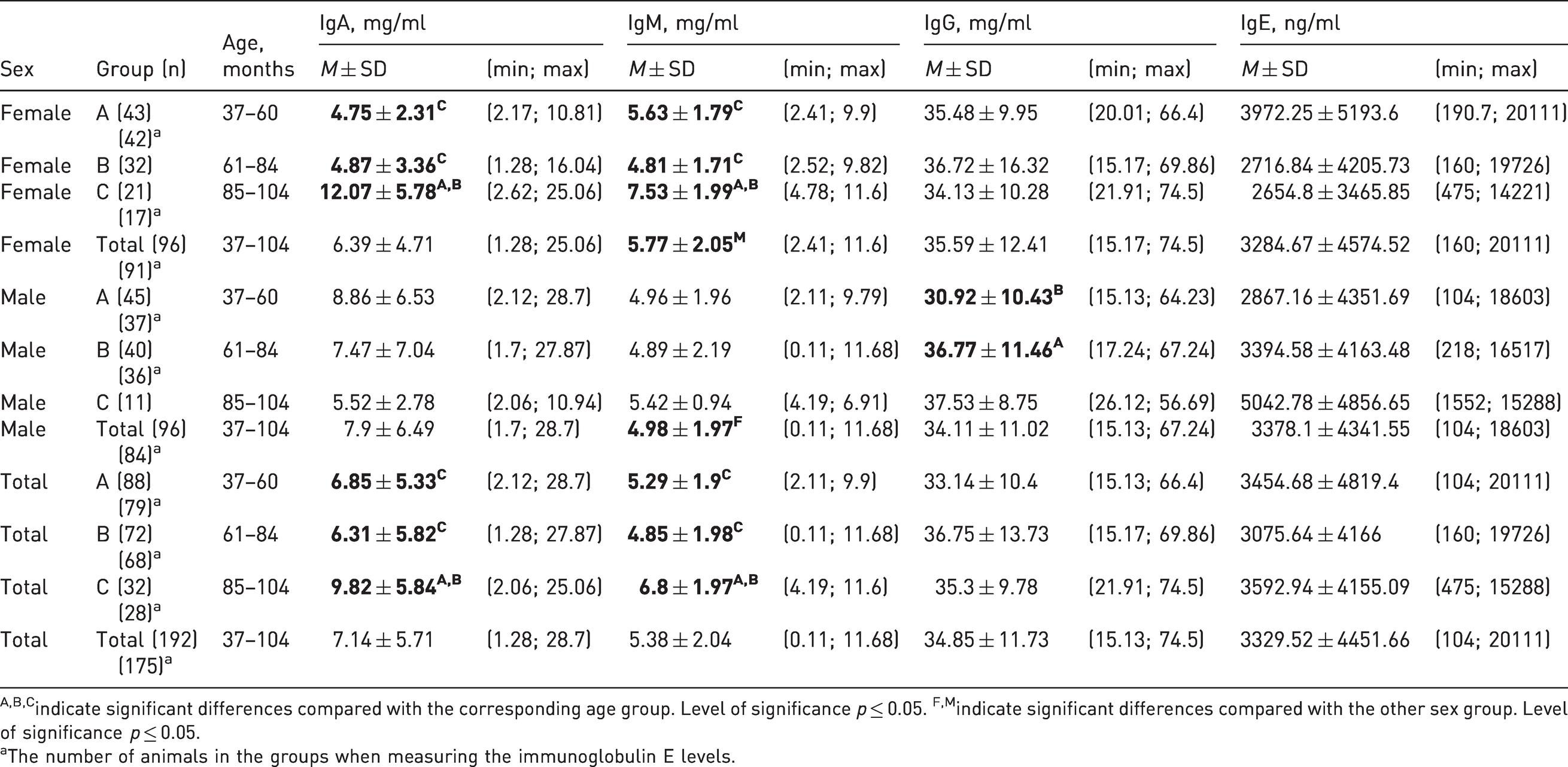
A,B,Cindicate significant differences compared with the corresponding age group. Level of significance p ≤ 0.05. F,Mindicate significant differences compared with the other sex group. Level of significance p ≤ 0.05.
aThe number of animals in the groups when measuring the immunoglobulin E levels.
Changes in the serum immunoglobulin A, M, G and E levels in animals of different sexes
When comparing the data obtained for groups of different sexes of the same age, the IgA levels in females aged 37–60 months were lower (4.75 ± 2.31 mg/ml), and those aged 85–104 months were higher (12.07 ± 5.78 mg/ml) than those in the male groups, whose IgA levels were 8.86 ± 6.53 mg/ml and 5.52 ± 2.78 mg/ml, respectively; the differences were statistically significant. For IgM levels, significant differences were observed in the age group of 85–104 months where the values of this parameter in males were lower than those in the female group and amounted to 5.42 ± 0.94 mg/ml and 7.53 ± 1.99 mg/ml, respectively. In the combined group (aged 37–104 months), the IgM level in the male group (4.98 ± 1.97 mg/ml) was lower than that in the female group (5.77 ± 2.05 mg/ml) (Table 2).
The IgG level in females aged 37–60 months was higher (35.48 ± 9.95 mg/ml) than that in the male group (30.92 ± 10.43 mg/ml). The IgE level in females aged 85–104 months (2654.8 ± 3465.84 ng/ml) was significantly lower than those in the male group (5042.78 ± 4856.65 ng/ml) (Figure 2).
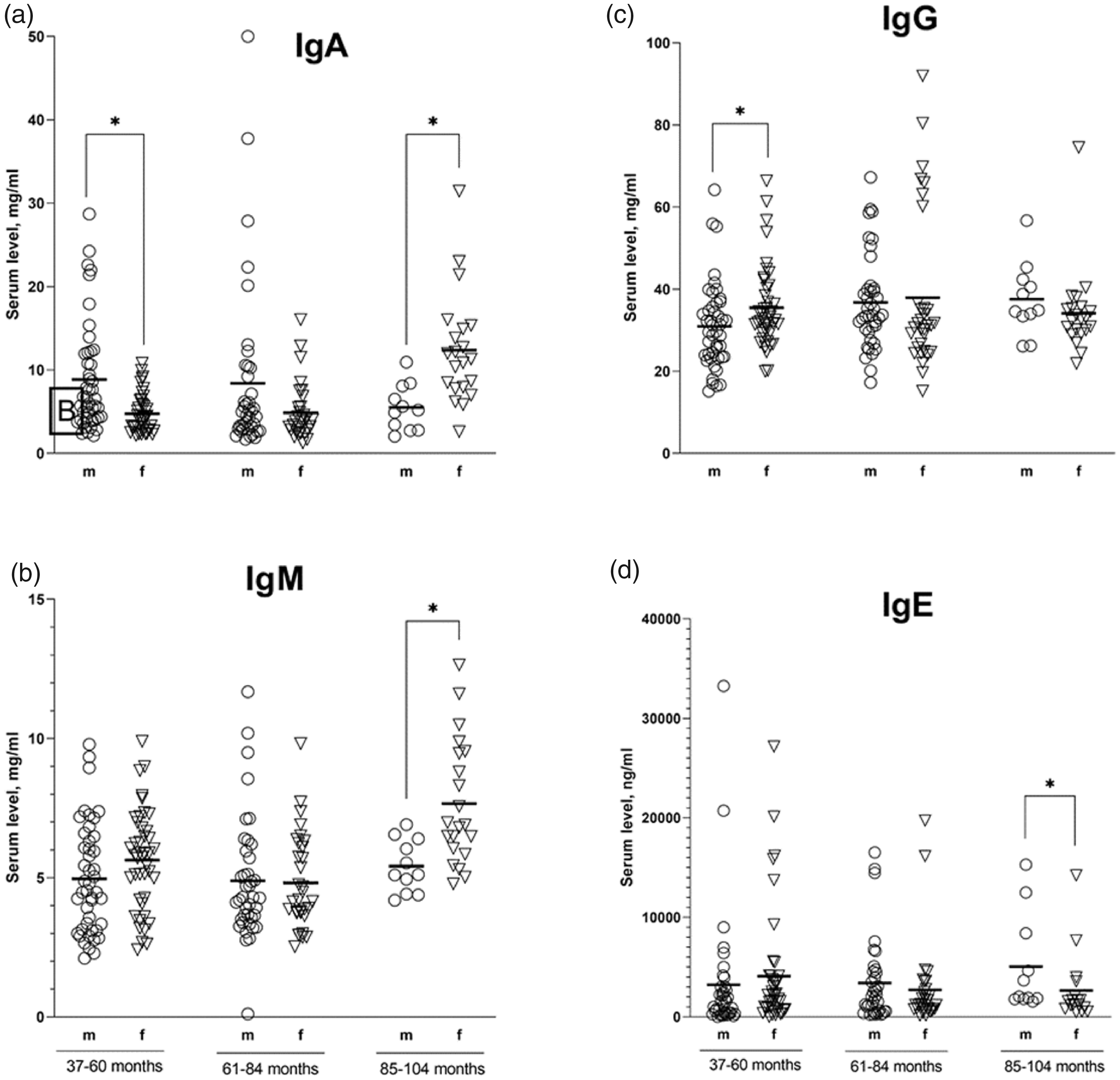
Serum immunoglobulin levels in male and female cynomolgus monkeys who were divided into three age groups. The serum levels of each isotype were assessed in serum samples from animals aged 37–60, 61–84 and 85–104 months (x-axis) and were expressed in mg/ml or ng/ml (y-axis). The individual data points are represented by sex-specific shapes. Coordinated horizontal bars represent the group mean values. All results were considered statistically significant at p < 0.05.
Female and male cynomolgus monkeys aged 61–84 months showed no statistically significant differences in the levels of all immunoglobulin classes. The serum immunoglobulin levels did not show a significant correlation with age in the combined group summarizing the data of both sexes and were weakly correlated with IgA levels in female groups and IgG levels in male groups. Spearman’s rank correlation coefficients were 0.39 and 0.38 for females and males, respectively, at a significance level of 0.01.
Discussion
The cynomolgus macaque (M. fascicularis) is a widely used species in non-clinical safety studies. The current trend toward the development of biologicals, such as monoclonal antibodies, has increased the need for studies on non-human primates. A number of publications have been devoted to the study of separate populations of lymphocytes and, thus, it is very difficult to rely on these indicators as a physiological norm.14–19 The current study aimed to characterize the potential differences in background data between macaques of different ages and sexes.
Analysis of age-related changes in peripheral blood lymphocytes from clinically healthy monkeys revealed significant differences in the populations of CD3+, CD8+ lymphocytes, NK cells and B-cells in females and males of different ages. Although changes in cytotoxic T-lymphocyte and T-lymphocyte counts over time in animals of different sexes showed contrasting trends, the recorded differences did not remain statistically significant when the values were combined. The results obtained were generally consistent with the data provided by other authors.1–5 Thus, the percentages of T cells (CD3+) in female and male cynomolgus monkeys aged 50.4–120 months 1 and 12–120 months2–5 were relatively constant. Further, the relative counts of CD4–CD8+ cells increased until 48–60 months, but with age, there were no significant differences. 1 Statistical analysis of 10 immunophenotype studies on cynomolgus monkeys 20 indicated that the absolute counts of CD3+, CD4+, CD8+ and B-cells were higher in males than in females, with the exception of NK cells. According to the data obtained in our study for the combined groups (37–104 months), there were no significant differences in the absolute counts of these cells between the males and females. In studies published previously, the sex of the animals was not considered in the comparative analysis. In individual studies, there were no significant differences between the groups of different sexes when evaluating the relative count of CD3+ lymphocytes in female and male cynomolgus monkeys aged 12–48 months 3 and 24–36 months. 4
The opposite trends of CD20+ lymphocyte counts in the groups of different sexes shown in this study, as well as the significant differences in the populations of B-lymphocytes in females and males aged 37–60 and 85–104 months and in the combined groups aged 61–84 and 85–104 months, are not fully consistent with that reported in the literature. 1 Thus, in contrast to the data presented, a decrease in the relative counts of CD20+ cells in M. fascicularis was observed in the combined group of females and males aged 50–60 months; with age, the values of this parameter remained relatively stable, 1 and there were no significant differences in the values of this parameter between the female and male groups.2–4 In a comparative study 21 on M. fascicularis from Mauritius (age 39–46 months), Cambodia (age 38–51 months) and a mixed Asian source (Cambodia, Vietnam and Indonesia; age 36–43 months), differences were identified in B-cell counts (CD45+CD20+) and CD8+ T-cell counts (CD45+CD3+CD8+). The B-cell and CD8+ T-cell populations were lower in Mauritius macaques than those in Asian macaques. The changes in CD20+ cells thus appear to correlate with the age and origin of the animals.
The relative counts of CD3+CD4+ lymphocytes and NK cells in groups of different ages and sex were relatively stable, which is consistent with the data presented in a similar study, 1 where the number of blood NK cells in primates of both sexes increased until maturation (48–60 months), and with age, there were no differences between the values of different age groups.
Evaluation of humoral immunity indices showed that the serum IgA and IgM levels in female cynomolgus monkeys aged 85–104 months were significantly higher than those in male groups of the same age, as well as in female groups of other ages; the IgA levels in females aged 37–60 months were significantly lower than those in males. In combined groups summarizing the values obtained in groups of both sexes, the IgA and IgM levels at the age of 85–104 months were significantly higher than those in younger animals (37–60 and 61–84 months, respectively), which was consistent with the results of another study which described similar trends in cynomolgus monkeys aged 60–108 months. 9
The IgG levels, established in males aged 61–84 months compared with those in males aged 37–60 months, were not consistent with the data presented previously, 9 indicating that the IgG concentration in primates aged 36–48 months was higher than that in the group aged 60–108 months. In one study, 21 there were no significant differences in the serum levels of IgG, IgM and IgA between Mauritius macaques and Asian macaques. In another study, 6 there were no changes in the IgA, IgG and IgM levels with age; however, the fundamental differences in the analytical methods used by the authors and the fairly small sampling should be considered. In a study on M. leonina, there were no effects of age (36–132 months) on the serum IgA and IgM levels. However, the serum IgG levels progressively increased with age and the serum IgA, IgG and IgM levels in females were higher than those in males. 22 These contradictory results probably occurred due to interspecies differences.
The average IgE levels did not show significant age differences for all analysed groups, whereas the absolute values ranged from 4 μg/ml to 23 μg/ml, which corresponds with the physiological norm range previously established for this parameter. 8
Thus, considering the differences in the principles of animal assignment by age groups and statistical analysis methods, as well as the animal geographical origin used in the studies, the results presented in this study cannot be fully comparable with those reported in the literature; however, they do have an important practical value and make a significant contribution to the development of ideas regarding the variability of physiological norms in terms of lymphocyte subsets and immunoglobulin levels in the peripheral blood of cynomolgus monkeys. There are two major limitations of the study: the first is the method used to determine immunoglobulin levels and the second is the animal origin. The reference ranges of the immunoglobulin levels determined by ELISA may differ from the ranges determined by other methods. Animal origin and environment could influence animal immunological parameters. Indicated limitations could hamper comparison with the results published by different research groups. Despite indicated limitations, the main study goals were achieved.
Overall, the weak age correlation of the absolute or relative parameter values in groups of different ages and sexes established by statistical analyses justifies the use of biological drugs in animals aged three to eight years for non-clinical immunotoxicity evaluation. However, randomization should be avoided, as it could result in the formation of experimental groups with extreme age values in the range. As the combination of female and male groups resulted in statistically significant differences in immunoglobulin content and lymphocyte subset relative counts, the patterns of parameter changes over time as revealed in groups of different sexes should be considered only during specific comparative studies.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
