Abstract
Laboratory rodents spend the entire day housed in standard cages that provide a restricted area for movements and might, therefore, limit physical activity. However, it has not been tested in immature rodents of ages ranging from weaning to adulthood (adolescence period) whether the restricted area per animal does actually reduce physical activity and impact the body composition. We analyzed the spontaneous physical activity and feeding behavior during the adolescence of mice kept in two different housing conditions (standard stocking density (SSD) versus low stocking density (LSD)). We aimed to compare the body composition between SSD and LSD groups before they reached adulthood. Differential housing began at four weeks of age and was maintained for four weeks until euthanasia at eight weeks of age. The SSD group had a floor space of 88 cm2 available per animal, while LSD mice were housed with a floor space of 320 cm2 per animal, increasing the individual radius for movement more than three-fold compared with standard requirements. Mice kept in SSD exhibit lower spontaneous physical activity than mice kept in LSD. Early-life exposure to reduced physical activity in mice housed in SSD resulted in greater visceral fat accumulation before adulthood. An environment enabling/stimulating physical activity should be established for rodents as early as possible. This study will be helpful in showing that mice kept in SSD are early exposed to a reduced physical activity already in the adolescence period. Our findings could raise reflections about the translatability of rodents kept in SSD to healthy active humans.
Introduction
Mice and rats are the most widely used animal models in biology research. 1 Standards and best practices have led to important advances in the way laboratory rodents are handled, treated or even housed.2–4 However, more attention should be paid to the day-to-day activities of rodents in their home cages, which could also influence research outcomes. 5 Rodents spend almost the entire day housed in cages that provide food ad libitum and a limited radius for movement.6,7 As the number of animals per unit area (stocking density) may influence behavioral predictors of welfare, 8 it is reasonable to assume that a smaller area available for each rodent would limit their potential radius of movement inside the cage. A high stocking density is a situation that could lead to animals being physically inactive throughout their life span. In line with this, some studies show that adult rodents kept in small cages or even in housing conditions taken as standard exhibited reduced muscle enzyme activities as well as decreased muscle strength on an inclined plane test, 7 decreased motor coordination, 9 increased adiposity 10 and reduced aerobic capacity and spontaneous physical activity (SPA) 11 compared with their counterparts kept in cages with a larger area available per animal (low stocking density). Other models, such as those with climbing prevention, 9 hindlimb unloading12,13 and immobilization cages 14 could be used for inducing physical inactivity, but they are more extreme in changing rodents’ life. While the existing animal housing studies are important to show that physical fitness and health outcomes of rodents may be compromised by cages with limited space (often taken as standard cages), they fail to provide a more complete and realistic picture of daily routines of rodents in their cages, especially in terms of physical activity. It is obvious that reduced physical activity is associated with decreased physical fitness and health outcomes, but these measures are not solely enough for diagnosis of active and inactive behavior. Therefore, a consistent evaluation of spontaneous physical activity is undoubtedly necessary. SPA (also known as non-volitional exercise) may include activities with or without horizontal locomotion (e.g. deambulation, foraging, grooming, fidgeting, rearing, spontaneous muscle contractions, and maintaining posture when not recumbent).15–17
As far as we know, a study of effect of housing conditions on SPA and body composition outcomes has never been done in rodents with ages ranging from the weaning period to adulthood (adolescence stage). 18 There is a need to determine the minimum age in which laboratory rodents are already exposed to reduced physical activity, which may be happening since they are weaned from their mother. Considering that this issue has not yet received due attention, our study was undertaken to fill this gap, addressing two research questions: 1) do stocking conditions taken as standard housing reduce SPA of rodents already in adolescence? 2) can a short-term exposure to reduced physical activity in early life lead mice to show fat accumulation before reaching adulthood?
Therefore, we aimed to analyze the SPA and feeding behavior during adolescence of isogenic male mice kept in two different housing conditions (standard stocking density (SSD), 88 cm2 per animal, versus low stocking density (LSD), 320 cm2 per animal). We also aimed to compare the body composition between SSD and LSD groups before they reached full adulthood. A point to be mentioned is that floor space for each mouse in SSD (i.e. 88 cm2) is in accordance with European and US-based guidelines for the Care and Use of Laboratory Animals, suggesting that our SSD group may be taken as standard in terms of area per animal.3,4,19–21
We hypothesized that, already at the first weeks of age, male mice kept in SSD would be less physically active (exhibiting lower SPA) than mice housed in LSD. We also hypothesized that in the mice housed in SSD (early exposed to reduced physical activity), the fat deposition would increase compared with mice housed in LSD. Our study will not only be helpful from a theoretical point of view, but also will have practical usefulness for researchers conducting animal experiments. An early reduction of SPA in adolescent mice could be not only a contributing factor to the obese phenotype, but also an influencing factor that can have significant impact on the translatability of rodent findings to healthy active humans.
Material and methods
Animal care
Sixty male mice (isogenic C57BL/6J) were studied. Male rodents were chosen to avoid potential confounding effects of estrogen increase at puberty and considering that the puberty of mice differs between male (∼5 weeks) and female mice (∼8 weeks). Physiology and behavior are known to be influenced by the estrous cycle in female rodents.22–24 During the experimental period, the mice were kept in a room with a controlled environment, including the temperature (23 ± 1°C), relative humidity (45–55%), noise ( < 80 decibels) and a photoperiod with a 12-h light/dark cycle (illumination from 6:00 a.m. to 6:00 p.m.). The mice used in this study were obtained in the same animal facility of University of Campinas, at the Multidisciplinary Center for Biological Investigation (CEMIB), part of the quality network of laboratory animals, adopting health monitoring and genetic quality in the availability of rodents. The local Research Ethics Committee (CEUA-UNICAMP, protocol number 4953-1/2018) approved all procedures and protocols. The authors confirm that they have followed national and international standards for the protection and management of laboratory animals. All experiments were conducted according to the European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes 3 and the National Research Council Guide for the Care and Use of Laboratory Animals. 4
Study design
Within a short time after weaning, animals were allocated into two types of housing conditions: SSD, n = 30, and LSD, n = 30. The allocation of mice to different housing conditions was done randomly by draw to prevent selection bias and balance the distribution of measured and unmeasured confounding variables between the groups. 25 No differences in body mass were noted between groups’ mass at the start of the experiment (pre-intervention), showing that the groups started from similar conditions (a reliable criterion for starting the study). Differential housing began at four weeks of age and was maintained during four weeks until euthanasia at eight weeks of age. Based on some studies, the age in C57BL/6 J mice may be divided into the following stages: 21–60 days of age (adolescence), 3–6 months of age (adult age), 10–15 months of age (middle age) and 18–24 months of age (old age).18,26 The SPA and feeding behavior were measured daily on a per cage basis. At the end of the experiment, 10 animals of each group (SSD, n = 10 and LSD, n = 10) were used for the determination of body composition (visceral fat depots and brown adipose tissue, as well as water, fat mass and fat-free mass of carcass).
Housing conditions
During all experimental time, the mice were collectively housed at the density of 15 animals per cage (88 and 320 cm2 per animal for SSD and LSD, respectively). In SSD, mice were kept (15 animals per cage, floor area of 1320 cm2) in polyethylene cages (dimensions: length: 40 cm; width: 33 cm; height: 16 cm). In LSD, animals were also kept in groups of 15 mice (floor area of 4800 cm2; dimensions: length: 80 cm; width: 60 cm; height: 33.3 cm). While it is recommended to include environmental enrichment (e.g. toys) to improve the rodents’ welfare, 27 it was not included in this study because a confounding factor could arise if mice had access to objects that could affect SPA. For this reason, we did not insert any material or toys inside housing in an attempt to study more specifically the effect on SPA of increasing the area per animal. All cages were lined with autoclave-sterilized sawdust that was replaced four times per week. Our choice to preserve social housing instead of providing a cage for each animal was made because chronic social isolation may induce anxiety and aggressive behavior in social rodents.28–30 Moreover, there are reports demonstrating that chronic social isolation alters metabolic parameters.31–33 It must not be forgotten that the behavior of rodents in their daily routines may be affected by social instability through changes in housing density or cage isolation. 34 Except for some situations, single housing is discouraged by animal welfare regulations.3,4 We adopted some strategies to induce peaceful living among rodents: (a) new mice (‘intruders’) were not placed in cages with already-established social groups; (b) there was no varied size cage-to-cage migration; (c) mice had been acclimatized because the same group of animals had been kept together since the arrival of the mice to the vivarium; (d) the food (standard chow, Nuvilab CR-1; Nuvital, Brazil) and fresh and clean water were accessible ad libitum to prevent competition and disturbances in social relationships. These strategies were planed following animal guidelines, as discussed above.3,4,35 While the formation of new social relationships is potentially stressful, a temporary dissolution of established relationships can be also equally important. 3 Even varied size cage-to-cage exposure affects behavioral and biomolecular parameters, thus diminishing research reproducibility. 36 It is also known that if the daily routine is disrupted or if resources such as food or resting spaces are limited, the hierarchy becomes disestablished and the number of aggressive interactions increases.
Measurement of SPA
The gravimetric method described by Biesiadecki et al. 37 was used for measuring SPA (experimental set-up in Figure 1). Technical details of the method are provided in the Supplementary material online. SPA recordings were performed for 19 continuous hours (12 h of dark period and 7 h of light period), starting at 11:00 a.m. and continuing until 6:00 a.m. All experimental procedures (e.g. cage cleaning, weighing) occurred between 6:00 a.m. and 10:00 a.m. No animal manipulation was done outside this time range to avoid human presence, which could disturb rodents in their daily routines. 38
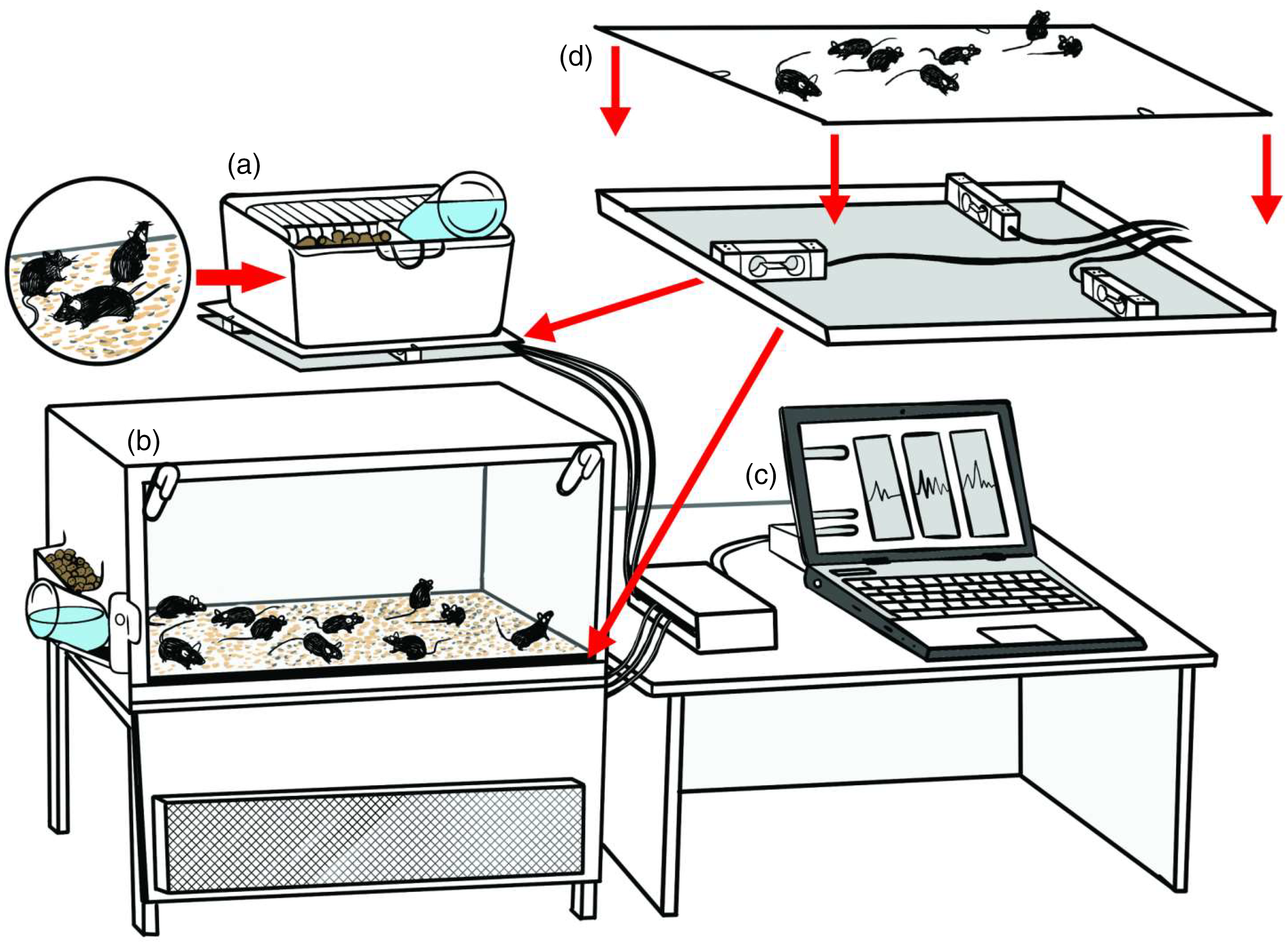
Spontaneous physical activity of mice was gravimetrically measured in a cage with standard stocking density (88 cm2 per animal (a)) and cage with low stocking density (320 cm2 per animal (b)). Both housings operate as a balance, where activities produced by mice induce a force on the platform that can be registered (by load cells) as a change in weight. As exemplified in (c), output signals from load cells were captured by an acquisition system for amplifying and conditioning the signals with sampling frequency of 200 Hz. In both housing types, three load cells of similar full scale (2 kg, PWZL, MK Controle e Instrumentação, Brazil) were coupled in a triangular configuration and fixed between two iron platforms (d).
Characterization of body composition
Ten mice per group were fasted overnight, and euthanasia was performed by cervical dislocation followed by decapitation in the shortest possible time. Cervical dislocation was performed by a well-trained executor with demonstrated experience in this technique. The remaining animals were used in another investigation. The visceral fat depots were removed and weighed. The brown adipose tissue of the interscapular region was rapidly dissected, cleaned of tissue debris and weighed. The eviscerated carcass was weighed before and after dehydration at a temperature of 100°C. The dehydrated carcass was crushed in the presence of organic solvent (benzene), and again dehydrated at a temperature of 100°C. Water and fat mass were obtained from the weight differences after dehydration and fat removal. The fat-free mass was defined as the remaining amount of carcass. The results were expressed as a percentage of the eviscerated carcass, following a method previously described. 11
Statistical procedures
All data are presented as mean ± standard deviation. According to the Shapiro–Wilk test, the data fit a normal distribution and homogeneity of variance was confirmed by Levene’s test. The sample size was determined with an a priori power analysis by G*power software. It was calculated that a sample size of 60 (30 mice per group) would be required to detect a clinically important difference in SPA using a two-way analysis of variance (ANOVA) test at the 5% significance level with 86% power, assuming an effect size of 0.4 (moderate). Measurements – such as SPA, body mass, food and water intake, taken multiple times over four weeks – were analyzed by two-way ANOVA to determine the effects of housing conditions (SSD versus LSD) and experimental time (four weeks). Moreover, another ANOVA layout was designed to determine whether SPA behavior (fluctuation throughout the day) would be influenced by housing conditions. For this, all SPA data obtained over the entire period of observation (including both recordings at dark and at light periods) were plotted for evaluating the interaction between housing conditions and time of day. Newman–Keuls post-hoc test was chosen. An independent t test was also used to compare the differences between groups for the variables measured at the end of the experiment (body composition). The significance level was set at p < 0.05 in all cases. The effect size (Cohen’s d) for pair-wise comparisons was determined by the formula: (mean of SSD group – mean of LSD group)/pooled standard deviation.
Results
SPA was higher for the LSD group than the SSD group when considering the entire four-week period (Figure 2). Detailed ANOVA results and post-hoc comparisons are shown in Figure 2. An effect of time of day was found on SPA data, indicating that SPA fluctuates in a pattern of peaks and troughs throughout the day, as expected for nocturnal rodents. In addition, SPA undergoes a divergent fluctuation throughout the day when comparing SSD and LSD groups, although not differing in highest (occurring at dark period) and lowest (occurring at light period) values over the day.
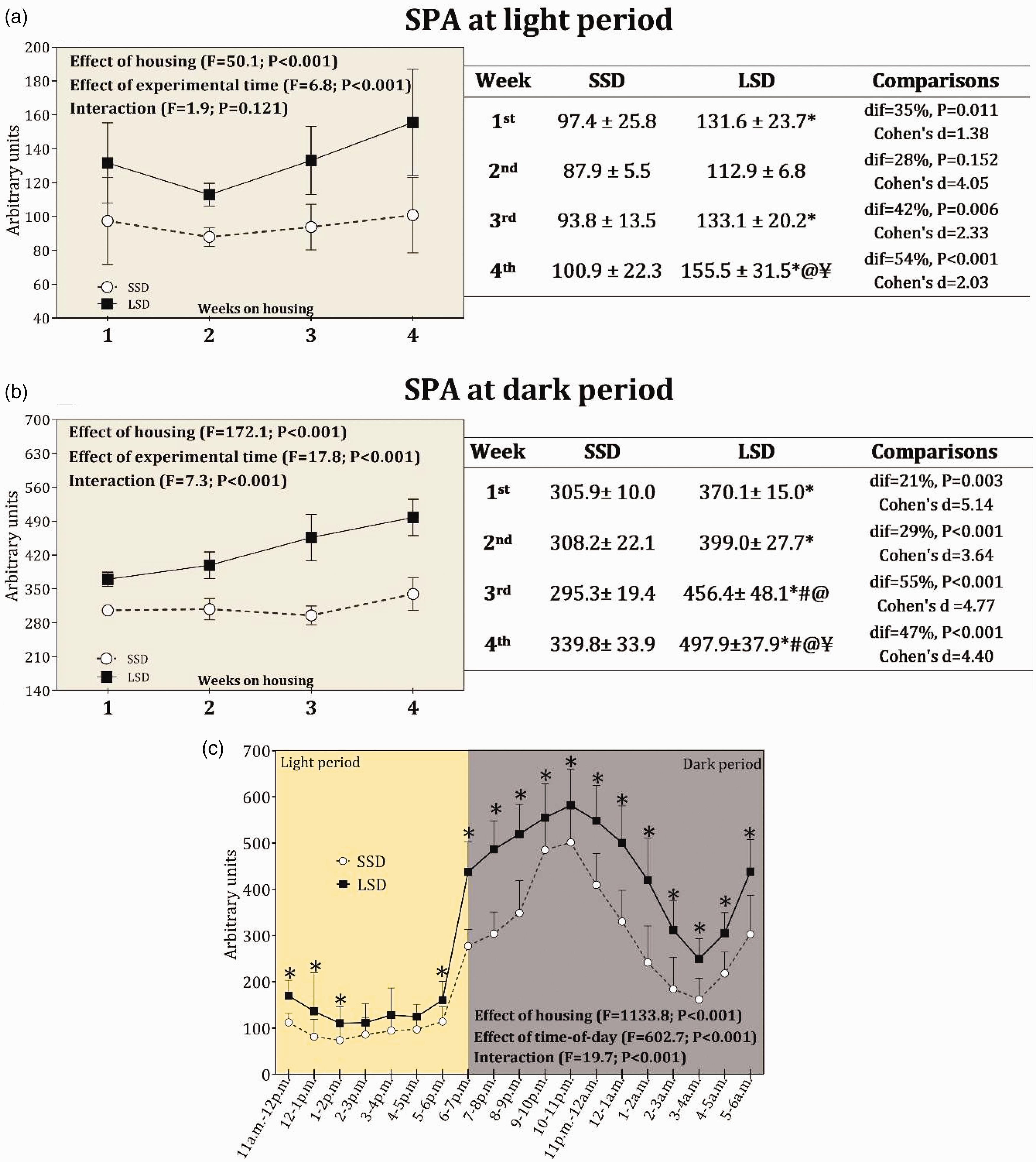
Spontaneous physical activity (SPA) records at light period (a) and dark period (b) over four weeks for standard stocking density (SSD; 88cm2 per animal) and low stocking density (LSD; 320cm2 per animal). SPA data are in arbitrary units for mice housed at the density of 15 animals per cage. The dif represents the percentage differences between groups where the SSD group has been set at 0%. (c) The time-of-day variation of SPA for each hour time interval from 11:00 a.m. to 6:00 a.m., totaling seven hours of light period (11:00 a.m. to 6:00 p.m.) and 12 hours of dark period (6:00 p.m. to 6:00 a.m.). This analysis considered all SPA data obtained over the entire four-week period. The symbol (
Regarding food and water intake, detailed ANOVA results and post-hoc comparisons are presented in Figure 3. ANOVA findings revealed a significant effect of housing conditions on normalized food and water intake, showing that mice kept in LSD present higher food and water consumption than mice kept in SSD, considering the entire eight-week period (Figure 3). ANOVA detected an effect of experimental time on normalized food and water intake, demonstrating that consumption of food and water decreased over the course of four weeks. ANOVA did not show a significant interaction between housing conditions and experimental time on normalized food and water ingestion (Figure 3), indicating that the decline of food and water intake over time was similar in both groups.
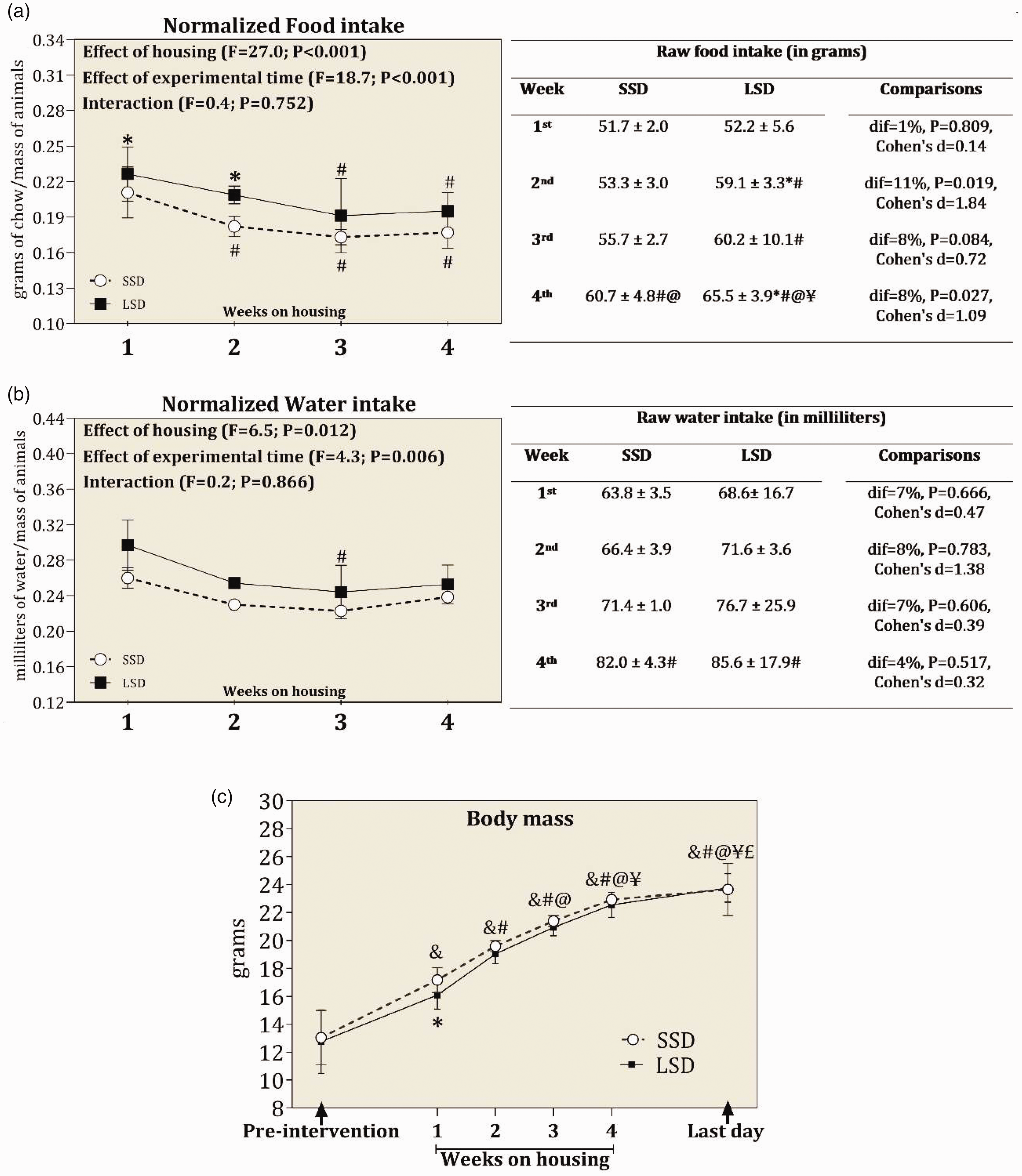
(a) and (b) Normalized and raw data of food and water intake recorded over the course of four weeks for standard stocking density (SSD; 88cm2 per animal) and low stocking density (LSD; 320cm2 per animal). (c) The body mass from pre-intervention to the last day of study. The dif represents the percentage differences between groups where the SSD group has been set at 0%. The symbol (
Data from body composition analyses at the end of the study are shown in Figure 4. No differences were found between SSD and LSD groups concerning the water of carcass, fat mass and fat-free mass, as exhibited in Figure 4. Mice kept in LSD showed reduced visceral fat depots compared with mice kept in SSD (t = −2.2; p = 0.039; Cohen’s d = 1.01) (Figure 4). No differences were found between SSD and LSD groups for brown adipose tissue (t = 0.0; p = 0.948; Cohen’s d = 0.03) (Figure 4).
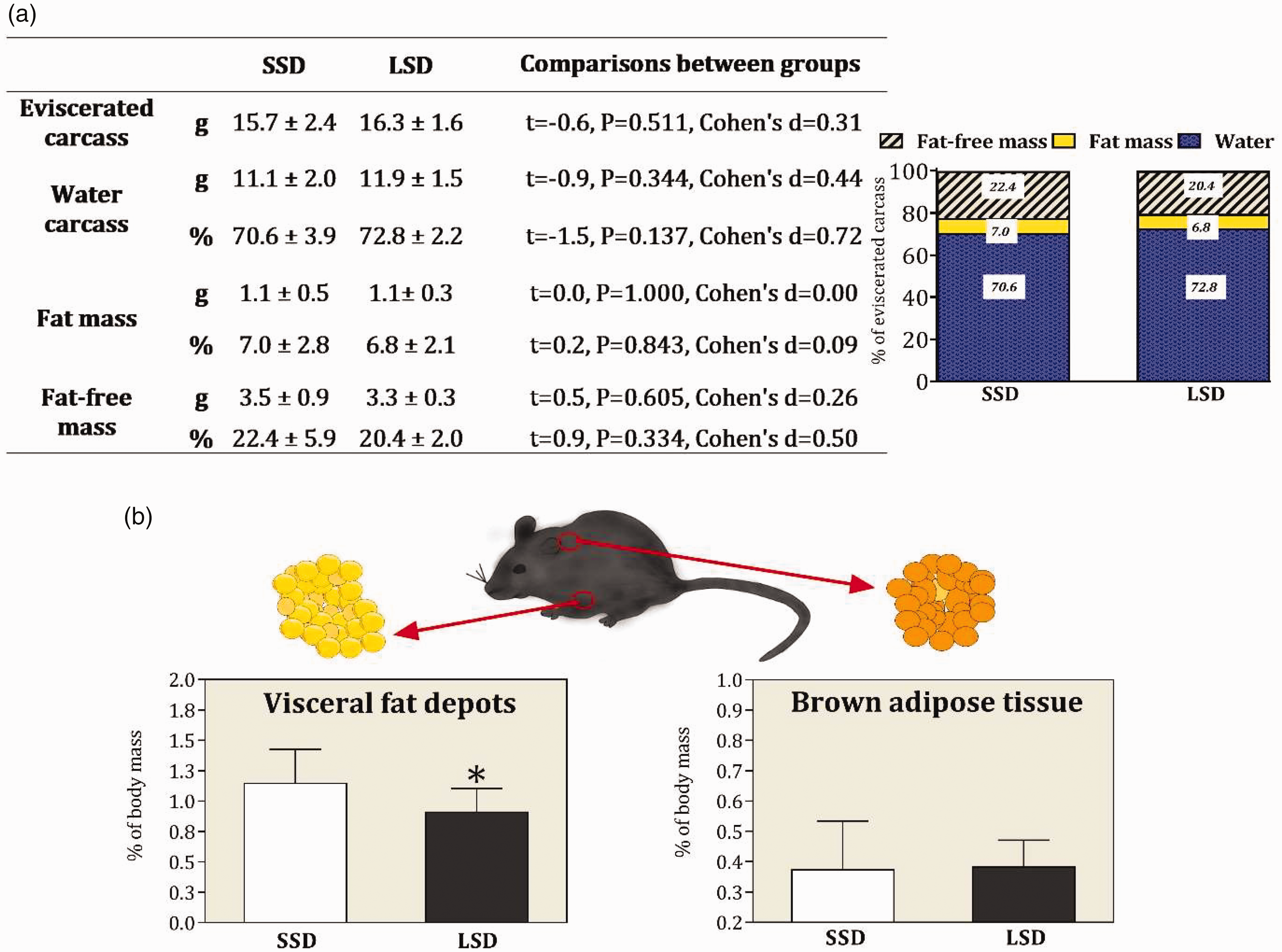
(a) The eviscerated carcass, water, fat mass and fat-free mass of carcass in absolute (g) and distribution of body components (%) for standard stocking density (SSD; 88 cm2 per animal) and low stocking density (LSD; 320 cm2 per animal) at the end of study. (b) The mass of visceral fat depots and brown adipose tissue of the interscapular region in relation to body mass. The symbol (*) indicates difference when compared with the SSD group.
Discussion
To the best of our knowledge, this is the first study to show that SSD leads male mice to be physically inactive already in adolescence. Furthermore, mice kept in SSD already have visceral fat accumulation before reaching adulthood. These findings help to clarify important questions in rodent studies, where the animals are used as a model for ‘normal healthy humans’. Our findings that mice kept in SSD already have an abnormal visceral fat deposition at the end of adolescence suggest that housing conditions affect certain end-points regarding body composition. This is worrying, given that, in a wide variety of studies, interventions are initiated when mice reached 8–12 weeks of age (at the beginning of adulthood), indicating that animals started from an unhealthy baseline status when using stocking density conditions as in this study. Our findings also provide an explanation as to why rodents housed in standard housing conditions have impairments in health/physical fitness status.5,39–41 In agreement with these discussions, it has been posited that sedentary rodents housed in ‘standard’ conditions may be reasonable models for the study of obesity in humans; however, the translatability of these animal models to healthy active humans has been questioned.5,40
Researchers should consider the impact of physical inactivity on their research models, particularly when modeling healthy active adolescent and/or adult humans. Moreover, veterinarians and researchers should establish a physical activity environment after animals are weaned or arrived from the breeding laboratory. An early enhancement of SPA may be important to the maintenance of a healthy phenotype in laboratory rodents. We consider favorable the use of other ways to stimulate physical activity in laboratory rodents, such as the use of running wheels, as an alternative to increasing cage size. Several adaptations can be achieved when a running wheel is introduced into the home cage; nevertheless, caution is required in interpreting measures obtained from running wheels. 42 Other voluntary strength training methods, such as burrowing (digging a substrate out of a tube) and unloaded tower climbing, 43 should be encouraged for stimulating physical activity in rodent models. The inclusion of environmental enrichment can also reduce anxiety, in addition to promoting cognitive and motor stimulation in rodents.44,45 Regarding our study, it is plausible to consider that our SPA values may be diminished, since we did not insert any material or toys inside the cage. Thus, higher SPA values would be expected if enrichment had been included.
An important observation was that adolescent male mice kept in LSD exhibited higher food and water consumption (when considering the entire four-week period) than mice kept in SSD, reinforcing the idea that housing conditions (and SPA) affect energy metabolism. It is important to note that an increase in SPA may be one reason (but not the only one) why mice kept in LSD were resistant to fat accumulation (exhibiting a lean phenotype). Our experimental design does not allow us to make statements about the evidence for causation, but a reduced visceral fat in mice kept in LSD suggests that muscle contractions generated by activities included within SPA are not negligible in promoting energy expenditure. This is further supported when we consider the absence of any exercise-induced training effect and no difference in brown adipose mass, which is linked to non-shivering thermogenesis. 46
For achieving a consistent measure of SPA, researchers must consider not only ‘what’ is measured but also ‘how’ and ‘where’ measurement occurs.15,47 One must be aware that SPA does not represent just a locomotion measure, obtained in an environment unfamiliar to the animals. Instead of this, SPA should be obtained from animals in their natural habitat (or home cage), and preferably in long-term monitoring without disturbing the normal life and behavior of animals. In an attempt to cover all these aspects in a single method, the gravimetric-based method was adopted in this study, in which all movements produced by mice can be registered as a change in weight. 11 In our experiments, SPA can be determined in animals in their natural settings (in loco) without the need for removing them from their own home cage (thus without obstructing their day-to-day life). With this, SPA can be regularly recorded throughout the experimental period (and not only for an isolated day), thus ensuring data consistency.
Considering that the environment is the most important factor that influences SPA, our efforts were focused on developing an environment that encourages physical active to help rodents move more. Our main challenge was to ensure that mice kept in LSD would have the opportunity to move with less restriction. We achieved this by manipulating cage dimensions. While in the SSD group a floor space of 88 cm2 was available per animal, LSD mice were housed with a floor space of 320 cm2 per animal, increasing the individual radius for movement/physical activity more than three-fold compared with standard requirements. While this difference in housing density could be considered a limitation, another confounding factor (i.e. difference in social interactions) would arise if more mice were added into the cage (in an attempt to match housing density). For the reasons given above, the same number of mice per cage (n = 15) was fixed in an attempt to match the amount of social interactions. An open question to be answered in future research is whether with a similar housing density, animals kept in a large cage (in terms of space) would still have better physical fitness than mice in a small cage. One way to investigate this would be to keep 55 animals (instead of 15) in our ‘large cage’ to establish the housing density similar to those found in SSD (88 cm2 per rodent). Had we been asked to make a guess, we would predict that SPA would be limited with more animals living in a large cage, regardless of more floor space.
We recognize that our study has limitations. We have no time course of the assessments, especially in analyzing mice kept in LSD that returned to SSD at later time points. Considering that excess adiposity tends to track into adulthood, it is of importance that future studies further investigate whether mice housed since weaning in a large cage would have better health in advanced ages or prolonged lifespan than mice housed in SSD. It would also be interesting to evaluate whether housing space could affect metabolic health markers 48 such as inflammation, insulin resistance, glucose metabolism and dyslipidemia, which are linked to fat deposition, as already reported here. A final point to be noted is that these results were obtained with male mice, and care must be taken in generalizing these results to female rodents. As a secondary benefit from this study, we thought that our animal-based model could be useful in providing reflections on reduced physical activity and health-related consequences in periods of COVID-19 lockdown measures, especially in children and adolescents.49–53 We believe that housing conditions offering smaller radius for movement could simulate a scenario of very low physical activity (as happens in times of COVID-19 lockdown measures), while a large cage with higher area per animal could emulate an environment that favors physical activity.
Conclusion
We have shown that male mice kept in SSD (88 cm2 per animal) had a more pronounced reduction in SPA than mice housed in LSD (320cm2 per animal). An early exposure to reduced physical activity (already in adolescence) in male mice housed in SSD resulted in greater visceral fat accumulation before reaching adulthood. From a practical point of view, our findings suggest that an environment enabling/stimulating physical activity should be established for rodents as early as possible.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772211065915 - Supplemental material for Early-life mice housed in standard stocking density reduce the spontaneous physical activity and increase visceral fat deposition before reaching adulthood
Supplemental material, sj-pdf-1-lan-10.1177_00236772211065915 for Early-life mice housed in standard stocking density reduce the spontaneous physical activity and increase visceral fat deposition before reaching adulthood by Pedro PM Scariot, Claudio A Gobatto, Emanuel EC Polisel, Ana EC Gomes, Wladimir R Beck and Fúlvia B Manchado-Gobatto in Laboratory Animals
Footnotes
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the São Paulo Research Foundation – FAPESP (grant numbers: 2019/05115-7, 2014/10336-9, 2015/01362-9, 2015/00272-6, 2017/10201-4, 2018/07099-6, 2019/08148-3) and the National Council for Scientific and Technological Development – CNPq (grant numbers: 307395/2013-8, 302827/2015-3, 307718/2018-2, 308117/2018-2).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
