Abstract
Spontaneous infections of the preputial glands represent overlooked health problems in mice that could raise welfare concerns and potentially confound scientific experiments. Agents involved in preputial gland infections have rarely been investigated, with opportunistic pathogens of laboratory animals usually detected in inflamed preputial glands. The aim of this study was to investigate the prevalence of bacterial infection in the preputial glands and the relationship between haematological and pathological changes and infection status. We analysed 40 preputial glands from 20 one-year-old C57BL/6NCrl male mice by using bacteriology, haematology and pathology. Bacteria were isolated from 16/20 (80%) mice, for a total of 32/40 (80%) examined preputial glands. Enterobacter cloacae, Pasteurella spp., Klebsiella spp. and Staphylococcus aureus were identified in 35%, 17.5%, 15% and 12.5% of the examined glands, respectively. Preputial gland inflammation was identified in 29/40 (72.5%) glands and was classified as chronic interstitial adenitis in 27 cases and suppurative adenitis in the remaining two glands. No haematological changes were found in mice with infected glands. Histologically, the presence of intralesional bacteria, intraluminal necrotic material, intraluminal keratin accumulation, interstitial inflammatory cell infiltrate and granulocytes (intraluminal and/or interstitial), along with total inflammatory score and total histopathological score, were significantly increased in infected glands and correlated with the bacterial load. Most severe inflammatory changes were identified after S. aureus infection, while ductal hyperkeratosis was significantly increased in glands infected with Klebsiella spp. In conclusion, preputial gland infection was a common event in one-year-old C57BL/6NCrl mice, and bacterial load correlated with pathological findings, while systemic effects were not highlighted by haematology.
Introduction
In mice, preputial glands are paired subcutaneous glands located laterally and cranially to the penis. These specialised holocrine sebaceous glands are known to play a key role in reproduction and other pheromone-based social behaviours. 1 Reported preputial gland pathological changes include ductal ectasia, hyperkeratosis, cysts, atrophy, hyperplasia, mineralisation, osseous metaplasia, inflammation and benign and malignant neoplasms. 2 While most preputial gland studies dealt with aging-associated macroscopic and histological abnormalities in different mouse strains, few articles have focused specifically on inflammatory processes, underlying bacterial infections that affect these glands and the potential systemic impact on the overall health of the affected animals. According to these reports, preputial gland adenitis is a condition that deserves to be investigated further with regard to its impact on animal welfare and scientific outcome. This pathological condition, when clinically observed, is a painful lesion characterised by uni- or bilateral subcutaneous enlargement adjacent to the prepuce, which can result in ulceration of the overlying skin and systemic consequences such as fever or amyloidosis as previously reported in mice with preputial gland adenitis caused by Corynebacterium mastitidis.3–12
Several factors, such as the multi-lobular structure of the organ, mouse strain, age, microbiological flora and social and environmental conditions are thought to be implicated in the development of preputial gland inflammation. 11 Preputial gland suppurative adenitis often results from septic complications of fight wounds around the external genitalia,2,6,7 though preputial gland abscesses were also experimentally induced by inoculating Staphylococcus aureus in the penile urethra. 13
Preputial gland suppurative adenitis was identified in 321/17,422 (1.8%) mice of several strains. 13 Preputial gland adenitis was observed in 13/2543 (0.4%) mice during a pathological survey on B6C3F1 mice. 4 Investigations of B6;129 mice revealed severe preputial gland adenitis in 6/50 examined animals. 14 Results from a survey on old mice affected by non-neoplastic diseases revealed that 3% of 16- to 36-month-old C57BL/6J mice housed at the University of Washington were affected by preputial gland cystic adenitis. 8 Finally, a lower overall prevalence (1.4%) of abscesses of preputial glands was observed in a more recent study that involved several mouse strains, such as C3H/W, C3H(wad)/W, C57BL/6W, C57BL/10W, 129S1/SvW, DBA/2W, CBA/W, WOM/W, BN/aW, AKR/W, BALB/cW and A.CA-H2(f)/W. 10
S. aureus was the most prevalent bacterial species isolated from chronic suppurative adenitis of the preputial glands.6,9,13 However, other microorganisms such as Staphylococcus epidermidis, Pseudomonas aeruginosa, Klebsiella pneumoniae, Klebsiella oxytoca, Pasteurella pneumotropica (recently reclassified as Rodentibacter pneumotropicus or R. heylii), Enterobacter cloacae and Escherichia coli were also isolated in pure and mixed cultures from inflamed glands.6,13 More recently, Corynebacterium mastitis was reported in preputial gland suppurative adenitis of oestrogen receptor β knockout (BERKO (129P2/OlaHsd*C57BL/6 Esr2 tm1Unc /Esr2 tm1Unc )) mice. 7 Based on these studies, different bacteria can be detected in preputial glands, but to our knowledge, the specific role that each agent plays on the pathogenesis of preputial gland adenitis has not yet been investigated.
In this context, we examined the preputial glands of 20 one-year-old C57BL/6NCrl male mice with the aim of investigating the relationship between pathological changes of the preputial glands and bacterial isolates, and assessing if systemic effects of the infection occurred. The study design included a thorough macroscopic and histological examination of the preputial glands to evaluate the presence of lesions, bacterial cultures to assess the infection status and haematology to evaluate potential systemic effects of adenitis.
Methods
Animals and husbandry
Twenty one-year-old C57BL/6NCrl male mice were investigated in this study conducted at the Istituto di Ricerche Farmacologiche Mario Negri IRCCS. The mice enrolled in this study belonged to the control group of a previous published study. 15 The research centre adheres to the principles set out in the following laws, regulations and policies governing the care and use of laboratory animals: Italian Governing Law (D.lgs 26/2014; authorisation no. 19/2008-A issued 6 March 2008 by Ministry of Health); Mario Negri Institutional Regulations and Policies providing internal authorisation for persons conducting animal experiments (Quality Management System Certificate – UNI EN ISO 9001:2015 – reg. no. 6121); the NIH Guide for the Care and Use of Laboratory Animals (2011 edition) 16 and the EU directive EEC Council Directive 2010/63/UE. Mice were purchased from Charles River Laboratories (Calco, Italy) and maintained in compatible social groups for one year, with a maximum of four mice per cage, under specific pathogen-free conditions in an individually ventilated caging (IVC) system (GM500; Tecniplast S.p.a, Buguggiate, Italy). Microbiological monitoring was performed on days 60, 120, 180 and 365. Fur, mouth swabs and faecal pellets were sampled from one cage, and a swab was taken from the exhaust prefilter of the air handling unit. The prefilter was changed at each time point after swabbing. All samples were submitted to Charles River Laboratories (Wilmington, MA) for infectious disease polymerase chain reaction (PCR) testing. Intra-cage samples were analysed according to the Mouse Surveillance PRIA panel (on day 60) or the Mouse FELASA Complete PRIA panel (on 120, 180 and 365 days). Prefilters were analysed according to the Environmental (EAD) Mouse Surveillance Plus PRIA panel (on days 60, 120, 180 and 365). 17 Environmental conditions were set at 20–24°C, 12-hour/12-hour light/dark cycle, with the lights on at 7:00 am, relative humidity of 45–65%, 75 air exchanges per hour in the IVCs, which worked in positive pressure mode and 18 air exchanges per hour in the room. Mice were allowed to feed ad libitum autoclaved standard rodent chow (2014 S Envigo/Teklad global diet rodent maintenance, 14% protein) and were provided with filtered autoclaved water. The cages containing 150 g of autoclaved corncob bedding (Follador, Treviso, Italy) were replaced every two weeks or more often if necessary. Animals were inspected daily, and veterinary care was provided when necessary. All animals were euthanised by CO2 exposure and necropsied immediately upon being dead in order to collect preputial gland specimens for bacterial culture and histopathology.
Haematology
At sacrifice, blood was drawn from the heart and immediately placed into tubes containing EDTA. Blood cell count was performed using a laser-based cell counter (Sysmex XT-2000iV, Kobe, Japan) validated to be used in mice to determine platelet numbers, erythroid parameters such as red blood cell count, haemoglobin, haematocrit, total white blood cell counts and number and percentage of each white blood cell type (neutrophils, lymphocytes, monocytes, eosinophils and basophils). Differential leucocyte counts automatically obtained were then matched with findings derived from the microscopic evaluation of May Grunwald Giemsa-stained blood smears. At microscopy, the observation of blood smears allowed differentiation between mature (segmented) neutrophils and immature (band) neutrophils as well as the identification of possible morphological modifications of blood cells.
Pathological examination
At sacrifice, a full necropsy was performed in a biosafety cabinet, and the preputial glands were collected aseptically. Animals were laid on their back and had their abdomen and inguinal region wet with 70% alcohol. Sterile instruments used for dissection and sampling procedures were placed in sterilant solutions for at least 15 minutes (Klercide™ sporicidal low residue peroxide) after each use and in between animals and flushed with sterile water to remove disinfectant residues before being reused. Preputial glands were removed without separating them from the overlying skin to preserve the integrity of the anatomical structures. Preputial glands were then sampled as follows: each lobe was transversally cut 2 mm behind its cranial border, and a swab was rubbed over the cut surface and placed in a tube containing Amies charcoal transport medium for microbiological analysis. Two different sets of sterile instruments were used to minimise the risk of contamination between the two lobes of the gland in each animal. Gross inspection was focused in assessing the following morphological features of the preputial glands: size and colour changes of each lobe, and amount, colour and viscosity of fluid leaked from the glands after cutting the cranial portion.
After removal of the cranial portion for microbiology, the remaining preputial gland lobes (including the overlying skin) were fixed in 10% neutral buffered formalin and cut to obtain three transverse slices, each 2 mm in thickness. After paraffin embedding, tissues were cut in serial sections 4 µm thick and stained with hematoxylin and eosin (H&E) and Gram staining. H&E-stained preputial gland sections were evaluated under a light microscope (DM2500; Leica, Wetzlar, Germany) for the presence of histopathological findings and assessed separately at the level of the ducts (i.e. duct dilation, amount of intraluminal necrotic material and keratin, amount and type of intraluminal inflammatory cells), acini (i.e. atrophy) and interstitium (amount and type of interstitial inflammatory cell infiltrate and fibrosis). The amount of bacteria was additionally recorded. All these features were separately graded according to a four-point scale (where 0 = absent, 1 = mild, 2 = moderate and 3 = severe), except for fibrosis, which was graded using a binary system (0 = absent, 1 = present; Supplemental Table S1). A total inflammatory score (intraluminal inflammatory cells + interstitial inflammatory cell infiltrate) and a total histopathological score (intraluminal necrotic material + intraluminal keratin + intraluminal inflammatory cells + interstitial inflammatory cell infiltrate + fibrosis) was then calculated. The presence of granulocytes in the duct lumen and/or interstitium was recorded as 0 if absent or 1 if present. Gram-stained sections were evaluated to assess the type (Gram positive or Gram negative) and morphology (cocci, bacilli or coccobacilli) of bacteria. Slides were examined and scored independently by two veterinary pathologists with expertise in laboratory animal pathology (C.R. and L.M.) and then reviewed together until consensus was reached.
Bacteriology
Swabs, collected from the cut surface of each lobe, were streaked onto sheep blood agar (Oxoid, Milan, Italy) for bacterial isolation and incubated at 37°C for 24 hours. Bacterial cultures that showed no growth after 24 hours of incubation were incubated for an additional 24 hours before being discarded.
The bacterial load of each specimen was quantified by counting the colony forming units (CFUs) on each plate and graded as follows: 0 = 0 CFUs, 1 ≤ 150 CFUs, 2 = 150–1000 CFUs and 3 ≥ 1000 CFUs.
Bacterial species were identified by observing macroscopic characteristic of colonies and then microscopic shape after Gram staining. Moreover, specimens were processed with conventional biochemical methods (i.e. coagulase, catalase and oxidase) and streaked onto MacConkey agar (Oxoid) and methylene blue agar (Oxoid), both selective and differential media for Gram-negative bacteria (E. coli and other Enterobacteriaceae). Mannitol salt agar (Oxoid) was used as a selective and differential medium for identification of the members of the genus Staphylococcus based on their ability to ferment mannitol. Finally, for microorganism identification to the species level, the miniaturised biochemical test API System® (BioMerieux, Marcy-l’Étoile, France) was used.
Statistical analysis
The Kruskal–Wallis test followed by Dunn’s multiple comparison test were used to detect differences in histopathological findings among glands with different bacterial isolates. The association between the frequency of macroscopic and histological findings and the infection status was evaluated using Fisher’s exact test. Spearman’s rho test was employed to analyse the correlation between the number of isolated bacterial colonies and selected histopathological findings, the total inflammatory score, and the total histological score assigned to each gland. Data were analysed using Graph Pad Prism v9.0 (GraphPad Software, San Diego, CA). p-Values <0.05 were considered statistically significant.
Results
Health monitoring
Health monitoring revealed the presence of S. aureus and K. oxytoca from intra-cage sampling on day 60 (no more tested at subsequent time points) and in the prefilters of the air handling unit (S. aureus on days 60, 120 and 180; K. oxytoca on days 60 and 180).
Bacteriology
Bacterial isolates were obtained from 32/40 (80%) examined preputial glands, for a total of 16/20 (80%) mice, which were all bilaterally infected. The following bacterial species were identified: E. cloacae in 14/40 (35%) glands, Pasteurella spp. in 7/40 (17.5%) glands, Klebsiella spp. in 6/40 (15%) glands and S. aureus in 5/40 (12.5%) glands. Among the positive specimens, plates contained no more than 150 colonies in five cases, 150–1000 colonies in 14 cases and more than 1000 colonies in 13 cases. Different bacterial isolates were obtained from the two paired glands in 8/16 (50%) mice that resulted bilaterally positive at bacteriology. Pasteurella spp. and S. aureus were identified in the paired glands of 2/8 mice, Pasteurella spp. and E. cloacae in 3/8 mice, E. cloacae and Klebsiella spp. in 2/8 mice and E. cloacae and S. aureus in 1/8 mice.
Haematology
Haematological results obtained from each mouse are reported in Supplemental Table S2 and summarised in Supplemental Table S3 according to the infection status of the preputial glands. No morphological abnormalities were detected in the blood smears. No significant differences in haematological parameters were observed between animals with non-infected and infected glands, and among animals with glands infected with different bacterial isolates.
Pathology
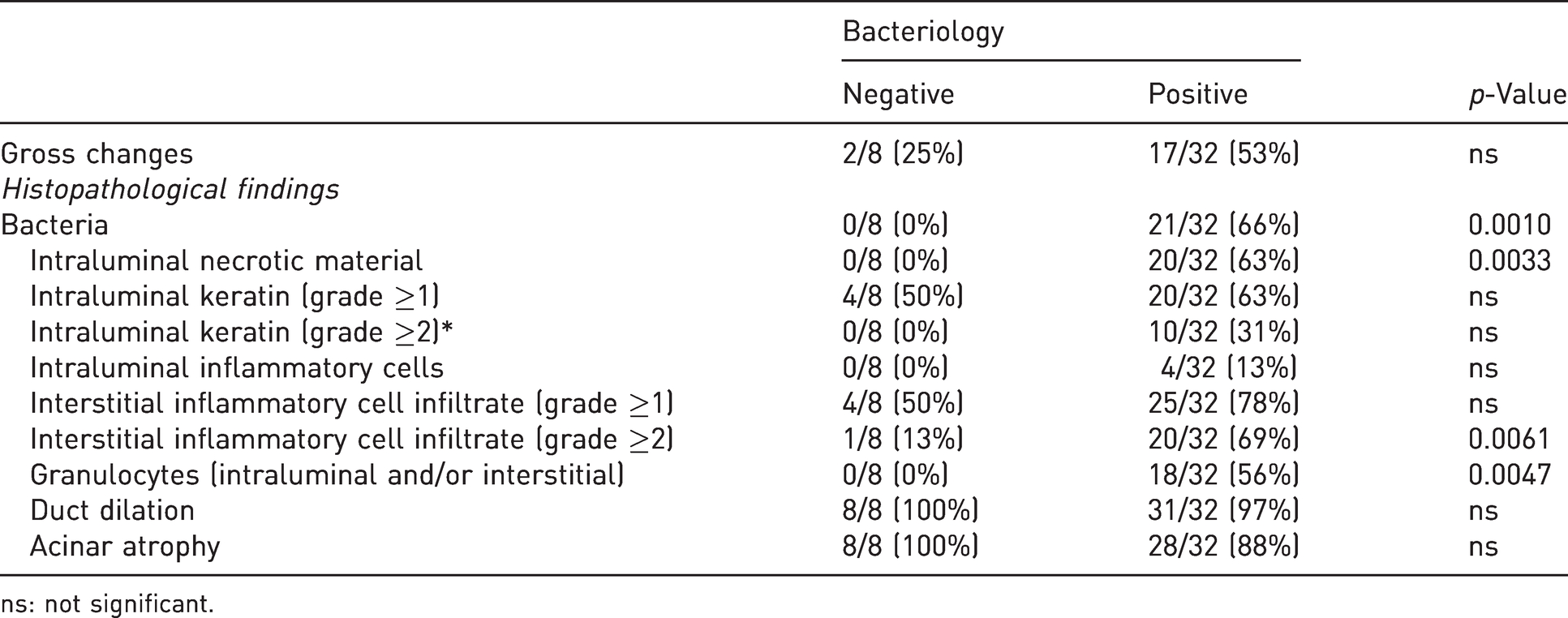
Gross and microscopic pathological results of each gland are reported in Supplemental Table S4. Results of the gross examination are summarised in Table 1. Gross changes were observed in 19/40 (47.5%) examined glands, and of these grossly affected glands, 17 (89.5%) were positive at bacteriology. Conversely, 21/40 glands (52.5%) were grossly unremarkable, but of these, only six (28.6%) were negative at bacteriology. Two animals had bilateral preputial gland enlargement that in one gland was associated with leakage of abundant clear-to-grey/yellowish fluid released by the sectioned gland. This abundant glandular secretion was also observed in other eight glands that were found to be normal at the external inspection. A creamy yellowish material, consistent with purulent exudate, was observed after sectioning of six glands, whereas one gland contained a dry solid whitish material, consistent with keratin debris. Among the 32 infected glands, only 17 (53%) were grossly affected, while only 2/8 (25%) non-infected glands appeared grossly enlarged. Keratin and purulent exudate were observed only in association with bacterial infection, while enlargement and abundant glandular secretion were also identified in non-infected glands. Overall, no significant differences in the frequency of gross lesions between infected and non-infected glands were present.
Gross pathological changes of preputial glands according to bacterial isolates.
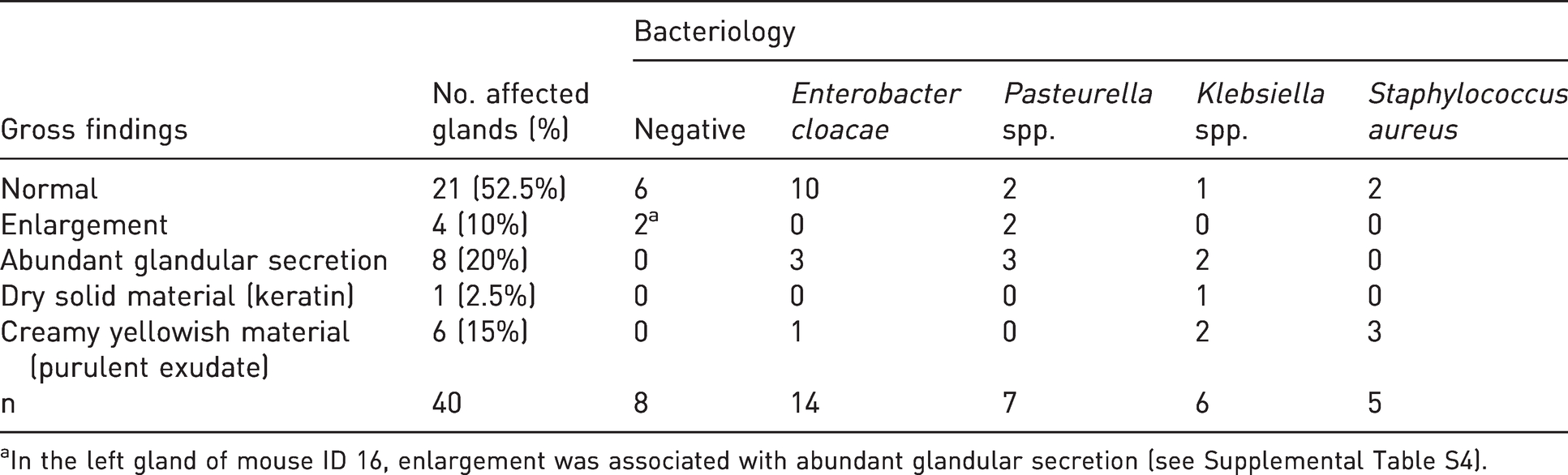
aIn the left gland of mouse ID 16, enlargement was associated with abundant glandular secretion (see Supplemental Table S4).
Results of the histopathological evaluation are summarised in Tables 2 and 3. Histologically, bacteria were detected in H&E-stained sections in 21/32 infected glands (6/14 E. cloacae, 5/7 Pasteurella spp., 5/6 Klebsiella spp. and 5/5 S. aureus culture-positive cases). Bacterial organisms were mainly located within the glandular lumen admixed with intraluminal necrotic debris, keratin and/or inflammatory cells (mainly granulocytes). No bacteria were found in the eight culture-negative glands. The amount of bacteria detected histologically within the glands was highest in glands infected with Klebsiella spp. and S. aureus. Results obtained from the evaluation of Gram-stained slides were overall consistent with the bacteriological results (Supplemental Table S3), although in two glands infected with E. cloacae, in four glands infected with Pasteurella spp. and in five glands infected with Klebsiella spp., variable amounts of Gram-positive bacteria were also present, usually as a minor population, except for one case of Pasteurella spp. that had 70% Gram-positive rods and three glands infected with Klebsiella spp. that had 50–90% Gram-positive coccobacilli. Gram-positive cocci were observed in 5/5 glands infected with S. aureus in the absence of other bacteria. Representative pictures of bacteria detected in glands infected with E. cloacae, Pasteurella spp., Klebsiella spp. and S. aureus are shown in Supplemental Figure S1.
Summary of histopathological findings detected in preputial glands according to bacterial isolates.
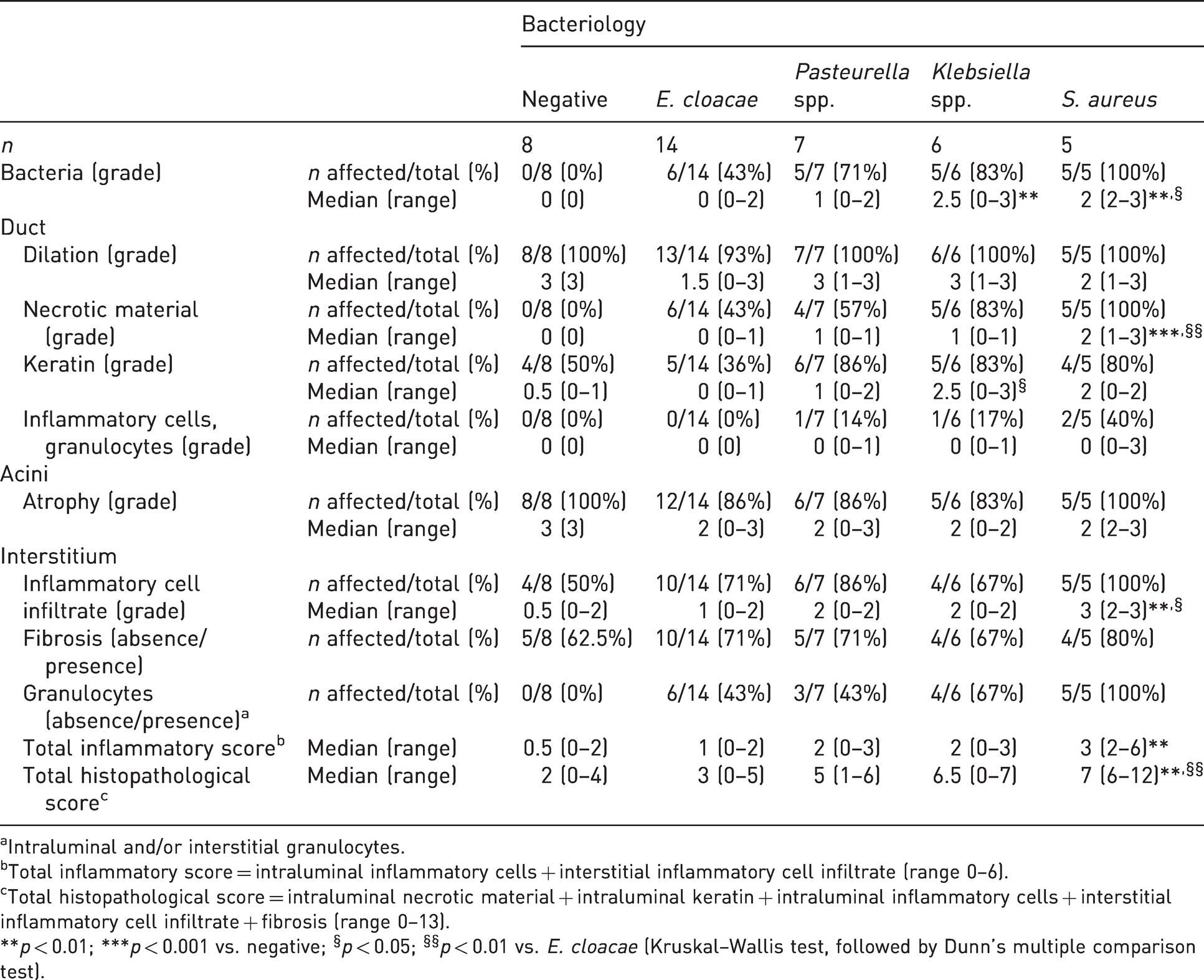
aIntraluminal and/or interstitial granulocytes.
bTotal inflammatory score = intraluminal inflammatory cells + interstitial inflammatory cell infiltrate (range 0–6).
cTotal histopathological score = intraluminal necrotic material + intraluminal keratin + intraluminal inflammatory cells + interstitial inflammatory cell infiltrate + fibrosis (range 0–13).
**p < 0.01; ***p < 0.001 vs. negative; §p < 0.05; §§p < 0.01 vs. E. cloacae (Kruskal–Wallis test, followed by Dunn’s multiple comparison test).
Histopathological morphological diagnoses of preputial glands according to bacterial isolates.
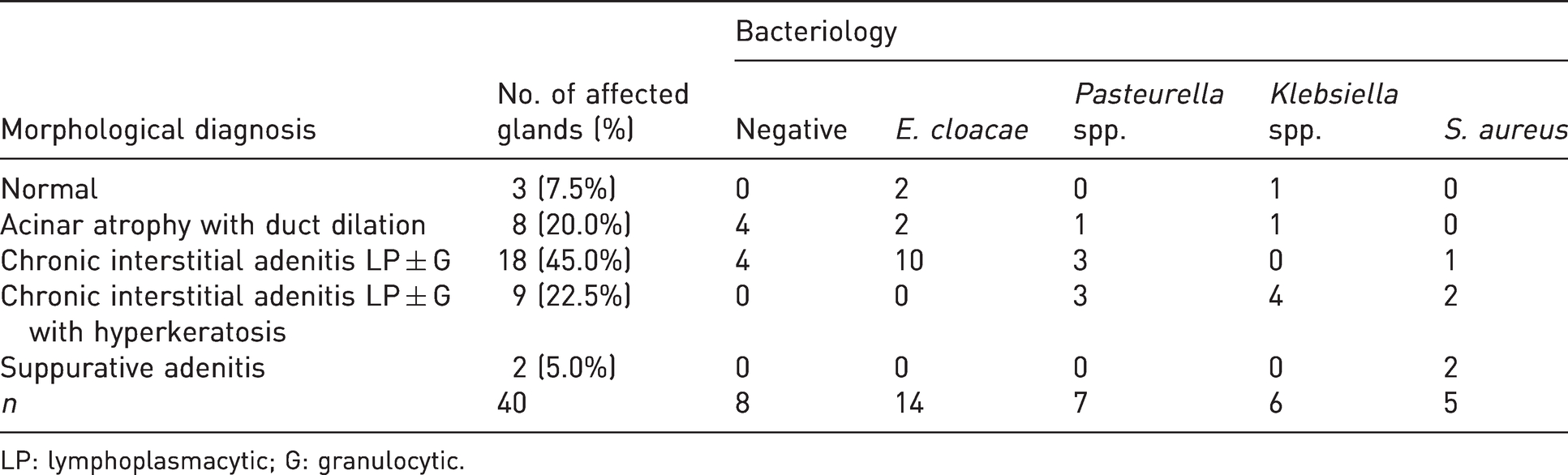
LP: lymphoplasmacytic; G: granulocytic.
Histopathological changes of the preputial glands were observed at the level of the ducts, acini and interstitium. The most common findings were duct dilation and acinar atrophy, detected in 39/40 and 36/40 (97.5% and 90%) examined glands, respectively. These two findings were usually associated and present with similar frequency regardless of the infection status.
Presence of interstitial inflammatory cells was observed in 29/40 (72.5%) glands. Interstitial infiltrates were composed of lymphocytes and plasma cells (in all cases) and granulocytes (in 17/29; 59%). While interstitial lymphoplasmacytic infiltrates were also detected in 4/8 negative glands, granulocytes were only observed in infected glands, with the highest frequency found in glands infected with S. aureus (100%). Intraluminal granulocytes were only occasionally identified (4/40; 10%), and the grade was slight in one case of glands infected with Pasteurella spp. and Klebsiella spp., and moderate to severe in two cases of glands infected with S. aureus. The grade of interstitial inflammatory cell infiltrate and the total inflammatory score were significantly increased in glands infected with S. aureus compared to glands that tested negative. Intraluminal necrotic material was detected in 20/40 (50%) glands and only in infected glands. In glands infected with E. cloacae, Pasteurella spp. and Klebsiella spp., it was mild in severity, while in gland infected with S. aureus, it ranged from mild to severe, and it was significantly increased compared to gland that tested negative and glands infected with E. cloacae.
Intraluminal keratin accumulation, resulting from hyperkeratosis of the ductal epithelial lining, was observed in 24/40 (60%) examined glands, ranging from mild to severe. While mild keratin accumulation was observed in both negative and infected glands, a moderate to severe degree of keratin accumulation was only observed in glands infected with Pasteurella spp., Klebsiella spp. and S. aureus and was significantly increased in Klebsiella spp. compared to negative glands. Fibrosis was present in 28/40 (70%) of examined glands. It was always mild in severity, and it present with similar frequency regardless of the infection status.
The total histopathological score was significantly increased in glands infected with S. aureus compared to negative glands and those infected with E. cloacae, and there was a progressive increase of this score across negative glands and those infected with E. cloacae, Pasteurella spp., Klebsiella spp. and S. aureus.
Based on histological findings, a morphological diagnosis was eventually made for each gland, according to the parameters reported in Supplemental Table S5. Preputial glands were thus classified as follows: normal, acinar atrophy with duct dilation, chronic interstitial adenitis lymphoplasmacytic (LP) ± granulocytic (G), chronic interstitial adenitis LP ± G with hyperkeratosis and suppurative adenitis (Figure 1). Frequency of morphological diagnoses and association with each bacterial isolate are reported in Table 3. Among the 40 preputial glands, only three (7.5%) were considered histologically normal, but two were positive for E. cloacae and one was positive for Klebsiella spp. Acinar atrophy with duct dilation was diagnosed only when there were no concurrent signs of inflammation, as it occurred in 8/40 (20%) glands. Four out of these eight atrophic glands were positive at bacteriology (two for E. cloacae, one for Pasteurella spp. and one for Klebsiella spp.). Chronic interstitial adenitis (LP ± G) was observed in 27/40 (67.5%) glands, and in nine of these, it was associated with hyperkeratosis. Hyperkeratosis was only detected in glands infected with Pasteurella spp., Klebsiella spp. and S. aureus. Suppurative adenitis was present in 2/40 glands and only in glands infected with S. aureus. In both cases, it was associated with surrounding suppurative panniculitis. Overall, preputial gland inflammation (including chronic interstitial adenitis and suppurative adenitis) was histologically observed in 29/40 (72.5%) examined glands.
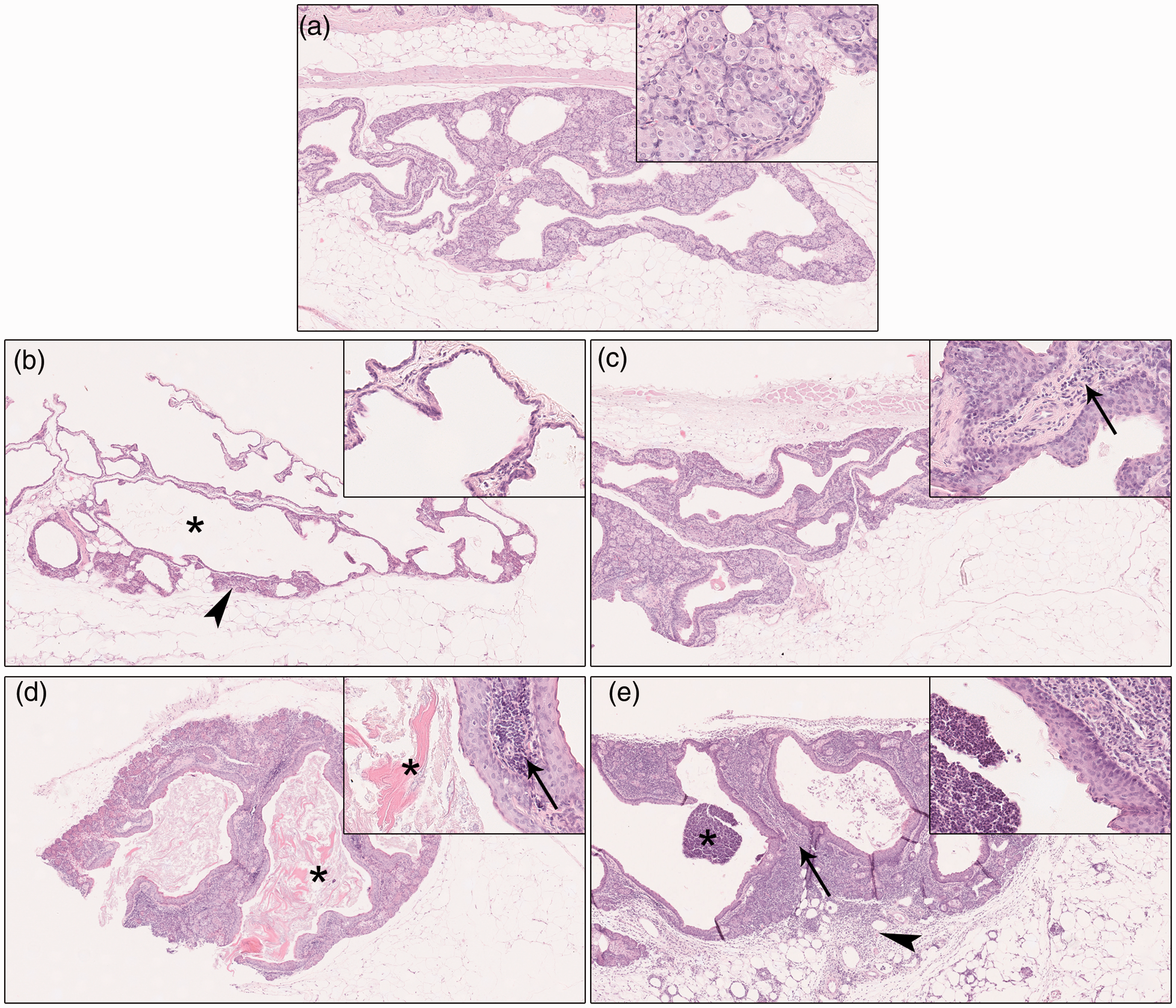
Classification of preputial glands based on histological findings. (a) Normal preputial gland. (b) Acinar atrophy with duct dilation: reduction of the gland parenchyma (arrowhead) associated with duct dilation (asterisk) and absence of concurrent inflammation. (c) Chronic interstitial adenitis: the gland interstitium is infiltrated by lymphocytes, plasma cells±granulocytes (inset, arrow), occasionally associated with interstitial fibrosis. (d) Chronic interstitial adenitis with hyperkeratosis: characterised by interstitial inflammation (inset, arrow) and marked accumulation of luminal keratin (asterisks). (e) Suppurative adenitis: neutrophilic inflammatory infiltrate within the interstitium (arrow) and duct lumen (asterisk). The inflammatory infiltrate extends into the surrounding adipose tissue (panniculitis) (arrowhead). Hematoxylin and eosin, 5× magnification. Insets: 20× magnification.
Associations between bacteriological results and pathological findings
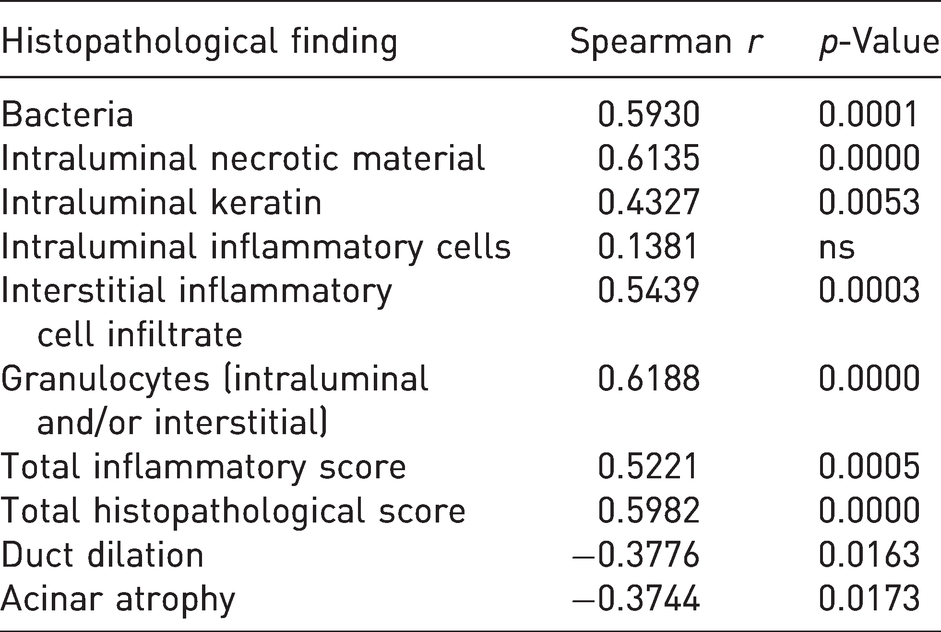
Fisher’s exact test revealed that the frequency of intralesional bacteria, intraluminal necrotic material, interstitial inflammatory cell infiltrate (from moderate to severe) and the presence of granulocytes within the duct lumen and/or interstitium was significantly increased in infected glands as compared to non-infected glands. Frequency of gross changes was not significantly different between non-infected and infected glands (Table 4). Additionally, significant positive correlations were found between the amount of isolated bacterial colonies and several histological findings (intraluminal necrotic material, intraluminal keratin, interstitial inflammatory cell infiltrate, granulocytes (intraluminal and/or interstitial), total inflammatory score, total histopathological score). Only the presence of intraluminal inflammatory cells was not correlated with the amount of isolated bacterial colonies. Acinar atrophy and duct dilation were negatively correlated with the amount of isolated bacterial colonies (Table 5).
Frequency of macroscopic and histopathological findings in non-infected and infected preputial glands.
ns: not significant.
Correlation between number of isolated bacterial colonies (grade) and histopathological findings.
Discussion
In this study, we investigated preputial glands from one-year old C57BL/6NCrl males by using bacteriological, haematological and pathological methods. Our aim was to assess the prevalence of bacterial infection, characterise the bacterial species present in these accessory sex glands and evaluate potential correlations between infection status and haematological and pathological changes in order to understand the potential impact of preputial gland infection on animal health.
Most remarkably, 80% of the examined mice were found to be positive at bacteriology. E. cloacae was the most prevalent bacterial species isolated from preputial glands (35%), and other infectious agents included Pasteurella spp. (17.5%), Klebsiella spp. (15%) and S. aureus (12.5%). Thus, in accordance with the literature, preputial gland infection was found to be determined by opportunistic pathogens also in the present study.6,9,13 E. cloacae, Klebsiella spp. and Pasteurella spp. are Gram-negative bacteria often harboured by healthy immunocompetent rodents as part of the gut microbiota.6,18 Pasteurellaceae, mainly Rodentibacter pneumotropicus, were also commonly isolated from the oral cavity, conjunctiva, urinary bladder and genital tracts of conventional and barrier-maintained rodents,6,19,20 while S. aureus is a Gram-positive bacteria commonly recovered from the skin, nasopharynx and intestine of laboratory mice.6,9
The high prevalence of preputial gland infections detected in our study is not in line with previous published reports where a lower prevalence of this condition was observed in laboratory mice, ranging from 0.4% to 3%.4,8,10,14 A reason for this discrepancy might be that data collected in previous surveys do not provide a reliable panorama of the true prevalence of preputial gland infections. Despite the large number of samples examined in such studies, the identification of infected and inflamed preputial glands relied exclusively on clinical observations or gross examination of animal carcasses, while ancillary exams were only restricted to samples that showed gross pathological changes. In the present study, 47% of infected glands were grossly unremarkable. Therefore, it is reasonable to conclude that subclinical forms could pass undetected during daily controls. An important issue raised by this result is that subclinically infected glands might represent a reservoir of opportunistic pathogens for other animals of the microbiological unit. Besides the welfare implications for aged mice, related to the potential development of preputial gland abscesses and ascending urinary tract inflammation, 21 infected males with high bacterial loads could be responsible for the transmission of infection to females at the time of breeding, potentially causing metritis, foetal resorption, abortion or stillbirth. 22 In this context, it is advisable to retire aged mice from reproduction, even though they are still fertile. In addition, an increased frequency of cage changes and more stringent sanitation procedures are warranted in long-term studies to improve housing conditions and to reduce the transmission of opportunistic pathogens through the contaminated microenvironment.
Preputial gland adenitis was previously reported to originate from septic complications of skin wounds in the inguinal area or the external genitalia. 7 However, in all 16 animals diagnosed with preputial gland adenitis, there were no lesions around the external genitalia or any microscopic changes consistent with wounds or fibrotic scars in the skin of the preputial gland area. These results strongly suggest that bacteria might have gained entry into the gland parenchyma via the ascending route, as experimentally documented by Hong and Ediger. 13
Histologically, the most common observed findings were gland atrophy and duct dilation, detected in 90% and 97.5% of examined glands, respectively. Preputial gland atrophy is a common age-related preputial gland change that can occur in association with ductal dilatation,2,5 and in our study, it was observed with or without concurrent inflammatory cell infiltration.
The second most common finding was inflammation, identified in 29/40 (72.5%) glands and classified as chronic interstitial adenitis in 27 cases and suppurative in the remaining two glands. Chronic lymphoplasmacytic interstitial infiltrates were very common and were identified in glands infected with all different bacterial isolates as well as in negative glands. Only when severity was moderate or severe was an association with infection identified. In 16/27 glands with chronic interstitial adenitis, variable amounts of granulocytes were found in the interstitium admixed to lymphocytes and plasma cells, suggesting the presence of a chronic active inflammatory process in these cases.
With regard to the different pathogenic potential of isolated bacteria, there was a progressive increase of the total histopathological score across negative glands and those infected with E. cloacae, Pasteurella spp., Klebsiella spp. and S. aureus, with most severe inflammatory changes identified in glands infected with S. aureus, as demonstrated by its isolation from the two cases of suppurative adenitis, both associated with surrounding panniculitis. Results from previous studies indicated S. aureus as the major pathogen in preputial gland adenitis.6,9,13 Nevertheless, in our study, S. aureus only accounted for a small number of cases (5/32 infected glands). This finding might be related to the caging system (IVC) and stringent husbandry practices applied in our facility that likely contributed to limit the spread of pathogens from cage to cage.9,23
Chronic interstitial adenitis with hyperkeratosis was diagnosed only in preputial glands infected with Pasteurella spp., Klebsiella spp. and S. aureus, suggesting that these opportunistic bacteria might be more pathogenic than E. cloacae. In particular, hyperkeratosis was most prominent in Klebsiella spp. infection. Hyperkeratosis of the duct epithelium can be considered an adaptive process of the epithelial cells secondary to a chronic irritating agent. 24
A strong correlation between the bacterial load and the severity of several histological findings (i.e. presence of intraluminal necrotic material, intraluminal keratin, interstitial inflammatory cell infiltrate, granulocytes (intraluminal and/or interstitial), total inflammatory score and total histopathological score) was found in the examined glands, supporting a causal relationship between bacterial infection and the development of preputial gland histopathological lesions.
The main strength of the present study relies on the thorough bacteriological and histopathological analysis of the preputial glands. We identified a broad spectrum of preputial gland lesions, and some of these appeared to be associated with a specific bacterial species, such as suppurative adenitis with S. aureus and hyperkeratosis with Klebsiella spp. However, some limitations should be acknowledged. Aerobic cultures used to detect microorganisms in this study bear intrinsic limits, as they cannot detect nonculturable bacterial species or microorganisms that require anaerobic or microaerophilic conditions. 25 For these reasons, we cannot totally exclude that undetected false-negatives among true-negatives exist in the current work. Moreover, the characterisation of Pasteurella spp. and Klebsiella spp. to the species level was not achieved through bacterial culture and biochemical methods in this study. The API system employed in our routine diagnostic activities is known to be unreliable for the diagnosis of Pasteurellaceae at the species level, and multiple methods, including PCR analysis and sequencing of the 16S rRNA gene should be used to discriminate among Pasteurellaceae.19,26 The small number of samples represents another limitation in the sense that these data do not necessarily reflect the prevalence of these infectious agents in our institution. Nonetheless, they provide useful information about which microorganisms might represent a potential risk in terms of animal welfare and quality of research. As regards the development of potential systemic effects induced by preputial gland infection and subsequent inflammation, we were not able to identify any change in haematological parameters correlated with bacterial infection or histopathological findings. This result is not surprising, since the infection was circumscribed to the genital tract, and most of the lesions were chronic.
In conclusion, preputial gland infection and inflammation were a common event in one-year old male C57BL/6NCrl mice, although in most cases it was subclinical and not associated with gross lesions and haematological changes.
Supplemental Material
sj-xlsx-1-lan-10.1177_00236772211055966 - Supplemental material for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages
Supplemental material, sj-xlsx-1-lan-10.1177_00236772211055966 for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages by Gianfranco Di Caro, Lucia Minoli, Marzia Ferrario, Gerardo Marsella, Gianpaolo Milite, Luca Crippa, Piera Anna Martino, Saverio Paltrinieri, Eugenio Scanziani and Camilla Recordati in Laboratory Animals
Supplemental Material
sj-xlsx-2-lan-10.1177_00236772211055966 - Supplemental material for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages
Supplemental material, sj-xlsx-2-lan-10.1177_00236772211055966 for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages by Gianfranco Di Caro, Lucia Minoli, Marzia Ferrario, Gerardo Marsella, Gianpaolo Milite, Luca Crippa, Piera Anna Martino, Saverio Paltrinieri, Eugenio Scanziani and Camilla Recordati in Laboratory Animals
Supplemental Material
sj-xlsx-3-lan-10.1177_00236772211055966 - Supplemental material for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages
Supplemental material, sj-xlsx-3-lan-10.1177_00236772211055966 for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages by Gianfranco Di Caro, Lucia Minoli, Marzia Ferrario, Gerardo Marsella, Gianpaolo Milite, Luca Crippa, Piera Anna Martino, Saverio Paltrinieri, Eugenio Scanziani and Camilla Recordati in Laboratory Animals
Supplemental Material
sj-xlsx-4-lan-10.1177_00236772211055966 - Supplemental material for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages
Supplemental material, sj-xlsx-4-lan-10.1177_00236772211055966 for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages by Gianfranco Di Caro, Lucia Minoli, Marzia Ferrario, Gerardo Marsella, Gianpaolo Milite, Luca Crippa, Piera Anna Martino, Saverio Paltrinieri, Eugenio Scanziani and Camilla Recordati in Laboratory Animals
Supplemental Material
sj-xlsx-5-lan-10.1177_00236772211055966 - Supplemental material for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages
Supplemental material, sj-xlsx-5-lan-10.1177_00236772211055966 for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages by Gianfranco Di Caro, Lucia Minoli, Marzia Ferrario, Gerardo Marsella, Gianpaolo Milite, Luca Crippa, Piera Anna Martino, Saverio Paltrinieri, Eugenio Scanziani and Camilla Recordati in Laboratory Animals
Supplemental Material
sj-pdf-6-lan-10.1177_00236772211055966 - Supplemental material for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages
Supplemental material, sj-pdf-6-lan-10.1177_00236772211055966 for Bacteriological and pathological investigations on the preputial glands of one-year-old C57BL/6NCrl mice maintained in individually ventilated cages by Gianfranco Di Caro, Lucia Minoli, Marzia Ferrario, Gerardo Marsella, Gianpaolo Milite, Luca Crippa, Piera Anna Martino, Saverio Paltrinieri, Eugenio Scanziani and Camilla Recordati in Laboratory Animals
Footnotes
Acknowledgements
We are grateful to M. Losa and A. Rigamonti for their technical assistance.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethical considerations
Legal and ethical requirements have been met with regards to the humane treatment of animals.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
