Abstract
In preclinical positron emission tomography animal studies, continuous blood sampling is used to measure the time course of the activity concentration in arterial blood. However, pigs have hypercoagulable blood that tends to clot inside plastic tubes. We tested several tube materials and lengths and the use of three-way connectors. We validated set-ups for automated blood sampling with and without blood recirculation that could run for 90 minutes without problematic clots and without any evidence of emboli formation during necropsy.
Introduction
In positron emission tomography (PET) research, blood sampling is the gold standard for obtaining arterial time-activity curves (TAC) needed for quantitative kinetic modelling of PET data. The SwissTrace Twilite system (STS; SwissTrace GmbH, Menzingen, Switzerland) has been successfully applied for automated blood sampling in rodents using recirculation through an arteriovenous shunt and in humans without recirculation. Pigs are widely used in preclinical PET research calculated in terms of both publication and citation numbers, 1 but the hypercoagulability of their blood 2 results in clotting inside plastic tubes. The tube set-up recommended by the vendor for use in humans is only functional for a few minutes in pigs before large blood clots obstruct the flow. Therefore, we explored several tube materials and set-ups to use with the STS for automated arterial blood sampling in pigs without heparinisation, and we tested them for 90 minutes – the duration of a typical dynamic PET experiment. We aimed at validating two set-ups: (a) without recirculation, and (b) with recirculation through an arteriovenous shunt.
Methods
Ethics statement
The protocol was approved by The Danish Animal Experiments Inspectorate (licence numbers 2018-15-0201-01609 and 2019-15-0201-01609), and all experiments were performed according to the current laws on animal experimentation and ethics.
Animals
We used seven clinically healthy pigs (females, 38–47 kg, Danish Landrace × Yorkshire) from a local breeder. They were group housed and acclimatised for at least one week. The environment was maintained on a 12-hour/12-hour light/dark cycle at 21–23°C and 50–55% humidity. The pigs were fed twice a day (Complete Slaughterhouse Pig Feed Mixture; Himmerland Grovvarer, Aarhus, Denmark) with free access to tap water. The pigs were fasted for 16 hours before premedication with 50 mg midazolam and 250 mg ketamine given intramuscularly (i.m.). After a few minutes of rest, 21G ear vein catheters were placed for induction and maintenance of anaesthesia with 50 mg and 400 mg/h propofol administered intravenously, respectively. The pigs were intubated (tube size 7-0) and mechanically ventilated with a tidal volume of 8–10 mL/kg (1 part oxygen and 2.2 parts air) 15 times per minute. The pigs were treated with 0.3–0.6 mg buprenorphine i.m. for analgesia. Cordis 6F sheaths (Cordis, Miami, FL) were surgically placed in the femoral arteries and veins. Pulse, oxygen saturation and body temperature were monitored. Respirator settings were adjusted continuously based on blood gas measurements. At the end of the experiments, pigs were euthanised with an overdose of 100 mg/kg pentobarbitone i.v.
Tube materials
Pigs 1 and 2 were used to test the efficacy of different tube materials. Blood from the femoral artery was circulated through the tubes to the waste bin. The tubes were then immediately flushed with water in attempt to detect clots. Some of the tubes were coated in-house with poly(MPC-BMA). 3 Based on the initial experiments (Table 1), we decided to proceed with the uncoated polyvinyl (PVC) tube (internal diameter 1.52 mm), which performed well in all tests, and is easier, faster and less expensive to use compared to the coated PVC and coated silicone tube. Using this tube, we achieved successful flow rates between 1.5 and 7 mL/min.
Main findings of the experiments performed using domestic female pigs.

Due to the COVID-19 pandemic, most of the tubes used in pig 1 were no longer available to order, and therefore substitutes were used for the rest of the pigs.
*Three pigs were used for other studies (data not shown) prior to these experiments.
ID: inner diameter; PVC: polyvinyl chloride; MRI: magnetic resonance imaging.
Set-ups with and without recirculation
In pigs 4–7, we first validated the set-up without recirculation followed by the set-up with recirculation (Table 1 and Figure 1). In pigs 4–6, PVC tubes were 1.5–2 m long, which is sufficiently long for PET/computed tomography studies. The set-up without recirculation was effective for 90 minutes, with high flow (5–7 mL/min) for seven minutes and low flow (1.5–2 mL/min) for the remaining 83 minutes. A few minor clots were observed that did not obstruct the blood flow inside the tube. The recirculation set-up could be used for 90 minutes, with high continuous flow (5–7 mL/min) and without any signs of clots. In pig 7, we successfully validated use of 3–4 m long tubes in both set-ups – important for use with PET/magnetic resonance imaging where the pump needs to be shielded and placed at a safe distance of at least 1.5 m from the scanner. During a gross macroscopic necropsy of pigs 4–7, the lungs and kidneys were found to have no signs of embolism. Only gross pathology was performed without any histopathology.
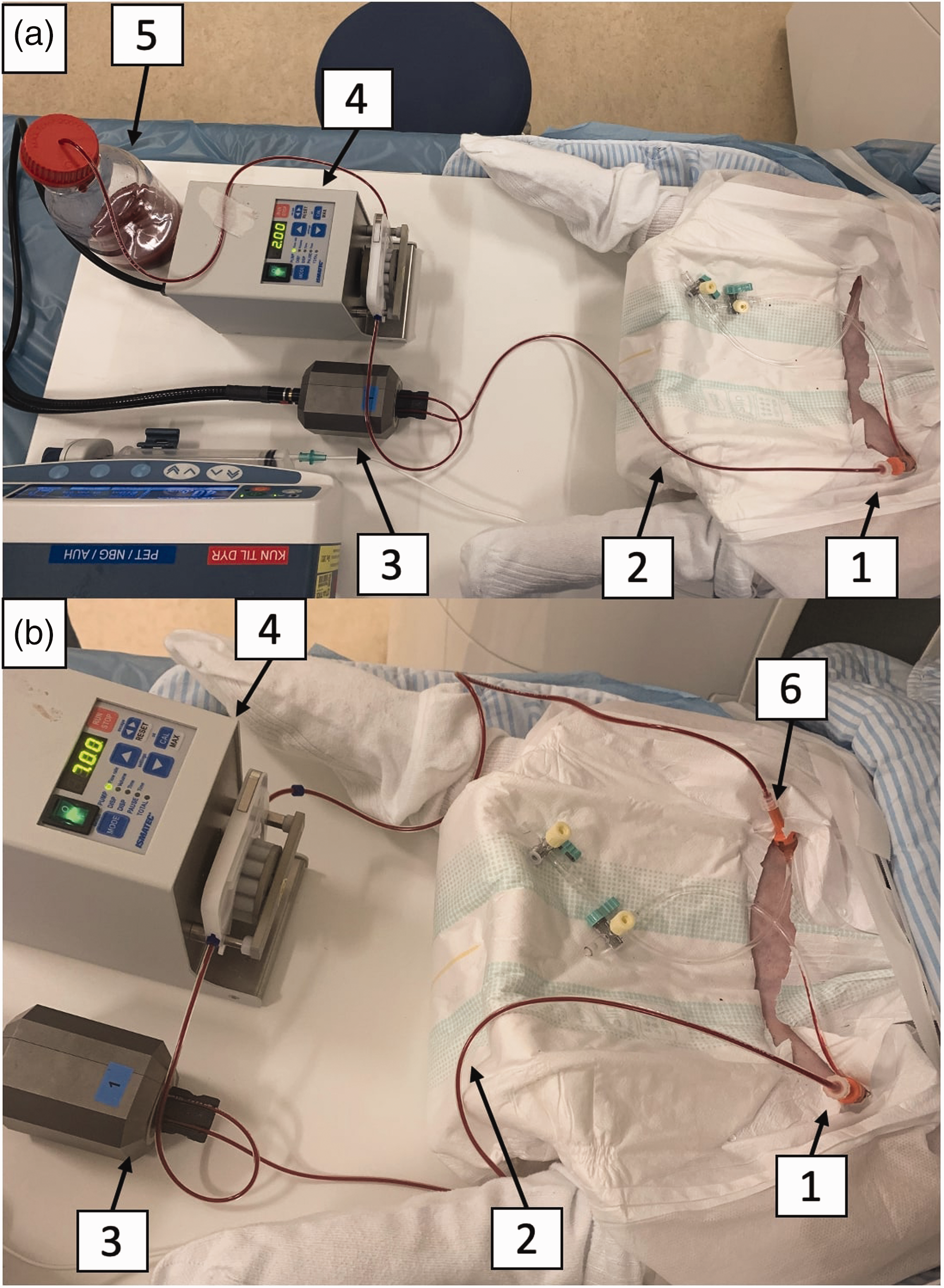
Set-up (a) without and (b) with blood recirculating through an arteriovenous shunt. Blood is continuously sampled from (1) the femoral arterial catheter through (2) a PVC tube (ID=1.52 mm) to (3) the detector head with the black 3D-printed template and via (4) the peristaltic pump (Ismatec, tubing pump, REGLO digital, two channels) back to the (A, 5) wastebin or (B, 6) femoral vein. Both set-ups can run for at least 90 minutes.
Three-way connector and test of blood gases
In preliminary tests, we found that set-ups with different combinations of tubes clotted within minutes (data not shown). Therefore, we focused on the use of long uncut tubes. However, in PET experiments, three-way connectors are often used for blood sampling for metabolites and blood gas analyses. In pigs 3 and 5, we tested a three-way connector (Discofix® C; B. Braun Melsungen AG, Germany) which unfortunately led to major clot formation leading and obstruction of the tube. As an alternative way to measure blood gases without a three-way connector, blood was sampled simultaneously from a three-way connector on the contralateral arterial catheter and through drops from the end of the blood sampling tube into the test syringe (before waste bin, step 5 in Figure 1(a)) in the set-up without recirculation. A total of nine paired blood samples from the two sites were analysed 4 and statistically compared using the Wilcoxon test. Analyses based on blood from the end of the blood sampling catheter led to significantly lower pCO2 values (on average 0.54 kPa lower, p = 0.038), no significant difference in pO2 (on average 0.6 kPa lower, p = 0.515), glucose (on average 0.04 mmol/L higher; p = 0.402) and lactate (on average 0.13 mmol/L higher, p = 0.091).
SwissTrace cross-calibration and dispersion correction
We calibrated the STS using the PVC tube filled with a long-lived 68Ge/68Ga (half-life 271 days) solution with known activity concentration. The cross-calibration factor was calculated as the known activity concentration divided by the measured background-corrected count rate. This method allowed a two-minute calibration before each experiment. Quantitative dispersion-free TACs were calculated using a transmission-dispersion model with two parameters: α (the fraction of tracer molecules sticking to the tube) and k (the rate by which tracer molecules are released from the tube). 5 For a flow rate of 7 mL/min, we measured the dispersion through a femoral arterial catheter + 50 cm PVC (internal diameter = 1.52 mm) tube, which is the length from the pig artery to the detector head. The tube-specific dispersion parameters (α = 0.5 and k = 7.6/min) 5 allowed us to calculate quantitative dispersion-free TACs.
Discussion and conclusion
Here, we presented two automatic blood sampling set-ups, with and without recirculation, which can be used in pigs for at least 90 minutes using uncoated PVC tubes. We later found using fluorescence microscopy that our poly(MPC-BMA) coating was not completely homogeneous. So, we cannot reject coating as a viable solution for materials that tend to cause clots, and we found improvement in the coated silicone tube, and for the coated PVC tube, the results were similar to the normal PVC tube. Therefore, future studies are necessary.
Without recirculation, PVC tubes were effective for 90 minutes with 5–7 mL/min flow during the first seven minutes and then with a reduced 1.5–2 mL/min flow. This is equivalent to 159–215 mL blood being sampled during a single scan, which is well below the acceptable maximum of 400 mL blood for a 40 kg pig in a survival study. Minor blood clots inside the tube are not problematic as long as the blood is not recirculating, since they do not obstruct the blood flow. A high initial flow rate (5–7 mL/min) is required for the best measurement of the dynamic TACs after the pig receives a bolus injection of PET tracer. After the initial tracer distribution, the TACs become less dynamic, and reduced tube flow could help to limit the amount of blood sampled from the pig. For this set-up, blood samples from the end of the PVC tube could be used with slight bias for blood gas values. Furthermore, it is an advantage that the placement of only a single catheter is required. With recirculation, the same tubes can be used with a continuous 5–7 mL/min flow for 90 minutes without clots or any evidence of emboli. This set-up was a closed loop, and a separate catheter is needed to sample blood gases and metabolites. It is an advantage that blood can be sampled during imaging sessions using multiple tracers without the risk of inducing anaemia in the pigs. The choice between recirculation and non-recirculation therefore depends on the length of time during which blood sampling is required.
Footnotes
Acknowledgements
Thanks to Michele Gammeltoft for proofreading.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: We are grateful to the Simon Fougner Hartmanns Family Foundation for funding the purchase of the STS.
