Abstract
Mice and rats are among the most used laboratory animals. They share numerous similarities along with differences, some yet unexplored. One of them is the morphometry of their adrenal glands, whose characteristics may be related to differences in energy management, immune response, drug metabolism, behaviour and temperament. The present study tries to fill this knowledge gap with the evaluation and comparison of adrenal gland anatomical/morphometric parameters of mice and rats. In groups of 10 (
Introduction
Mice and rats are two species of great interest as laboratory animals. Despite the obvious similarities, there are many differences, some of which relate to temperament, stress response and behaviour. For the understanding of these differences, it is appropriate to investigate the quantitative and/or qualitative parameters of particular characteristics of the physiologically related organs, such as the adrenal glands. In literature, a number of studies exist about their morphometric parameters in mice1–4 and rats.5–15 To our knowledge though, there are no studies comparing morphometric parameters between the two species.
The adrenal or suprarenal glands or simply adrenals, are complex endocrine organs regulating multiple physiological processes, such as metabolism, stress and immune response, and the function of the cardiovascular system.1,5,16–20 They are divided into two distinct layers consisting of two distinct types of endocrine tissue, cortex and medulla. The cortex, the outer layer, originates from mesodermal mesenchymal cells, whereas the medulla, the inner layer, originates from the ectodermal cells of the neural crest just like sympathetic neurons.21,22 Therefore, the medulla acts as a sympathetic paraganglion,23,24 with the difference that the released catecholamines do not locally, affecting one organ, but widely, affecting the whole body. Neural crest cells migrate to the centre of the formatting adrenal cortex and differentiate to chromaffin cells. Thus, cortex and medulla can be considered as two independent endocrine glands bounded in one body during the embryonic development.
The accepted general model of adrenocortical zonation is that it comprises three main zones: zona (z.) glomerulosa (ZG), z. fasciculata (ZF) and z. reticularis (ZR) each having a distinct function, secreting mineralocorticoids (aldosterone), glucocorticoids (mainly corticosterone in rodents) and androgens, respectively. 18 ZR border is more clearly delineated in rats compared with mice. In mice appears an additional zone, z. X (ZX).25–27 This zone, which gradually regresses, is possibly associated with androgen secretion, although its exact role is largely unknown.28,29 In virgin female mice, ZX regresses gradually and disappears in 3–7 months of life. Whereas in males, it disappears before postnatal day 40.25,30,31 ZX in the mouse adrenal cortex is an equivalent to the foetal cortex in primates.29,32,33
The cells of the cortex originate from a stem cell population in the outer cortex or capsule, and migrate centripetally, changing their appearance as they progress through the zones. 29 Adipose tissue can be observed in various places in the rat adrenal tissue all over the ZF (small deposits), and also in the mouse, but only near the borders of medulla, probably representing the remnants of the ZX.
Materials and methods
Animals
For the needs of this study, 20 (
Twenty rats aged between four and five months, 10 male, weighing 347–401 g and 10 female, weighing 193–248 g, as well as 20 mice, aged between 50 and 75 days, 10 male weighing 38–42g and 10 female, weighing 27–35 g, were used. All animals were bred in our facilities, maintained under specified pathogen-free conditions, and monitored as recommended. 34 Animals’ health was monitored according to FELASA recommendations 35 using IDEXX GmbH (IDEXX BioAnalytics, Germany) testing. Mice are kept in IVC EHRET cages in a facility with HEPA filters. The air is 100% fresh with 10 to 15 air changes/hour. Access is controlled. Room temperature of 20–22°C, and humidity of 45–55, are checked daily. In the room there is a SafeFlow 1.2 hood (BioAir Instruments, Pavia, Italy). We use UV-light for a limited time for sanitation of the equipment kept inside the hood.
Rats are kept in open cages. Access is controlled and all the other characteristics are the same as in the murine colony.
Both species had ad libitum access to standard rodent food 4RF25 from Mucedola (Scobis, Italy) and water. The sanitation is undertaken every week from experienced personnel. A stable temperature was ensured by a HVAC system controlled by a thermostat. Lights were less than 325 lux and were programmed for 12 h:12 h light to dark. Noise was kept <85 db and there were no ultrasonic noise and vibration. The only procedure performed in the animals used for the study was the animal identification. Animals are kept in a density that is recommended from the EU Directive 2010/63/EU on the protection of animals used for scientific purposes.
Fixation
Animals were anesthetized via the intraperitoneal administration of a ketamine (Imalgene; Merial S.A, France; 50 mg/kg) and xylazine (Rombun, Bayer, Germany; 5 mg/kg) mixture and sacrificed by trans-cardial orthograde perfusion via the left heart ventricle of normal saline, followed by 4% paraformaldehyde (Sigma-Aldrich, USA) for
Histology
Dehydration in a graded series of ethanol and xylene was followed by embedding in paraffin. Sections, 8µm thick, transverse to the longitudinal axis, were collected. Sampling was undertaken during sectioning of the adrenal glands. Every 20th section of the series was processed with traditional histological techniques, stained with haematoxylin and eosin and measured for morphometric evaluation with an optical microscope (Zeiss Axioplan, Germany), using a digital camera (Basler AG, Germany) connected to computer equipped with Manual Whole Slide Imaging Software (Manual WSI Scanner 2019a v1.6, Micro Visioneer, Germany), under a 10× lens. The program allowed the digital pictures taken during the scanning of the slide to be united harmoniously in a composite image of the whole section.
Morphometric analysis
Morphometric analysis was performed using image analysis software (Image Pro Plus 6.2, Media Cybernetics, USA), and the areas of capsule, cortex (along with its constituent zones) and medulla were quantified (Figure 1A and 1B). In this study six female mice were virgin and used for the ZX measurements. ZX disappears relatively early in male mice, 2 while it is absent in rats. Therefore, no comparisons of ZX between species and sexes were made.
Statistical analysis
Dimension data were analysed, and the effects of sex and side within each species were compared with the use of a factorial General Linear Model provided in the IBM SPSS statistic (v.25) software (IBM Corp., USA). Linear correlations between adrenal descriptive parameters were based on determination of Pearson’s product moment correlation coefficient (assumption of Normality was verified). The level of significance was set at 5%.
Results
All animals had two normal glands. Noteworthy, in two male rats the left gland was found rostral from the typically described anatomical location at the cranial pole of the kidneys. 1 Rat adrenals varied from bean shape in most cases, to lentil shape less frequently. The mouse adrenals were generally ovoid.
The image analysis software applied on whole slide/gland section scans acquired with a manual slide scanning system provided measurements of the areas of interest surface in mm2 of each sub-area of the gland. From the collected data, mean values were calculated, and comparisons were made between sexes of the same species and right/left gland. Right/left gland comparison did not reveal statistically significant differences and therefore are not analysed. Quantitative comparisons between the two species were not made, because of the unequivocal difference in body sizes.
Ratios, that is quotients of specific pairs of measured parameters, were calculated and compared between species and between sexes in the same species.
The linear correlation between the magnitudes of medulla and cortex areas, as well as between the cortex area and the area of each one of its constituent zones, was explored by species and sex, with use of the Pearson’s product moment correlation coefficient (r). Data analysis provided some intriguing results. Length of the adrenals was calculated by multiplying the number of sections with the section thickness, and volume by multiplying the average gland surface with the average length. It is reported that ZX persists in some strains more than days in male mice.4,29 In our study ZX was not found in 50–75 day-old male mice.
Morphology and geometry of cells
In female and male mouse adrenal gland (Figure 1 (C) and (D)), in the more superficial ZG, small, polygonal, cells were observed (Figure 1 (C1) and (D1)) which form distinct spirals. In ZF, cells appear hypertrophic with oval to polygonal shape, formatting columns, though not so clearly structured, typical for this zone (Figure 1, (C2) and (D2)). ZR is absent; while at the medulla border intense vacuolization is observed due to the degeneration of ZX (Figure 1 (C3) and (D3)).
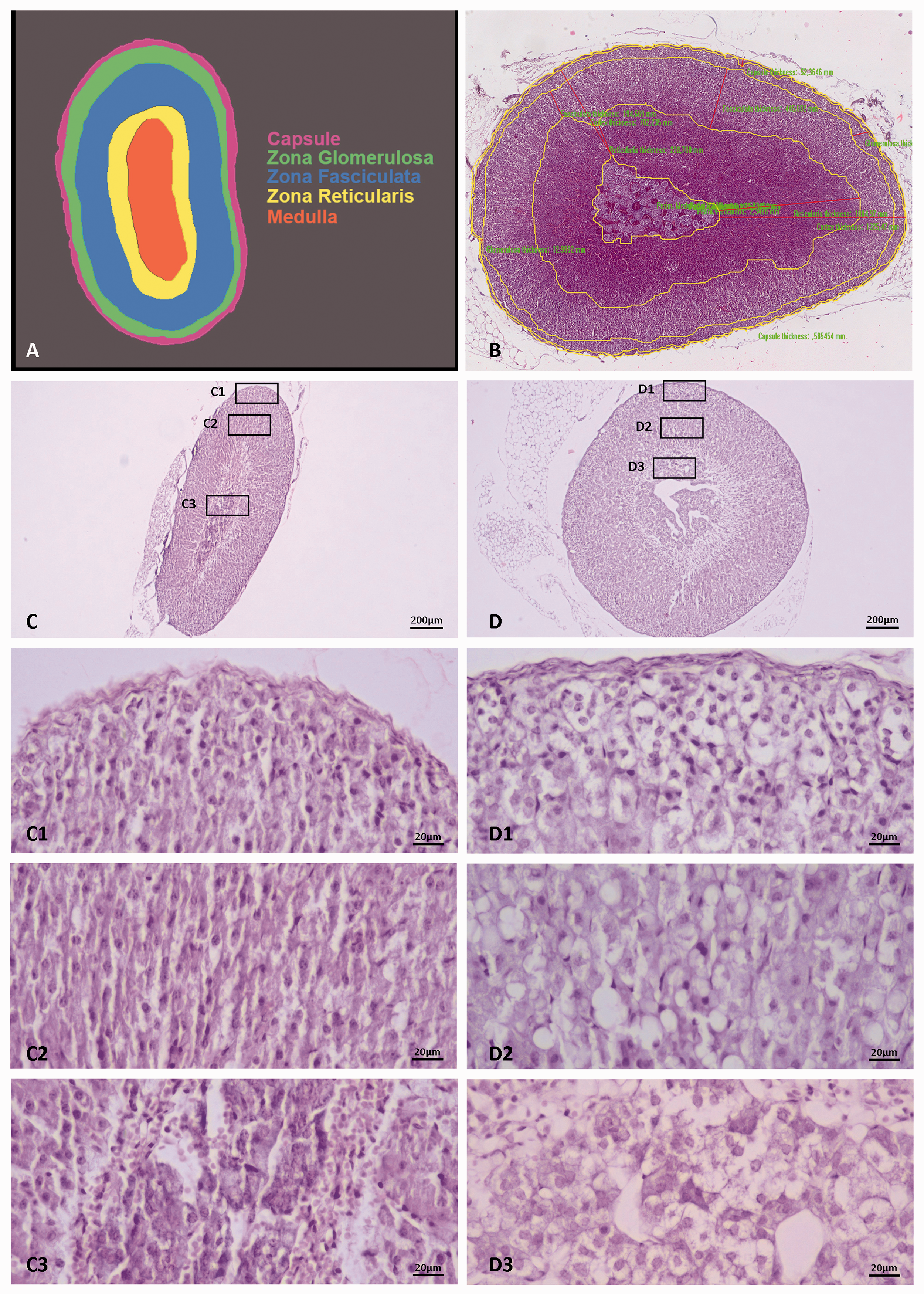
A. Schematic representation of the adrenal gland zonation B. Microphotograph showing the adrenal zones delineated by the image analysis software. Microphotographs of C. female and D. male mouse adrenal glands. Magnifications of cortex zones and medulla (rectangular frames) reveal cellular morphology. Zona glomerulosa C1 and D1, zona fasciculata C2 and D2 and medulla C3 and D3.
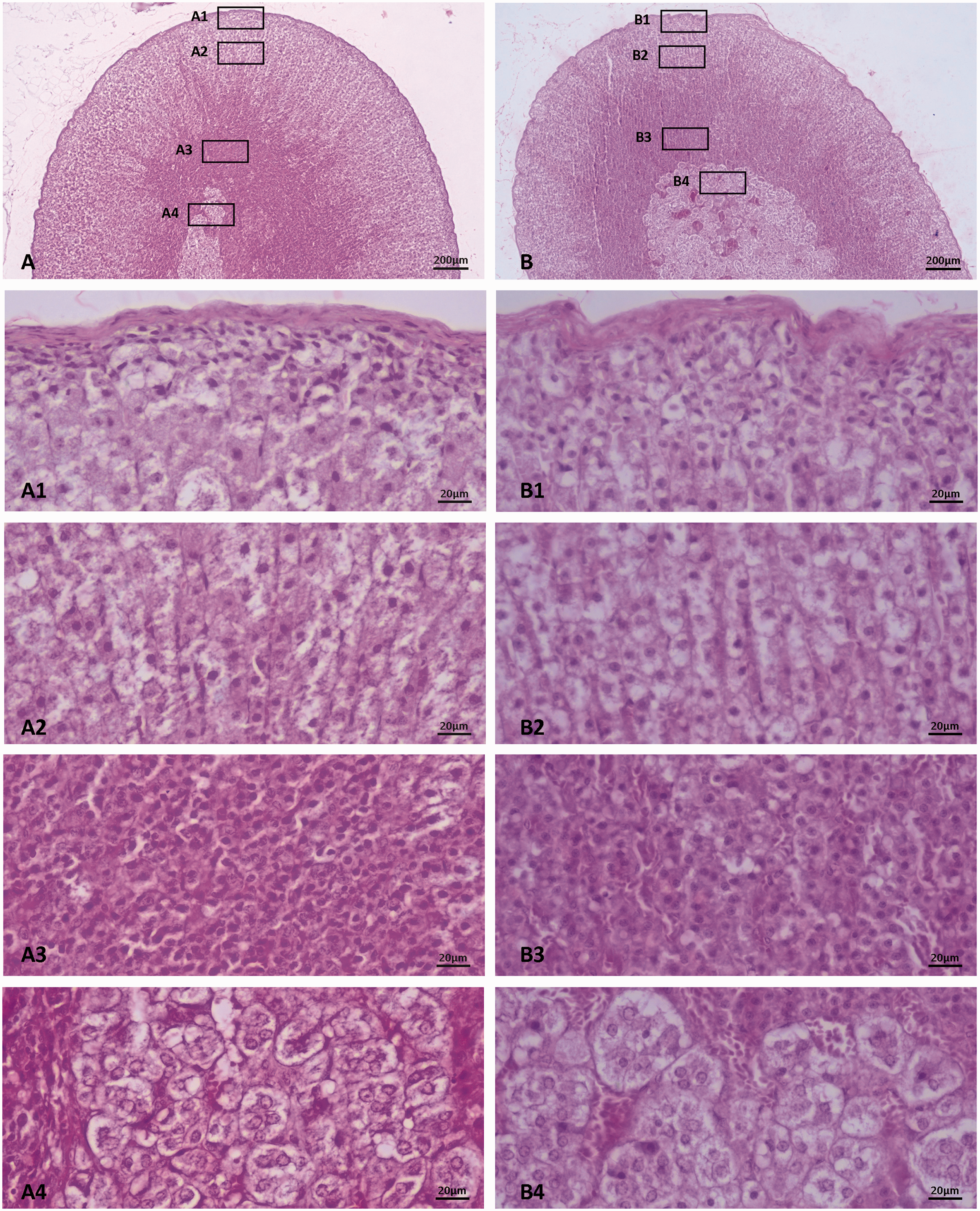
Microphotographs of A. female and B. male rat adrenal glands. Magnifications of cortex zones and medulla (rectangular frames) reveal cellular morphology. Zona glomerulosa A1 and B1, zona fasciculata A2 and B2, zona reticularis A3 and B3 and medulla A4 and B4.
In female and male rat adrenals (Figure 2 (A) and (B)), underneath the capsule, in the ZG, small, polygonal, cells are observed (Figure 2 (A1) and (B1)). At first, they seem disordered, but persistent observation reveals spirals, less clear though compared with mice. In ZF, cells compared with those of mice seem rather oval than polygonal (Figure 2 (A2) and (B2)). Nuclei are centrally located as in the ZG cells, while their size is larger. ZR cells are small, condensed and near the medulla border they seem apoptotic (Figure 2 (A3) and (B3)). In the medulla, chromophilic cells form clusters. Their nuclei are not so intensely stained as in mice (Figure 2 (A4) and (B4)).
Areas
The surfaces, in mm2 (mean values ± standard deviation), measured on mouse and rat gland sections, revealed statistically significant differences as summarized in Table 1.
Selected morphometric parameters of the measured areas (mean±SD) of adrenals in male and female rats (n=20) and mice (n=20). All measurements are in mm2 (square millimeters).
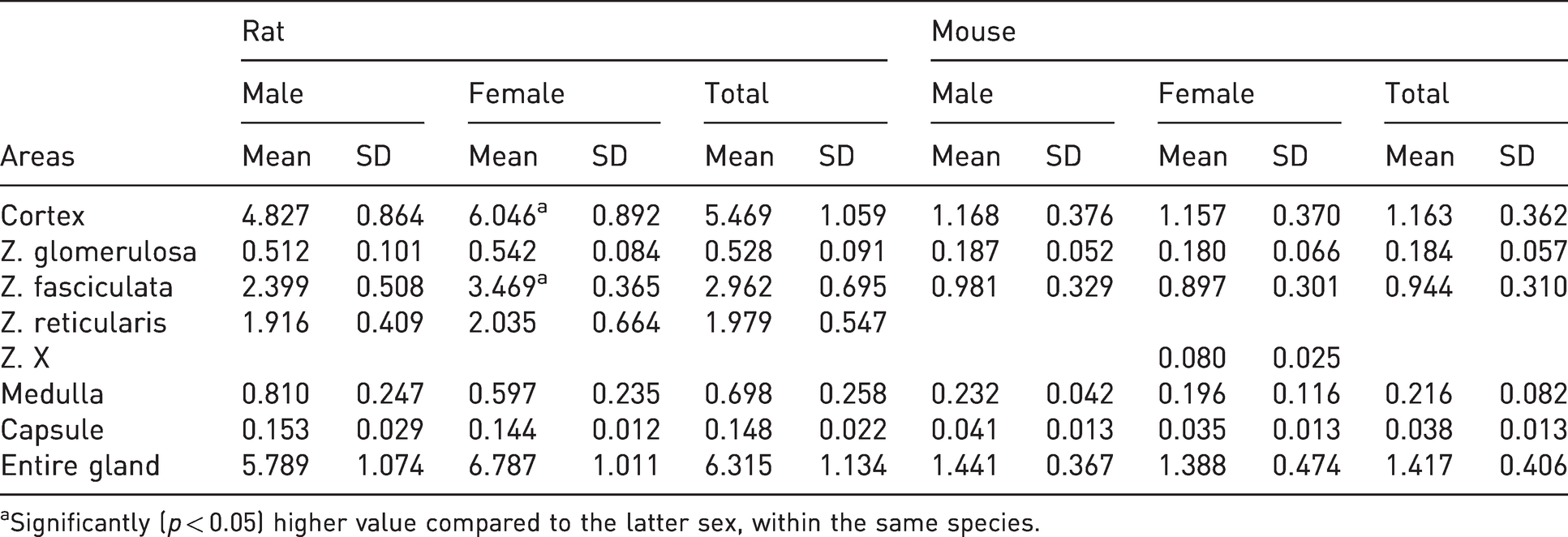
aSignificantly (
The entire transverse gland surface was larger in female compared with male rats, although this difference was not statistically significant (
Ratios
The quotients (values ± standard deviation) between the measured surfaces and the statistically significant differences are summarized in Table 2. The units for the calculation of ratio values were metric.
Selected morphometric parameters (mean ± SD) of adrenals in male and female rats (n = 20) and mice (n = 20).
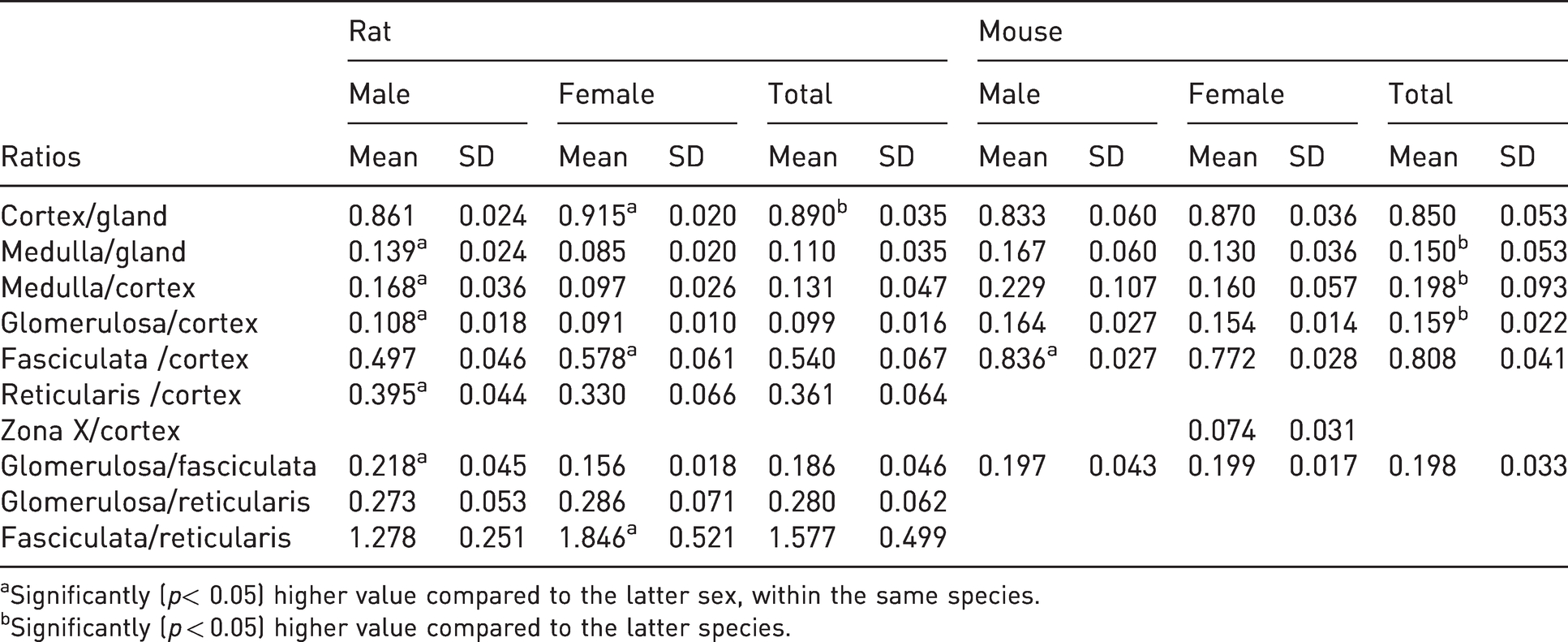
aSignificantly (
bSignificantly (
Cortex area to entire gland area ratio was significantly higher in rats compared with mice (
Correlations
Linear correlations between medulla and cortex areas, as well as between the cortex area and the area of each one of its constituent zones in rats and mice (r values) are summarized in Table 3.
Linear correlation between medulla and cortex areas, as well as between the cortex area and the area of each one of its constituent zones in rats and mice (r values in bold font imply statistical significance).
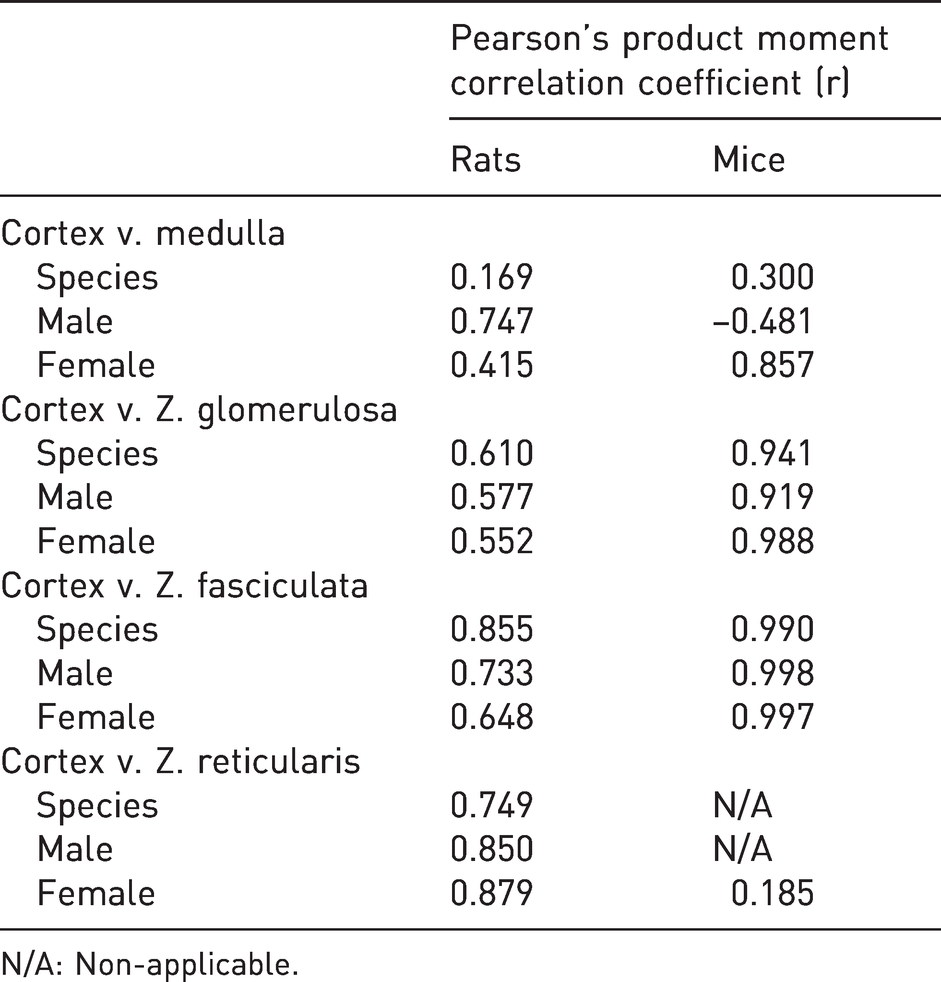
N/A: Non-applicable.
In rats, a statistically significant positive linear relationship between cortex and medulla areas was observed in male animals only (
A statistically significant positive linear relationship between the cortex and ZG (i.e. a larger cortex is accompanied by a larger ZG) was observed in rats, sexes combined (
Length and volume
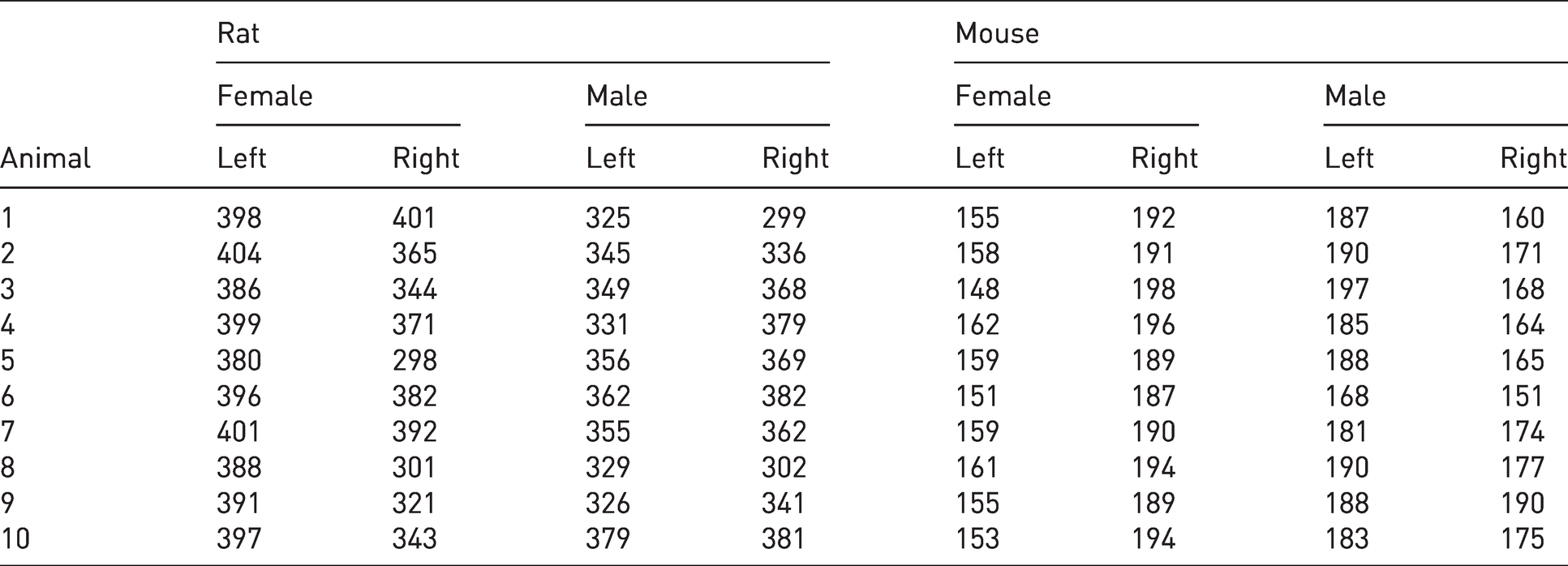
The calculated values of length and volume for mouse and rat adrenal glands as well as the number of sections per adrenal gland are presented in Tables 4 and 5.
Length and volume (mean ± SD) of the entire adrenal gland. Length was calculated by multiplying the number of sections to the section thickness (8 µm), whereas volume was calculated by multiplying the average area by the average length in each gland of each animal. The effects of both sex and side, as well as any sex × side interaction, were determined by use of the General Linear Model within each species. Length in mm (millimeters) and volume in mm3 (cubic millimeters).
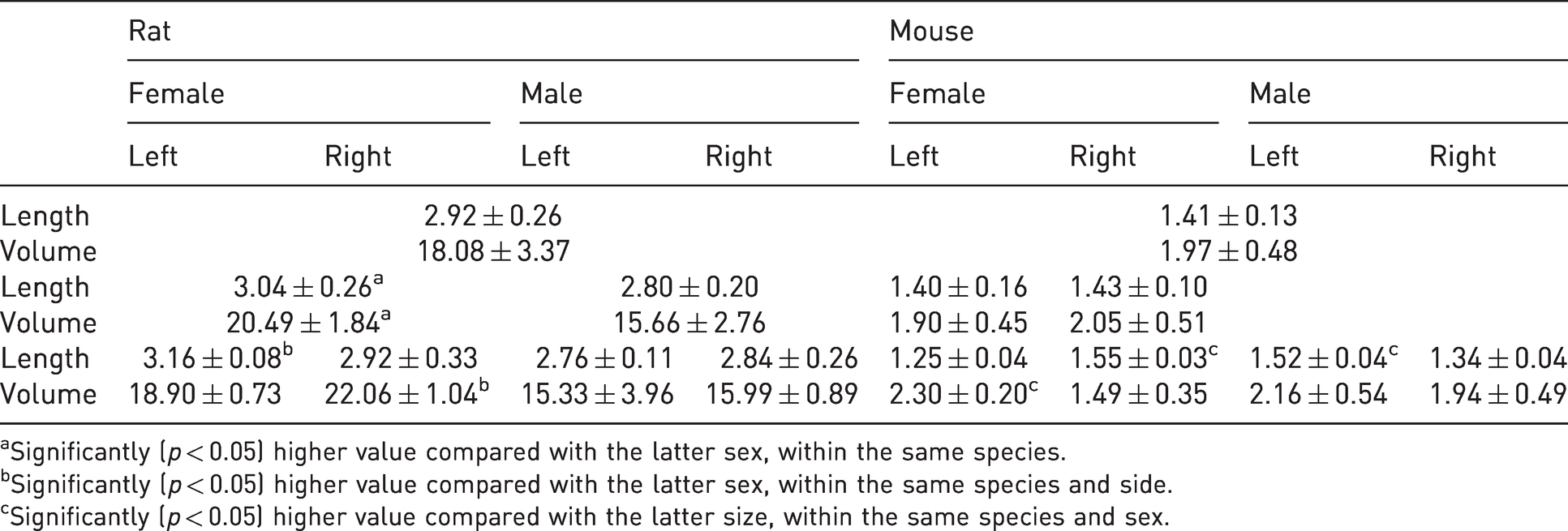
aSignificantly (
bSignificantly (
cSignificantly (
Number of 8μm thick sections per adrenal gland.
Adrenals in female rats, sides combined, were significantly longer than the adrenals in male rats (
In mice, female and male animals did not generally differ in either the length or the volume of their adrenals (
Discussion
According to Bielohuby et al. 1 and Tanaka and Matsuzawa, 3 at all developmental stages the adrenal glands of female mice were significantly heavier than those of their male counterparts, whereas our findings did not reveal any statistically significant difference between sexes in mice (in fact female adrenal length and volume were slightly smaller compared with males). In addition, the aforementioned authors reported that cortex and medulla volume and cortical thickness were significantly greater in female compared with male mice. Our results indicated that cortex area was almost equal in the two sexes and medulla area was only slightly larger in male mice. Furthermore, Bielohuby et al. 1 reported that all cortex zones had larger volumes and Tanaka and Matsuzawa 3 greater thickness in female mice. In the first study the differences were statistically significant in ZF and ZX, but not statistically significant in ZG, whereas no statistical significance was declared in the second study. On the other hand, in our study the slight prevalence of male compared with female mice concerning the areas of ZG and ZF was not significant. No comparison could be performed for ZX because of its absence in the studied animals. It is possible that the differences in the results can be partially explained by differences in the methodological approach.
Deacon et al. 2 studied the adrenals of male Swiss albino strain mice and reported the areas (surfaces in mm2) of the entire gland (1.77 ± 0.06), medulla (0.47 ± 0.03), ZG (0.23 ± 0.01), and ZF (0.62 ± 0.03). In addition, they reported the area of juxtamedullary degeneration area (0.45 ± 0.02). These results are generally in parallel with those reported in our study: entire gland (1.441 ± 0.367), medulla (0.232 ± 0.042), ZG (0.187 ± 0.052) and ZF (0.981 ± 0.329). The divergence regarding ZF could be explained by its division by Deacon et al. 2 into two zones: ZF and juxtamedullary degeneration zone.
The calculated significantly larger female adrenal volume compared with male rats (Table 4) are congruent with the findings of Hatai, 8 who reported as early as in 1913 the equivalent sex dimorphism concerning body weight in rats.
Majchrzak and Malendowicz 10 described the same difference, but they pointed out that it occurs after postnatal day 63. They also reported that in young adult female rats the volumes of medulla and all cortical zones were bigger compared with male rats, but for ZR the difference was not significant. Our data, although concerning areas and not volumes, abide with the findings of Majchrzak and Malendowicz 10 about the cortical zones but are in contrast with their findings about medulla. They attribute the sexual dimorphism of rat adrenals to qualitative rather than quantitative changes in adrenocortical cells due to a stronger inhibition of adrenocortical growth in maturing male than in female animals.
The outcomes of the current study concerning rats are in general agreement with those of Ashworth et al., 5 who reported that female rat adrenal area was significantly larger compared with that of the male. This finding complies in general with our results, although the difference revealed by our measurements was not significant. Furthermore, Ashworth et al., 5 comparing the medulla/gland ratio found it higher in male rats, though not significantly, whereas in our results the difference was statistically significant. It must be noted, that Ashworth et al. 5 compared the ratio of cortex zones to the entire gland area. In the present study the comparison of cortex zones to the cortex area was adopted. Nevertheless, the results of both studies are comparable. They reported that ZG/gland area ratio was significantly higher in male, ZF/gland area ratio was only slightly higher in male and ZR/gland area ratio was significantly higher in female rats. Our data indicate that ZG/cortex area and ZR/cortex area ratios were significantly higher, whereas ZF/cortex area ratio was significantly lower in male rats. ZG and ZR are implicated in the production of aldosterone and (in species that express 7 alpha-hydroxylase) androgens, respectively, whereas ZF is responsible for the production of endogenous glucocorticoids, hormone-acting molecules that – among others – regulate stress response, energy management, functionality of the immune system, behavioural status and the expression of genes, some of which (e.g. CYP450 genes) are responsible for drug metabolism. Glucocorticoids are known, albeit not uniform, inducers of many CYP450 isoenzymes. 36 The differentially increased ZF/cortex ratio in female rats and male mice could signify a possible species–sex differential shift in glucocorticoid-regulated functions, such as the aforementioned drug metabolism.
Stress activates the hypothalamus–pituitary–adrenal axis and, as a result, the secretion of corticosteroids from the adrenal cortex increases. Corticosteroids, such as cortisol (mainly in humans) and corticosterone (mainly in rodents), serve as biomarkers for stress and depressive disorders. There is a strong correlation between serum cortisol and corticosterone during acute or repeated restraints, chronic unpredictable stress and acute stressful ordeals. 37 The ratio of ZF/cortex was found to be higher in female compared with male rats. Perhaps female rats need more glucocorticoids than male rats in order to cope with their physiology and social behaviour.
The results recorded by Moroz et al. 12 who calculated in young adult (70 day old) male rats the ratios of medulla/gland area (12.6%), cortex/gland area (87.4%) and within cortex ZG/cortex area (9.17%) are in total agreement with the measurements of the present study (13.9, 86.1 and 10.8% respectively), whereas their results for ZF/cortex area 69.01% and ZR/cortex area 21.82% differ slightly from ours (49.75 and 39.5% respectively).
Chakraborty et al. 6 calculated the ratios of medulla/gland volume (14.03%) and, within cortex, the ZG/gland volume (7.65%), ZF/gland volume (62.31%) and ZR/gland volume (15.73%) in young adult female rats. Our measurements in young adult female rats provided the ratios: medulla/gland area 8.5%, ZG/gland area 8.32%, ZF/gland area 52.8% and ZR/gland area 30.19%. It is evident that the minor differences are the result of the different morphometric approaches of the two studies. Malendowicz 11 presented volumes and the ratios of medulla (female 5.3% v. male 6.16%), ZG (female 11.15% v. male 11.22%), ZF (female 52.36% v. male 51.84%) and ZR (female 30.72% v. male 30.34%) to gland volume, comparing them between sexes in Wistar rats. The study did not reveal any significant sexual differences, whereas our approach provided ratios that differ significantly between female and male rats: medulla/gland area (female 0.085 v. male 0.139), ZG/cortex (female 0.091 v. male 0.108), ZF/cortex (female 0.578 v. male 0.497) and ZR/cortex (female 0.33 v. male 0.395). The methodological divergence of the two studies may be the reason behind the differences in results. Rohr et al. 15 reported volume ratios of cortical zones to cortex in male Wistar rats: ZG/cortex (10%), ZF/cortex (63.5%) and ZR/cortex (26.5%), which are comparable with our results.
Rat adrenal gland volume (18.08 ± 3.37 mm3) and medulla/entire gland area ratio (0.11 ± 0.035) measured in our study were comparable with those reported by Coupland and Tomlinson 7 (13.67 ± 2.7 mm3 and 0.098 ± 0.003 respectively). On the contrary, these results concerning rat adrenal volume differ completely from those described by Kurtulus et al. 9 (4.81 ± 0.95 mm3). Furthermore, the other findings of Kurtulus et al. 9 on rat adrenal length (300–450 nm) cortex (4.13 ± 1.02 mm3) and medulla volume (0.69 ± 0.06 mm3) are in such sharp contrast with our results and those of other studies5–7 that raise scepticism about their validity.
Summarizing the results of the present study, female rat adrenal glands were found to be significantly longer, with a larger cortex area and so consequently bigger (larger volume) compared with the male rat. Entire gland and medulla areas were also larger in female rats, though not significantly. On the contrary, in mice, the aforementioned parameters did not differ significantly between sexes. Comparison of ratios revealed more statistically significant differences between sexes in rats and what is more interesting between mouse and rat. In brief, medulla/gland and medulla/cortex were higher, whereas the cortex/gland was lower in male compared with female rats and in mice compared with rats. Furthermore, the ratio ZG/cortex was found to be higher in mice compared with rats.
The literature abounds in research about mice and rats. Remarkably there is a dearth of studies comparing basic anatomical features such as morphological aspects of the two species’ adrenal glands. The collected data may be proven useful in understanding and clarifying some of these differences about development, behaviour, physiology, pharmacology and pathology.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary materials
All data are available upon request.
