Abstract
An integrated rearing system for Cantareus aspersus under environmental conditions in an experimental orchard is proposed. In this study, the natural behaviour, circadian rhythms and suitable rearing conditions of the edible snail were optimised to produce homogeneous growth and low variability. The growth was standardised, and growth pattern fit was assessed with various models. One thousand fry were cultured in the orchard, and a random sample of 100 snails were measured weekly for 23 weeks. The rearing system had the following characteristics: snails and earthworms were included in the experimental rearing orchard; a homogeneous group of juvenile snails of the same age and size and high rearing density (500 snails/m2) was used; snails were fed with layers mash ad libitum; and mixed and fringes vegetation was planted in the orchard. A commercial size of 60% of snails was achieved in 21 weeks and 95% in 23 weeks. The different models showed a good fit, and the quadratic model obtained the best fit. This experimental snail orchard proposal can be extended to other areas, although it must be corrected according to different environmental conditions and fit to other species of interest. This experimental model could constitute a viable alternative to traditional models of animal experimentation with mammals, and given its high adaptability, it could be applied in different fields of science.
Introduction
Cantareus aspersus (Syn. Helix aspersa) is the most extended edible snail and a useful tool in animal experimentation in several scientific fields.1–4 It has the advantage that it is able to adapt to different environmental conditions.5–7 Although its use is conditioned by high genetic diversity, a significant number of genera and species have marked differences amongst them. 8 Snails show a very marked circadian rhythm that is influenced by many genetic and environmental factors, resulting in high individual variability and asymmetrical distribution in individual weights.9,10 When unfavourable rearing conditions arise, the snail can inhibit its growth for a long period of time, from two to four months.
The effects of these factors on growth have been broadly studied and include genetic factors, 8 environmental conditions,5,11 stocking density, effect of crowding on growth and circadian rhythm,2,12,13 food consumption and metabolism,1,10,14,15 habitat preferences 16 humus effects,17,18 interspecific relations, 12 among others.
The reproduction and nursing of C. aspersus take place in buildings, under controlled ambient conditions. Fattening takes place in orchards and outdoor greenhouses, whenever climatic conditions allow. The classification of growth stages is variable and depends on the rearing system.2,8,9,18–20 The following stages are common: fingerlings – from hatching until the fry reach 5 mm; juvenile snails – from 5 to 25 mm approximately, with a first fast growth stage (young) and a fattening one (non-adult); adult snails ≥20 mm, the shell peristome is reflected and they have reached sexual maturity. In previous studies, the process of rearing of juvenile snails in a laboratory under semi-controlled conditions was identified. 21 Regarding outdoor rearing farms, it would be necessary to promote an appropriate farming system for any given species oriented to providing optimal living conditions, supporting growth and survival, minimising risks and optimising production.10,18
A lack of knowledge of the rearing conditions, where different factors interact, results in a lack of growth. If no growth patterns are achieved, this is a major barrier for the use of the snail in farming and experimental models. What would an orchard rearing system that determines the existing knowledge on the edible snail look like? Would it be possible to get the animals to grow homogeneously and in short periods of time? Answers to these questions would support the standardisation and modelling of growth in an experimental snail orchard, with models that could be extended to other countries. The system proposed in the experimental orchard seeks favourable rearing conditions according to the habitat preferences, activity and circadian rhythm of the land snail. It also facilitates animal experimentation, isolating fixed effects from future research and avoiding masking these factors under unfavourable rearing conditions. Therefore, the aim of this research was to propose an integrated rearing system of C. aspersus in an experimental orchard under environmental conditions. The fit of the rearing system will allow continuous growth to be obtained and a pattern of growth to be established.
Methods
Biological materials
Edible snails from the market were used as breeders (C. aspersus). The experimental snail orchard was developed in the Agropecuary Provincial Centre of ‘Diputación’ (Cordoba, Spain). The climate was continental Mediterranean with Atlantic influences, having mild winters without extreme temperatures. 22 The laying period started on 1 September 2016 and finished on 1 October 2016. The batches of eggs were incubated for 17 days under controlled conditions. At the end of the incubation period, a tray containing around 4000 hatchlings was available. Of these, 1100 snails measuring 3.2 ± 0.5 mm were selected. The snails were activated and kept for one week in the breeding boxes in laboratory conditions. 21 After a week, 1000 snails, with an average shell diameter of 4.6 ± 0.6 mm, were spread in the experimental orchard. Snails outside this range were not used. The experimental orchard was divided with a net into four similar sections measuring 0.5 m2 in area, and 250 snails were placed in each one. A sample of 100 snails were individually measured weekly for 23 weeks from November 2016 to April 2017.2,5,10,14,21 They were collected weekly from different parts of the orchard, with 25 snails chosen at random from each section, and once the experiment started, no snails were excluded.
Establishment of experimental snail orchid
The open field system is the most frequent snail farming system. A mixed system is composed of open parks, with planting suitable for snail feeding and net-covered greenhouses, where outdoor growth and fattening of juvenile snails takes place. Rearing methods have been described widely. The duration of the production cycle depends on the biological cycle of the cultured species and the climatic conditions.1,2,6,8,9 Figure 1 shows the experimental snail orchard. On a concrete surface, an area measuring 2 m2 was fenced (2.7 m length and 0.75 m width) with a 60 cm high galvanised iron perimeter supported by iron posts. A layer of gravel, 10 cm deep, was placed to prevent the land from flooding and was covered with soil with a depth of 50 cm. According to the soil texture calculator of the USDA, soil texture can be defined from loam to clay loam, with 26% clay, 46% silt and 28% sand. The soil was low in phosphorus (8.1 ppm), potassium (110 ppm), magnesium (276 ppm) and organic matter (1.34%). Humus was added to improve the water-holding capacity and organic matter content of the culture area. 23 Supplemental Table S1 shows the chemical composition of the humus used. The orchard was covered with a bird protection mesh, and it was oriented according to prevailing wind directions (east to west). Three plant types were sown at high density in mixed fringes: fringes of Cynara cardunculus sp. (cardoon), Brassica olaracea subsp. sabellica (green cabbage), and Beta vulgaris subsp. cicla (beet).
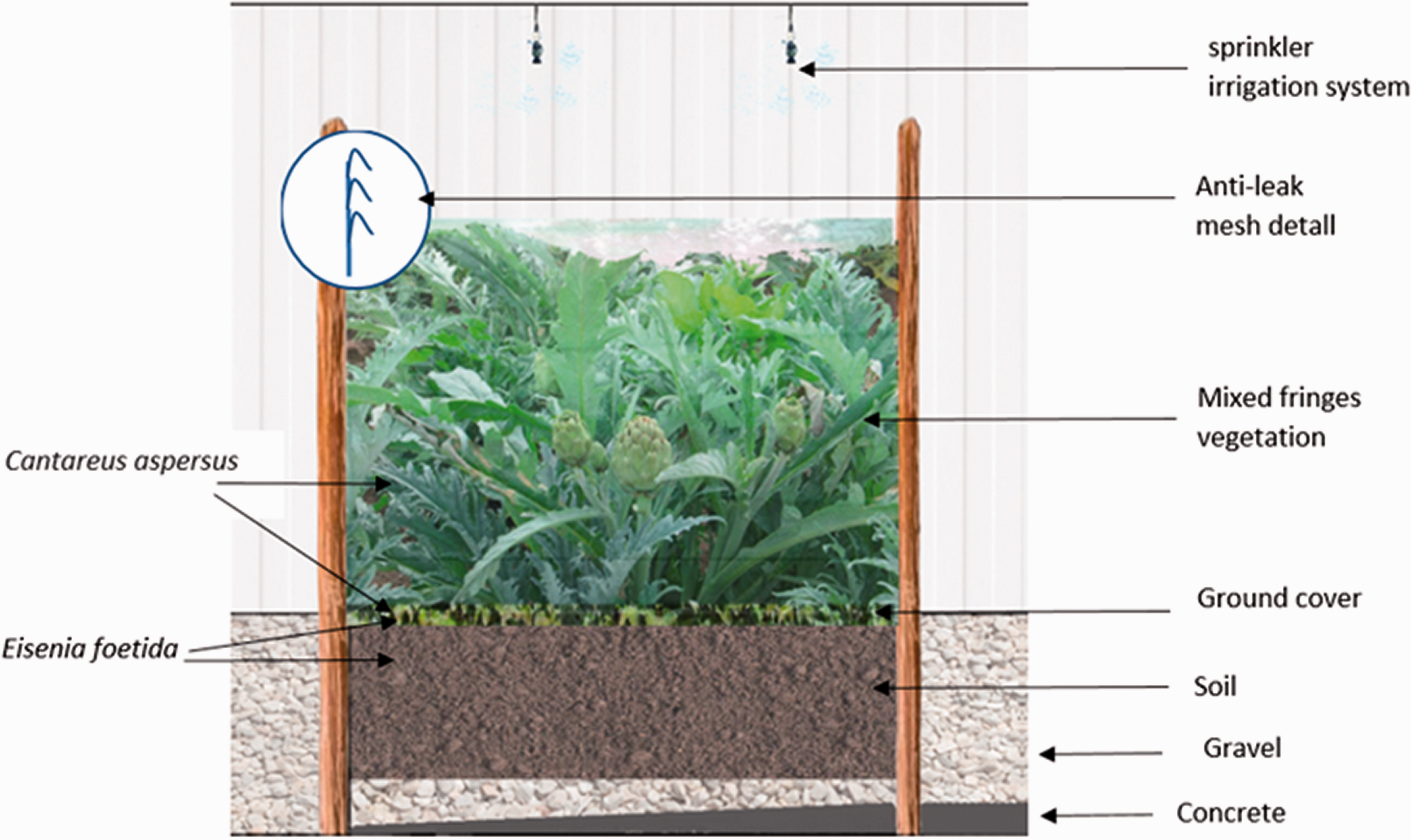
Cross-section of the experimental orchard (2.7 m long and 0.75 m wide). *Mixed fringes vegetation of Cynara cardunculus sp. (cardoon), Brassica olaracea subsp. sabellica (green cabbage) and Beta vulgaris subsp. cicla (beet).
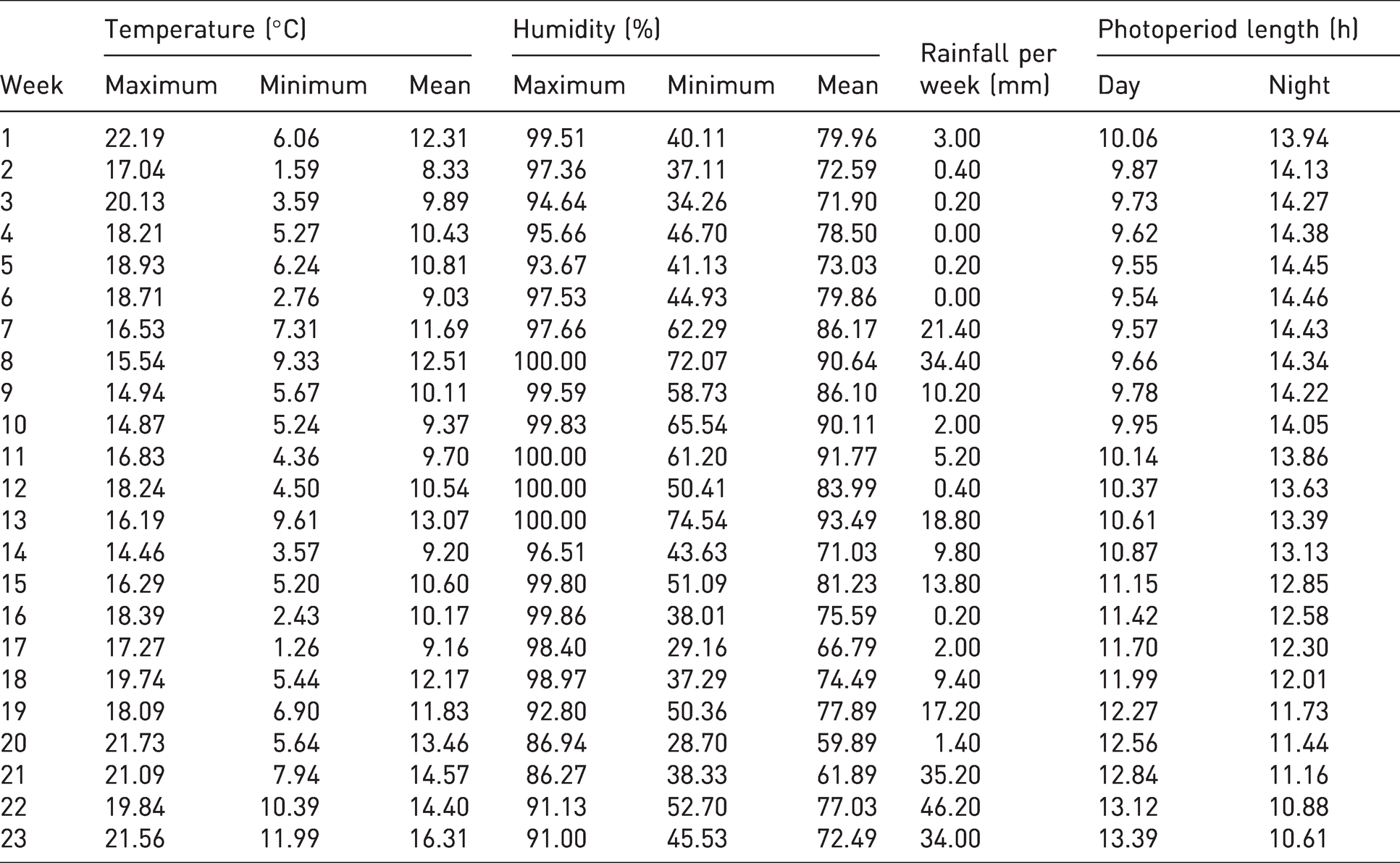
The sprinkler irrigation system was activated every 12 hours (8:00am and 8:00pm) for 5–20 minutes. Supplemental Table S2 shows the physicochemical water characteristics. Relative humidity was regulated with water twice daily, 21 allowing two main activity periods of snails to be established: during the daytime (the resting phase) with 10.93 hours of natural light, lower relative humidity and higher temperature, and overnight (the active phase) with 13.07 hours of darkness, higher humidity and lower temperature. The average temperature during the experimental period was 11.91°C (range 6.37–18.82°C). Table 1 shows the environmental conditions.
Weekly changes in environmental conditions for snail-rearing orchard.
A ground cover comprised of a vermicomposting bed integrated with residues and earthworms was incorporated in the experimental orchard. Approximately 500 g/m2 (n = 100–120) of Eisenia fetida, ranging in length from 4 to 8 cm, was added. Vermicomposting is considered a potential option in the hierarchy of integrated solid waste management, involving the stabilisation of organic material by the joint action of earthworms and microorganisms. 23 During the experiment, the ground cover was renewed every month.
Once the experimental orchard was prepared, it was sown with the snail brood. The snails were reared with a density of 500 individual snails per square metre.2,16 The feed was poured and dispersed manually over the leaves of the tallest plants. The animals were fed ad libitum every day at 8:30pm after evening watering with flour made up of commercial layer mash to which 20% calcium carbonate was added. The main constituents of the feed were dry matter (92.6%), crude protein (14.4% of dry matter), lipids (3.1% of DM), carbohydrates (51.8% of DM), neutral detergent fibre (11.8% of DM) and ash (30.7% of DM, including 10.3% calcium).
Measurements and statistical analyses
Morphometric traits used to describe snail growth usually include the live weight and the shell dimensions (length, height and width).10,24 Other authors have proposed measuring the mass of the snails. 25 Snail live weight is the most frequent trait for measuring growth,1,16,26–28 although the diameter of the shell is also used.24,29 Both traits are easy to obtain, direct and fast, and this process can be easily automated. 8 Moreover, it has been shown that there is a very strong correlation between live weight and shell diameter or length, as well as a high correlation among the three shell-size measurements.10,19,24 However, there are practical differences between both measures. Live weight can have high variability and asymmetrical distribution of individual weights born from the same clutch.1,8,10,30 The fit of the weight–age growth pattern is complex and lacks precision.1,31 In contrast, shell dimensions present low variability, have large repeatability, adjust to a normal distribution and facilitate the construction of growth models.17,24,29,32–34
In addition, a high correlation between shell growth and sexual maturity has been investigated,28,35 with a relationship between shell growth and peristome thickness reported. A snail is considered to be an adult when the shell peristome (shell edge) is reflected, that is, when shell growth is completed. 2 Snails with a growing shell and developed reproductive system (young) produce only sperm. Snails with a developed reproductive system and cessation of shell growth (adult) produce both sperm and eggs.
In this research, the shell diameter or length was used, which was a direct, fast and stable measurement, applying the protocol described below. The measurements were carried out when the snails were resting, each Friday during a two-hour period (from 10:00am to 12:00pm). First, each snail was picked up and placed on a squared sheet. With the help of a two-pointed compass, the shell diameter was marked on the paper (Figure 2(a)). Next, a digital calliper with a precision of 0.1 mm (Figure 2(b)) measured the shell diameter.

Shell diameter measurement.
To determine growth pattern fit, the normality of the distribution of the shell diameter each month was verified using Kolmogorov–Smirnov, Cramer–Von Mises, and Anderson–Darling tests. Bartlett’s test was performed to verify equality of the data variance (homoscedasticity). The weekly growth was compared by analysis of variance, establishing the week as a fixed effect. For the comparison of means, the Tukey method was used. Growth was modelled using asymptotic and non-asymptotic models. 36 Different growth models were fitted to explain the growth of C. aspersus: the von Bertalanffy’s growth model, the Gompertz’s growth model and the Richard’s growth model, logistic, quadratic and linear hyperbolic models.21,24
In the adjustment of the models, the NLIN procedure was used in SAS v9 (SAS Institute, Cary, NC). For the calculation of the parameters of the models, the method of Marquardt was used. The different growth models were fitted with iterations by means of the Levenberg–Marquardt method for nonlinear least squares. The selection of the best model took place by using the coefficient of determination (R2), the mean squared error (MSE) and Akaike information criterion (AIC). The AIC is a measure of the quality of a selected statistical model that best fits the data using the maximum likelihood technique. 37
Results
The animals grew progressively and continuously each week, with significant differences between weeks (p < 0.001; Table 2). Snails started with a low standard deviation (0.99) that increased progressively as the snail aged. The coefficient of variation was <20%. Nearly two-thirds (60%) of the snails achieved their commercial size (diameter = 28 mm) by week 21. No significant differences among sections were found (p > 0.05; Supplemental Table S3).
Snails’ shell diameter from 1 to 23 weeks (mm).
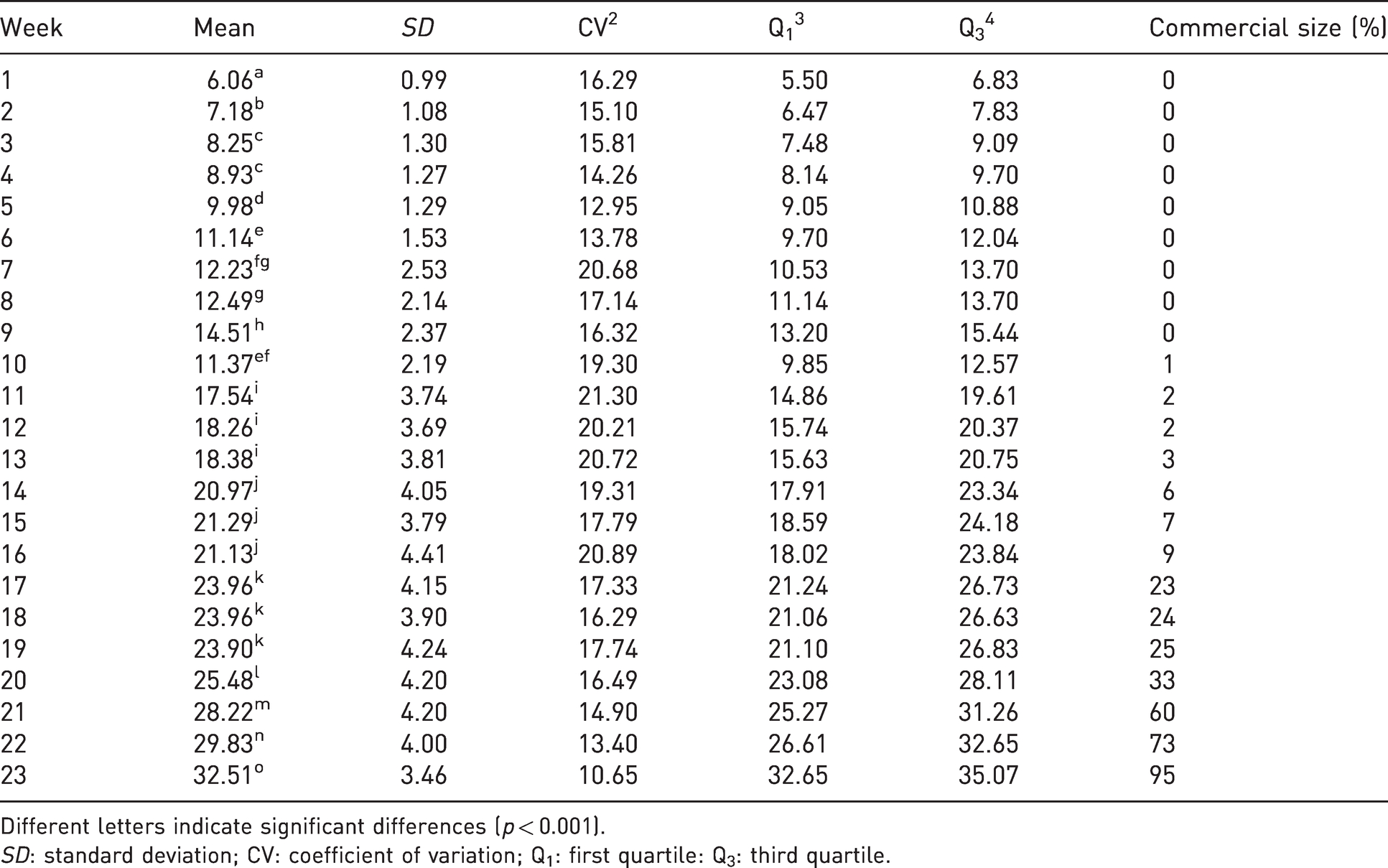
Different letters indicate significant differences (p < 0.001).
SD: standard deviation; CV: coefficient of variation; Q1: first quartile: Q3: third quartile.
The normality of the distribution of shell diameter was verified monthly with the three fit tests. The result obtained is shown in Supplemental Table S4. At months 1, 5 and 6, the statistics of adjustment to the normal distribution showed a non-significant statistical difference with respect to the normal distribution, which indicates that the length data recorded conform to a normal distribution. At months 2 and 3, the data did not fit a normal distribution. At month 4, the Cramer–Von Mises and Anderson–Darlin statistics indicated a significant difference regarding the fit of the data series to the normal distribution. However, the Kolmogorov–Smirnov statistic showed a non-significant difference with respect to the normal distribution.
Table 3 shows the growth parameters estimated for C. aspersus. The coefficient of determination (R2), MSE, AIC and the Durbin–Watson differences were calculated for each data set in each estimated model. The six proposed models were accepted and showed very similar values regarding the adjustment parameters. The coefficient of determination had the upper value for quadratic, logistic, and Richard models. Likewise, quadratic and logistic models presented the lower SEM. The quadratic model only requires one iteration against 11 of the logistic models. Finally, the lowest AIC values were estimated using quadratic and logistic models, which were considered the best models describing the growth of C. aspersus. The parameters of the quadratic equation from Table 3 were used to build the growth curve of C. aspersus (Figure 3).
Growth models and parameters estimated for C. aspersus.
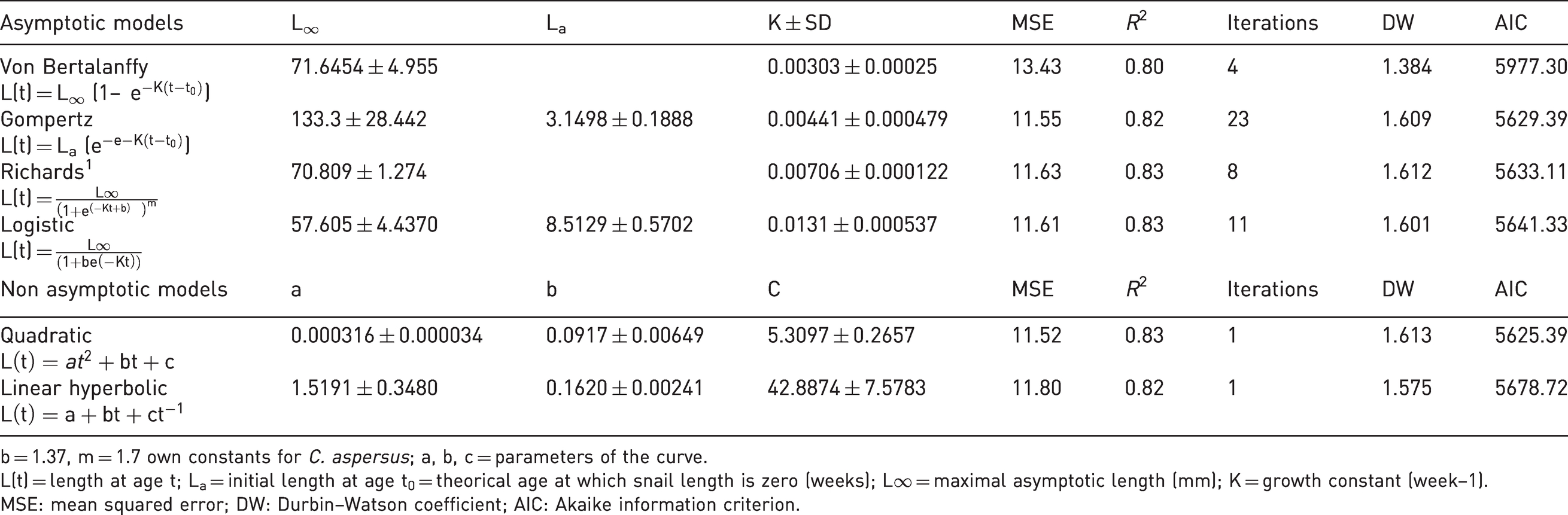
b = 1.37, m = 1.7 own constants for C. aspersus; a, b, c = parameters of the curve.
L(t) = length at age t; La = initial length at age t0 = theorical age at which snail length is zero (weeks); L∞ = maximal asymptotic length (mm); K = growth constant (week–1).
MSE: mean squared error; DW: Durbin–Watson coefficient; AIC: Akaike information criterion.
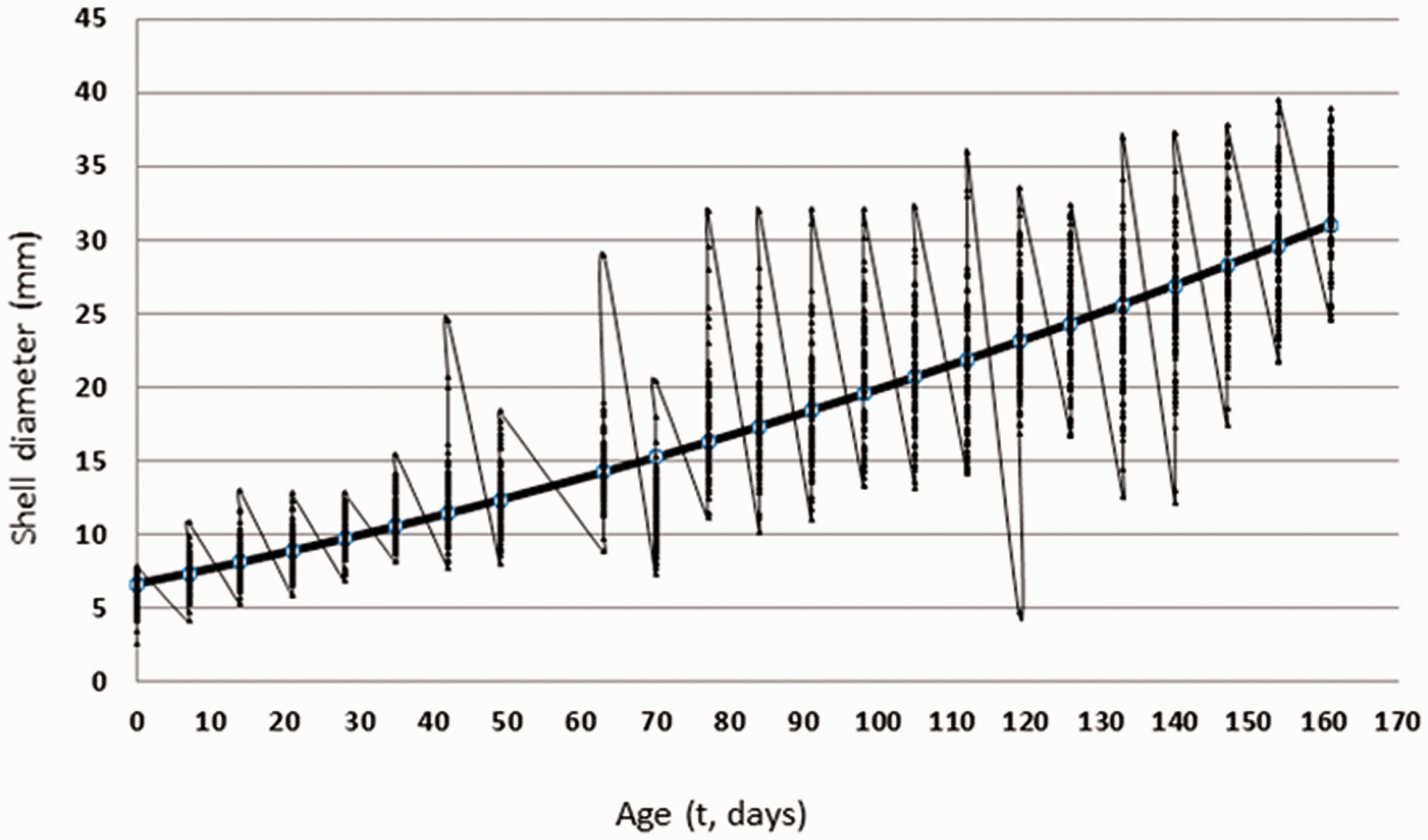
Estimated quadratic growth curve and observer values for C. aspersus.
Discussion
The results ensured the smooth running of the snail rearing system in the orchard in accordance with their behavioural activity and circadian rhythm, with two differenced phases: the active phase was nocturne, wet and with a low temperature; the resting phase was diurnal, dry and with a higher temperature. Irrigation in the morning favoured the dew point, acting as a signal for the phase shift. After watering with the sprinkler, the snails quickly rehydrated and began their resting period.11,37–40 An integrated rearing system was applied, producing homogeneous and rapid growth with high density of rearing and fattening from feed. Vegetation in was sown at the fringes and used as a useful and shelter surface for the C. aspersus. The production system factors (food, density, cleanliness, husbandry, etc.) can act either independently or synergistically to create a favourable or unfavourable environmental condition for raising snails. 18 In this sense, a direct relationship between snail handling and low growth rate on animal productivity and welfare has been described.24,41
In the orchard, the welfare of the animals was sought, favouring the snails’ behavioural activity based on their circadian rhythm and avoiding those stress factors that inhibit their growth. The curve shape and growth were considered as suitable indicators of the animal welfare and rearing system proposed.
42
The system had the following characteristics:
An integrated rearing system. In the orchard, feeding relationships were established between C. aspersus and E. fetida. We did not observe any negative interactions between the earthworms and the snails. The integration was done to recycle resources efficiently. The products or by-products of one species serve as a resource for the other – waste from the snails and plants is used by the earthworms. In addition, generated humus was used by the plants and snails.
23
In this case, the integration serves to make the best use of the resources and favours interspecific synergies.12,39 When snails are rearing in the presence of humus, they feed regularly on the humus and grow significantly faster. Humus increases the availability of organic nutrients in the diet, improving its digestibility, thus increasing nutritional efficiency as well as feed conversion ratio and protein efficiency ratio. It is possible that the increased digestibility was due to the action of microorganisms and the high catalytic capacity of enzymes that there were in the humus.17,43,44 However, further research is needed to confirm the nutritional contribution of the microorganisms present in the humus. Homogeneous and rapid growth with a concentrated diet. The C. apersus in this study came from a homogeneous sample of the same age and size. The snails grew progressively and continuously at a high growth rate with low individual variations. Intensive snail rearing, where snails were fed ad libitum on layers mash, was used. In the orchard, 60% of the snails achieved commercial size (diameter = 28 mm) in 21 weeks and 90% in 23 weeks compared to the 18 months needed naturally. Similar results were obtained under intensive culture conditions.10,27 The continuous growth observed during the experimental period can be explained by the favourable experimental conditions (relative humidity, temperature and food quality), which matched the circadian rhythm and individual preferences of C. aspersus. The results are slightly lower than the 240 days needed to reach commercial size on farms in Turkey.6,26 The fastest proportional growth occurred in the youngest snails and decreased over time.
11
High stocking density was used (500 snails/m2), avoiding negative effects on growth. During the first three weeks, the stocking density had a positive effect on growth.27,41,45 According to these authors, negative effects of stocking density on growth start to impact from the first month of life. Different indoor rearing densities (250–500 animals/m2) were tested.
2
It was observed that at low densities, the animals consumed more and were bigger and most of them became adults. However, the total amount of biomass produced is greater at higher densities. A higher population density decreased growth (p < 0.001).
45
In the same sense, other authors recommend an initial stocking density of 485 individuals per square metre for rearing juvenile H. lucorum, but after four weeks, stocking density should be decreased, thereby creating a healthier environment and supplying balanced food among individuals.
26
The adverse effects of high-density rearing were attributable to the crowding and accumulation of nitrogenous waste products.21,45 Mucus acts as a growth retardant.8,38 The increase density of population has a negative effect on growth due to chemical interactions between snails via envisaged mucus trucks. The effect due to chemical or behavioural interactions could be amongst the reasons of this negative effect.12,27 To avoid the build-up of excreta and other waste products, various husbandry practices were applied in the experimental orchard. High availability of a useful surface was favoured, and concentrations of animals were avoided at one point, using the leaves of the taller plants and foliage, which were used as tatters and drinkers. Likewise, the irrigation system favoured the cleaning of the plants, and the action of the earthworms reducing the effects of excreta and other waste products. High-density strip vegetation with mixtures of various species increased the useful surface for C. aspersus. A large number of rest and activity areas was favoured according to the snails’ preferences.
22
In addition, a high density of vegetation served to cushion extreme temperatures, both those <5°C and >25°C, which act as growth inhibitors.
20
The location of the orchard in a mild climate (continental Mediterranean) also favoured the results obtained. The relative role of environmental conditions on the growth of snails has been described previously.
11
The length of the growing season and temperature were additional factors in determining the differences in the snails’ growth and population dynamics between the sites.
The rearing and husbandry system proposed in the experimental orchard allowed rapid, homogeneous and standardised growth to be achieved for C. aspersus, with low individual variation and a normal distribution of size. The growth curve showed an inflection point at week 17, with the animals reaching commercial weight, increasing from 9% to 23%. The genetic potential of C. aspersus is wide and requires further research. Furthermore, snails’ selection of fast growth could allow strong improved in short time. 2
The results of this research showed a consistent positive relationship between shell diameter and age of C. aspersus under environmental conditions and a high determination coefficient (R2=83%).
In this research, the growth of C. aspersus was standardised. The models obtained were in line with the laboratorial results for juvenile snails.17,21 Although this is the first time that the experiment has been done for the whole snail’s life cycle under environmental conditions, the models obtained, quadratic and logistic, were highly consistent and present fit values very similar to other species. 46 The growth models obtained were suitable indicators of animal welfare and good growth in experimental snail orchards.
This research supports the use of C. aspersus in orchards under environmental conditions as an alternative to traditional models in animal experimentation. The snails showed high adaptability, and the built growth patterns were robust. So, the growth of the snails can be used as an appropriate and reliable indicator. The orchard model can be extended to other countries. Moreover, it would be necessary to adapt the model to the conditions of each climate because the relationships between shell diameter and age may also be largely affected by environmental factors such as temperature, experimental conditions, habitats, seasons and geographical location.6,7 In addition, the rearing system and the growth functions should be adjusted to other genera of close snails (Otala lactea, Otala punctata, Iberus gualtieranus alonensis, and Theba pisana, among others).
Supplemental Material
sj-pdf-1-lan-10.1177_00236772211043978 - Supplemental material for Integrated rearing system proposal for Cantareus aspersus in experimental orchards: Growth models
Supplemental material, sj-pdf-1-lan-10.1177_00236772211043978 for Integrated rearing system proposal for Cantareus aspersus in experimental orchards: Growth models by Anton Garcia, Cecilio Barba, Manuel Aragón, Jose M León, Carmen De-Pablos-Heredero and Ana Gonzalez in Laboratory Animals
Footnotes
Acknowledgements
We want to thank the Agropecuary Provincial Centre of Diputación of Córdoba, University of Cordoba (Spain), and AGR267 Group at Córdoba University for their support during this research.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
