Abstract
Complete blood count, serum chemistry values, and biological reference intervals were compared between two age groups (34–49 and 84–120 days old) of healthy male and female laboratory raised natal multimammate mice (
Introduction
Reference intervals (RIs), results that represent 95% of a healthy population, have an important role in interpreting clinical results of experimental laboratory animals to values from healthy populations.13,14 Normal baseline hematologic value averages and ranges have been published for 40 and 90 day old
The goal of this study was to develop species- and method-specific hematologic and serum biochemistry RIs for two age groups of male and female laboratory-bred natal multimammate mice (
Material and methods
Animals
Clinically healthy, laboratory-bred (originally wild caught in Mali and transported to the facility in 2013),
18
specific pathogen free, intact male (
Blood collection and analysis
Blood was collected via terminal cardiocentesis completed in multiple cohorts from non-fasted, anesthetized, natal multimammate mice using isoflurane anesthesia to effect between the times 08:00 and 11:00. The centesis site was cleaned with 70% ethyl alcohol prior to blood collection. After blood collection, natal multimammate mice were immediately euthanized while anesthetized via cervical dislocation using a commercial mechanical cervical dislocator (Stoelting Co., Wood Dale, IL, USA).
Blood was collected in a 1-mL slip-tip tuberculin syringe (BD syringes, Becton, Dickinson and Company, Franklin Lakes, NJ, USA) with attached needle (BD PrecisionGlide™ Hypodermic Needle 23 G x 1”, Becton, Dickinson and Company, Franklin Lakes, NJ, USA). The needle was removed prior to expelling blood into collection tubes. Blood was aliquoted into a serum separator tube (300–600 µL, Micro tube 1.1 mL Z-Gel, STARSTEDT AgG & Co., Nümbrecht, Germany) and an EDTA tube (200–500 µL, Micro tube 1.3 mL K3 EDTA, STARSTEDT AgG & Co., Nümbrecht, Germany).
EDTA blood-filled tubes were manually inverted per manufacturer recommendations and then placed on a tube rocker at room temperature until analyzed using a ProCyte Dx Hematology Analyzer (Idexx Laboratories Inc., Westbrook, ME, USA). Hematological parameters assessed include: red blood cell (erythrocyte) count (RBC), hemoglobin concentration (HGB), hematocrit (HCT), mean red blood cell volume (MCV), mean cell hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), platelet count (PLT), mean platelet volume (MPV), reticulocyte count (Ret #), total white blood cell (leukocyte) count (WBC), neutrophil count (NEUT #), lymphocyte count (LYMPH #), monocyte count (MONO #), eosinophil count (EO #), and basophil count (BASO #).
Serum separator tubes were manually inverted and allowed to clot at room temperature for at least 20 min according to manufacturer recommendations and then centrifuged at 10,000
A total of 236 EDTA blood samples and 182 serum samples were analyzed. Sex and age numbers per group can be seen in tables below.
RI analysis
RIs for the above analytes were determined according to ASCVP guidelines
13
using Reference Value Advisor.
19
Reference limits were calculated using the non-parametric approach unless sample size fell below
Statistical analysis
Statistical analysis software (Prism 8.20 for Windows, GraphPad Software, La Jolla, CA, USA) using two-way analysis of variance with Sidak multiple-comparison testing was used to compare hematologic and serum biochemistry values according to sex (male
Results
Hematology
Hematologic values were compared between age and sex for
Hematological reference means ± standard deviation (SD) and ranges for male and female natal multimammate mice (
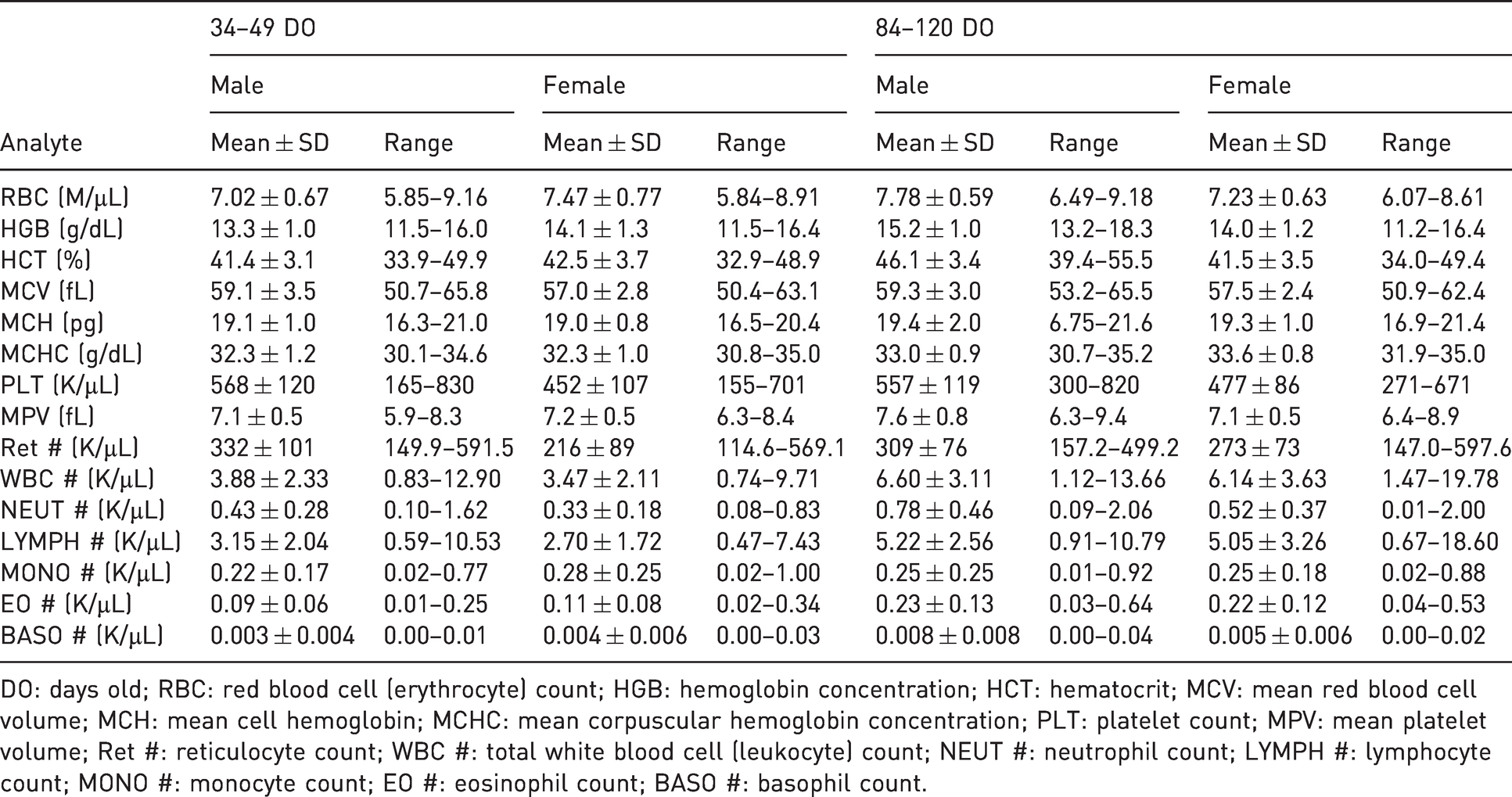
DO: days old; RBC: red blood cell (erythrocyte) count; HGB: hemoglobin concentration; HCT: hematocrit; MCV: mean red blood cell volume; MCH: mean cell hemoglobin; MCHC: mean corpuscular hemoglobin concentration; PLT: platelet count; MPV: mean platelet volume; Ret #: reticulocyte count; WBC #: total white blood cell (leukocyte) count; NEUT #: neutrophil count; LYMPH #: lymphocyte count; MONO #: monocyte count; EO #: eosinophil count; BASO #: basophil count.
Hematologic reference intervals and 90% lower and upper confidence interval limits of each analyte for male and female natal multimammate mice (
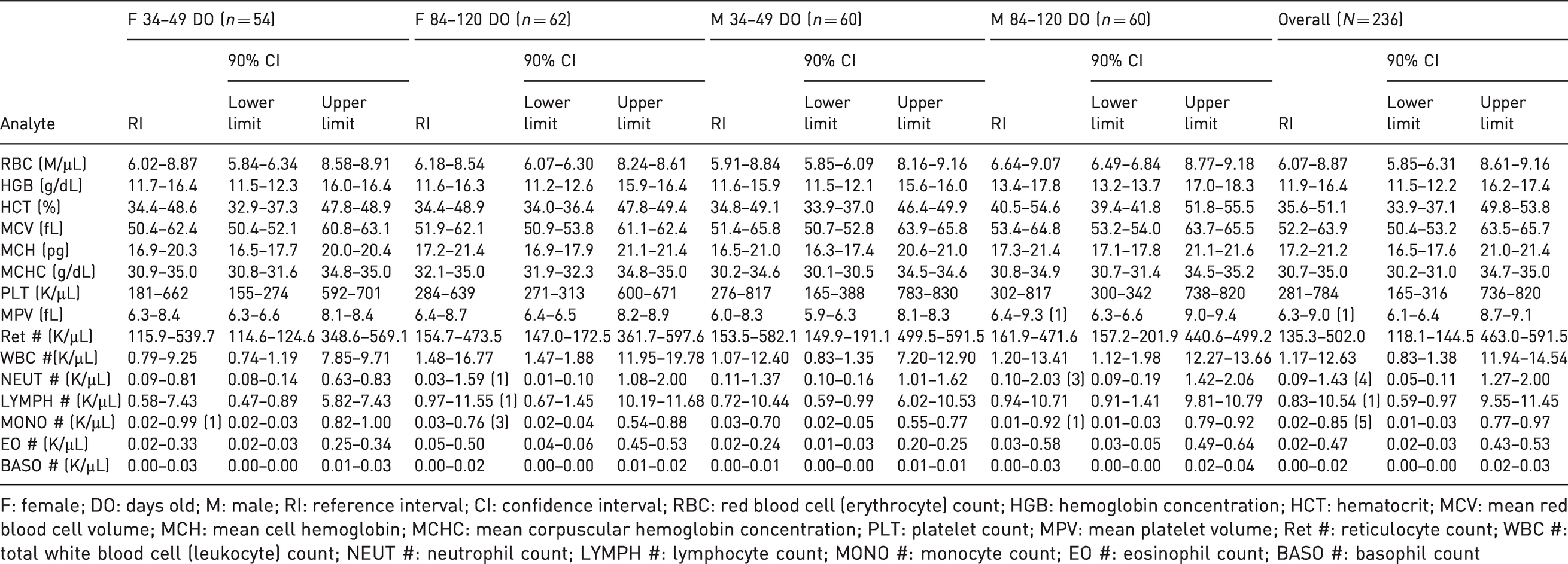
F: female; DO: days old; M: male; RI: reference interval; CI: confidence interval; RBC: red blood cell (erythrocyte) count; HGB: hemoglobin concentration; HCT: hematocrit; MCV: mean red blood cell volume; MCH: mean cell hemoglobin; MCHC: mean corpuscular hemoglobin concentration; PLT: platelet count; MPV: mean platelet volume; Ret #: reticulocyte count; WBC #: total white blood cell (leukocyte) count; NEUT #: neutrophil count; LYMPH #: lymphocyte count; MONO #: monocyte count; EO #: eosinophil count; BASO #: basophil count
Sex (male
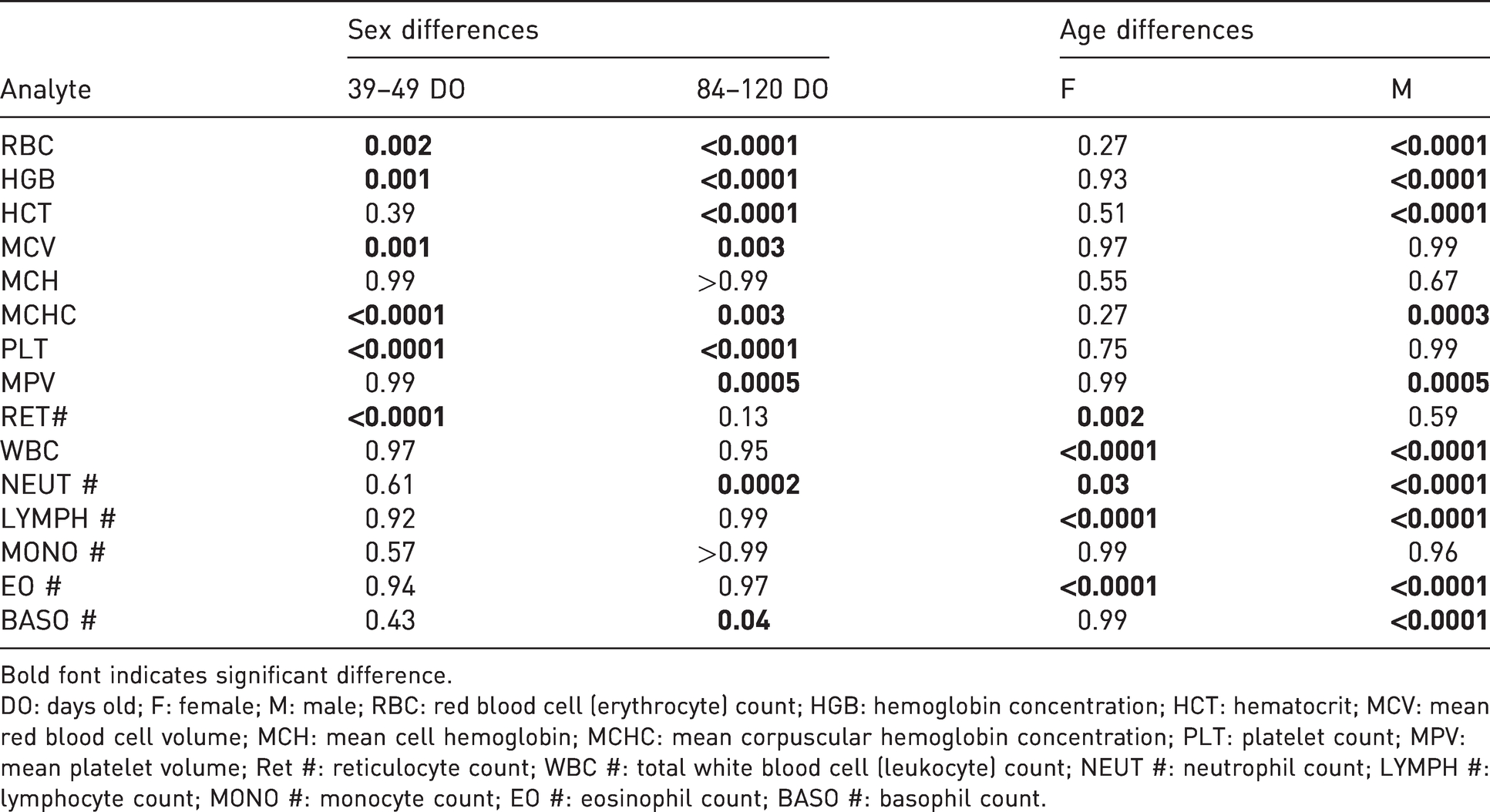
Bold font indicates significant difference.
DO: days old; F: female; M: male; RBC: red blood cell (erythrocyte) count; HGB: hemoglobin concentration; HCT: hematocrit; MCV: mean red blood cell volume; MCH: mean cell hemoglobin; MCHC: mean corpuscular hemoglobin concentration; PLT: platelet count; MPV: mean platelet volume; Ret #: reticulocyte count; WBC #: total white blood cell (leukocyte) count; NEUT #: neutrophil count; LYMPH #: lymphocyte count; MONO #: monocyte count; EO #: eosinophil count; BASO #: basophil count.
At 34–49 days old, female
Statistical differences in hematology values between the age groups were observed with both females and males. Female
Serum biochemistry
Serum biochemistry values were compared between age and sex for
Serum biochemistry reference means ± standard deviation (SD) and ranges from male and female natal multimammate mice (
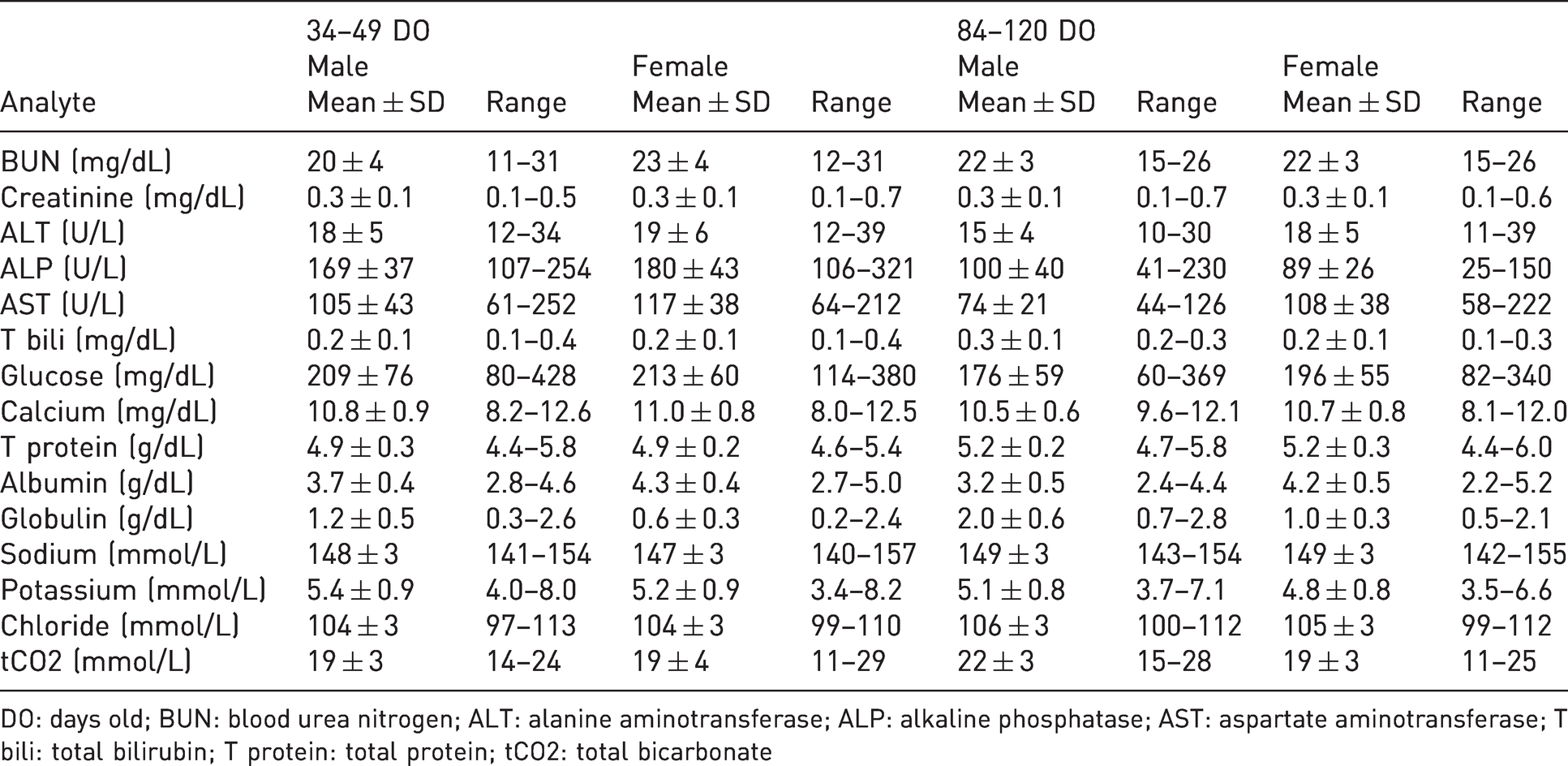
DO: days old; BUN: blood urea nitrogen; ALT: alanine aminotransferase; ALP: alkaline phosphatase; AST: aspartate aminotransferase; T bili: total bilirubin; T protein: total protein; tCO2: total bicarbonate
Serum biochemistry reference intervals (RIs) and 90% lower and upper confidence interval limits of each analyte for male and female natal multimammate mice (
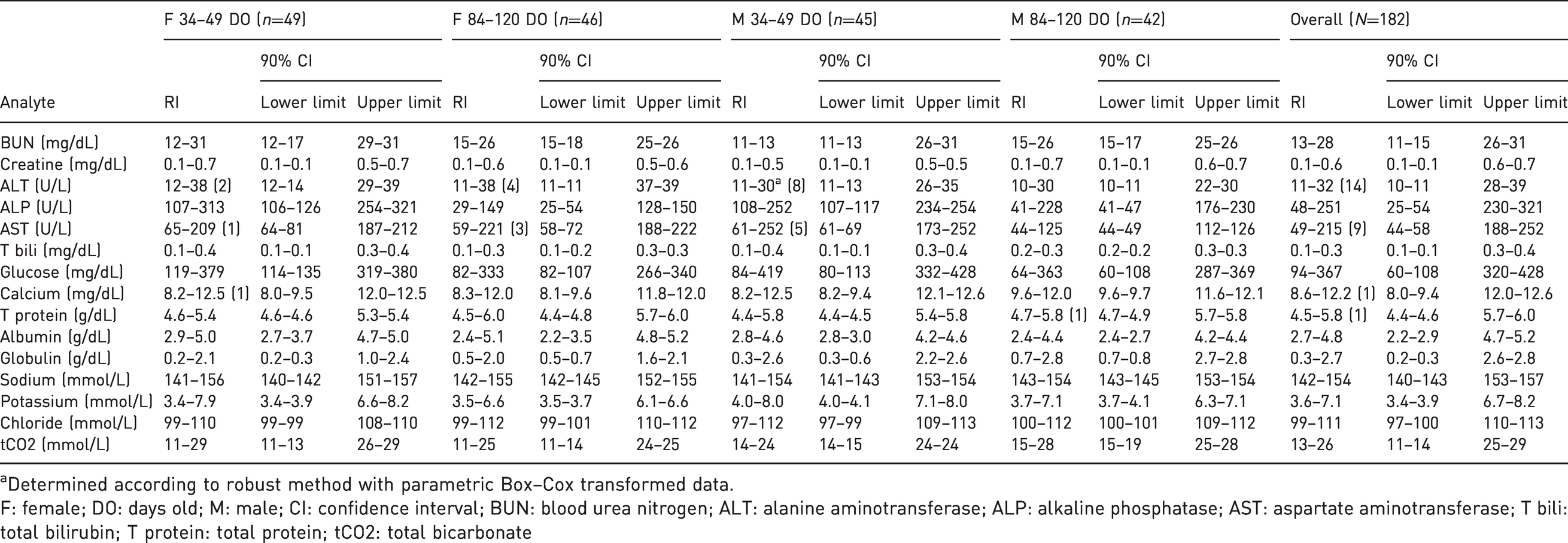
aDetermined according to robust method with parametric Box–Cox transformed data.
F: female; DO: days old; M: male; CI: confidence interval; BUN: blood urea nitrogen; ALT: alanine aminotransferase; ALP: alkaline phosphatase; AST: aspartate aminotransferase; T bili: total bilirubin; T protein: total protein; tCO2: total bicarbonate
Sex (male
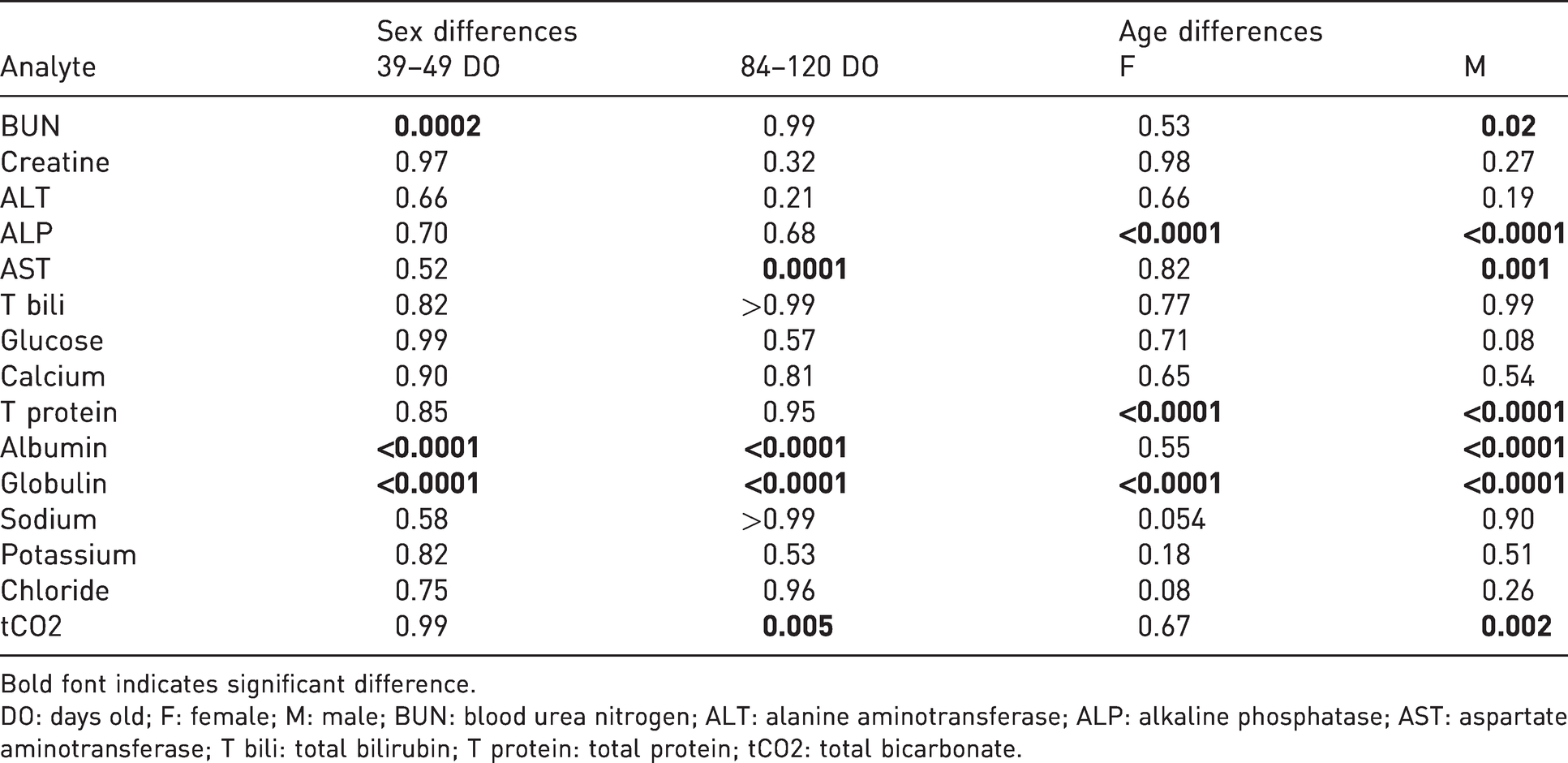
Bold font indicates significant difference.
DO: days old; F: female; M: male; BUN: blood urea nitrogen; ALT: alanine aminotransferase; ALP: alkaline phosphatase; AST: aspartate aminotransferase; T bili: total bilirubin; T protein: total protein; tCO2: total bicarbonate.
At 34–49 days old, female animals had significantly higher BUN (
Statistically significant differences in serum biochemistry values between the age groups were observed for both female and male
Discussion
Data collected from this study revealed significant differences between sex and age groups for both hematology and serum biochemistry values. These findings show the importance of species- and method-specific RI for both sexes at multiple different age ranges, which has also previously been emphasized.13,20 The age ranges in this study were chosen because they are the most common age groups used in infectious disease research at our facility and are separated by sexual maturity. This study provides a statistically robust complete hematology and clinical chemistry RIs for both sexes at different ages, unlike current published reports.
Hematologic findings described here are in similar ranges to previously published findings for
Differences in hematologic parameters between sexes of the same age have been described previously for other rodents,22,23 but not with
WBC count means for both sexes and age groups described in this study were lower than previously reported by Kagira et al. 15 but were similar to those of Wozniak et al. 16 This could be due to blood collection location and colony health as Kagira et al. collected blood from the tail vein while our study and Wozniak et al. collected blood via cardiocentesis. Previous research has shown that blood collected from the tail (room temperature or heated) or other peripheral blood collection locations of laboratory mice or rats results in significantly more WBCs compared with blood collected from the heart.24,25 Additionally, Wozniak and colleagues’ colony and our colony are free of common rodent pathogens according to environmental PCR samples and dirty bedding sentinel serology results, while published hematology data reported by Kagira et al. was generated from colonies recently procured from the wild with intermittent parasitic infestations. 15 Infection with certain pathogens may elicit inflammation resulting in significant changes within the leukogram. More specifically, parasitic infections may cause eosinophilia. These leukogram differences between the published results and our study illustrate the necessity for specific pathogen free colonies. Due to a lack of differential counts, a comparison of the affected leukocyte population (neutrophils, lymphocytes, eosinophils) between Kagira et al. and our study is impossible. Other potential explanations for the difference between WBC counts between the two studies could be husbandry (feed and bedding) and age differences.
Serum biochemistry values described are similar to previously reported values for
Differences in ALP between the age groups was expected as young, growing animals tend to have higher measurable ALP than adults due to elevations in the bone isoform of ALP. Studies have shown that this is true in mice and rats.
26
Similar to previous reports of
RIs were calculated using a previously published set of macroinstructions called Reference Value Advisor (RVA)
19
referenced by the ASCVP guidelines.
13
Some of the parameter’s 90% CIs determined by the RVA are wider than recommended by the ASVCP guidelines.
13
It is recommended that CI should not exceed 0.2 times the width of the RI. Increasing animal numbers could improve the width of the CI, due to CI width being dependent on sample size.
13
ASVCP guidelines recommend at least 120 samples per partition to determine 90% CI non-parametrically, unless bootstrap method is used.
13
Obtaining 120 reference samples per sex and age could be considered unethical as it would be time consuming and costly with little benefit and stress to more animals. The goal of this study was to obtain
The ASCVP guidelines are modeled after the Clinical Laboratory and Standards Institute recommendations with specific language for veterinary species and consider partitioning criteria, selection criteria, exclusion criteria, and pre- and post-analytical procedures. Previous publications assessing reference ranges of
In conclusion, findings from this study provide serum biochemical and hematologic parameter baseline reference means, ranges, and intervals for laboratory raised
Footnotes
Acknowledgements
The authors would like to acknowledge the dedication and hard work of the Rocky Mountain Veterinary Branch technicians for carefully monitoring and caring for the animals during this study and members of the NIAID International Center for Excellence in Research (ICER) in Mali, especially Nafomon Sogoba and Sidy Bane, for their support of the field and laboratory work. Underlying research material related to the paper is available upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: financial support for this study was supported by Intramural Research Program and the International Center for Excellence in Research of the National Institute of Allergy and Infectious Diseases, National Institute of Health.
