Abstract
Bats play a key role as reservoir hosts of many emerging viral diseases with zoonotic potential. However, little is known about the laboratory reference intervals (RIs) of bats, especially Egyptian fruit bats (
Introduction
Bats are classified in the order Chiroptera and have attracted interest based on their role as reservoir hosts for highly pathogenic and emerging zoonotic viruses.
1
Egyptian fruit bats (
The Egyptian fruit bat is considered a valuable experimental animal and is one of the most investigated bats because of its role as a virus reservoir and its particular use of the sensory systems.4,5 They are also often kept in zoological parks in various countries. Although in the literature there are many studies on the laboratory data concerning Megachiroptera,6–10 haematological and biochemical reference intervals (RIs) for Egyptian fruit bats are lacking. Although the serum protein electrophoresis of these animals may provide useful biological information it has not been evaluated before.
Most biological data from
The aim of this study was to establish haematological, biochemical and electrophoretic RIs, obtained with automated instruments, from captive Egyptian fruit bats. We believe that our results should help scientists to study the role of bats as a reservoir host for viral infections, and to evaluate the possible negative effects of viruses on the overall health status of bats as well as on specific systems or organs. Our study could also help veterinarians and zoologists in the health monitoring and disease prevention of both wild and captive bats, especially the
Material and methods
Animals
Twenty-one Egyptian fruit bats (11 females and 10 males) were obtained by the Veterinary Teaching Hospital of the University of Milan from a private owner in northern Italy, in order to perform gonadectomy. The bats had been housed in a metal aviary (height 4 m, width 3 m, length 4 m) and fed
During hospitalization for the surgical procedures, the bats were housed together in a large metal aviary (height 2.5 m, width 1.5 m, length 2 m) in a temperature- and humidity-controlled room (22–25°C and 60% humidity) with a light/dark alternation period of 8/16 hours. Bats were fed
Before surgery, bats were taken out of the aviary one by one and gently placed in a canvas bag and weighed with laboratory precision scales (Precisa BJ610C, Precisa Instrument, Dietikon, Switzerland). The bats were randomly administered two different anaesthetic protocols (protocol 1, intramuscular injection of dexmedetomidine and ketamine; protocol 2, intramuscular injection of dexmedetomidine, butorphanol and midazolam; anaesthesia was maintained with isoflurane in both protocols; drug dosages are not shown as they are the subject of another publication).
Before surgery, a complete physical examination was performed under anaesthesia and wingspan length and body size (head to tail length) were recorded. The age of the bats was unknown, since they lived together and no marking methods for identification had been applied. However, the bats’ age was determined during surgery based on anatomical observations, considering a mature gonadal or prepubescent gonadal development for males and the presence or absence of the foetus in the uterus for females. Accordingly, bats were identified as adult females (
Sampling
From each bat, 0.9–1.0 ml of peripheral blood (corresponding to the maximum of 1% of the body weight) was collected under general anaesthesia from the cephalic vein along the leading edge of the patagium. All samplings were performed with the owner’s consent as part of a routine health assessment. Specifically, blood was drawn for preoperative laboratory testing. According to the guidelines of our institution (University of Milan) for routine diagnostic analyses, formal approval from the Institutional Animal Care and Use Committee was therefore not required (Ethics Committee decision 29 October 2012, renewed with the protocol n° 02-2016). Immediately after sampling, the blood was divided into two aliquots:
Haematology
For each bat, CBC was performed on EDTA fresh whole blood, using an impedance cell counter (MS4vet Melet Schloesing, Osny, France), which requires a small volume of blood for the analysis (<30 µl). The following haematological variables generated by the instrument were recorded: erythrocyte count (RBC), total leukocyte count (WBC), platelet count (PLT), haemoglobin concentration, mean corpuscular volume (MCV), haematocrit (HCT), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC) and red cell distribution width (RDW).
For each sample, the leukocyte differential count provided by the instrument was checked microscopically on blood smears stained with a modified Romanowsky rapid stain (Dif-stain kit, Titolchimica S.P.A., Rovigo, Italy) by counting at least 100 nucleated cells. The absolute number of each leukocyte population was then calculated based on the total WBC number and on the percentage of each cell population, as provided by the manual differential. The blood smears were also checked for any other possible morphological peculiarity. The percentage of polychromatophilic erythrocytes was determined by counting polychromatophils and mature erythrocytes in five randomly selected 1000× microscopic high-power fields (HPFs). The percentage of polychromatophils was then calculated as the number of polychromatophils/5 HPFs divided by the number of RBC/5 HPFs, which was then multiplied by 100 to obtain the percentage.
Clinical chemistry
Biochemistry profiles were performed on sera using an automated chemistry analyser (Cobas Mira; Roche Diagnostics, Basel, Switzerland) and reagents provided by Real Time Diagnostic System (Viterbo, Italy). Quality control was performed before each assay with two levels of human control sera (Precinorm U and Precipath U; Real Time Diagnostic System). The instrument was calibrated with human-based calibrators (Calibrator; Real Time Diagnostic System). The serum concentration or activity of the following analytes was measured: urea (urease method), glucose (GOD-POD), total protein (Biuret), alkaline phosphatase (ALP, kinetic IFCC), creatinine (Jaffe), alanine aminotransferase (ALT, kinetic IFCC), total calcium (ortho-cresolphthalein), inorganic phosphate (molybdate), creatine kinase (CK, CK-NAC) and triglycerides (GOD-PAP).
Serum protein electrophoresis
Agarose gel electrophoresis was performed as previously described 14 using an automated system (Hydrasis, Sebia Italia Srl, Bagno a Ripoli, Florence, Italy) and kits provided by the manufacturer. Briefly, a 0.8% agarose gel was run in a Tris-barbital buffer at pH 8.6, with a migration time of 7 min at 800 V. Gels were stained with amidoschwarz, destained and dried for scanning by the gel scanner. Data were then transferred to the software program and visually inspected to correct for possible errors in fractionation generated by the software (Phoresis, Sebia Italia Srl). Absolute concentrations (g/l) for each electrophoretic fraction were calculated based on total serum protein and on the percentage corresponding to the area under each peak.
Statistical analysis and generation of RIs
Specific RIs were generated using an Excel spreadsheet with the Reference Value Advisor (v 2.1) set of macroinstructions.
15
The software performs the following computations recommended by the International Federation of Clinical Chemistry-Clinical and Laboratory Standards Institute:
16
descriptive statistics; tests of normality (Anderson–Darling with histograms and Q–Q plots); Box–Cox transformation; test of symmetry; and outlier analysis. The generalized Box–Cox transformation was performed by the software, according to the following equation:
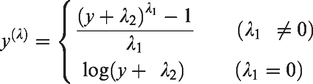
For each analyte, the software automatically generates a spreadsheet with the RI report generated using both standard and robust methods on both the native and the transformed data. In accordance with the ASVCP guidelines, of all the generated RIs, those with the lowest ratio between the confidence interval width and the reference interval width were chosen. Specifically, when 20 or more reference values were available and if the native data had a Gaussian and symmetrical distribution, the reference intervals were calculated with the robust method on non-transformed data. The robust method on transformed data was used if the native data did not show a Gaussian distribution, but symmetry was achieved after Box–Cox transformation. When fewer than 20 reference values were available, the reference intervals were not determined and only the mean, SD, median, minimum and maximum values were reported. The reference values obtained for each bat regarding these latter parameters are included as supplementary material.
Additional statistical analyses were performed using Analyse-it v. 2.30 (Analyse-it Software Ltd, Leeds, UK). For all the variables considered, the possible differences between male and female bats, between sexually immature and adult bats, and between the two anaesthetic protocols were also investigated with a non-parametric test for independent data (Mann-Whitney U test). In addition, the possible correlation between the analytical variables and bats’ weight, the polychromatophil percentage and haematocrit values and between the glucose concentration and all the other analytes were evaluated with Spearman’s test.
For all the statistical analyses, the level of significance was set at
Results
No statistically significant differences between males and females or between adult and juvenile bats were recorded in terms of weight, body size and wingspan length.
The results and RIs concerning the haematological parameters are reported in Table 1. In all the specimens, the microscopic evaluation of the blood smears revealed mild polychromasia (defined as the percentage of polychromatophilic erythrocytes, Figure 1(a)) ranging from 0.5 to 2.5%, and did not correlate with the haematocrit value (Spearman’s Rho (
Haematological reference intervals (RIs) for Egyptian fruit bats (
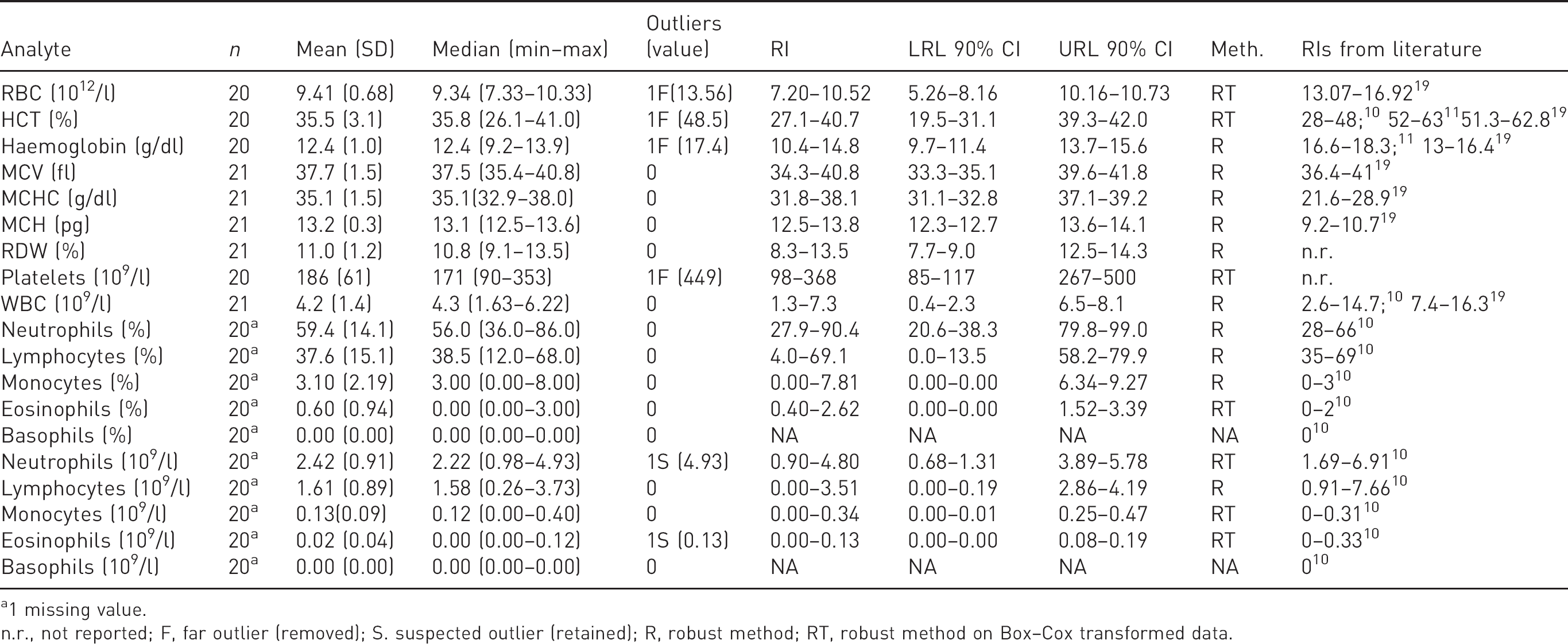
1 missing value.
n.r., not reported; F, far outlier (removed); S. suspected outlier (retained); R, robust method; RT, robust method on Box–Cox transformed data.
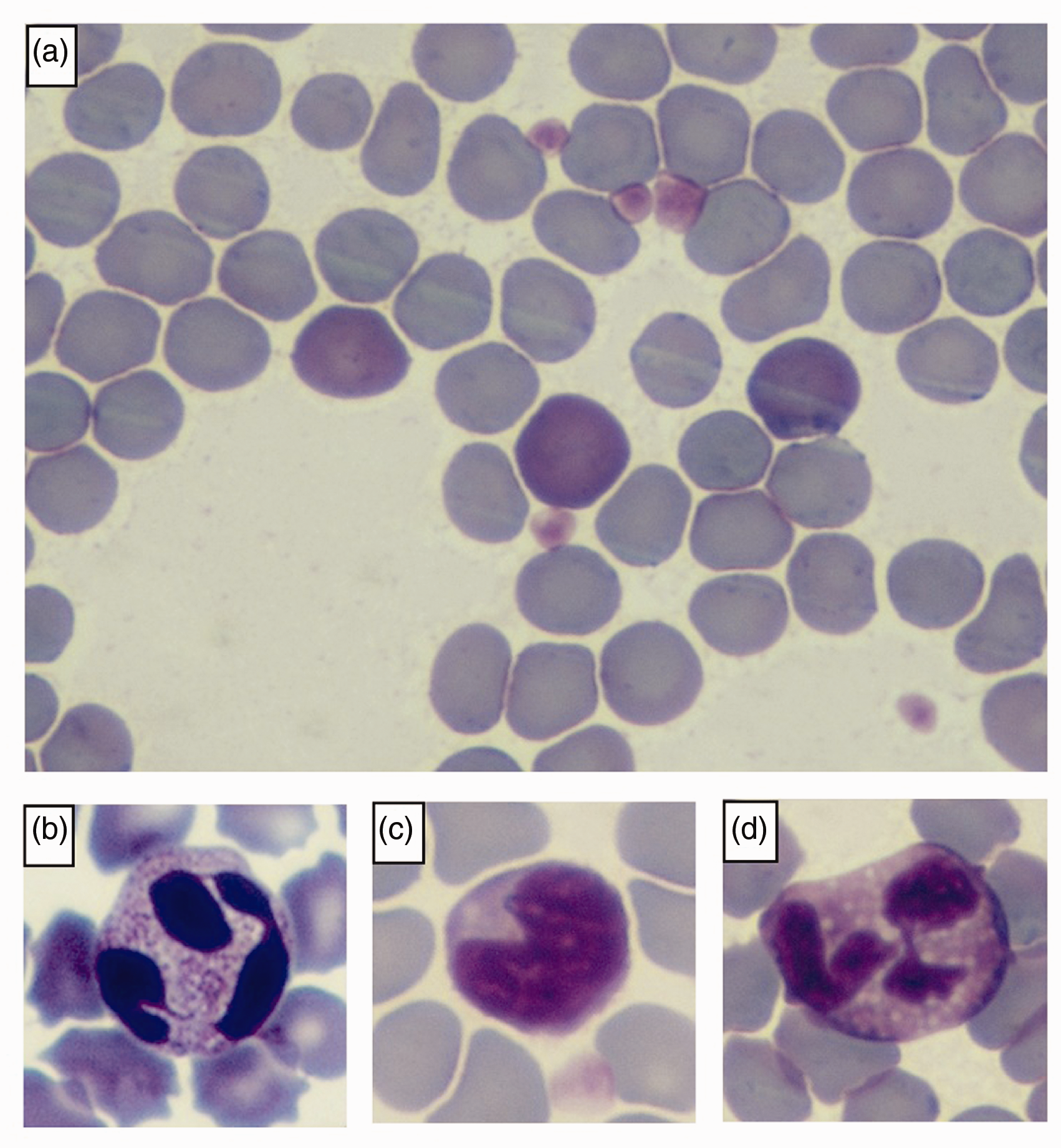
Microscopic features of blood smears from healthy
The morphology of the neutrophils was characterized by a fine eosinophilic granulation in the cytoplasm (Figure 1(b)). Lymphocytes and monocytes were similar to those of other mammals, in some cases showing a nuclear indentation (Figure 1(c)). Eosinophils, observed in 16 out of 21 bats, showed a cytoplasm rich in homogeneous round, light lavender, small granules (Figure 1(d)), whereas basophils were not detected in this study. Haemoparasites were not observed at morphological examination in any of the blood smears. The RIs for the haematological variables overlapped with those of most domestic mammalian species
18
and no differences were recorded based on the sex or the age of bats. The mean RBC count was higher in the animals anaesthetized with protocol 1 than with protocol 2, although the difference was not significant after Bonferroni correction (
The results and RIs concerning clinical chemistry analysis are reported in Table 2. Due to the insufficient sample volume, less than 20 reference values (RVs) were available for ALP (two missing values), total calcium (two missing values) and triglycerides (seven missing values). In accordance with the ASVCP guidelines, RIs for these parameters were not generated, and raw data are supplied as supplementary material (Supplementary table 1).
Biochemical reference intervals (RIs) for Egyptian fruit bats (
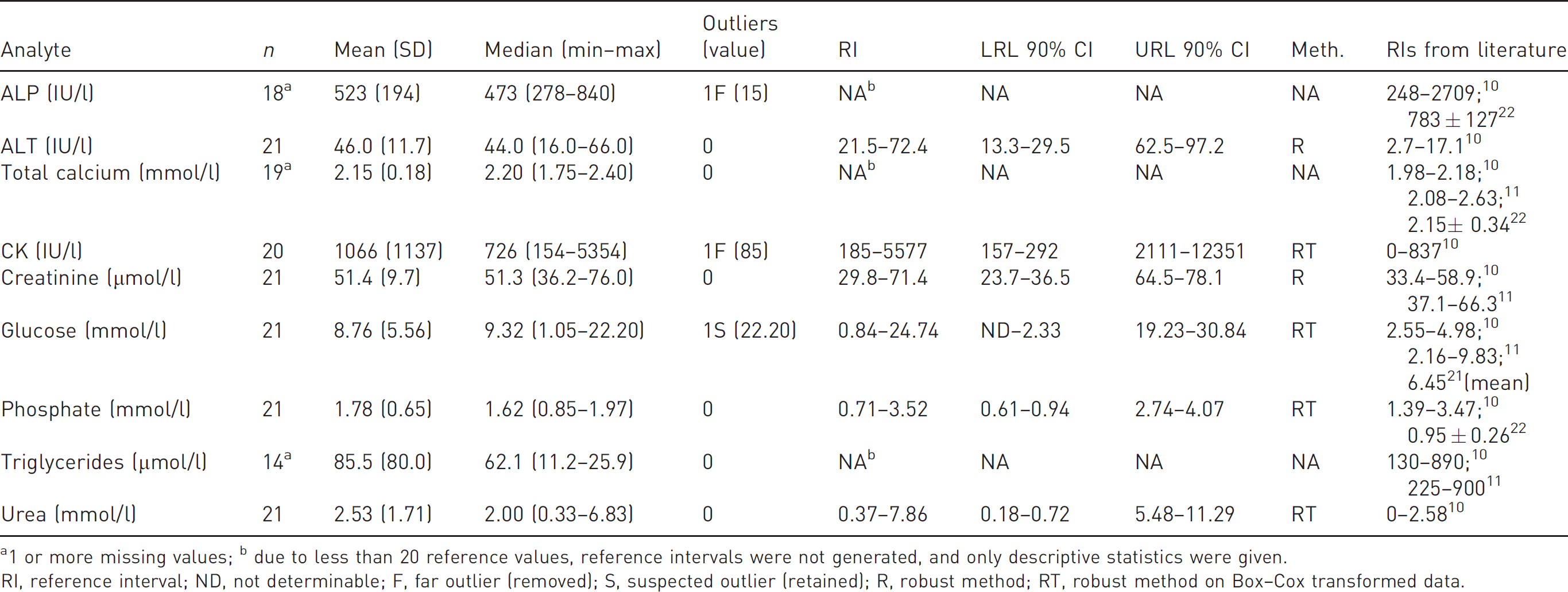
1 or more missing values; b due to less than 20 reference values, reference intervals were not generated, and only descriptive statistics were given.
RI, reference interval; ND, not determinable; F, far outlier (removed); S, suspected outlier (retained); R, robust method; RT, robust method on Box–Cox transformed data.
For most of the analytes investigated, biochemistry values were similar to those reported for other mammalian species 20 and for bats.10,11,21,22 The few exceptions were as follows: a wider RI for glucose was determined, however the mean value observed in this study overlapped data reported in other studies;10,11,21 mean and median values of CK activity were considerably higher than those reported for most other mammals and showed a five times higher upper reference limit (URL) than those already reported in bats; 10 triglycerides were considerably lower compared with values of other mammals, 20 and also slightly lower than those observed in bats;10,11 and finally, the mean urea concentration was similar to those of other mammals but the URL was three times higher than those of bats. 10
No statistically significant differences were recorded for any of the considered analytes between male and female bats, juvenile and adult bats, or between the two anaesthetic protocols, and for all the correlations after the Bonferroni correction. However, juvenile bats showed a higher ALP activity than adults (
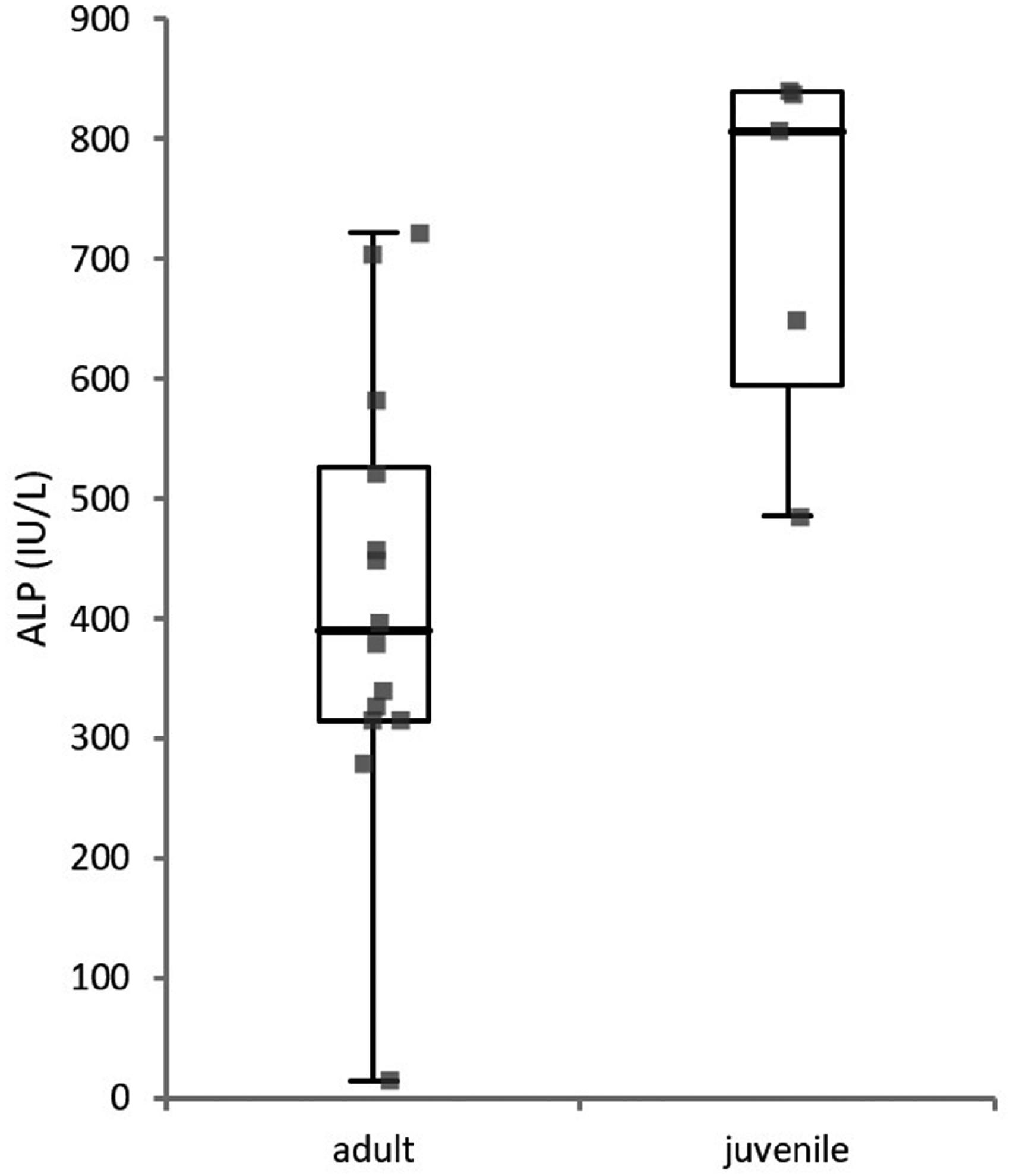
Alkaline phosphatase activity differs according to the age of bats (
Electrophoretic results and RIs are reported in Table 3. As shown in Figure 3, the fractionation of serum proteins highlighted a clear subdivision into five fractions (albumin, α-, β1-, β2- and γ-globulins). A moderately higher albumin content (and, as a consequence, of the albumin/globulin ratio) compared to other mammals
20
was recorded, with similar values to those reported in bats by Hall et al.
10
In contrast these latter authors observed a two times higher lower reference limit for total globulins compared to that recorded in our study. No differences between males and females or between juvenile and adult bats were recorded, whereas the mean absolute β2-globulin concentration was higher in bats anaesthetized with protocol 1 than with protocol 2 (
Electrophoretic reference intervals (RIs) for Egyptian fruit bats (
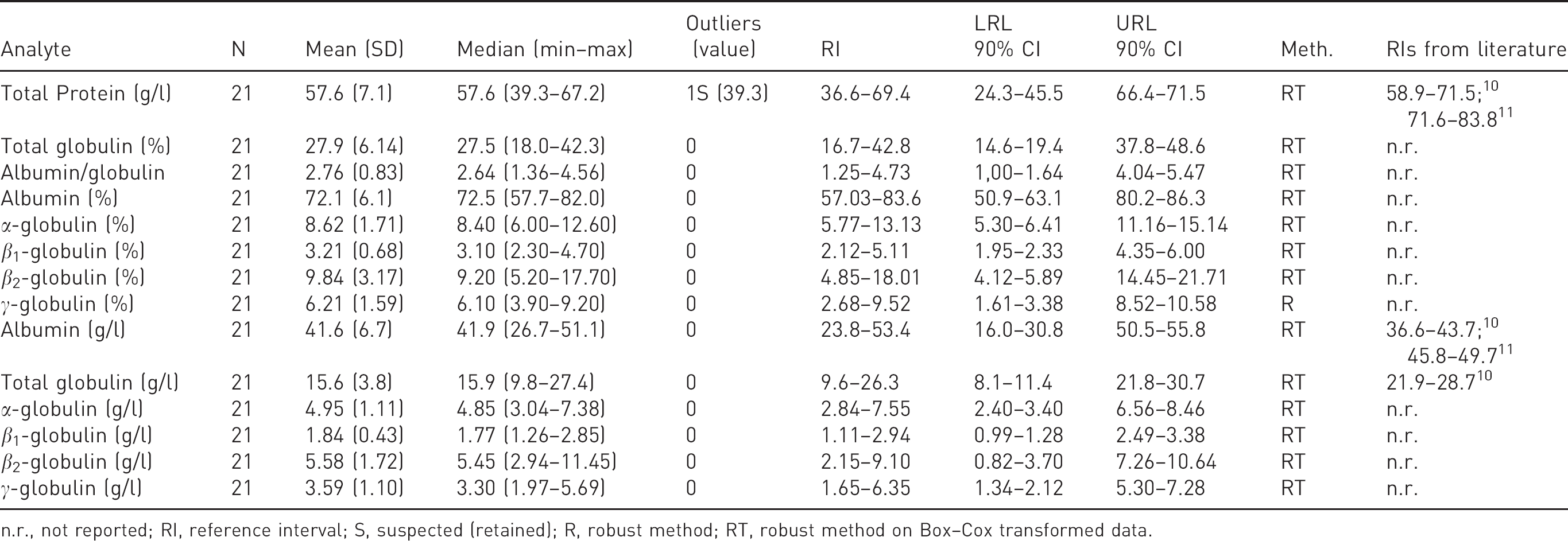
n.r., not reported; RI, reference interval; S, suspected (retained); R, robust method; RT, robust method on Box–Cox transformed data.
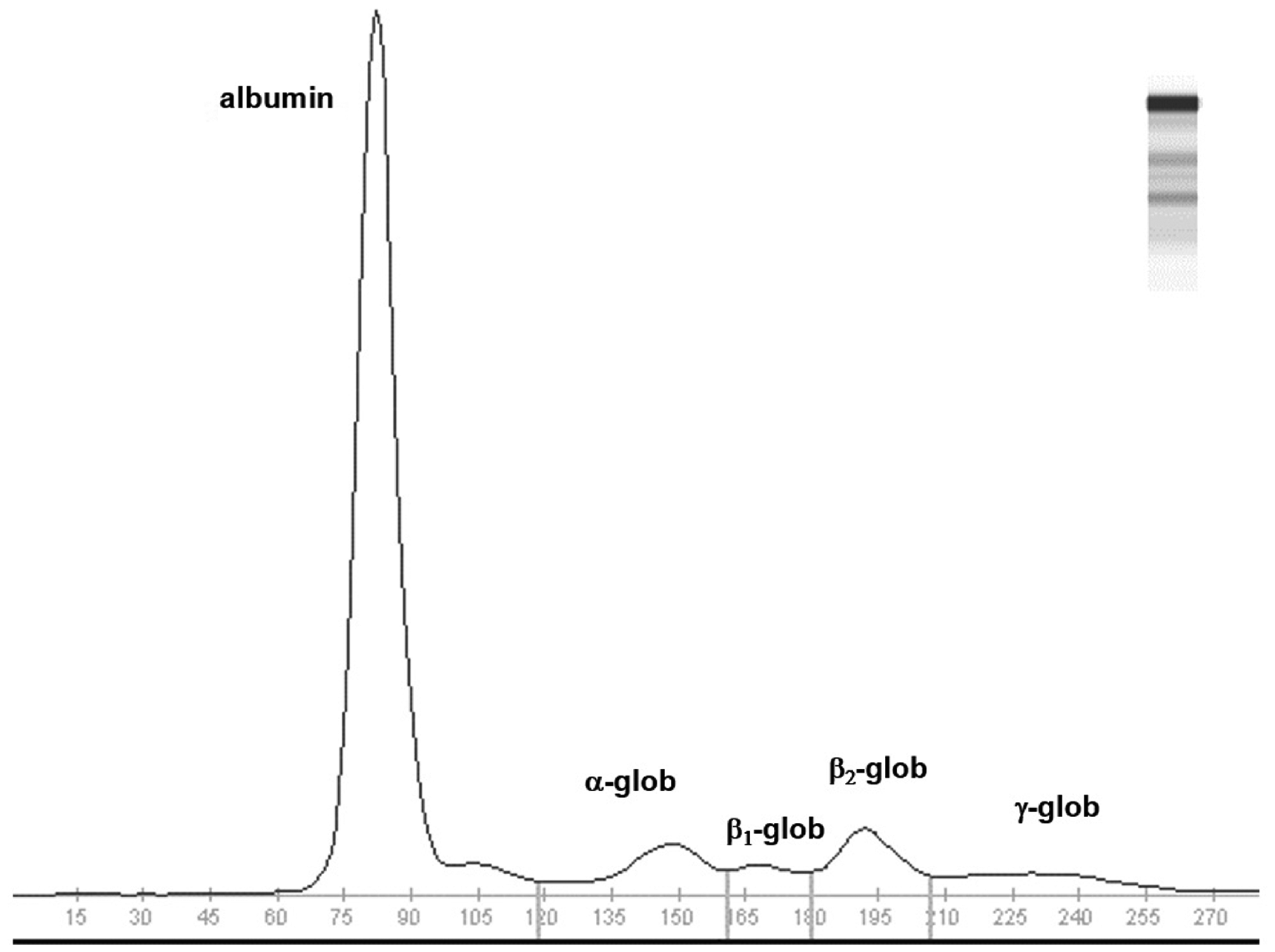
Electrophoretograms obtained with agarose gel electrophoresis on bat serum (
Discussion
Currently, there is no complete biological dataset including CBC, serum biochemistry and electrophoresis for the Egyptian fruit bat
Most of the data collected in the present study were comparable both to those reported in other mammals and in bats,6,10,11,19 with a few exceptions. In some cases, unfortunately, the low number of animals enrolled affected the statistical significance of the differences observed.
Blood cell morphology did not show significant abnormalities, except for mild polychromasia, which was also observed in flying foxes. 10 In domestic mammals, anisocytosis and polychromasia are usually associated with regenerative anaemia, 23 whereas a high concentration of reticulocytes is normal in young rodents and rabbits. 24 Since in this study polychromasia was detected regardless of the haematocrit values, it seems to be associated with a normal erythrocyte maturation in this species, in line with rodents and hedgehogs.24,25 Haemoparasites have been reported in wild bats in Africa, America and Europe26–28 but none of the bats in our study revealed haemoparasites in blood smears. Molecular tests are necessary to definitively rule out the infection, but none of the bats showed clinical signs of disease possibly associated with haemoparasites. Moreover, in northern Italy most of the known vectors are not present, making the spread of these pathogens less likely. 27
The haematocrit and haemoglobin concentrations were slightly lower than those reported in wild Chiroptera.10,11,19 This finding, in the absence of anaemia, could be due to a variable degree of dehydration in wild bats compared to captive bats, which have more regular access to water and food. Wild bats may also suffer from more evident stress than captive bats, which are more used to human contact, reflecting a higher haematocrit value due to stress-related spleen contraction. 29 The differences observed with the previous study 19 may be due to the different sampling technique, the anaesthesia effect, or the haematological instrument used.
Leukocyte counts were lower than those reported in wild bats which, in turn, may be more prone to infections than captive ones, leading to leukocytosis. Acute stress with epinephrine release may also determine an increase in lymphocytes. In this study, only three out of 21 bats had lymphocytosis, and all of them were juvenile bats which are less used to human contact than adults, confirming that leukocytosis was more related to stress than to a pathological condition.
The glucose concentration varied considerably. The range obtained in this study was wider than that previously reported in bats.10,11,21,30 This high variability was probably related to different factors inducing both an increased or decreased glucose concentration: glucose in Egyptians fruit bats is higher when measured at the end than at the beginning of the nightly feeding activity.11,21 In our study, the animals waiting for surgery were transported and kept in a cage with free access to food, with a short time period between food intake and sampling, possibly influencing glucose levels. In addition, the anaesthetic drugs may have an impact on the glucose level. For example, α‐adrenergic receptor agonists increase blood glucose in animals, due to the inhibition of insulin secretion. 31 In the present study, however, dexmedetomidine was used at the same dosage in all the animals. Hyperglycaemia could also be induced by the stress associated with the manual restraint used just before the anaesthesiological procedures. Triglycerides, in turn, appeared lower than in other mammals and bats.10,11,20
CK activity was variable and notably higher than in domestic mammals and in some
In line with previous findings, 11 we did not find a significant correlation between body mass and urea concentration, although the values recorded were slightly higher than those observed in another study on flying foxes. 10 Energy shortage has been suggested as a possible cause of the increased BUN concentration in Egyptian fruit bats. 11 The negative correlation found with glucose concentration, may support this hypothesis.
The ALP activity was higher in sexually immature bats compared with adults (Figure 2). This may be biologically significant, since in other mammals the bone ALP isoenzyme is known to increase the total plasmatic ALP activity of growing animals, 23 as also found in other Pteropodidae.8,10
Further haematological and biochemical differences have been found according to the sex and age categories in
To our knowledge, this is the first investigation into serum protein electrophoresis (SPE) in bats. SPE showed a clear subdivision into five fractions (albumin, α-, β1-, β2- and γ-globulins), as commonly observed in several mammals, with a higher albumin content than in other mammals. 34 Usually, high albumin is associated with dehydration, but in our study animals had free access to water. This high albumin may thus be specific to this species as also observed in similar bats.10,30 At the visual inspection of the electrophoretograms (Figure 3), no abnormal peaks in the other fractions were observed, as with other healthy mammals.34,35 There was no distinct separation between α1- and α2-globulins, thus suggesting that α-globulins should not be further fractionated. Again, this could be a peculiarity of this species.
In conclusion, although the results of this study were from a limited number of animals, ASVCP guidelines were followed to generate the RIs,
17
using suitably robust statistical tests for small sample size. The results could be beneficial as a reference biological dataset to monitor the health status of captive
Supplemental Material
sj-pdf-1-lan-10.1177_0023677220948542 - Supplemental material for Haematological, serum biochemical and electrophoretic data on healthy captive Egyptian fruit bats (Rousettus aegyptiacus )
Supplemental material, sj-pdf-1-lan-10.1177_0023677220948542 for Haematological, serum biochemical and electrophoretic data on healthy captive Egyptian fruit bats (
Footnotes
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
