Abstract
A cohort of captive-bred red-eared slider turtles, Trachemys scripta, was received from a commercial vendor. Shortly after arrival, several turtles presented as lethargic with subjectively pale skin and multifocal areas of cotton-like tufts in the mouth area and distal extremities. The water was treated with a commercial anti-fungal and anti-bacterial preparation of Victoria Green B and acriflavine. Despite treatment, 10 turtles were euthanized and others demonstrated persistent clinical signs. A live turtle was submitted to a commercial diagnostic laboratory for microbiologic and histologic evaluation. Seven cultures were obtained from this turtle and numerous organisms grew from each culture, including Flavobacterium sp. Blood film analysis demonstrated intracytoplasmic gamonts of Haemogregarina sp. within erythrocytes. On necropsy, internal organs appeared to be slightly more adhered within the coelomic cavity than normal. The urinary bladder was markedly distended with turbid, dark yellow urine. Microscopic evaluation of the tissues revealed significant parasitism with Myxidium sp., Spirorchis sp. and Neopolystoma orbiculare. No fungal organisms were identified on histology or grown in culture. While there are scattered reports of these pathogens in freshwater turtles, none of the cases reported describe such extensive co-infections. It is likely that complicated infection and shipping stress exacerbated clinical signs typically seen with these organisms. Efforts to minimize stress and administration of prophylactic antiparasitic agents during the acclimation period may aid in reducing the consequences of internal parasitism in aquatic turtles.
Introduction
Red-eared slider turtles (Trachemys scripta elegans) (RES) are commonly used for physiologic and infectious disease research. Commercial sources of these animals are limited to the pet trade and there are no purpose-bred suppliers for research institutions. Because of this, animals are essentially of unknown pathogen status as these sources do not conform to the high health standards of most rodent vendors. 1 Here we describe a case wherein a captive-bred shipment of RES presented nine days after arrival to our facility with general malaise and multifocal areas of white, cotton-like patches on the skin. The tank system was subsequently treated with a commercial antifungal and antiseptic agent. Still, 10 turtles were humanely euthanized with an overdose of an injectable commercial euthanasia solution (Euthasol, Virbac AH, Inc., Fort Worth, TX, USA) due to persistent clinical signs. This represented a cohort mortality of 20%. Shipping stress combined with high parasitic loads further complicated by opportunistic mixed bacterial infection of superficial skin injuries incurred during shipping likely resulted in the high morbidity and mortality seen in this cohort of RES.
Case presentation
A shipment of 50 captive-bred RES originating from Florida was ordered from a commercial vendor under an IACUC approved protocol. The animals were shipped from the vendor in ventilated plastic containers outfitted with moistened paper towels and contained up to four turtles per container. These containers were secured within a ventilated Styrofoam-insulated shipping box and padded internally with shredded newspaper. Animals were shipped via overnight courier in the spring of 2017. At our institution, hair cells harvested from the cochlea of RES are used to study signal transduction, because the relatively large neurons and synaptic clefts are amenable to single cell, electrophysiological recordings.
Upon arrival at the AAALAC-accredited facility, all turtles were in overall good clinical condition with several turtles demonstrating minor abrasions to the skin, likely secondary to trauma. These turtles did not require veterinary medical intervention. The turtles were housed in a 300-gallon enclosure filled with 150 gallons of municipal water. Water delivered to the turtle tank passed through a mixing valve to adjust the temperature of the water to 25–27℃ (78–82°F). After passing through the mixing valve, the water was filtered by two 25 µm polypropylene filter cartridges (Pentek PD-25-934, Pentair, Korea) and two 10 µm polypropylene filter cartridges (Pentek PD-10-934, Pentair, Korea) to remove sediments. Turtles were provided with an appropriate basking surface that is illuminated with UV-B emitting light bulbs. Turtles were fed a commercial aquatic turtle diet (Aquatic Turtle Diet, Mazuri, Richmond, IN, USA) three times per week according to manufacturer directions.
Per institutional husbandry standard operating procedures, there was a complete change of water three times a week. After the water was changed, sulfathiazole (SulfaPlex, Seachem Laboratories, Madison, GA, USA) was added to the aquatic system per manufacturer’s recommendations. Nine days after arrival, several turtles presented as lethargic with pale skin. Upon examination, these turtles had a dry, flaky carapace with multifocal areas of white, cotton-like patches on the head, neck, legs and distal extremities along with skin ulcerations on the hind limbs (Figure 1).
Presenting clinical signs: (a) dry carapace with focal areas of exfoliation; (b) tuft of pale-tan to white area on the orofacial region; (c) epithelial ulcerations on the distal hind limb.
A fungal infection was initially suspected, and the system was treated with a commercial antifungal and antiseptic agent (API Fungus Cure, Mars Fishcare North American, Chalfont, PA, USA) containing Victoria green B and acriflavine per manufacturer instructions. Water exchange was increased to daily flushing. Despite treatment, the condition of the turtles steadily worsened. Five days after the original presentation, 10 turtles had been euthanized and additional turtles presented with white tufts on the skin and a dry, flaky shell. Thus, a decision was made to submit a live, clinically-affected turtle to a commercial diagnostic laboratory for microbiologic sampling and histopathological evaluation. This turtle demonstrated small skin erosions on all four limbs, the tail and the head, along with epithelial ulceration on the distal aspect of one limb. The turtle, while affected, was deemed healthy enough for shipment.
The turtle was alert and in good condition when it was received by the diagnostic laboratory. Prior to necropsy, samples were collected for bacterial culture from the blood, faeces, coelom, urine, toe lesions and tail lesions. The turtle was then euthanized via overdose of injectable barbiturate. At necropsy, the coelomic cavity was aseptically opened and swabs were collected for microbial culture. Blood was removed from the heart with a sterile syringe for blood culture and a blood film was prepared and stained for review.
All organs were examined and samples were collected and fixed in neutral-buffered 10% formalin. Tissues of the head and limbs were decalcified. Representative samples of tissues were embedded in paraffin, sectioned at 5 µm and stained with haematoxylin and eosin. Additional sections of the skin were stained with PAS and Ziehl-Neelsen stains, and sections of kidney with Giemsa stain.
Gross necropsy findings
The necropsy began by splitting the carapace from the plastron to expose the coelomic cavity. The internal organs appeared to be slightly adhered to the plastron. The urinary bladder was greatly enlarged with a paper-thin wall and distended with turbid dark yellow urine. No urinary outflow obstruction was found as urine flowed freely from the vent. Scant gastrointestinal contents were found, and fat bodies were very small.
Histopathologic findings
Microscopic evaluation of the tissues from the turtle revealed significant parasitism. Myxidium sp. were seen in lumens of renal tubules resulting in tubulointerstitial nephritis associated with intralesional myxozoa and tubular lumens were filled with inflammatory and necrotic debris (Figure 2(a) and (b)). Organisms consistent with Myxidium sp. were also identified free floating in the urine. Ova consistent with Spirorchis sp. were observed in the oesophagus, heart, lungs, liver, kidneys, intestines, stomach, spleen, skeletal muscle, dermis and brain (Figure 2(c)).
Representative histopathologic sections demonstrating typical parasite distribution and subsequent lesions. (a) Myxidium sp., white arrow, kidney, haematoxylin and eosin stain (H&E), 600× magnification. Tubulointerstitial nephritis is associated with purple-stained intralesional organisms. (b) Myxidium sp., white arrow. Kidney, Giemsa, 600× magnification. Organisms are highlighted purple with distinct dark blue polar capsule within the tubular lumens. (c) Spirorchis sp., stomach, H&E, 100×magnification. Ova are present in the mucosal lamina propria with associated granulomatous inflammation. (d) Neopolystoma orbiculare, urinary bladder, H&E, 100× magnification. Trematode ova are interspersed throughout the bladder wall with secondary inflammation and expanded submucosa.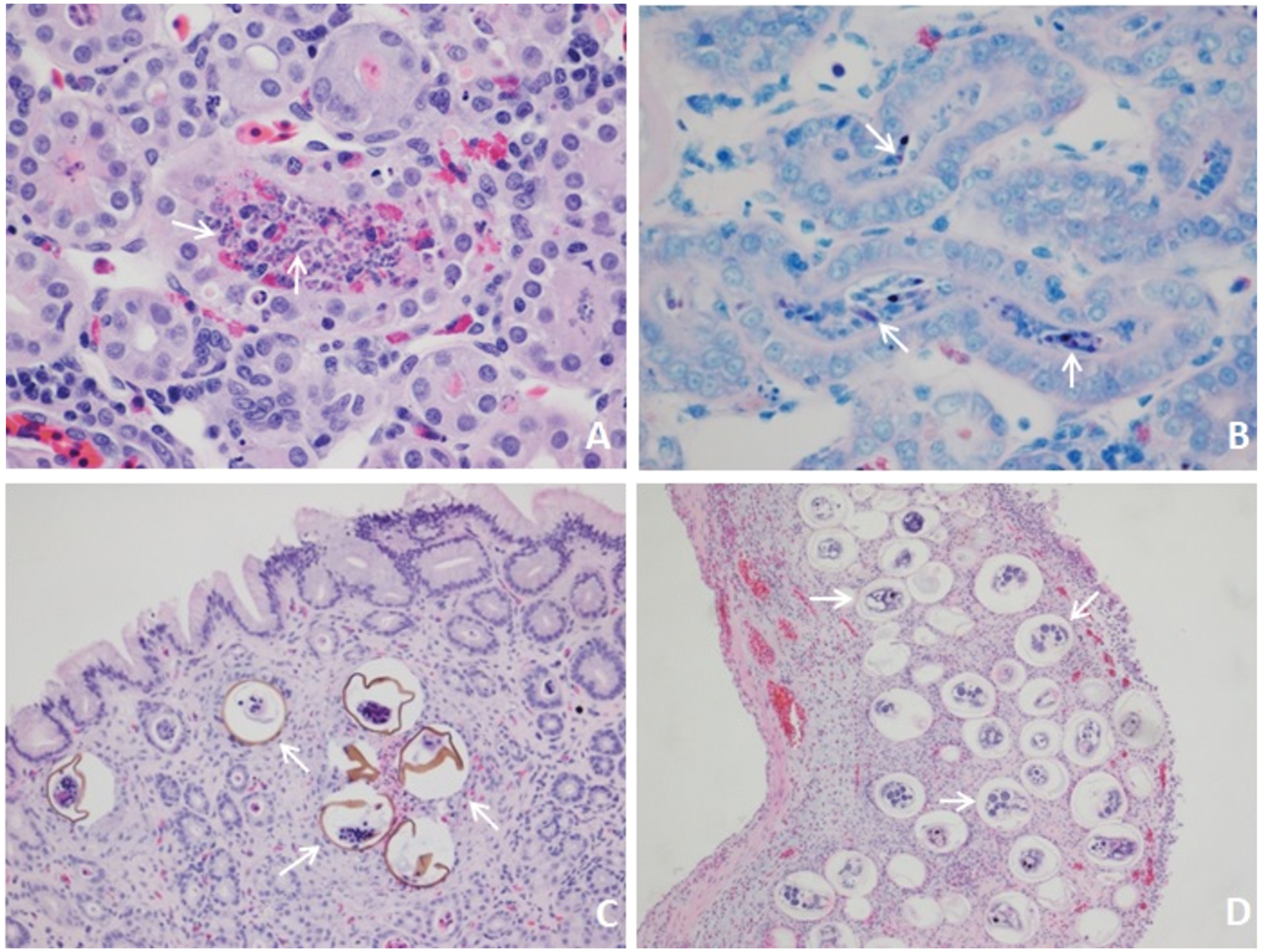
The urinary bladder submucosa was markedly expanded with a myriad of trematode ova and attendant inflammation (Figure 2(d)). The causative agent was identified as Neopolystoma orbiculare. The urothelium demonstrated multifocal erosions and sloughing. The lumen of the bladder contained cellular debris, bacteria and organisms distinct from the Myxidium sp. present in the urine and the spirorchid species observed in the stomach and other tissues.
There was no histologic evidence of underlying visceral/systemic bacterial infection or other infectious disease. Thin, variable Gram-positive filamentous bacteria had colonized the superficial serocellular crust covering the damaged epidermis; however, no fungal organisms or acid-fast bacteria were found with special staining on histologic sections from the skin.
Microbial findings
The blood film revealed slightly enlarged erythrocytes with intracytoplasmic slightly curved to straight cylindrical gamonts consistent with the genus Haemogregarina. All bacterial culture samples resulted in positive growth for a variety of microbial isolates (Figure 3).
Microbial culture results. A positive result is indicated by the symbol +.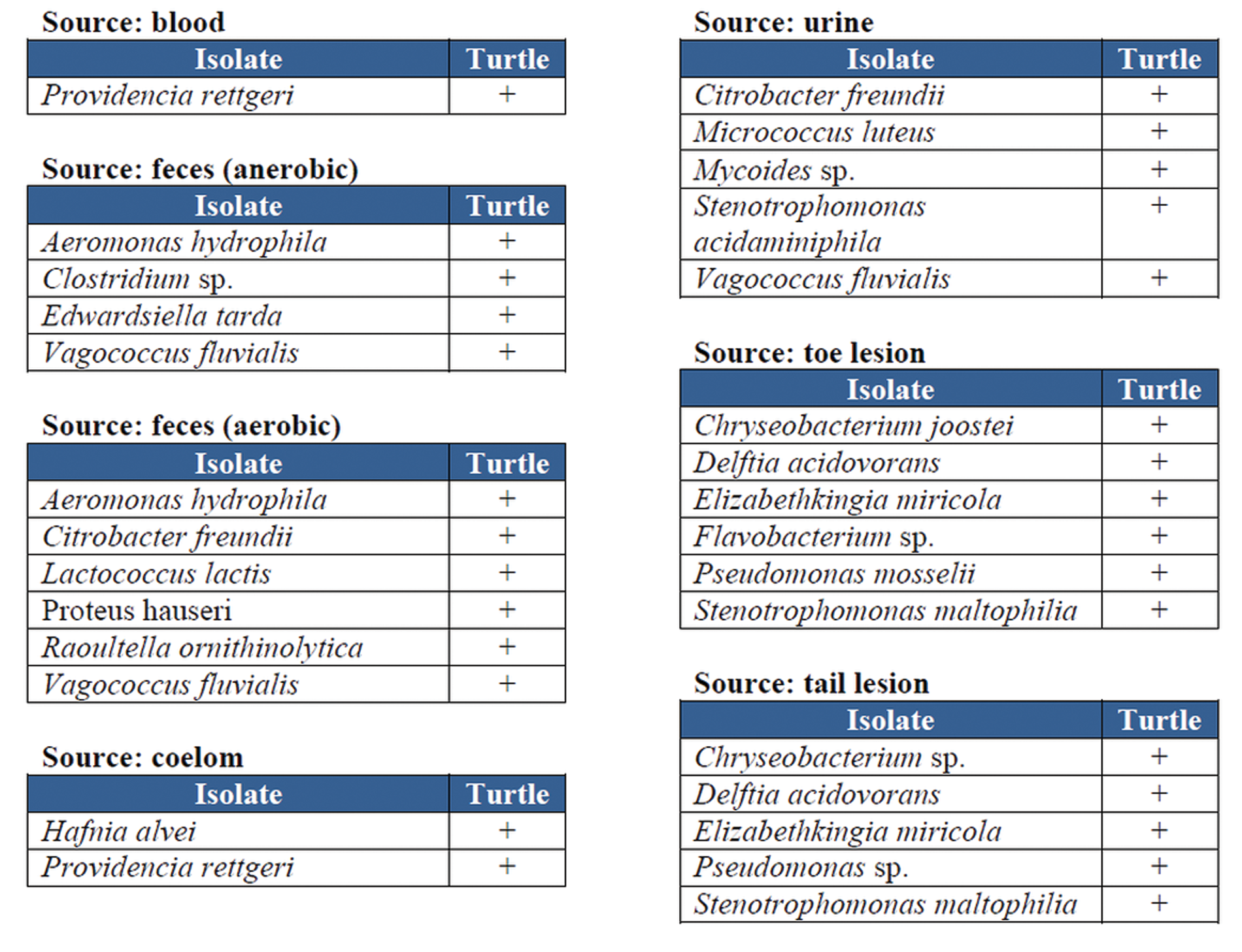
Discussion
This case report, describing an increase in morbidity and mortality in a colony of RES associated with Myxidium sp., Spirorchis sp. and Neopolystoma orbiculare, and infection with opportunistic bacteria, illustrates some of the challenges associated with managing aquatic turtles in the laboratory setting. Mainly, there are no commercial sources of purpose bred RES for research. Other sources include wild-caught and captive-bred animals intended for the pet trade; both of which supply animals of unknown pathogen status. Quarantine and animal conditioning programmes for aquatic turtles can present unique and difficult obstacles, especially when compared to similar procurement procedures for more traditional laboratory animal species of unknown or dissimilar health status.
One difficulty is elimination of potential pathogens. Antiparasitic treatment with ivermectin in chelonians is contraindicated as this agent can cross the blood–brain barrier resulting in neurologic disease and death. Alternatively, fenbendazole has a wide margin of safety and is often well tolerated in these species. 2 Institutional standard operating procedures for this facility dictate that newly arrived RES be prophylactically treated with sulfathiazine added the water in order to eliminate a wide range of bacteria. This treatment would not have been effective against the three parasites identified in this case series.
Myxosporeans are common parasites of fish and occur less frequently in reptiles. 3 Thus far, all myxozoan infections reported in reptiles have been by a member of the genus Myxidium and all of these reports have occurred in aquatic turtles. 4 Infection with Myxidium can contribute to disease and comorbidity; however, infection with this parasite not reported to be a primary pathogen, and infection with this organism is not known to be a primary cause of death.3,4
Spirorchis parvus is common in North American freshwater turtles 5 and primary disease and mortality has been associated with spirorchid infection in several species of chelonians. The migration of eggs from mesenteric arterioles through the walls of the stomach and intestine causes a disruption of epithelial barriers and likely permits infiltration of bacteria and secondary infections, as suspected seen in a case series describing thee spontaneous deaths of freshwater turtles diagnosed with spirorchid species. 7
Neopolystoma orbiculare is a common parasite of RES with a reported prevalence of over 40% in animals commercially supplied for the pet trade. 8 Neopolystoma spp. usually resides in the urinary bladder, cloaca and conjunctival sac of freshwater turtles. 9 In our case, a large burden of Neopolystoma orbiculare was identified in the urinary bladder. This parasite did not induce macroscopic changes in the urinary bladder, but microscopic evaluation did reveal a markedly expanded submucosa, inflammation and sloughing of the urothelium.
Flavobacterium sp. was cultured from the turtle submitted for testing. This bacterium often acts as an opportunistic pathogen invading tissues previously damaged by trauma. Histological examination of cutaneous tissues demonstrated lesions consistent with mechanical trauma. Skin damage likely occurred during shipping. Similar presentations and clinical timelines of secondary bacterial infections, specifically with Flavobacterium sp., have been reported in captive turtles resultant from traumatic ulcerative dermatitis. 10
Blood parasites consistent with the genus Haemogregarina were observed in blood film analysis of erythrocytes. These parasites are found ubiquitously throughout North America and appear to have little clinical impact on aquatic turtles. 11 Wild-caught turtles have a high rate of infection with Haemogregarine parasites resulting in little obvious untoward effect on overall health. Thus, Haemogregarine blood parasitism in this case report was not considered to a primary differential for clinical illness and infection did not result in any significant pathological effects.
As there are no sources of specific pathogen-free RES, working with these animals presents a management challenge. It is important to remain vigilant about colony health status, even when animals are procured from commercial sources regardless of the animals’ wild-caught or captive-bred origination. Appropriate quarantine programmes and health surveillance for aquatic turtles of unknown health status should be instituted, as is commonplace for rodent procurement, prior to release into the animal facility’s general animal population. Increased water turnover rates and conditioning with prophylactic antimicrobial and antiparasitic treatment upon arrival to the facility may decrease pathogen spread; however, treatment with some common antiparasitic agents, such as ivermectin, are contraindicated in chelonians. Other medical approaches to diminish pathogen burden such as Victoria green B (malachite green) as a topical fungicide and acriflavine as a topical antiseptic agent may be of limited use as treatments are variably effective. Fortunately, many aquatic parasitic organisms require intermediate hosts to complete their lifecycles; thus, in the laboratory setting, the potential for propagation of these organisms is essentially nonexistent.
It may be beneficial for individual animal health and cohort vigour to stagger more orders of fewer animals rather than have one large shipment group. Reduced shipping density may lessen shipment-related stress and subsequent immune function alteration, can decrease the possibility of host–parasite encounters and allows for quarantine of smaller groups. These smaller groups can have a more focused interventional approach in the face of a morbidity or mortality event thereby mitigating attrition. Smaller shipment cohorts may also reduce the incidence of mild skin trauma occurring from other individuals in the shipment cohort. This could lessen potential susceptibility to opportunistic microbial infection resultant from these abrasions and decrease pathogen transmission.
It is recommended that a broad-spectrum antimicrobial water additive along with increased water turnover rates be instituted for all newly procured aquatic turtles of unknown pathogen status. Although often nonpathogenic, internal parasitism in RES may act as a confounding factor for certain experimental models. Treatment with fenbendazole or praziquantel can be considered based on facility standards and the potential confounding effects on research the parasitism and treatment may have. In this case presentation, it is ultimately suspected that in light of routine institutional quarantine protocol, shipping stress combined with distinct co-parasitism along with diverse secondary bacterial infection, including Flavobacterium sp., overwhelmed the RES colony, which resulted in increased morbidity and mortality.
Footnotes
Acknowledgement
Thank you to Dr Susan Caraker at IDEXX BioResearch (Colombia, MO) and to Dr Jaime Landolfi at University of Illinois College of Veterinary Medicine for providing necropsy and histopathologic reports, and to JL for photomicrographs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
