Abstract
Few reports evaluate the clinical effects of opioids in sheep during experimental surgical procedures. Catecholamine-mediated haemodynamic changes resulting from surgical noxious stimulation are blunted by opioids. The aim of this study was to evaluate the efficacy of three opioid-based analgesic protocols in avoiding a 20% increase in heart rate (HR) and/or mean arterial blood pressure (MAP) during experimental intervertebral disk nucleotomy in sheep. Eighteen female Brogna sheep were anaesthetized with propofol and maintained with a fixed end-tidal isoflurane concentration of 1.5 ± 0.1%. Sheep were assigned to one of three groups that intravenously received methadone 0.3 mg/kg (group M), fentanyl 2 µg/kg followed by 10 µg/kg/h (group F), or buprenorphine 10 µg/kg and 30 minutes later ketamine 1 mg/kg followed by 5 mg/kg/h (group BK). Intravenous fentanyl at 2 µg/kg would have been used for rescue analgesia in case HR and/or MAP had increased. During surgery, HR and MAP values did not increase over 20% in all groups. All animals maintained the percentage change between -4 and 7% for both variables; only one sheep in group BK had an increase in MAP superior to 20% after ketamine administration before surgical stimulation. In group M, HR decreased over time and in group BK, MAP tended to increase during surgery. All the opioid-based protocols tested were able to control the cardiovascular response to noxious stimulation in sheep undergoing spinal surgery, although ketamine may have represented a confounding factor.
Introduction
Sheep are commonly used as in vivo model for experimental orthopaedic procedures involving spinal or long-bone surgeries.1–8 Analgesic protocols that include opioids are now more common during this type of surgery and among those compounds, buprenorphine is the most widely used. 1 Buprenorphine at 6 µg/kg administered intravenously (IV) was seen to increase the thermal threshold in sheep, although the same dose caused no change in response to mechanical stimuli.9,10 More recently, there has been renewed interest in the systemic use of opioids other than buprenorphine, such as methadone and fentanyl, in sheep.5,6,7,11–14 One study observed that intramuscular (IM) buprenorphine 10 µg/kg had a lower efficacy than fentanyl patch at 2 µg/kg/h in relieving postoperative pain after unilateral tibial osteotomy in sheep. 6
Fentanyl 10 µg/kg/h was shown to decrease end-tidal isoflurane requirement by 22.6%, compared to the control group, in sheep undergoing orthopaedic surgery, and recovery quality was scored as good to excellent in most of the animals. 5 Methadone 0.5 mg/kg injected IV was associated with dexmedetomidine or xylazine in sheep, and its effects on sedation and some cardiovascular variables were similar to those observed after the administration of the alpha-2 agonist alone.11,12 Another study used methadone as a premedication agent in combination with midazolam, before right heart bypass in sheep. 15 Nevertheless, none of those studies evaluated the analgesic effects of methadone.
Roizen and colleagues observed in human patients that skin incision caused a release of noradrenaline, and this may be reduced dose-dependently by volatile anaesthetic or by the concurrent administration of morphine. 16 A lower increase in arterial blood pressure due to skin incision was obtained at higher alveolar concentration of the anaesthetic or with incremental doses of morphine. Sympathetic-induced changes in heart rate (HR) and mean arterial blood pressure (MAP), proportional to the surgical stimulation, have been demonstrated in human patients undergoing abdominal surgical procedures. 17 Also in isoflurane-anaesthetized sheep undergoing experimental tibial surgery, clinically significant increases in HR and MAP were observed during intense surgical stimuli. 3 In that study, an increase in HR or MAP between 13 and 30% was associated with an electroencephalographic pattern compatible with arousal or paradoxical arousal during noxious stimulation.
To our knowledge, no study has so far assessed the ability of different opioid-based protocols to minimize cardiovascular changes due to surgical stimulation in sheep. The aim of this study was to evaluate the efficacy of three different opioid-based analgesic protocols in preventing increases in HR and/or invasive MAP in isoflurane-anaesthetized sheep undergoing experimental spinal surgery. We hypothesized that all the treatments evaluated might prevent a 20% increase in HR and/or invasive MAP, considered as indirect clinical signs of sympathetic activation induced by surgical stimulation.
Animals
Eighteen Brogna, non-pregnant female sheep were enrolled in a prospective controlled study and underwent spinal surgery consisting in a transpedicular intervertebral disk nucleotomy from L1 to L4–L5 as part of another experimental study. Sheep weighed 47 ± 8 kg and were 3 years old.
The study was performed with the approval of the Animal-welfare Body of the University of Padova (OPBA Authorization 7/2014) and the Italian Ministry of Health, according to European (Directive 2010/63/EU) and Italian regulations (Legislative Decree 26/2014).
Subcutaneous (SQ) ivermectin (Ivomec ovine, Merial Italia Spa, Milan, Italy) 200 µg/kg was administered as an endoparasite treatment at least 2 weeks before the procedures.
Sheep were housed in pens containing six animals each, and fed with a commercial pellet (Compli Sheep, Tecnozoo, Piombino Dese, Padua, Italy) and hay diet. Prior to the experimental procedure, clinical examination, complete blood count and serum chemistry were performed, on the basis of which sheep were considered healthy.
Sheep were evenly distributed between three groups on the basis of the IV analgesic protocol used: group M, 0.3 mg/kg of methadone (Comfortan, Dechra, Turin, Italy); group F, fentanyl (Fentadon, Dechra, Turin, Italy) 2 µg/kg followed by 10 µg/kg/h; and group BK, ketamine (Ketavet 100, Intervet Productions Srl, Aprilia, Italy) 1 mg/kg followed by 5 mg/kg/h and buprenorphine (Buprenodale, Dechra, Turin, Italy) 0.01 mg/kg. Sheep were weighed the morning of the day scheduled for the procedure.
Materials and methods
Food, but not water, was withheld for 12 hours before the experiment. Medetomidine (Sedator; Dechra, Turin, Italy) 8 µg/kg was administered IV in the jugular vein. Ten minutes later, sheep were moved to the presurgery area, where the marginal auricular vein and the auricular artery were cannulated with a 20 gauge over-the-needle IV catheter. After 30 minutes, anaesthesia was induced IV with propofol (Propofol Kabi; Fresenius Kabi Italia s.r.l., Italy) 4 mg/kg. Airways were secured with endotracheal tubes, and the animals were connected to a circle breathing system and mechanically ventilated in a pressure control mode (Datex-Ohmeda 7900 SmartVent; GE Healthcare, Finland), at a peak pressure of 15 cmH2O and a respiratory rate adjusted to maintain normocapnia between 37 and 45 mmHg. Fresh gas flow was set to 4 L/minute and after 10 minutes decreased to 1 L/minute, with a fraction of inspired oxygen of 0.5. Sheep were positioned in sternal recumbency on a heating pad, and a gastric tube was inserted through their mouth in order to minimize rumen distension. An isoflurane target end-tidal concentration (FE’Iso; IsoFlo; Zoetis, Rome, Italy) of 1.5 ± 0.1% was set and maintained throughout the procedure. A multiparameter monitor (Datex S/5; GE Healthcare, Helsinki, Finland) was used to record HR, invasive arterial blood pressure, respiratory rate, haemoglobin oxygen saturation and end-tidal gas analysis.
After 15 minutes of stable target FE’Iso, sheep received one of the three treatments. All drugs were injected IV over 2 minutes, at least 10 minutes before skin incision, while buprenorphine was injected at least 30 minutes before incision. Ketamine was infused for no longer than 2 hours; Ringer’s lactate solution at 5 ml/kg/h was infused during the entire procedure. Three surgical periods were identified: from skin incision to complete spine exposure, which was defined as Tspine; the nucleotomy period, called Tnucleo; and the period from the beginning of tissue reconstruction to the end of the skin suture, defined Tsuture.
During surgery, an increase in HR and/or invasive MAP of more than 20%, compared to the value measured 5 minutes earlier and lasting more than 30 seconds, was considered indicative of inadequate intraoperative analgesia.2, 13 In such a case, in order to provide rescue analgesia, the study design established that fentanyl at 2 µg/kg had to be administered IV and that the same dose had to be repeated at 5 minute intervals, until the parameters would have returned to previous values. At the end of the procedure, after the return of spontaneous ventilation, the sheep were disconnected from the breathing system and positioned in sternal recumbency with the head elevated. Once the animals were able to support their head and vigorous swallows were observed, the endotracheal tube was removed, and the animals were returned to their pen and monitored for behavioural signs of pain or abnormal posture. Amoxicilline-clavulanate (Synulox; Zoetis, Rome, Italy; 25 mg/kg, twice daily, SQ) was administered on the day of the surgery and repeated for the first 7 days after surgery. Carprofen (Rimadyl; Zoetis, Rome, Italy; 3 mg/kg once a day, SQ) and tramadol (Altadol; Formevet Spa, Milan, Italy; 4 mg/kg, twice daily, IM) were administered at the end of surgery and continued for the following 3 days, or longer if needed.
Statistical analysis
Percentage change in HR and MAP was expressed as median (min-max) and calculated every 5 minutes as: 100 × (Xt − Xt-5)/Xt-5. Normally distributed data were expressed as mean ± SD, otherwise as median (min-max). The anaesthesia time points considered for the analysis were: Tpre, which includes values recorded for 15 minutes of stabilization after having reached target FE’Iso; Tadmin, which includes values recorded 5 minutes after starting drug administration; Tskin, that includes measures obtained 5 minutes after skin incision; Tspine; Tnucleo; and Tsuture. Variations of HR and MAP over time were evaluated within each group with one-way analysis of variance. A Tukey’s multiple comparisons test was used to detect statistical difference between Tpre and the other time points considered.
Based on the results of a similar study, 2 sample size was calculated to obtain a mean difference of 20% ± 10% compared to baseline, with a power of 0.8 and α of 0.05. Data were analysed using GraphPad Prism 6.0 (GraphPad software, CA, US) and P < 0.05 was considered statistically significant.
Results
All sheep completed the study without complications during surgery or the postoperative period; the procedure lasted between 125 and 270 minutes. Rescue analgesia with fentanyl was never required and the target concentration of volatile anaesthetic was achieved in all the animals. Figure 1 shows the percentage change of HR and MAP in each surgical time point considered, for every single group. At skin incision, HRs in group M decreased more than 20% in two animals, while in group F and group BK, they minimally changed after opioid administration (Figure 1). Six animals in group M and group F, and five sheep in group BK, remained below the target limit for MAP (20% increase) at all time points (Figure 1). One animal in group BK had an increase of 46% in MAP immediately after ketamine loading dose administration, although the changes observed during the rest of the procedure remained below 20% (Figure 1).
Percentage change in heart rate and invasive mean arterial blood pressure at different procedure time points in isoflurane-anaesthetized sheep undergoing transpedicular intervertebral disk nucleotomy. Animals intravenously received methadone 0.3 mg/kg (group M), fentanyl 2 µg/kg followed by 10 µg/kg/h (group F) or ketamine 1 mg/kg followed by 5 mg/kg/h co-administered with buprenorphine 0.01 mg/kg (group BK) for intraoperative analgesia. Data are reported as medians, interquartile ranges, and minimum and maximum values. Anaesthesia time points reported on the x-axis are: Tpre, measurements recorded for 15 minutes of stabilization after having reached target FE’Iso; Tadmin, values recorded 5 minutes after starting drug administration; Tskin, which includes measures obtained 5 minutes after skin incision; Tspine, from skin incision to complete spine exposure; Tnucleo, from spine exposure to the end of nucleotomy; and Tsuture, from tissue reconstruction to the end of the skin suture.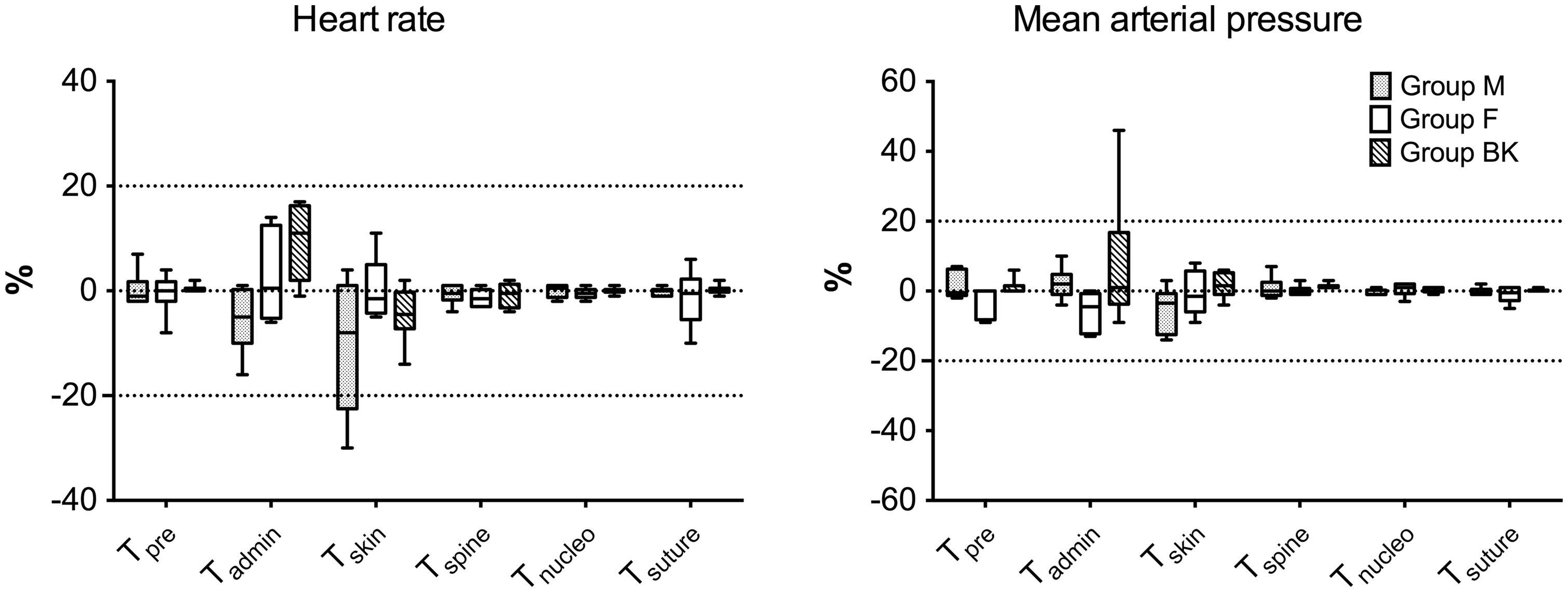
Moreover, in group M, HR decreased over time and differed statistically from Tpre at Tskin, Tspine and Tsuture (Figure 2(a)). In group F and group BK, cardiac frequency remained constant throughout the procedure: no statistical differences were observed between Tpre and other time points (Figure 2(b) and (c)).
Mean ± SD of intraoperative heart rate (on the left) and invasive mean arterial blood pressure (on the right), measured at different procedure time points in isoflurane-anaesthetized sheep undergoing transpedicular intervertebral disk nucleotomy that intravenously received methadone 0.3 mg/kg ((a) and (d)), fentanyl 2 µg/kg followed by 10 µg/kg/h ((b) and (e)) or ketamine 1 mg/kg followed by 5 mg/kg/h co-administered with buprenorphine 0.01 mg/kg ((c) and (f)) as intraoperative analgesia. Anaesthesia time points reported on the x-axis are: Tpre, measurements recorded for 15 minutes of stabilization after having reached target FE’Iso; Tadmin, values recorded 5 minutes after starting drug administration; Tskin, that includes measures obtained 5 minutes after skin incision; Tspine, from skin incision to complete spine exposure; Tnucleo, from spine exposure to the end of nucleotomy; and Tsuture, from tissue reconstruction to the end of the skin suture. *P < 0.05 from Tpre.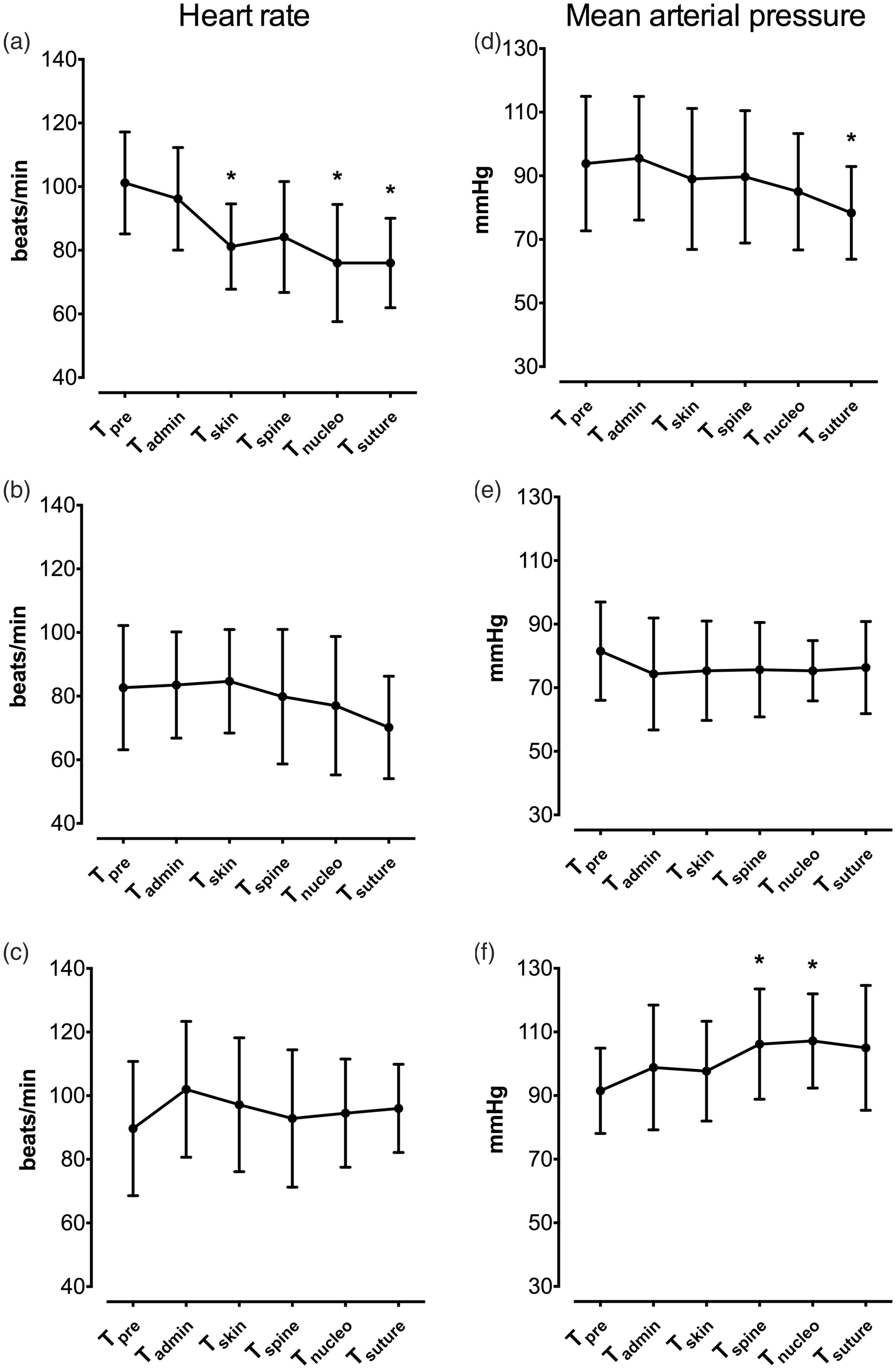
In group M, MAP tended to decrease throughout the anaesthesia and values measured at Tsuture were statistically different from those at Tpre (Figure 2(d)). Group F showed no statistical differences in MAP (Figure 2(e)). Blood pressure increased in group BK from Tpre over time, with statistically significant difference at Tspine and Tnucleo (Figure 2(f)).
Oesophageal temperature remained between 36.9 and 38.0℃ with no differences in mean values among groups (P = 0.397).
Recovery was smooth in all animals and no differences in extubation time (time between disconnection from the anaesthetic machine and removal of the endotracheal tube) were observed among groups, resulting in 9 ± 3, 11 ± 4 and 12 ± 5 minutes in groups M, F and BK, respectively (P = 0.396). All sheep were able to stand and walk within 35 minutes after extubation.
Discussion
Among indirect intraoperative indicators of nociception, changes over time in HR and arterial blood pressure are commonly used.2,3,16–20 A sudden increase of more than 20% in these parameters suggested a poor analgesic coverage during orthopaedic surgery in sheep. 3 The opioid-based protocols evaluated in our study maintained HR and/or MAP percentage change inferior to 20%.
The cardiovascular response to noxious stimulation after systemic methadone administration has not been studied in sheep, despite its use in painful experimental procedures in this species. 15 The doses reported in the literature for methadone range from 0.1 to 0.5 mg/kg for IV, intramuscular or epidural routes, and the impact of this opioid on HR seems to be negligible.11,12,14,15,21 Methadone at a dose of 0.3 mg/kg administered by epidural route provided more than 210 minutes of analgesia without signs of bradycardia. 21 In awake sheep, IV administration of 0.5 mg/kg methadone and acepromazine caused sedation that lasted 60 minutes without affecting HR. 14 The same dose of methadone, co-administered with xylazine or dexmedetomidine, caused bradycardia, which was not different from what observed with the alpha-2 agonist alone.11,12 In our study, methadone administered to sheep at 0.3 mg/kg was able to minimize the percentage increases in HR and MAP during surgery, thus suggesting an adequate control of the cardiovascular response secondary to surgical stimuli. Nevertheless, a decrease in HR was observed after methadone administration and this might be attributed to the vagally mediated effect, as reported in anaesthetized dogs receiving a full μ-opioid receptor agonist. 22
Fentanyl infusion has been used for intraoperative analgesia in sheep undergoing major surgery by either IV or transdermal administration routes. IV infusion rates reported in different studies vary from 2 to 10 µg/kg/h, usually preceded by a loading dose of 5 µg/kg,5,13 but no detailed information is available on the intraoperative cardiovascular response to noxious stimulation at these doses. In sheep receiving fentanyl at 10 µg/kg/h during orthopaedic surgery, a progressive decrease in isoflurane consumption from 1.5 to 0.9% was required to keep HR and MAP constant over the anaesthetic period. 5 In our study, although the end-tidal concentration of isoflurane and the fentanyl infusion rate were not modified, those parameters did not change, probably because the surgical noxious stimulation did not vary over time.
Two studies have clinically evaluated the effect of transdermal fentanyl patch at a drug-releasing rate of 2.0-2.6 µg/kg/h in sheep undergoing lumbar intervertebral spinal fusion or unilateral left tibial osteotomy, but the evaluation of the analgesic efficacy was limited to the postoperative period.6,7 Although both trials used isoflurane for the maintenance of anaesthesia, the mean end-tidal concentration delivered was reported only in one study. Ahern and co-workers reported a mean value of isoflurane end-tidal concentration of 2.2% at the end of the tibial osteotomy. 6 In our study, the partial pressure of isoflurane in the expired gases was maintained at 1.5 ± 0.1%, which represents the minimum alveolar concentration (MAC) for this species. The lower isoflurane consumption reported in our study, compared to that observed by Ahern and colleagues, might be explained by the different fentanyl infusion rate used, although differences in the intensity of noxious stimulation should be also considered. Moreover, transdermal absorption during anaesthesia may be influenced by several factors that could have affected fentanyl plasma concentration in Ahern’s study. 6
Mean extubation time was similar between our study and that observed by Funes and co-workers, despite different infusion times of 150 and 100 minutes, respectively. 5 Furthermore, as previously reported, recovery from anaesthesia was smooth without any signs of excitement or sedation recurrence.5,7 This may support the hypothesis that fentanyl in sheep does not accumulate significantly after an infusion lasting between 2 and 3 hours, as it was demonstrated to do in dogs after 4 hours of fentanyl infusion at 10 µg/kg/h. 23
Buprenorphine is considered a partial agonist of μ-opioid receptor and, when compared to the full agonist fentanyl, it is less adequate for the management of postoperative orthopaedic pain in sheep. 6 Analgesic activity of IV buprenorphine 0.01 mg/kg has been evaluated in sheep by mechanical and thermal threshold tests, and a dose-dependent decrease in response was measured only for the thermal threshold test.9,10 Because of the conflicting results on nociceptive threshold tests and the elevated concentration of isoflurane reported by Ahern and co-workers during orthopaedic surgery, 6 buprenorphine at 0.01 mg/kg was expected to provide poor analgesia for intervertebral disk nucleotomy. There is recognized evidence that subanaesthetic doses of ketamine provide analgesia, preventing hyperalgesia and blunting central sensitization, when they are used as adjuvant in analgesic protocols, both in humans and animals. 24 An infusion of ketamine at a subanaesthetic dose (10 µg/kg/minute) was thus co-administered in our study to improve nociception control. The same rate of ketamine infusion, associated with lidocaine at 20 µg/kg/minute, was used as adjuvant in isoflurane-anaesthetized sheep undergoing meniscal surgery. 4 In the same study, fentanyl at 2 µg/kg/h was provided by transcutaneous administration as intraoperative analgesia. Compared to the control group, which received only a transdermal fentanyl patch, isoflurane requirement was about 23% less when ketamine and lidocaine were used. In group BK, no animals showed a HR percentage increase superior to 20%, whereas only one sheep had an increase of more than 20% in MAP immediately after the administration of ketamine bolus. This increase can be considered an effect of ketamine injection since, at that time, no noxious stimulation had been applied. Moreover, before ketamine bolus, no signs of abnormal cardiovascular parameters had been noted. Similar to what we have observed in sheep, dogs under isoflurane anaesthesia receiving ketamine infusion at different plasma concentration showed an increase in cardiovascular parameters. 20 The cardiovascular response to ketamine might have been secondary to a central sympathomimetic effect that this drug seems to cause even at subanaesthtic doses in sheep and dogs, but not in humans.20-24
This study has some limitations. First, the concurrent administration of sedatives and anaesthetics may have affected the cardiovascular response secondary to the noxious surgical stimulation. Medetomidine may attenuate the sympathetic activation due to surgery, and its effect is influenced by the dose, and the time between administration and the application of the noxious stimulus. In sheep undergoing experimental tibial osteotomy, an intramuscular dose of 10 µg/kg caused a decrease in HR that was reversed by surgical stimulation 40 minutes after its injection. 8 In the same study, MAP was not influenced by medetomidine administration. Considering that, in our study, a low dose of medetomidine was used and the surgery began 80 minutes after its injection, an effect of the alpha-2 agonist on the cardiovascular system seems to be unlikely. Propofol and isoflurane may have contributed to suppress the response to noxious stimulation, although they should have influenced all the animals in the same way, as the doses used were similar. The lack of any evident sympathetically mediated cardiovascular response to surgery may also be due to a mild surgical stimulation, which could be less intense than expected. The use of a control group undergoing the same surgical procedure without analgesics could have certainly provided evidence of the noxious stimulation intensity and the effects of the three protocols, but such a condition would have been considered unethical; consequently, it was not contemplated. Indeed, this opportunistic study was part of a long-term orthopaedic experimental trial, and untreated intraoperative nociception is known to be detrimental with regard to animal welfare, and is potentially associated with postoperative distress and complications. In addition, in view of the 3 R principles (replacement, reduction and refinement), collecting both surgical and anaesthetic information from the same animal allows the reduction of the number of animals involved and refinement of the analgesic approach.
Another limitation is the use of cardiovascular variables as the sole sign of intraoperative nociception. In isoflurane-anaesthetized sheep undergoing tibial osteotomy, electroencephalographic analysis was evaluated beside HR and systemic arterial blood pressure as an indicator of intraoperative nociception. However, the results of that study showed that haemodynamic changes were more reliable and correlated with the intensity of intraoperative nociceptive stimulation better than the electroencephalographic pattern. 3 Moreover, to the authors’ knowledge, there are not objective methods for the acquisition of real-time information to assess surgical nociception in anaesthetized sheep.
Conclusion
All the opioid-based protocols evaluated were able to minimize the cardiovascular response to noxious surgical stimulation in isoflurane-anaesthetized sheep. This study indicates that, with the tested protocols, HR and MAP do not change by over 20% during the surgical stimulation. The cardiovascular activation caused by ketamine may represent a confounding factor, and this drug should be administered before the application of noxious stimuli if the sympathetic response to surgery has to be monitored. Further studies are warranted in order to evaluate the analgesic efficacy of these protocols in the early postoperative period.
Footnotes
Acknowledgements
The results of this investigation were presented at the Association of Veterinary Anaesthetists autumn meeting 2017, and are published as abstract in the proceedings of the conference. The authors would like to thank Dr Sara Bordin for proofreading assistance and constructive comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
