Abstract
It is important to develop a feasible animal model of regional anesthesia other than sciatic nerve blockade for pharmacokinetic investigations of new local anesthetics or analgesia in upper extremity surgery. Herein, we explored a nerve stimulator (NS)-guided brachial plexus block (BPB) in a rat model. The anatomy of the brachial plexus in rats was delineated in cadavers, and various BPBs were examined. The puncture point was located 0.5–1.0 cm below the lateral one-third of the clavicle. The efficacy and safety of the NS-guided BPB were evaluated using an injection of 2% lidocaine or 0.5% bupivacaine in 16 live animals; saline injection was used as a control. Both sides of the brachial plexus were located successfully using the NS-guided technique. Sensory blockade (nociception assessment) and motor blockade (grasping and straightening tests) appeared after application of the two classical local anesthetics, but not normal saline. The motor and sensory blockade induced by bupivacaine exhibited a longer duration than that induced by lidocaine (p < 0.05). All rats recovered uneventfully from general anesthesia and BPB. No abnormal results were found in pathological studies or behavioral observations. Thus, a rat model of NS-guided BPB was established, and BPB induced an overall reversible sensory and motor blockade in the thoracic limbs. Evaluation of the efficacy and safety demonstrated that this rat BPB model was feasible, reproducible, and safe.
Introduction
Regional anesthesia is widely used as a powerful anesthetic method in clinical surgery.1,2 Furthermore, there is growing research interest in investigating the mechanism, safety, duration, and distinct sensory and motor blockades of new local anesthetics and additives for inducing regional anesthesia. Thus, a feasible and safe animal model of regional anesthesia is essential for such studies.
Some regional anesthesia models have been developed in rats, cats, dogs and sheep.3–7 Sciatic nerve blockade models in rats are the most widely used animal models in experimental studies because of their simplicity and reproducibility. However, a sciatic nerve block only provides dorsal limb anesthetization of the hind limb, whereas it cannot provide complete anesthetization of the hind limb (i.e., the dorsal and ventral limb) without including a femoral or saphenous nerve block (ventral limb anesthetization of the hind limb). Brachial plexus block (BPB) is a commonly used peripheral nerve block for the upper extremities that provides complete anesthetization of the entire upper limb. The anatomical characteristics of the rat brachial plexus allow for the development of a rat BPB model. 8
Therefore, we developed a rat BPB model. We used rat cadavers to investigate the BPB and verified the effectiveness and safety of the BPB in living rats.
Materials and Methods
Cadaver Experiment
Animals
The Committee of Scientific Research and Institutional Animal Experimental Ethics Committee, West China Hospital, Sichuan University, Chengdu, China, approved all protocols (Ethics No.: 2015014A). Thirty fresh cadavers of adult Sprague-Dawley rats (weighing 200–300 g) that died or were euthanized for reasons unrelated to this study were allocated to this study.
Dissection of the brachial plexus
The brachial plexus (BP) morphology of 20 fresh cadavers of adult rats (euthanized with propofol) was investigated. Rats were positioned in a dorsal recumbent position with the blocked limb extended straight. The skin, fascia, and muscle were removed throughout the thoracic and forelimb areas. The BP on the left and right side of each cadaver was dissected from the spinal cord to the thoracic limb.
Results of BP dissection
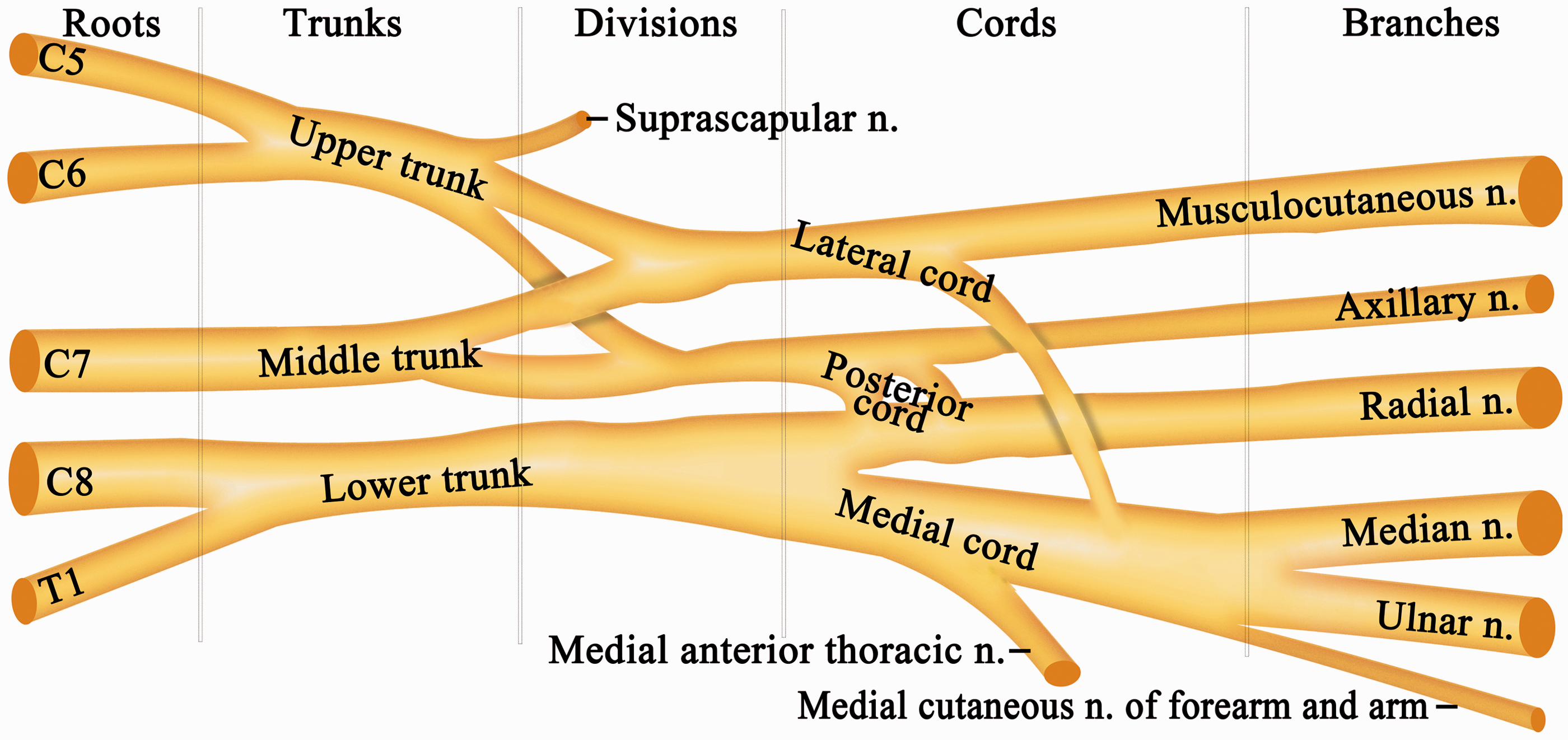
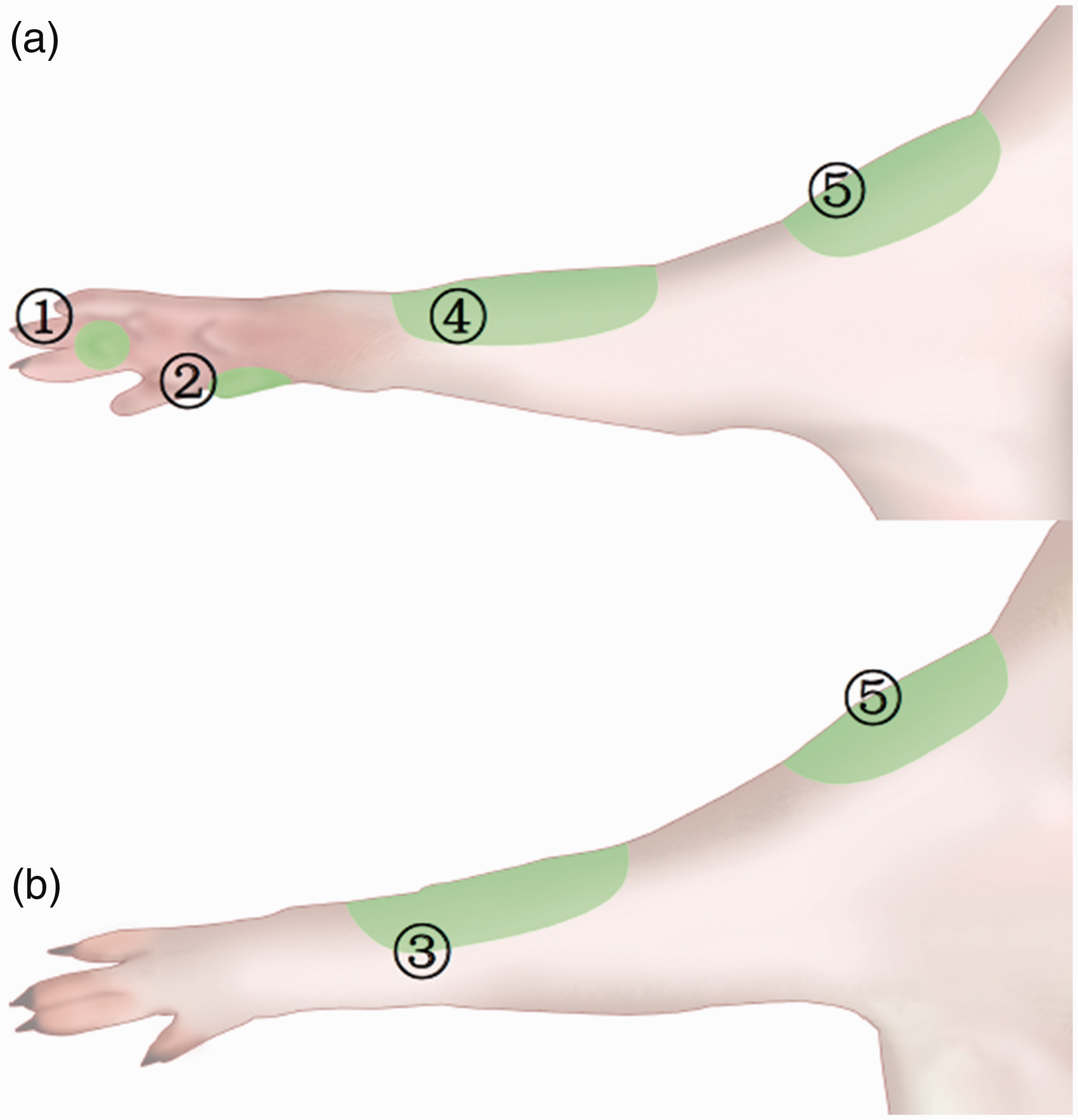
We identified the roots, trunks, divisions, cords, and terminal branches of the BP (Figures 1 and 2). The BP emerged at five different levels, C5, C6, C7, C8, and T1, similar to humans (Figure 1). The C5 and C6 branches merged to form the upper trunk, the C7 branch separately and continuously formed the middle trunk, and the C8 and T1 branches passed over the first rib and merged to form the lower trunk (Figure 2). The trunks divided into the anterior and posterior divisions. The cords continued and became the five major nerves of the upper extremity: the musculocutaneous nerve, the axillary nerve, the radial nerve, the median nerve, and the ulnar nerve. Figure 2 shows a schematic depiction of these nerves. The cutaneous branch of the five major sensory nerves innervated the area shown in Figure 3(a) and (b).
Anatomy of the position of the BP. ① lamina ventral, ② the first rib, ③ the cords of BP, ④ clavicle, ⑤ deltoids. BP: Brachial plexus; Cv: cervical vertebra. Schematic drawing of the roots, trunks, divisions, cords, and terminal branches of the BP in our study. n.: nerve. The skin area for evaluating mechanical nociceptors. (a) The ventral thoracic limb. (b) The dorsal thoracic limb. ① The median nerve, the skin overlying the palmaromedial aspect of digit 2. ② The ulnar nerve, the fourth digital pad. ③ The radial nerve, the skin area below the dorsal aspect of the antebrachiocarpal joint. ④ The musculocutaneous nerves, the skin area below the ventral aspect of the antebrachiocarpal joint. ⑤ The axial nerve, the dorsal and ventral aspects of the deltoid muscles.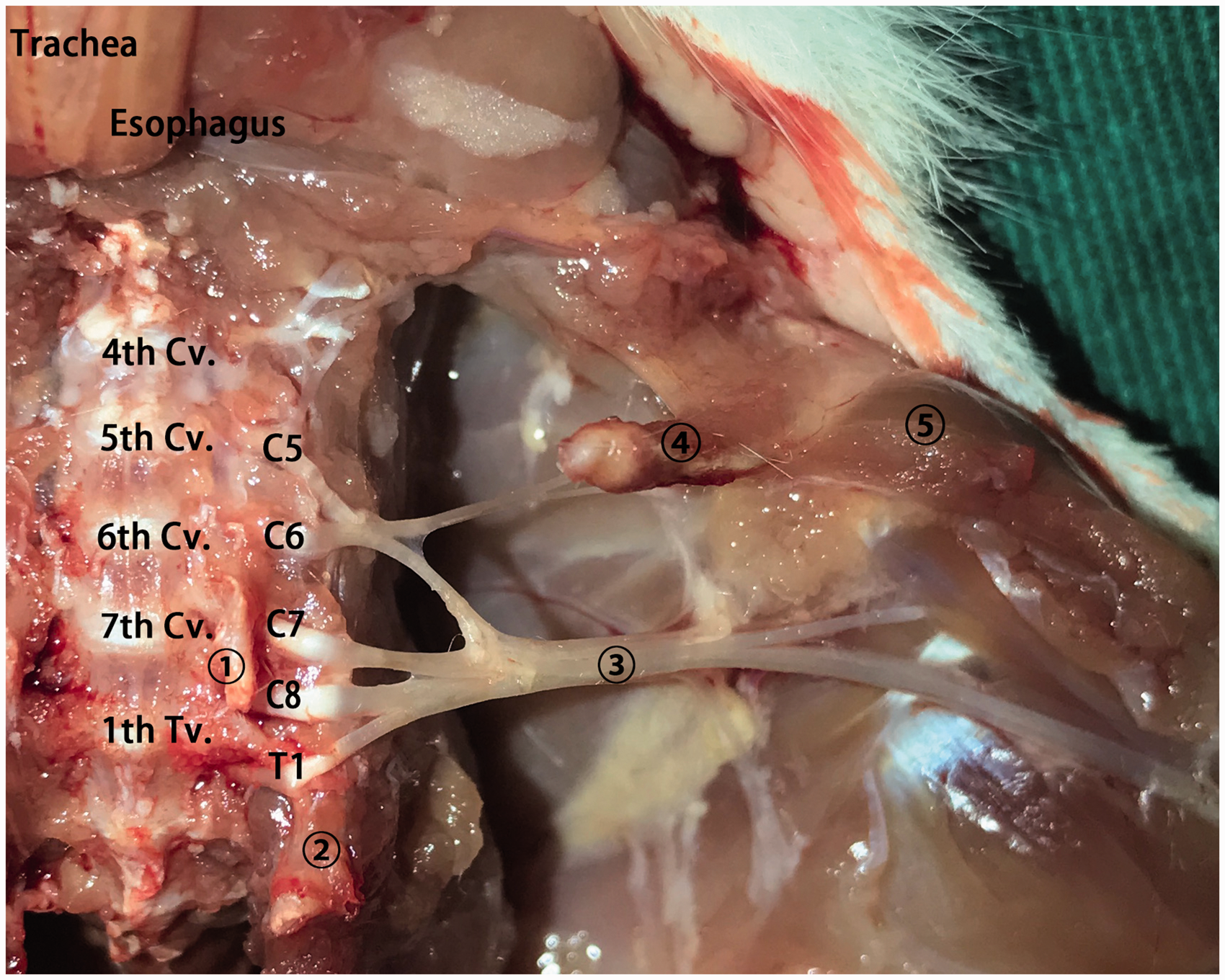
The beginning of the trunks was located posterior to the external jugular vein, which ran diagonal from the lateral neck, crossing to the medial clavicle (Figure 4). The divisions (anterior and posterior) and cords (lateral, medial and posterior cords) of the BP were concentrated and situated 0.5–1.0 cm below the middle and lateral clavicle. There are no major vessels at this position, except the axillary artery or vein, in addition to the lung. The branches spread out from this position. The axillary vessels ran along and beneath the cords but were not surrounded by the three cords like human vessels.
Schematic drawing of the position of the BP. The dashed line indicates the line passing the point of the lateral one-third of the clavicle parallel to the mid-sternal line of the rat. The position at the three circles indicates the blockade target position. The gray rectangle indicates the safe areas for puncture. n.: nerve; a: artery; v: vein.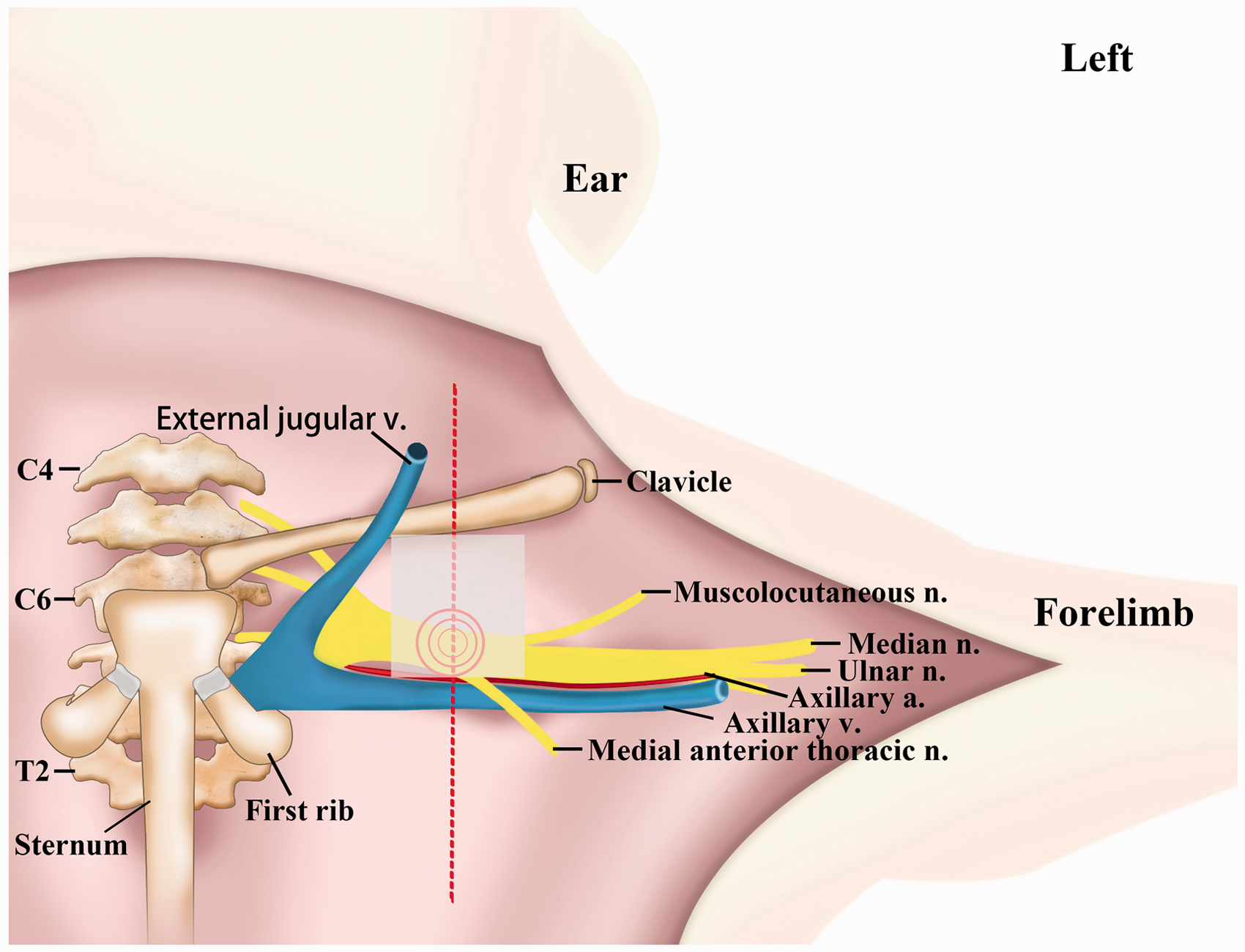
This dissection revealed that the space from the lateral one-third of the clavicle to the axillary vessels (distance of approximately 0.5–1.0 cm) was a safe puncture area (Figure 4). The anterior and posterior areas of this space were composed of muscles and fascia. Therefore, we defined the puncture point for rat BPB at 0.5–1.0 cm below the lateral one-third of the clavicle (Figure 4). The depth of the cords of the BP to the skin was approximately 0.7 cm.
Staining Studies
A percutaneous, perineural injection technique was developed for the divisions and cords of the BP using the injection method described in the results of BP dissection. Ten additional adult rat cadavers (weighing 200–300 g) were obtained to determine the feasibility and accuracy of this method. The rats were positioned in a dorsal recumbent position with the limb to be blocked extended straight, and the landmarks (0.5–1.0 cm below the lateral one-third of the clavicle) were determined (Figure 5(a)). A syringe needle (0.8 mm × 38 mm, Becton Dickinson SA, Mequinenza, Fraga, Spain) was used to create a puncture, and a non-traumatic peripheral nerve block (PNB)-stimulating needle (Uniplex Nanoline, 22 G × 50 mm, Pajunk GmbH Medizintechnologie, Geisingen, Germany) was used to inject 0.2 ml of methylene blue diluted in saline at 1:25 with the needle inserted vertically to a depth of approximately 0.7 cm. Dissection was immediately performed to expose the BP.
Rats were placed in a dorsal recumbent position with the limb to be blocked extended straight. (a) The body landmarks for BPB in rats. Arrows indicate the point for puncture at 0.5–1.0 cm below the lateral one-third of the clavicle. (b) The stained and unstained (deep injections) BPs in cadaver rats.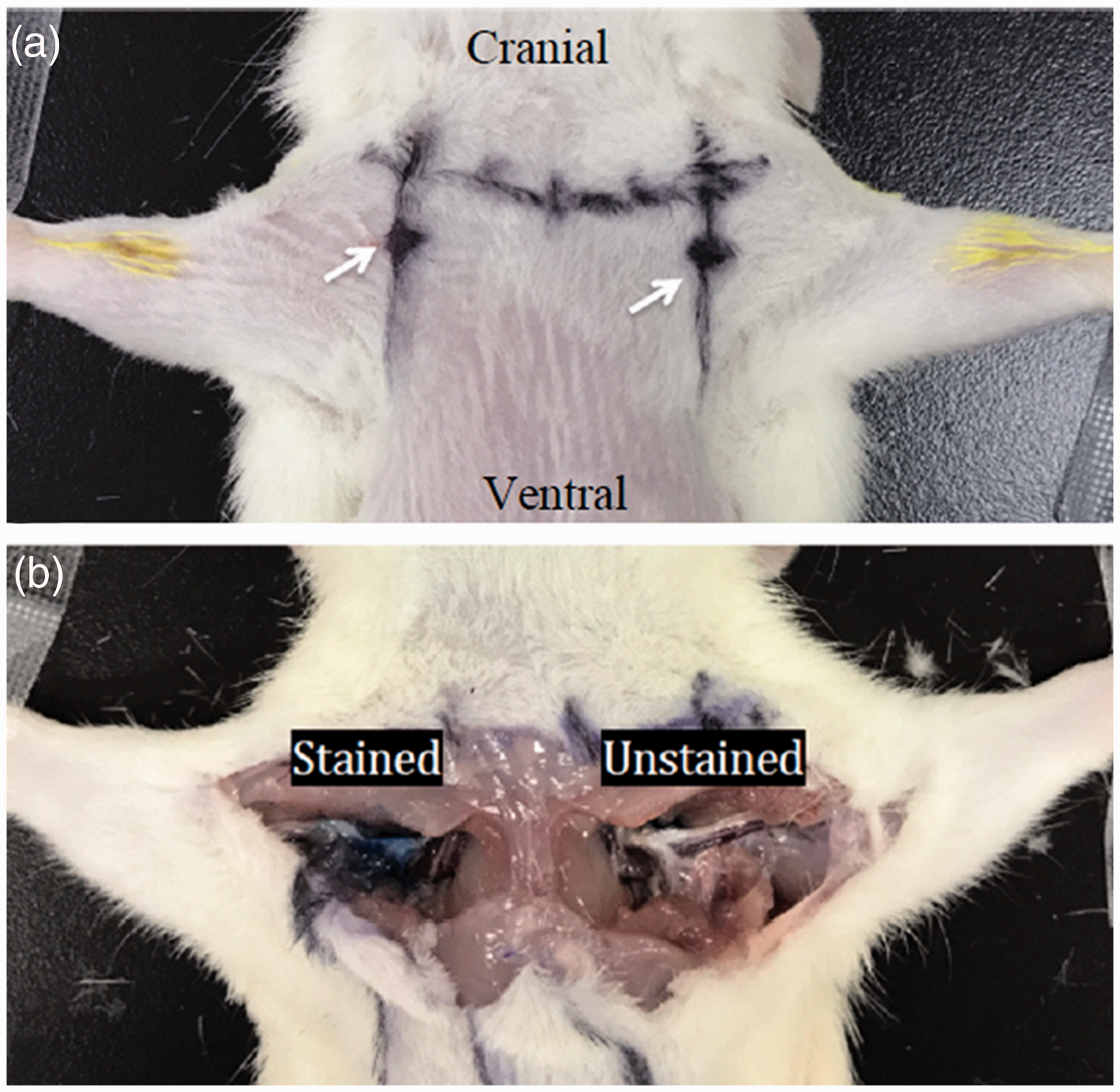
Staining results
Twenty stained positions were recorded; 14 BP cords were stained, and six BPs were not stained (Figure 5(b)). Two of the BPs, which were located in the pectoralis minor muscle, lacked blue dye due to too shallow insertion of the needle. The other four BPs, which were located in the posterior of the BP cords, lacked blue dye due to too deep insertion of the needle. No vessel ruptures or intrathoracic punctures occurred during the 20 injections.
Experiments in live rats
Animals
This study was performed in strict accordance with the guidelines for the care and use of laboratory animals, 9 and the Committee of Scientific Research and Institutional Animal Experimental Ethics Committee, West China Hospital, Sichuan University, Chengdu, China, approved the study (Ethics No.: 2015014A). Sixteen healthy Sprague-Dawley rats (8 males and 8 females) aged 3–4 months with a mean body weight of 238 g (range 220–260 g) were provided by Dashuo Laboratory Animal Co., Ltd (Chengdu, China). Rats were free of Hantavirus, Coronavirus, Dacryoadenitis virus, Mycoplasma spp., respiratory and enteric bacteria, ectoparasites, and endoparasites. The animals were housed in polycarbonate cages (five rats per cage) at an ambient temperature of 26℃ under a 12-h light/dark cycle. Rats were provided with nest-building material and allowed free access to a commercial pellet diet and pure water. Dashuo Laboratory Animal Co., Ltd (Chengdu, China) provided the diet, which conformed with the Chinese laboratory animal nutrient guidelines (GB 14924.3-2010) and hygienic standards (GB 14924.2-2001).10,11 Rats were handled daily for 3 days to become familiarized with the environment and to observe their physiological functions and limb activity.
Nerve stimulator-guided BPB
The results of the staining studies revealed six failed blocks (6/20) using blind injection. Therefore, we tested the nerve stimulator (NS)-guided BPB in 16 live rats. General anesthesia was induced with 5% isoflurane in 100% oxygen followed by a combination of 1.5–2% isoflurane in 50% oxygen for maintenance. Respiratory and pulse rates were closely observed. The fur of the rats was removed by shaving the chest and forelimbs. The skin areas innervated by the five major sensory nerves of the distal thoracic limb were marked using picric acid. A cotton swab was dipped into a 3% picric acid solution and applied to the skin (Figure 3). Rats were randomly divided into two groups (eight per group) based on a computer-generated randomized list. Rats received 2% lidocaine/saline (Group Lido) or 0.5% bupivacaine/saline (Group Bupi). One forelimb was randomly selected for saline or local anesthesia in each rat. Randomization of limbs was performed using a coin toss. The same investigator who was blinded to the drugs injected performed all injections.
The rats were positioned in a dorsal recumbent position with the limb blocked, and the landmark of the puncture point (0.5–1.0 cm below the lateral one-third of the clavicle) was determined (Figure 5(a)). A puncture was created at this point using a syringe needle (0.8 mm × 38 mm, Becton Dickinson SA, Mequinenza, Fraga, Spain) under surgical aseptic conditions. A non-traumatic PNB-stimulating needle (Uniplex Nanoline, 22 G × 50 mm; Pajunk GmbH Medizintechnologie, Geisingen, Germany) connected to a nerve stimulator (Stimuplex HNS 12, B Braun Melsungen AG, Melsungen, Germany) was inserted vertically through the hole in the skin. Stimulation was performed at a frequency of 1 Hz for 0.1 ms beginning at a current of 0.5 mA. The needle was slowly advanced until twitches of the forelimb and forepaw were observed, and the current was then decreased. Twitches should be maintained at 0.2 mA but abolished at lower stimulus intensities to avoid intraneural injection. 12 Drugs (0.2 ml) were injected when no blood or air appeared after gently withdrawing the syringe plunger. The twitches were immediately abolished following the drug injection. 12 BPB for the other forelimb was performed using the same protocol. Inhalational anesthesia was stopped immediately after surgery of the other forelimb was finished.
Blockade evaluation
The motor and sensory functions of the blocked forelimb were assessed qualitatively immediately after recovery of the righting reflex (i.e., defined as T0). Assessments were performed at 15-min intervals until normal motor function was restored. The same investigator who was blinded to the injection process performed all assessments.
Motor blockade
Motor block was assessed using digit grasping and the straightening test. A three-point subjective rating scale was used: 1 = normal, normal ability to grasp a grid mesh with a weight of 200 g or straighten the digits; 2 = partial blockade; and 3 = complete blockade, no active digit flexion or straightening. 13
Sensory blockade
Mechanical hyperalgesia in the dermatomes innervated by the five major sensory nerves of the thoracic limb (Figure 3) was evaluated using the same method for assessing sensory blockade in cats. 6 The evaluation was always performed in the same sequence: median, ulnar, radial, musculocutaneous, and axially nerves.
An alligator clip was used to elicit nociception. Only the first two jaws of the clip were used to clamp. The jaws were continually closed for a maximum time of 5 s until a positive response was elicited, and the following three-point rating scale was used: 1 = normal, normal response to the stimulus, with rapid withdrawal of the limb, weight shifting and/or head movement toward the testing site and/or vocalization; 2 = partial blockade, delayed or attenuated response to the stimulus; and 3 = complete blockade, absence of a response to the stimulus.
The following times (min) were also recorded in this study: onset time of blockade = time from the drug injection to complete blockade (score of 3); offset time of blockade (total duration time of blockade) = time interval after complete blockade (score of 3) was fully achieved and gradually reduced to partial blockade (score of 2).
Post-treatment evaluation and pathological study
Four rats (two from each group) were observed for 14 days after BPB to determine possible complications. The remaining rats (six rats per time-point) were euthanized using 2 ml propofol at 3 and 8 days after drug injection, and the BP nerves (12 per time-point) were retrieved. No injury of the surrounding tissues was observed in the BP nerves. The BP nerves were evaluated pathologically for mechanical injury and potential drug influence. The cords of the BP (the drug injection site) were removed and fixed in 10% formaldehyde at 4℃ overnight. Cords were cross and coronally sliced, and hematoxylin and eosin staining was used to determine nerve injury. A blind histological examination was performed. The percentage of the unstained area (%UA), in which the myelin was retreated, destroyed or dissolved, was used to evaluate the histological damage of nerve fibers. The formula was %UA = unstained area/entire area of the nerve fiber × 100. 14
Statistical analysis
Statistical analyses were performed using Prism 6 (GraphPad, San Diego, CA, USA). Statistical tests were two-tailed, and data are presented as the means ± SD. The onset and offset times of the motor and sensory blocks were analyzed using the Mann-Whitney test and two-way analysis of variance (ANOVA). The %UA was analyzed by using one-way ANOVA. Differences were considered significant at p < 0.05.
Results
All 32 BPB in the 16 rats were accomplished in 2–5 min. All rats recovered after isoflurane delivery was stopped, and the righting reflex was restored within 2 min of isoflurane cessation. Complete motor and sensory blockades were observed in all forepaws after 2% lidocaine and 0.5% bupivacaine injection at T0. The onset times were 5.50 ± 2.07 min after 2% lidocaine injection and 4.89 ± 1.73 min after 0.5% bupivacaine injection (p = 0.578).
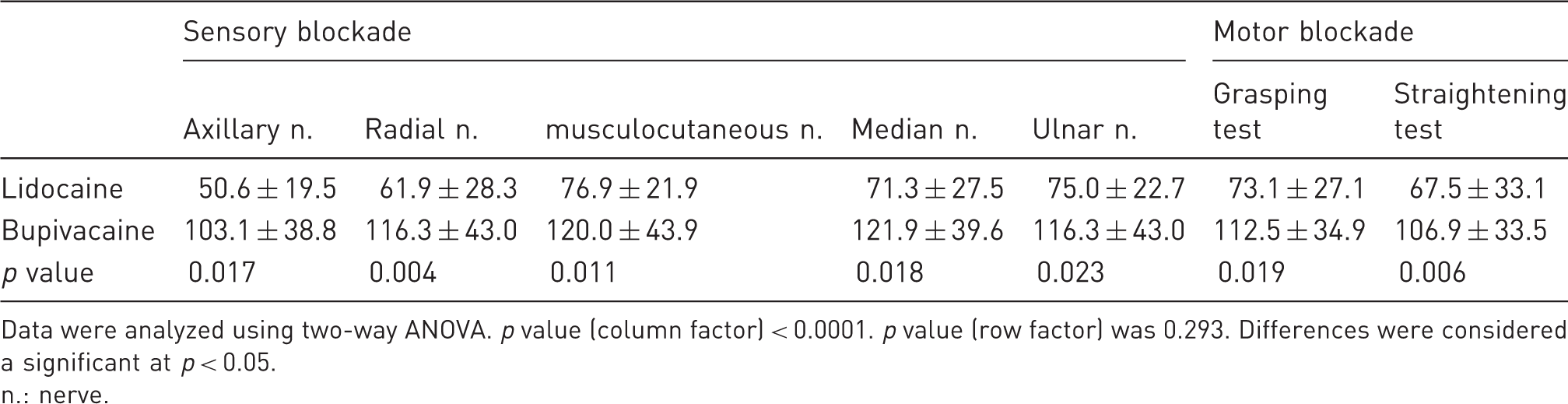
The offset times of the motor and sensory blockades induced by 2% lidocaine (p < 0.0001, Figure 6(a)) and 0.5% bupivacaine (p < 0.0001, Figure 6(b)) were significantly different compared with saline. The offset times of the motor (grasping, straightening) and sensory (axillary, radial, musculocutaneous, median, and ulnar nerves) blocks in the 0.5% bupivacaine group were significantly longer than those in the 2% lidocaine group (p < 0.05, Table 1). Table 1 shows the total duration of blockades induced by 2% lidocaine and 0.5% bupivacaine. No obvious differences were found between the offset times of the sensory and motor blockades induced by 2% lidocaine or 0.5% bupivacaine (Table 1).
The offset time of BPB in rats (each group, n = 8). (a) Comparison of offset times in Group Lido (2% lidocaine vs. saline, *p < 0.001). (b) Comparison of offset times in Group Bupi (0.5% bupivacaine vs. saline, *p < 0.001). n.: nerve; grasping: the grasping test; straightening: the straightening test. BPB Brachial plexus block. Offset time of brachial plexus nerve block with 2% lidocaine or 0.5% bupivacaine in rat. Data were analyzed using two-way ANOVA. p value (column factor) < 0.0001. p value (row factor) was 0.293. Differences were considered a significant at p < 0.05. n.: nerve.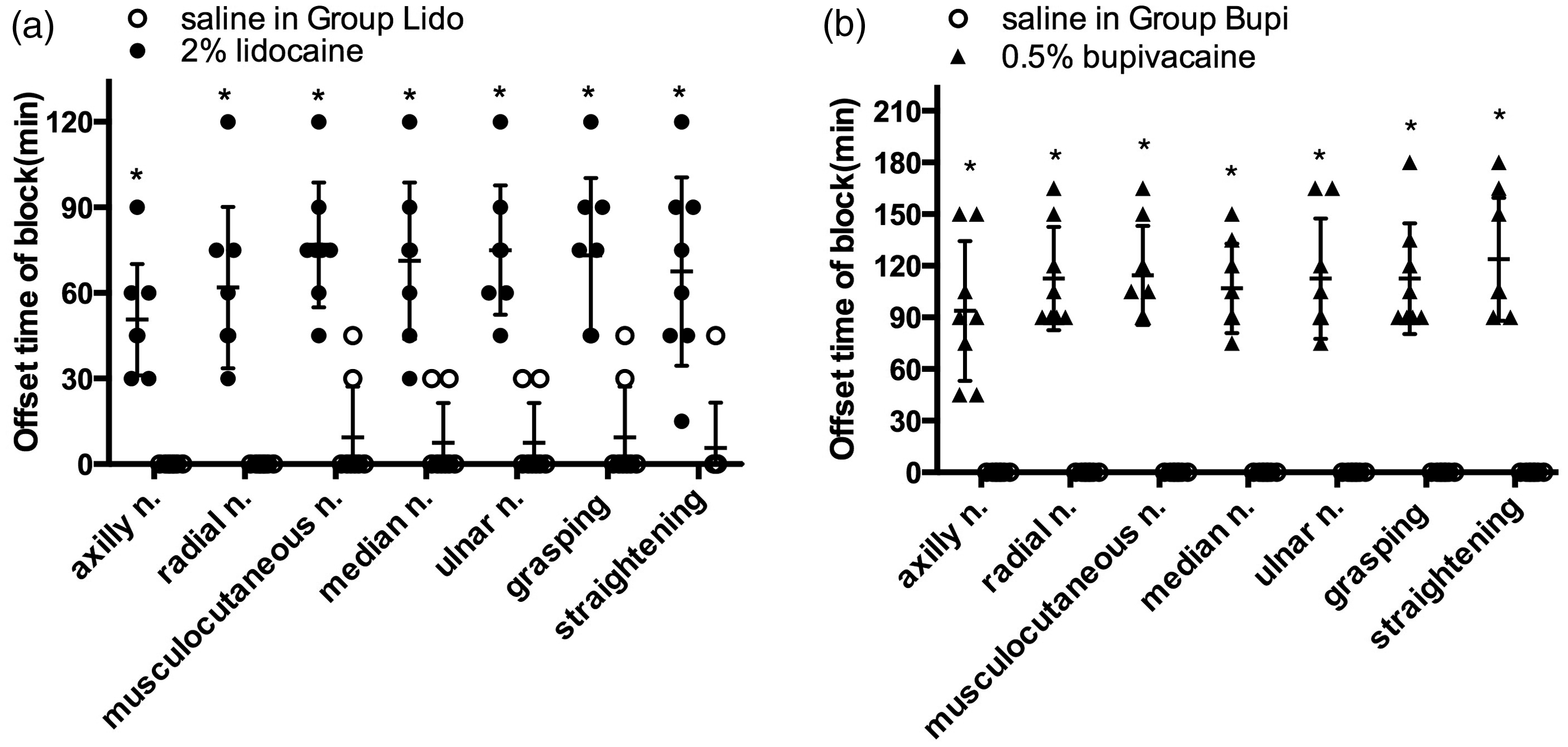
Two saline-injected limbs in the lidocaine group exhibited complete and partial motor and/or sensory blockade for 30 min (Figure 6(a)). There were no significant differences in % UA among the lidocaine, bupivacaine, and saline injections. No abnormal behavior, color change, ulcers, or necrosis of the thoracic limbs were observed.
Discussion
The present study developed and investigated a rat model of BPB. Completely reversible BPBs were observed in all limbs that received 2% lidocaine or 0.5% bupivacaine but not in the limbs that received normal saline. These results demonstrate high success and reliability rates in the rat BPB model. The anatomical and BPB results revealed that the rat BP innervated the cutaneous and muscular structures of the entire thoracic limb, and the NS-guided BPB provided complete anesthesia (motor and sensory block). The motor and sensory blockade induced by bupivacaine exhibited a longer duration than that induced by lidocaine. This characteristic is consistent with the pharmacodynamic function of these two drugs, as was demonstrated in other models. 14 Therefore, this BPB model is effective and reproducible and is suitable for evaluating the efficacy, safety, and pharmacokinetics of new local anesthetics and additives. This model will aid in the investigation of the chemical and pharmacological injurious effects of substances injected onto peripheral nerves and the effects of nerve block on specific outcomes.
All NS-guided BPBs in our study were accomplished in 2–5 min, which indicates that this method is simple and easy to perform. No skin injury, hematoma, pneumothorax, intrathecal block or nerve damage was observed in our model. Pathological studies revealed no mechanical injury or potential drug influence. This model differs from previously reported BPB models that did not discuss complications or reported relatively high complication rates.5,15 The possible reasons for these differences included that the anatomical characteristics of the rat BP and the NS-guided technique improved the safety of the rat BPB model. The rat model of NS-guided BPB is a feasible, safe, effective and reproducible model.
Autopsy was performed to examine easily identifiable surface landmarks and to define an effective entry approach for developing a rat BPB model. Our cadaver study revealed widespread morphological homology between the rat and human BP (Figure 1). However, some slight differences were observed between the rat BP and the human BP. The axillary vessels ran along and beneath the cords but were not surrounded by the three cords. There were no clear medial, posterior, or lateral cords in the rat BP, which was also reported in a previous study. 8 The trunks of the human BP pass between the anterior and middle scalene muscles at the level of the supraclavicular space, over the first rib, and continue beneath the clavicle. The sternum and clavicle in rats are at a higher position than in humans. The suprasternal border in rats is located at the 6th cervical vertebra (Figure 1) but is in front of the 1st thoracic vertebra in humans. The divisions and cords of the rat BP are located beneath the clavicle.
The results of the BP dissection in rats revealed that the space from the lateral one-third of the clavicle to the axillary vessels (distance approximately 0.5–1.0 cm) was a safe puncture area. The anterior and posterior areas of this space were composed of muscles and fascia. Our staining study demonstrated that 6 of the 20 BPs from the cadavers were not stained with methylene blue, which indicates inaccurate blind injections. Therefore, we used the nerve stimulator-guided method to improve the success rate of BPB. All BPBs were successful with the guidance of the nerve stimulator in the live rats, and no severe injuries or side effects were observed.
The duration of bupivacaine on rat BPB in this study was 103.1–121.9 min (Table 1). This duration of action was shorter than that of bupivacaine described in rat anesthesia and analgesia formulary. 16 However, one reason for the shorter duration in this study was the difference in the definition of block duration. We defined the onset time as the time from drug injection to complete blockade (score of 3) and the offset time as the time interval after achieving complete blockade and to reversion to a partial blockade (score of 2), but not to normal function (score of 1). Brummett et al. used 0.5% bupivacaine in 0.2 ml for rat sciatic nerve block. 17 The duration of complete blockade (101.3 ± 20.7 min) was similar to that reported in our study, but the time to restoration of normal function (223.1 ± 22.4 min) was similar to the duration described in the formulary.
Sensory or motor blockade after placebo injection was observed in 2 of the 16 rats in the present study (Figure 6(a)), but this block was shorter than the blocks from lidocaine and bupivacaine (Table 1). The unexpected blockade was not associated with permanent neurological injury or functional impairment in the hind limbs, and pathological study of the BP nerves revealed no demyelination or Wallerian degeneration. The possible reasons behind the saline-induced block should be further investigated. This phenomenon is sometimes referred to as a possible physiological block, which is unrelated to pharmacological effects. A psychological reason may also be considered because pain may be transferred via olfaction in co-housed mice. 18 Whether there is a social transfer of neural blockade in rodents is not known.
Ultrasound (US)-guided BPB is prevalent in upper extremity surgery. However, NSs are cheaper and easier to obtain and operate than US systems. A randomized controlled trial indicated no difference in the efficacy of axillary BPB performed by novices when using ultrasound guidance vs. the NS technique. 19 Axillary blocks with US guidance exhibited similar success rates and complication rates as NS guidance. 20 Our study demonstrated the feasibility of a BPB rat model and confirmed the efficacy and safety of BPB under NS-guided conditions. Further studies should be performed to investigate the US-guided BPB model in rats and to compare US and NS guided-BPB in rats for the development of a safer and more convenient BPB method. Vuckovic et al. 21 demonstrated that measuring the injection pressure was an effective method for evaluating the safety of BPB in the axilla of rats. This technique would be a good supplement for NS or US-guided BPB to improve injection safety.
One limitation of the current approach is that the onset times of the motor and sensory blocks were not elucidated. There was no statistically significant difference between the onset times of lidocaine and bupivacaine. Motor and sensory function was blocked prior to recovery from anesthesia. The actual onset time was shorter than the recorded onset time. The recorded onset time was 5.50 ± 2.07 min after 2% lidocaine injection and 4.89 ± 1.73 min after 0.5% bupivacaine injection. BPB block without general anesthesia could be useful in the determination of the onset times of local anesthetics. The high concentration of local anesthetic (2%, lidocaine) may also contribute to this limitation. A lower concentration of local anesthetic may be beneficial for observing the exact onset time and recovery sequence of motor and sensory blocks. A previous study demonstrated a motor deficit anterior to nociception block in rats after local anesthetic injection (0.05-1%, lidocaine). 22 A graded series of doses and concentrations of local anesthetics should be used to further evaluate the model.
Conclusions
We developed a feasible and promising BPB model in rats using cadavers and live rats that can be used for analgesia in upper limb surgery in rodents to evaluate the efficacy, safety and pharmacokinetics of new local anesthetics and additives.
Footnotes
Acknowledgments
We thank colleagues in the laboratory of Translational Neuroscience Centre, West China Hospital, Sichuan University, for their assistance with the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Nature Sciences, Beijing, P.R. China, (grant number 81271201).
