Abstract
Severe combined immunodeficiency (SCID) is defined by the lack of an adaptive immune system. Mutations causing SCID are found naturally in humans, mice, horses, dogs, and recently in pigs, with the serendipitous discovery of the Iowa State University SCID pigs. As research models, SCID animals are naturally tolerant of xenotransplantation and offer valuable insight into research areas such as regenerative medicine, cancer therapy, as well as immune cell signaling mechanisms. Large-animal biomedical models, particularly pigs, are increasingly essential to advance the efficacy and safety of novel regenerative therapies on human disease. Thus, there is a need to create practical approaches to maintain hygienic severe immunocompromised porcine models for exploratory medical research. Such research often requires stable genetic lines for replication and survival of healthy SCID animals for months post-treatment. A further hurdle in the development of the ISU SCID pig as a biomedical model involved the establishment of facilities and protocols necessary to obtain clean SPF piglets from the conventional pig farm on which they were discovered. A colony of homozygous SCID boars and SPF carrier sows has been created and maintained through selective breeding, bone marrow transplants, innovative husbandry techniques, and the development of biocontainment facilities.
Introduction
The Iowa State University (ISU) severe combined immunodeficiency (SCID) pigs were unexpectedly discovered during a viral challenge study in collaboration with Kansas State University. 1 Animals that died early in the trial revealed a complete lack of antibodies and dysplastic immune tissues (thymus, spleen, and lymph nodes). Genetic analysis revealed a significant association with the chromosome 10 region containing the Artemis gene, which is associated with human SCID.2,3 Two separate point mutations within the Artemis gene segregate in a Mendelian recessive mode of inheritance in which homozygotes or compound heterozygotes produce the SCID phenotype. 2 Consistent with human Artemis SCID patients, early in life the ISU SCID pig is devoid of B and T cells,4,2 but has a population of natural killer (NK) cells that demonstrate normal function in vitro. 5
While SCID is naturally occurring in humans, mice, dogs, horses, and now pigs,3,6,7,1 it has also been introduced into rodent and porcine hosts for use as biomedical models through targeted genetic modification.8–12
The ISU SCID pig permits the growth of two kinds of human cancer cell lines when injected into the ear, 13 establishing its value for research in cancer drug testing. The ISU SCID pig has also been used to explore the roles of the innate and adaptive immune system during a disease challenge, contributing to research focusing on mechanisms of disease for influenza A. 14 Although there is widespread interest in developing SCID pigs as a research model, little research is available on the care of large-animal SCID models.
Specific pathogen-free (SPF) SCID mice colonies are typically cared for in a “barrier facility” with group-housing boxes under high-efficiency particulate air (HEPA)-filtered conditions. 15 Staff interacting with animals follow strict personal protective equipment (PPE) requirements and animals are maintained with autoclaved feed and bedding. 16 Large-animal facilities have many more parameters to balance considering the size and requirements of pigs. We have been unable to keep the immune-compromised SCID pig alive for more than 80 days in a conventional pig-holding facility or within an isolated standard animal room. Similar viability of SCID pigs including failure to thrive phenotypes were described after engineering defective recombination activating genes (RAG) SCID pigs that had to be terminated before 30 days of age. 11 To improve the research potential of the SCID pig model by producing longer-lived, healthy SCID pigs, development of biocontainment facilities and appropriate methodologies to raise SPF SCID pigs are needed.
Current approaches for obtaining SPF piglets from non-SPF sows include snatch farrowing (SF) or cesarean-derived piglets, of which both may involve colostrum deprivation (CD). 17 The risks of CD are well understood, and include mortality, reduced protection from clinical disease, and significantly lower hematocrit and hemoglobin levels compared to naturally sucking piglets.18–22 Colostrum consumption is positively associated with piglet immunity, survival, and growth.21,23 Common practices for obtaining SPF piglets from non-SPF sows include supplementing piglets with milk replacer or a bovine colostrum alternative. 24 The production of dried replacement bovine colostrum is an involved process, 25 and does not protect the piglet from porcine-specific pathogens with antibodies they would normally receive from suckling a vaccinated or unvaccinated sow. An interesting study 22 compared circulating immunoglobulin (Ig)G levels between naturally suckling and CD piglets: The CD piglets were fed either milk replacer, bovine colostrum, or pig IgG. Naturally suckling piglets achieved the highest levels of total IgG, while supplementation with bovine or pig IgG was sufficient for 100% survival of piglets. However, only 30% of milk replacer-alone CD piglets survived. 22 While much of the CD piglet-rearing literature was developed prior to the availability of SCID pigs, these results indicate that various options may be available to rear SCID pigs isolated from their nursing sow, which is the main source of disease transmission to the piglet.
This manuscript describes the facilities and protocols developed to greatly decrease opportunities for contamination and raise SPF SCID and non-SCID pigs derived from non-SPF sows. This work also provides examples of facilities and protocols to produce disease-naive pigs, which may be important for disease challenge studies. The farrowing protocols herein described provide means for creating founding SPF SCID carrier dams, methods to introduce new genetics or replace animals within an established SPF colony, and provide recommended guidelines for researchers seeking to develop a SCID pig model containment facility.
Animals
Institutional Animal Care and Use Committee (IACUC) statement
All care and animal procedures were approved by the ISU IACUC and adhere to the United States Department of Agriculture guide to Large Animals and the Animal Welfare Act. Euthanasia via captive bolt or intravenous overdose of sodium pentobarbital was performed by trained staff.
SCID pigs and non-SCID littermates
Health testing for the Lauren Christian Swine Research Center (LCSRC) from 2015 to 2017. Carrier dams are sourced from the LCSRC, where animals are routinely monitored for herd health by testing for the pathogens listed. All testing was performed by the Veterinary Diagnostic Laboratory.
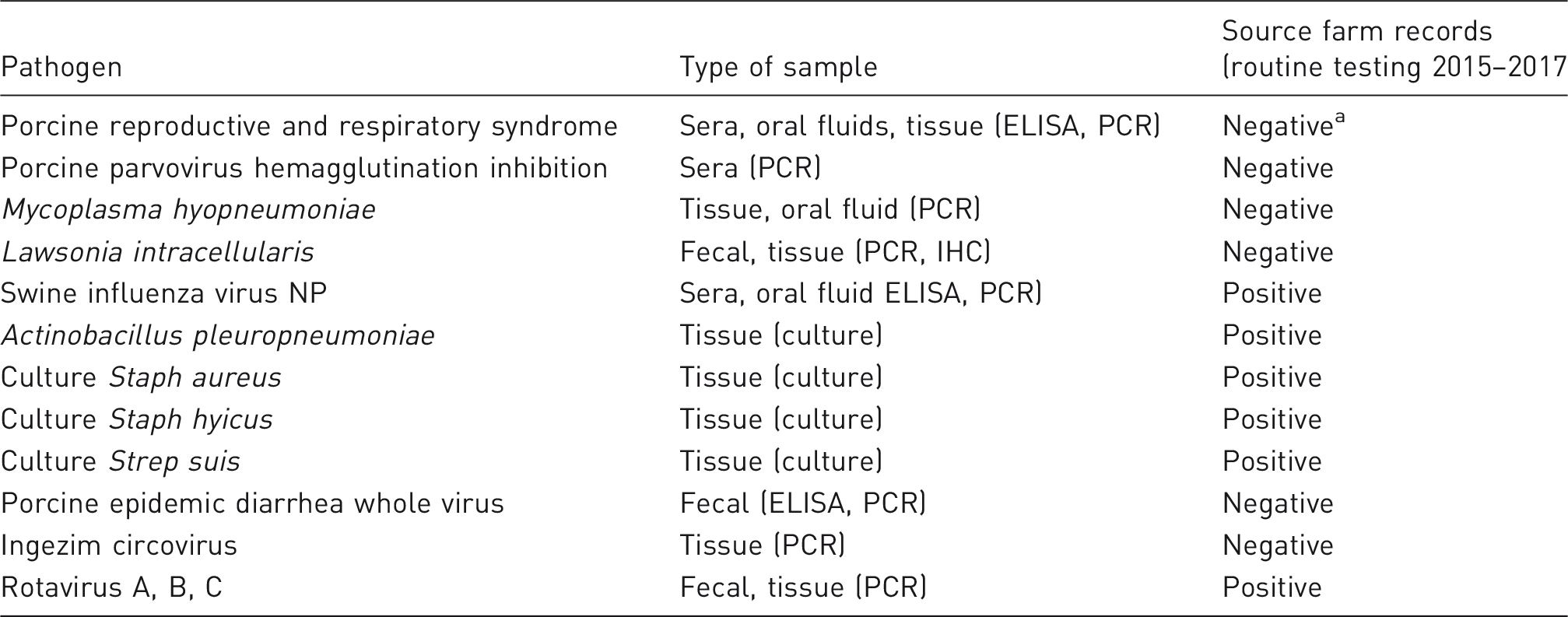
Single individual ELISA positive out of 30 sera samples in 2015; was not confirmed with PCR. Thought to be a contamination error or a false positive.
NP: nucleoprotein; ELISA: enzyme-linked immunosorbent assay; PCR: polymerase chain reaction; IHC: immunohistochemistry.
Bone marrow transplantation (BMT)-rescued SCID sires
To create litters composed of 50% SCID piglets, we rescued SCID boars via BMT, which resulted in a genetically SCID, phenotypically normal animal that could be used for breeding. Since successful BMT boars have intact immune systems, they can be housed in clean, more traditional pig facilities at lower cost. To accomplish this, SCID piglet-containing litters were major histocompatibility complex-typed to identify recipient and donor littermate matches. 26 SCID recipients received unfractionated bone marrow cells (isolated from all long bones of the donor) intravenously delivered via an ear vein. Successful engraftment was verified by complete blood cell count, flow cytometry, and response to vaccination.
Materials and methods
Maintenance and construction of facility
Bubble fabrication
Custom design of bubbles was completed with expertise from ISU, SCID project leadership, Laboratory Animal Resources (LAR) supervisors, and Biobubble Inc, which created the final design, and fabricated and installed the main isolation equipment. Incoming conditioned city water is ultraviolet (UV)-irradiated and filtered (5 micron before UV irradiation and 0.5 micron after UV irradiation) (Figure 1(d)).
General bubble components. (a) Entrance of long-term bubble; pigs are housed on either side while middle section is designated for dressing in PPE. (b) Staff shown collecting cord blood on newly snatch-farrowed piglets in the STB. (c) HEPA filtration unit filters all incoming air that enters the bubble. (d) Water is filtered and irradiated before entering the bubble. (e) All staff don PPE prior to entering either bubble. PPE: personal protective equipment; STB: short-term bubble; HEPA: high-efficiency particulate air.
Room sterilization
Prior to animal occupation, rooms are sterilized (Halo’d) using a HaloMist™ Disinfectant Fogging Solution (halosil.com/halo-disinfection-system), which entails a hydrogen peroxide/silver mixture aerosolized treatment of the room and any resident equipment.
Maintenance and care of animals
SCID carrier dams
Two sources of pregnant dams were used in this project: the LCSRC and from within the biocontainment facility. Artificial insemination is used at both locations. Before leaving the LCSRC, pregnant carrier sows are tested by blood draw for porcine reproductive and respiratory syndrome (PRRS), porcine circovirus (PCV2), and porcine epidemic diarrhea virus (PEDV) through the ISU Veterinary Diagnostic Laboratory (VDL). Test-negative sows are washed at the farm, delivered by pre-sanitized truck and trailer, washed again, and housed in a high-sanitation room with other separately penned gestating sows within the LAR facility. A week before farrowing, animals are transferred to a recently cleaned and Halo’d room containing a farrowing crate, and sows are cleaned with chlorhexidine. Once piglets are removed (delivered for SF (see below), or weaned for naturally farrowing) sows are transferred back to the sow room. If sows are to be used for artificial insemination within the biocontainment facility where no live boar was available, they are monitored for behavioral estrus (standing heat) with a cloth sprayed with boar pheromones (Part #: 320506, QC Supply, Schuyler, NE, USA). If natural heats are not detected, adjusting the estrous cycle in preparation for insemination of sows was accomplished by administering 15 mg/day altrenogest (Matrix, Merck, NJ, USA) orally for 18 days followed by one dose of PG600 (Merck, Part #: 147448) administered intramuscularly 24 hours after last Matrix dose, and a single dose of Ovugel (JBS United, Sheridan, IN, USA, NDC #: 51233-101-50) administered intravaginally approximately 80 hours after PG600 injection. Behavioral estrus was detected typically 24–48 hours post-Ovugel administration, at which point animals are artificially inseminated using fresh or frozen semen from the desired sire.
Neonatal piglet care
Naturally farrowed piglets are monitored to ensure they are suckling. SF piglets are delivered by personnel wearing PPE (see below) without touching the floor or crate using sterile towels and transferred into sterile rodent boxes with air-filtered lids (Innovive, Part #: RS1-H) for transport to the short-term bubble (STB), where they are dried with Mistral micronized clay (Part #: 541005, QC Supply, NE, USA), weighed, and placed in heat lamp-warmed, piglet-housing decks (Birthright™, Yellow Sow, Ralco, MN, USA). Within the first 24 hours of life, SF piglets were fed a minimum of 250 ml 27 pooled pig colostrum collected previously by hand by staff at the LCSRC from farrowing sows, which was then pasteurized at the ISU dairy farm using standard bovine practices. Confirmation of pasteurization was accomplished with a Bulk Tank Analysis/Standard Plate Count at the ISU VDL, which includes the detection of Streptococcus agalactiae, Staphylococcus aureus, coliforms, gram-negative rods (lactose negative), non-agalactiae Streptococci, coagulase-negative Staphylococci, Corynebacterium, and Bacillus, Mycoplasma, among others. Pasteurization of colostrum dramatically decreases, but does not eliminate, microbial counts. For example, one unpasteurized colostrum sample had colony-forming units (CFU) counts of 3.9 × 105 CFU/ml while post-pasteurization averages 3.8 × 103 CFU/ml. Piglets were fed with a 60 ml syringe and attached sterile tubing (Part #: 366.78750.4, Midwest Veterinary Supply, MN, USA) (Figure 2(b)). After 250 ml colostrum consumption, piglets are converted to irradiated milk replacer, and eventually starter feed.
General piglet care
All litters are processed 24–48 hours post-birth; piglets were ear notched, their needle teeth clipped and tails docked, and given 1 ml each of iron (Iron Dextran cat# DU3067, Durvet, MO, USA) and EXCEDE (ceftiofur crystalline free acid, used to manufacturer’s specifications, Zoetis, NJ, USA). Piglets are weighed daily until achieving a body weight of 20 kg, and weekly thereafter up to 100 kg. All piglets are weaned from the sow or from milk replacer at 24–35 days of age and transitioned to an irradiated starter feed. All feed given to pigs other than colostrum (gestation, starter, and milk replacer powder) is gamma-irradiated (5 kGy, Iotron Inc). Long-term carrier animals are vaccinated following standard pig farm practices including Circumvent PCV-M (Intervet), FarrowSure Gold B (Pfizer), and LitterGuard LT-C (Pfizer). To aid in the safety and efficiency of animal handling, as well as increased animal enrichment, pigs in our facility are clicker-trained with marshmallow rewards for regular care such as weighing, hoof trimming, vaccinations, snaring, and additional sample collection.
IgG enzyme-linked immunosorbent assay (ELISA)
A sandwich ELISA was used to quantify pig serum IgG concentration. The protocol, adapted from Bethyl Laboratories Porcine IgG ELISA protocols, used capture antibody of polyclonal goat anti-porcine IgG (Bethyl Labs #A100-104 A, 10 mg/ml). Unconjugated pig IgG reference serum (Bethyl Labs #RS10-107) was used as a standard for serially diluted experimental serum samples. Detection antibodies to pig IgG-Fc conjugated to horseradish peroxidase (Bethyl Labs #A100-104 P) were used with 2,2'-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) substrate buffer in citric acid. The reaction was stopped with 5% sodium dodecyl sulfate.
Monitoring for disease and SPF testing
Biosecurity, staff, and PPE
Our facility features different levels of biosecurity, and PPE guidelines are dependent on the level of biosecurity required. Staff interacting with animals cannot travel from one level to a higher biosecurity area without 24 hours’ down time and at least one shower and change of clothing. To enter the highest biosecurity region (either the long-term bubble (LTB) or STB), staff must have not been in contact with outside pigs for at least seven days and a minimum of five showers. Staff remove outer clothing, change into freshly laundered scrubs and socks, then don protective disposable outer shoe covers, zip-up full-body long-sleeved plastic suit (Part #: 4902,Valumax, PA, USA), surgical mask, hair net, a pair of long cuffed gloves, and a second pair of short cuffed gloves. Although gloves are non-sterile, gloved hands are then sprayed with a hydrogen peroxide disinfectant and staff immediately enter the high-biosecurity area. For staff moving between animal housing areas, innermost scrubs are protected within full-body plastic suits in hallways and new suits are donned before entering a new animal room. Rubber slip-on shoes are worn between animal containing units, and changed for rubber boots that remain inside pig-residing areas.
Monitoring bubble and research pig health
All pigs are checked by staff twice per day, with written records updated daily. In accordance with biosecurity management, staff enter the cleanest space first (usually the LTB), and as described above change outer suits and gloves before entering the STB. Only outer gloves are changed for entry into isolated non-SCID sow rooms (or housed naturally farrowed litters). If any animal in any given location is not Bright, Alert, Responsive (BAR) or Quiet, Alert, Responsive (QAR), an on-call veterinarian is called. Depending on the symptoms present and severity, animals are closely monitored with scheduled veterinarian check-ups, and if necessary, veterinarian-prescribed treatments.
Monitoring LCSRC pig health
Animals are routinely tested six times a year unless concerns or symptoms warrant additional testing for the specific pathogens listed (Table 1). The number of animals selected for testing was based on the estimated prevalence and a 95% confidence level that the disease will be detected 28 or on clinical signs at the time of a disease investigation. Sampling was conducted by the Swine Medicine Education Center. Diagnostics are performed on sera, oral fluid, fecal, and tissue samples. All testing was performed by the ISU VDL.
SPF testing
Testing for specific pathogen-free (SPF) status from the Veterinary Diagnostic and Clinical Pathology laboratories. Animals are tested for specific porcine pathogens and clinical parameters listed. Testing is conducted from serum, EDTA-treated whole blood, fecal, and/or nasal swab, and diagnostics are performed by the Veterinary Diagnostic or Clinical Pathology Laboratories. a

Samples submitted to the Clinical Pathology Laboratory.
EDTA: ethylenediaminetetraacetic acid; NP: nucleoprotein; ELISA: enzyme-linked immunosorbent assay; PCR: polymerase chain reaction.
Results
Large animal biocontainment bubbles
The novel SCID pig “bubbles” create a clean environment where SCID pigs can be raised free of pathogen threat (Figure 1(a) and (b)). Air sterility is maintained by a Biobubble containment structure consisting of a positive-pressure HEPA-filtered airflow system (Figure 1(c)) and UV-treated water system (Figure 1(d)). All personnel entering the bubbles or interacting with SCID colony animals wear PPE that minimizes human exposure as described in the Materials and methods section (Figure 1(e)). See https://vimeo.com/221432512 or contact the Tuggle lab for more information or detailed standard operating procedures.
SF/Cesarean section
Protocols for obtaining SPF piglets from non-SPF sows were developed and tested. Piglets obtained by cesarean section are not exposed to vaginal microbiota from their sow. To retain a reproductively sound carrier sow for production of future litters, and produce piglets exposed to maternal microbiota, we developed an SF technique based on prior work. 24 Farrowing sows were moved into a sterile room, washed, and monitored for signs of labor. As piglets are delivered, they are caught directly from the vaginal canal in sterile towels by gowned staff and immediately have their umbilical cords clamped prior to being placed in a sterile rodent box with filter tops for delivery to staff in the bubble.
Upon arrival into the bubble, piglets have cord blood collected and are processed. To provide as safe an exposure to microbiota as possible, piglets are fed pasteurized pooled pig colostrum that was collected by hand from farrowing animals at the farm from which the carrier sows are originally sourced. To date, pooled pasteurized colostrum has been administered to five litters and approximately 27 SF piglets. Various pools included samples from 13–45 sows and had an average total IgG concentration of 5.56 (±0.13) mg/ml. Since conventional human bottles are inadequate for feeding piglets and difficult to regularly clean in the bubble environment, we feed our piglets with a disposable catheter attached to a 60 ml syringe (Figure 2(a)). We tape the tapered end of the tube to our finger and allow the piglet to suckle while delivering colostrum with the syringe. This feeding apparatus is disposable, and allows us to carefully monitor and record colostrum volumes consumed by each individual (Figure 2(b)). Piglets are fed pasteurized colostrum every three hours until they have consumed 250 ml, at which point they are fed irradiated milk replacer using the same method. Once sufficient colostrum has been consumed by all piglets in the litter (within the first 24 hours of life), milk replacer is also provided in a separate feed dish inside the farrowing decks. Piglets typically require supplemental tube feeding for the following 48–72 hours as they transition to dish feeding.
Snatch-farrowed piglet feeding apparatus. (a) A sterile urinary catheter tube is taped to a gloved finger and attached to a 60 ml tube for feeding. (b) Porcine colostrum or milk replacer is administered as piglets suckle on the finger with tube attached.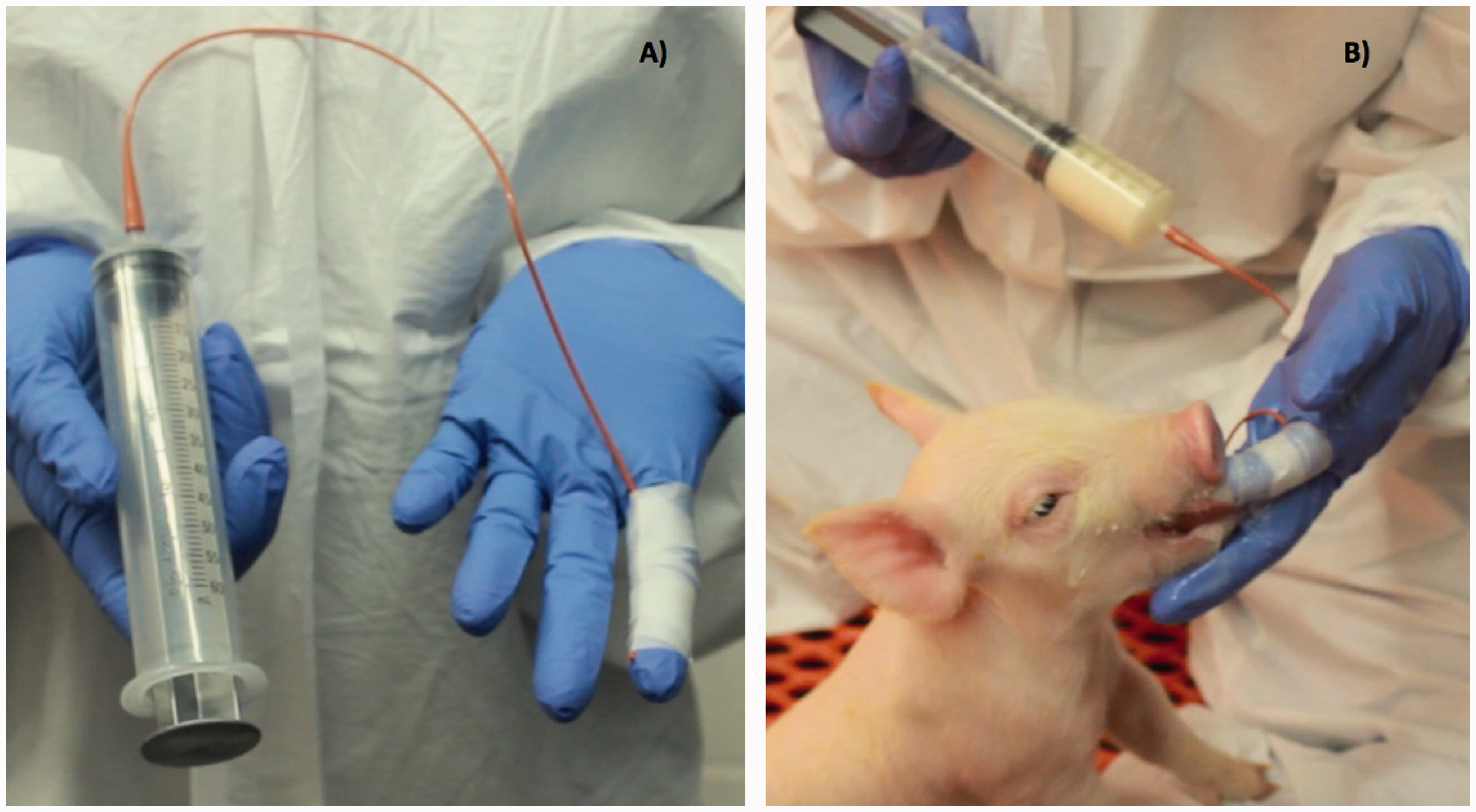
To confirm we were delivering antibody in pasteurized colostrum to the bloodstream before gut closure at approximately 24 hours of age, we collected serum from cord blood (pre-colostrum) and from the jugular, cephalic, or ear vein during the first one to four days of life to determine IgG levels by ELISA (Figure 3(a)). Non-SCIDs and SCIDs that were naturally farrowed (NS_NF and S_NF, respectively) had total IgG levels of 13.06 ± 3.63 mg/ml and 19.05 ± 4.78 mg/ml, while non-SCID and SCID SF piglets (NS_SF and S_SF, respectively) had 7.19 ± 0.51 mg/ml and 11.36 ±4.23 mg/ml (Figure 3(a)). To further compare naturally farrowed versus SF piglets, we examined growth curves for the first 50 days of life from SCID and non-SCID piglets (Figure 3(b)). Although tube-fed piglets weighed less for the first 30 days, their growth curves were consistent with the trend of naturally suckling piglets and weight differences are not noticeable after day 35, when piglets were weaned from dam and milk replacer (shown as vertical line in Figure 3(b)).
ELISA IgG and growth curve data comparing naturally sucking and tube-fed piglets. (a) The box and whisker plot shows serum levels of IgG (mg/ml) from cord blood of SCID (gray) and non-SCID (black) piglets prior to consumption of colostrum compared to SCID and non-SCID samples post-colostrum delivery both from tube-fed and naturally suckling piglets. (NS: non-SCID; S: SCID; NF: naturally farrowed; SF: snatch-farrowed, respectively.) (b) Weight (kg) over time for average of combined SCID and non-SCID piglets that were tube-fed (gray) or naturally suckled (black) is shown. The gray vertical line denotes typical 35-day wean date (from sow or off-milk replacer). Shaded region shows 95% confidence interval. ELISA: enzyme-linked immunosorbent assay; IgG: immunoglobulin G; SCID: severe combined immunodeficiency.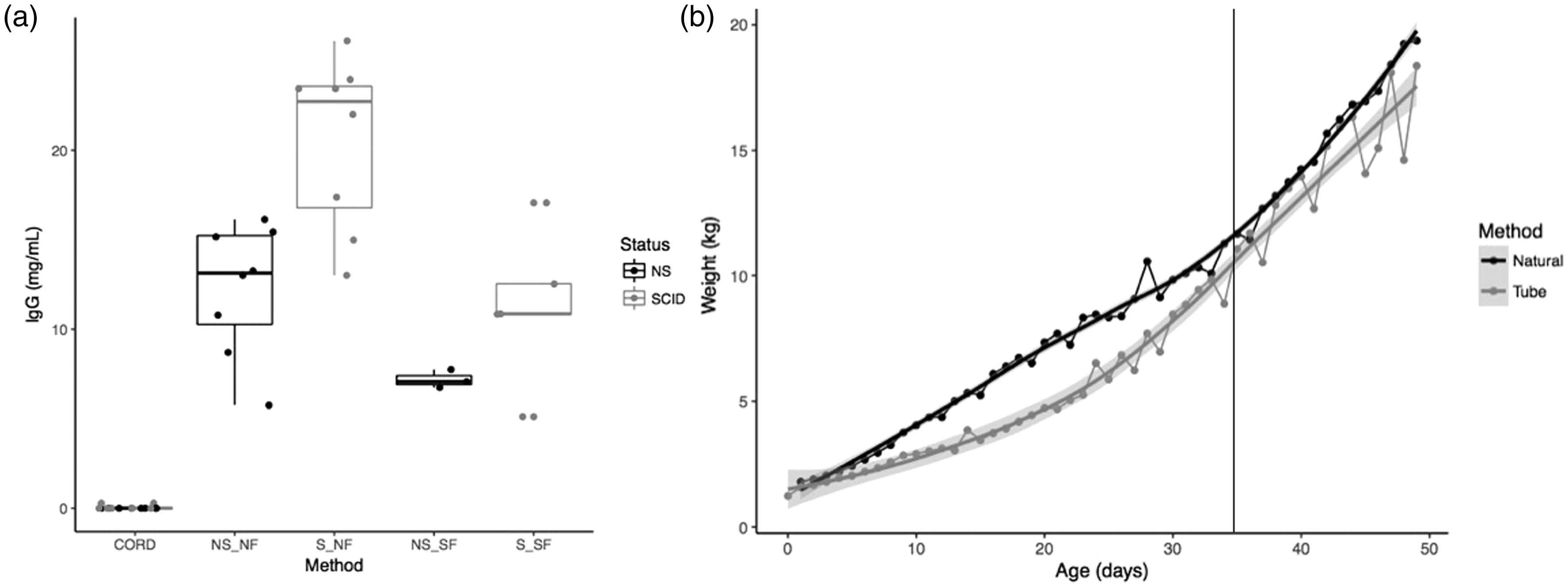
Animal flow
To further reduce the risk of contamination, we established a time- and testing-based quarantine system whereby piglets are tested for SPF status before changing location within the facility. To accomplish this, we utilized two bubbles: the smaller STB that receive SF or cesarean-derived piglets, and the LTB where SPF females can be raised to sexual maturity. The two bubbles are located in adjacent wings of the same building. SF piglets pass through the vaginal canal during farrowing and are thus exposed to maternal microbiota and possible contaminants before entering the STB. They are tested at 1 month and 2 months of age for SPF status (see Materials and methods, Table 2). If either test results in the positive identification of a pathogen in an individual, litter, or pen mate, the pig cannot progress to the LTB. If SPF status is achieved, female piglets are cleared for transfer to the LTB (Figure 4(a)).
Animal transfer through facilities. (a) The figure shows animal flow from farrowing through short- (STB) and long-term bubbles (LTB). Litters are either naturally farrowed where SCID males are BMT rescued and used for subsequent breeding, or snatch farrowed where carrier females are kept SPF clean and moved to the LTB to sexually mature and ultimately be bred to BMT boars to produce naturally farrowed SPF litters in the LTB. (b) HEPA-filtered transfer cart used to move carrier female pigs from the STB into the LTB. SCID: severe combined immunodeficiency; BMT: bone marrow transplantation; SPF: specific pathogen-free; HEPA: high-efficiency particulate air.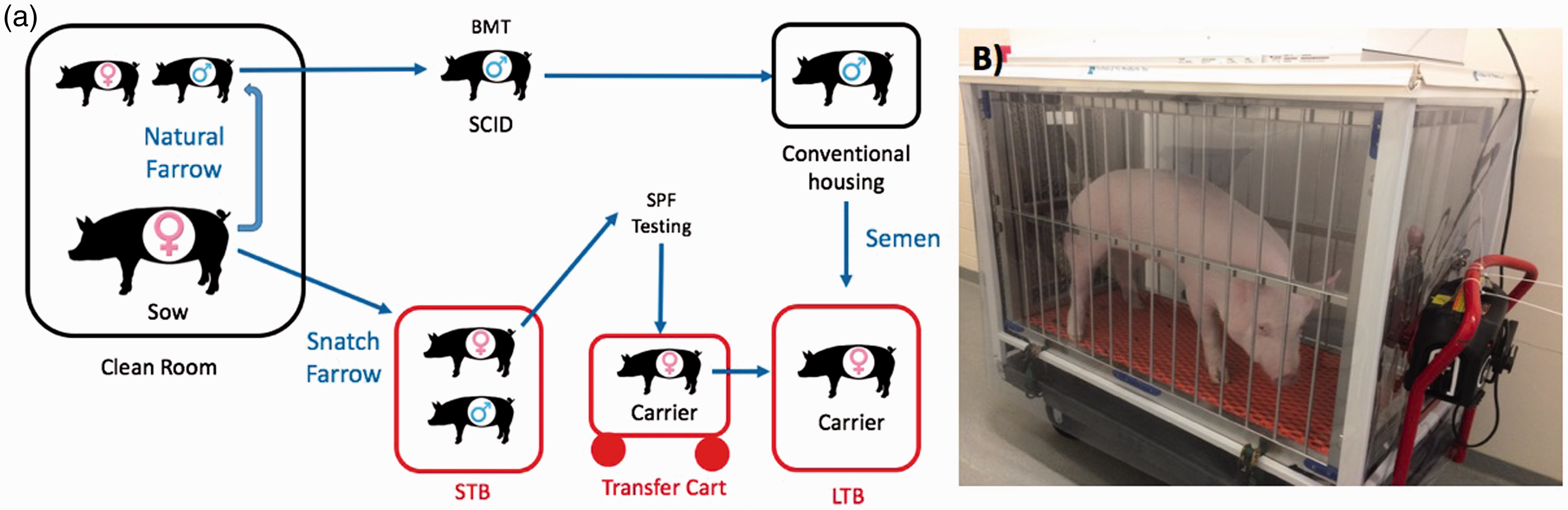
To decrease the exposure to potential contaminants in the environment outside the positive-pressure bubble, we designed and constructed a battery-operated, positive-pressure, HEPA-filtered transfer cart that allows us to wheel a pig from an internal pen in the STB, to the biocontainment transfer cart, and then into an internal pen in the LTB (Figure 4(b)).
Disease prevalence
Routine testing of animals at the LCSRC where carrier dams are originally from established that LCSRC pigs carry and are exposed to several common pathogens (Table 1). Although the ISU SCID pig is susceptible to virtually all bacterial and viral threats, historically the two most common infections documented in our colony are Streptococcus suis and Staphylococcus hyicus, which are common among commercial pig herds as well as present at the LCSRC (Table 1). Although antibiotic treatment is effective in pigs with a normal immune system, we have not observed any SCID piglets to clear the infection, and infected SCID piglets typically fail to thrive and are euthanized for humane care reasons. The development of the bubble and farrowing system has significantly decreased the prevalence and severity of these infections in SF SCIDs. While 71% (5/7 litters) of naturally farrowed litters have presented with diagnosed or suspected incidences of S. hyicus and/or S. suis, we have identified only two individual cases (2/27 or 8% of piglets) of infection in animals SF into the STB, and zero events of infectious disease in our LTB to date.
Additional evidence that high levels of biocontainment are necessary for maximum health was provided by two healthy SCID females that were SF into the STB and maintained SPF status at one and two months. These gilts were later moved out of biocontainment into non-bubble clean rooms, where they were cared for following strict clean PPE guidelines, irradiated feed, and the aforementioned monitoring practices. However, within two months after transfer, both animals had positive confirmation of S. hyicus and one additionally was positive for S. suis.
Discussion
The potential contributions of a large-animal SCID research model to the development of regenerative medicine and cancer therapies, as well as improving our understanding of the mechanisms of immunity, is enormous. The practices described herein focus on the refinement component of the three Rs (replacement, reduction, and refinement) with an emphasis on improving the overall management, health, and welfare of the ISU SCID pig model.
The described SCID pig bubbles create a clean environment where SCID pigs can be raised free of pathogen threat. To complete future biomedical studies utilizing SCID pigs, we have developed these facilities for maintaining an SPF SCID pig colony and concurrently developed methods using SF to produce SPF piglets from non-SPF sows. Our SF protocols and feeding of pasteurized colostrum successfully delivered IgG to neonatal piglets, for which growth follows normal rates seen in naturally suckling piglets. Although some exposure to potential contaminants is possible through contact with the vaginal canal of the sow, our high-sanitation practices are validated by observed reduced disease incidence and severity.
For the long-term SCID pig colony, our goal is to raise females to reproductive age in the LTB, where they can be artificially inseminated with semen from our BMT SCID boars to create SPF litters with 50% SCID and 50% non-SCID piglets for further research. This will decrease labor and the use of resources required by SF and raising piglets in the STB. To date, we have successfully raised four SCID carrier gilts to sexual maturity and farrowed a litter within the LTB. The SF practices and unique facilities described herein allow us the flexibility of introducing new genetics into our herd to control inbreeding, and provide us with methods to readily replace animals if necessary or repopulate the LTB in the event of contamination. Furthermore, the development of protocols that couple SF with biocontainment facilities provides valuable information to collaborators who do not have access to full-scale LTBs, but still require SCID piglets for biomedical research.
Footnotes
Acknowledgments
Such a complex project would not be possible without the help of a huge team of highly dedicated individuals. We wish to thank the entire LAR staff (especially Dale Hinderaker and Eldon Whitaker), the LAR veterinarians (especially Dr Kathleen Mullin, Dr Amanda Ahrens, and Dr Giuseppe Dell’Anna), the Swine Medicine Education Center, the SCID pig team of ISU (especially Jackie Jens, Elizabeth Snella, Adrianne Kaiser-Vry, and Austin Putz), and lastly the Iowa State University Farm staff (especially Gary Kuper).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (grant number 1R24OD019813-1) and the Iowa State University Vice President for Research.
