Abstract
Genetic alterations can unpredictably compromise the wellbeing of animals. Thus, more or less harmful phenotypes might appear in the animals used in research projects even when they are not subjected to experimental treatments. The severity classification of suffering has become an important issue since the implementation of Directive 2010/63/EU on the protection of animals used for scientific purposes. Accordingly, the breeding and maintenance of genetically altered (GA) animals which are likely to develop a harmful phenotype has to be authorized. However, a determination of the degree of severity is rather challenging due to the large variety of phenotypes. Here, the Working Group of Berlin Animal Welfare Officers (WG Berlin AWO) provides field-tested guidelines on severity assessment and classification of GA rodents. With a focus on basic welfare assessment and severity classification we provide a list of symptoms that have been classified as non-harmful, mild, moderate or severe burdens. Corresponding monitoring and refinement strategies as well as specific housing requirements have been compiled and are strongly recommended to improve hitherto applied breeding procedures and conditions. The document serves as a guide to determine the degree of severity for an observed phenotype. The aim is to support scientists, animal care takers, animal welfare bodies and competent authorities with this task, and thereby make an important contribution to a European harmonization of severity assessments for the continually increasing number of GA rodents.
Introduction
Relevance of GA animals in biomedical research
The use of genetically altered (GA) animals in biomedical research is steadily rising. In Berlin, a total number of 242,193 mice, rats and zebrafish were used for animal experimental procedures in 2015. 1 Roughly 40% of these animals had been genetically modified. Even though the majority of GA rodents present no obvious changes in their phenotype, broad-based phenotypic screening of 303 GA mouse lines by the European Mouse Disease Clinic (EUMODIC) consortium determined that about 12% of homozygous mutants have a subviable phenotype. 2 Without appropriate care and attention, these animal models are at an increased risk of experiencing pain, suffering or distress, caused exclusively by their genetic alteration. Similarly, the competent authority in Berlin reported harmful phenotypes for 11% of laboratory animals (mice, rats, zebrafish) included in scientific studies 1 in 2015.
European law has been adapted to current developments in research, and presently addresses the potential burden of GA animals. According to Article 3 of the Directive 2010/63/EU, the ‘creation and maintenance of a genetically modified animal’ that is likely to develop a harmful phenotype is subject to approval. Thus, new protocols have to be adapted within breeding facilities, and all existing as well as newly generated GA lines must be classified into severity categories.
Systematic assessment of potentially harmful phenotypes in Germany
In 2013, the German National Committee for the Protection of Animals used for Scientific Purposes (German Centre for the Protection of Laboratory Animals, Bf3R) organized two expert workshops to define criteria for severity assessment of GA laboratory animals. A set of forms for the assessment of GA rodents at different ages has been developed on the basis of the EU working document on GA animals. 3 According to these recommendations, which were published in 20144,5 and updated in 2016, 6 seven females and seven males shall be assessed over the period that they are kept in a facility.
The suggested procedure for the assessment mainly focuses on clinical symptoms without the need for specialized equipment and can therefore be easily integrated into any husbandry routine. However, if a line-specific phenotype requires a deeper understanding of the progress of a disease or some other feature, or if early humane endpoints need to be defined, additional investigations might be necessary. Following the 3R (replacement, reduction and refinement) principle, the set of parameters should be selected either on the prospective assessment or, when unexpected side-effects emerge, on a case-by-case basis.
No extra animals should be bred for basic welfare assessment. Moreover, animals should not be kept longer than initially intended for breeding and scientific purposes.
For the assessment, genotypes that are likely to develop a harmful phenotype should be selected and observed. For example, when using GA embryonic stem cells to generate a new mouse line, a potentially harmful phenotype is most likely to occur in animals homozygous for the mutation. Although dominant phenotypes are more common than generally expected, 7 in most cases they can only be identified with sophisticated analytical methods. During clinical evaluation, normally conducted in rodent breeding facilities, haploinsufficiency will mostly remain undetected and therefore should be excluded from a standard welfare assessment. However, as soon as additional findings become available for an individual line, a reassessment of the severity degree may become necessary.
For each established line a final assessment form 6 should be in place summarizing the outcome of the assessment. This form should provide information on the phenotype, genotype, degree of severity, and specific refinement strategies, if needed.
The use of standardized and defined terminology such as animal welfare 8 or veterinary 9 terms ensures a comprehensible, objective and consistent assessment and transfer of welfare data.
What is to be considered as a harmful phenotype?
According to Directive 2010/63/EU, a level of pain, suffering, distress or lasting harm equivalent to, or higher than that caused by the introduction of a needle in accordance with good veterinary practice10,11 is considered to be harmful. Translation of this definition to phenotypes of GA rodents is challenging. The German Animal Welfare Act states that pain, suffering and harm have to be considered within an animal experiment without further definition of the specific level of pain, suffering or harm. To draw the line for lasting harm in GA animals to be relevant for animal welfare, the presented guidelines consider ‘harm’ as a permanent condition that compromises an animal's wellbeing according to the intention of Directive 2010/63/EU. However, what can be considered an appropriate way to deal with obvious deviations from wild types that do not negatively compromise an animal's wellbeing under current standard husbandry conditions for laboratory animals? In the UK, such procedures are often described as ‘below threshold’ or ‘subthreshold’, whereas in Germany this category does not exist. Instead, we chose to specify these lines as non-harmful phenotypes to emphasize that no burden is expected. Furthermore, exempting GA lines from basic welfare assessment is recommended,4–6 if there is no reason to expect a harmful phenotype.
According to German law the following lines are classified as having a non-harmful phenotype by prospective severity assessment:
Lines in which the addition/administration of inductors triggers the altered phenotype (no impact on wellbeing before the induction/activation of the genetic alteration). Lines in which the type of genetic modification does not cause a burden (e.g. recombinase expressing or reporter lines). Wild-type lines with and without standardized background or recombinant inbred strains.
If, contrary to these assumed classifications, a clinically detectable phenotype becomes obvious in such a GA line, the animals have to undergo basic welfare assessment in order to define the degree of severity.
Discussions have also been raised about the definition of GA animals and which criteria determine a genetic alteration. International standards of nomenclature define the rules for designation of animals harbouring experimentally-induced mutations. 12 According to this standardized designation, strains with established backgrounds are considered as genetically unaltered (wild type). Nevertheless, it should be mentioned that most inbred strains suffer from inbreeding depression and alterations in specific physiological traits due to homozygosity of unfavourable alleles with recessive inheritance patterns (e.g. blindness in the famous mouse inbred strains C3H, CBA, FVB and SJL). Furthermore, currently no authorization is needed for the breeding of strains developed by selection for a particular phenotype which are mediated by multiple (mostly unknown) genes and are homozygous for their recessive alleles. Prominent examples are NZBW1 (systemic lupus erythematosus), NOD (diabetes) or AKR (leukaemia) mice. It is important to note that refinement efforts have to be undertaken when breeding these animals in order to meet legal requirements.
Approach of Berlin animal welfare officers
The Working Group of Berlin Animal Welfare Officers (WG Berlin AWO) consists of some 40 animal welfare officers (AWOs) active in academic institutions and industries across Berlin and Potsdam. AWOs mainly support scientists and facility managers with their expert knowledge in laboratory animal science, animal welfare and legal requirements. In particular, they oversee the husbandry of laboratory animals and animal experiments and promote a culture of care 13 regarding the continuous implementation of the 3Rs. Since Berlin has a large diversity of research areas, the WG Berlin AWO provides a valuable pool of expertise and experience from various research backgrounds.
Due to the significance of GA rodents in the life sciences and the challenge of implementing the new legal requirements according to the EU directive, a breakout group was founded in 2014 to develop a guidance document on severity assessments and classifications of GA rodents.
Preparation of the guidelines
As a first step, existing severity classifications of GA mouse and rat lines were collected and reviewed. Data had been compiled from the principal research institutions in Berlin which are active in various fields of the life sciences, and employ animal models with genetic alterations on a large scale. In total, data on some 180,000 rodents were collected between 2013 and 2015.
In parallel, pertinent literature and legal requirements were intensively reviewed and compared with practical experience from the research facilities that were involved.
The guidelines presented here summarize the workflow of welfare assessment of GA animals in Berlin, Germany, and provide examples of severity classifications in mice and rats. These recommendations are supposed to facilitate the practical implementation of severity determination and refinement strategies into the process of breeding laboratory rodents for experimental purposes. The guidelines are divided into two main parts. The introductory section addresses the assignment to a severity degree as well as the investigation and assessment of harmful phenotypes. An illustrative list of symptoms provides examples for the severity classifications of phenotypic characteristics of GA lines.
A survey was developed to collect information on the applicability of the guidelines, tested by experts in the field of laboratory animal sciences. Moreover feedback of either a general or a specific nature has been included in the current guidelines.
Results
Survey on the applicability of the guidelines and scope of participants
Experts in the field of animal welfare and laboratory animal science were invited to evaluate the first version of the guidelines (version 1.0) at the international FELASA and EUSAAT congresses 2016 in Brussels and Linz. In addition, personnel were asked by the respective AWOs of Berlin's research institutes to test the guidelines. Hence, an anonymous survey asking about the applicability of the guidelines was developed and sent to the testers. In total, 35 qualified persons participated in the survey. More feedback was received by email or through personal exchanges with colleagues. The survey comprised 17 questions. General information on the testers and institutional backgrounds were collected from questions 1 to 5. Questions 6 to 9 asked about the current management of severity assessment. Questions 10 to 17 referred to the content of the guidelines and asked for feedback after specific testing.
Information on the testers and institutional backgrounds
Twenty-two of the 35 participants in the study were located in Germany, 10 in other EU countries including Austria, Ireland, The Netherlands, Poland, Spain, Sweden and the UK. The three remaining participants responded from Switzerland, Norway and Chile.
Twenty-seven participants worked at universities or other research institutes. Backgrounds of the other participants were: commercial breeders of laboratory animals, competent authorities, 3R centre, FELASA and the pharmacological industry. The majority of the participants were scientists, some of whom were members of animal welfare bodies (AWBs). Most of the other participants were AWOs, and animal facility managers. One was a veterinarian employed in a laboratory animal facility.
The capacity of the institutional animal facilities ranged from fewer than 1000 animals to more than 10,000 animals.
Current management of severity assessment
According to the spectrum of participants, the examination of GA animals was performed with the collaborative effort of scientists, animal care takers and AWOs. Interestingly, only one out of 16 participating facilities in which scientists were responsible for the severity assessment of GA rodents, claimed to use institutional guidelines apart from documents provided by the Bundesinstitut für Risikobewertung (BfR–The Federal Institute for Risk Assessment),4–6 or information derived from the EU directive including EU working documents.3,10 In answer to the question of who finally classified the severity degree of GA animals, various statements were made. The most common answer, accounting for 39% of cases, was AWOs, followed by scientists, AWBs, animal facility managers, and animal care takers.
Feedback on the guidelines (version 1.0)
Thirty-one testers stated that the introductory section of the developed guidelines helped them to understand the concept of severity classification. However, some questions remained open.
In particular these questions concerned the statistical calculation of the number of animals required to perform assessments, as well as setting thresholds for the classification as a harmful phenotype. Both were intensively reviewed for the improvement of the presented guidelines. Furthermore, 23 testers reported that they had implemented or planned to implement parts of the presented guidelines at their institutes, referring particularly to the schemes of legal requirements (see Figure 1) and practical implementation of severity assessment.
Legal requirements for severity assessment. Obligations depend on the likelihood of a harmful phenotype. Even if a genetically altered (GA) line retains a risk of developing a harmful phenotype, authorization is required. This applies to new lines if the phenotype is unknown or if the genetic alteration indicates any impairment of wellbeing. Accordingly, established or imported lines require authorization if a harmful phenotype is known. In case of insufficient information or the unexpected development of harmful symptoms, a welfare assessment has to be conducted.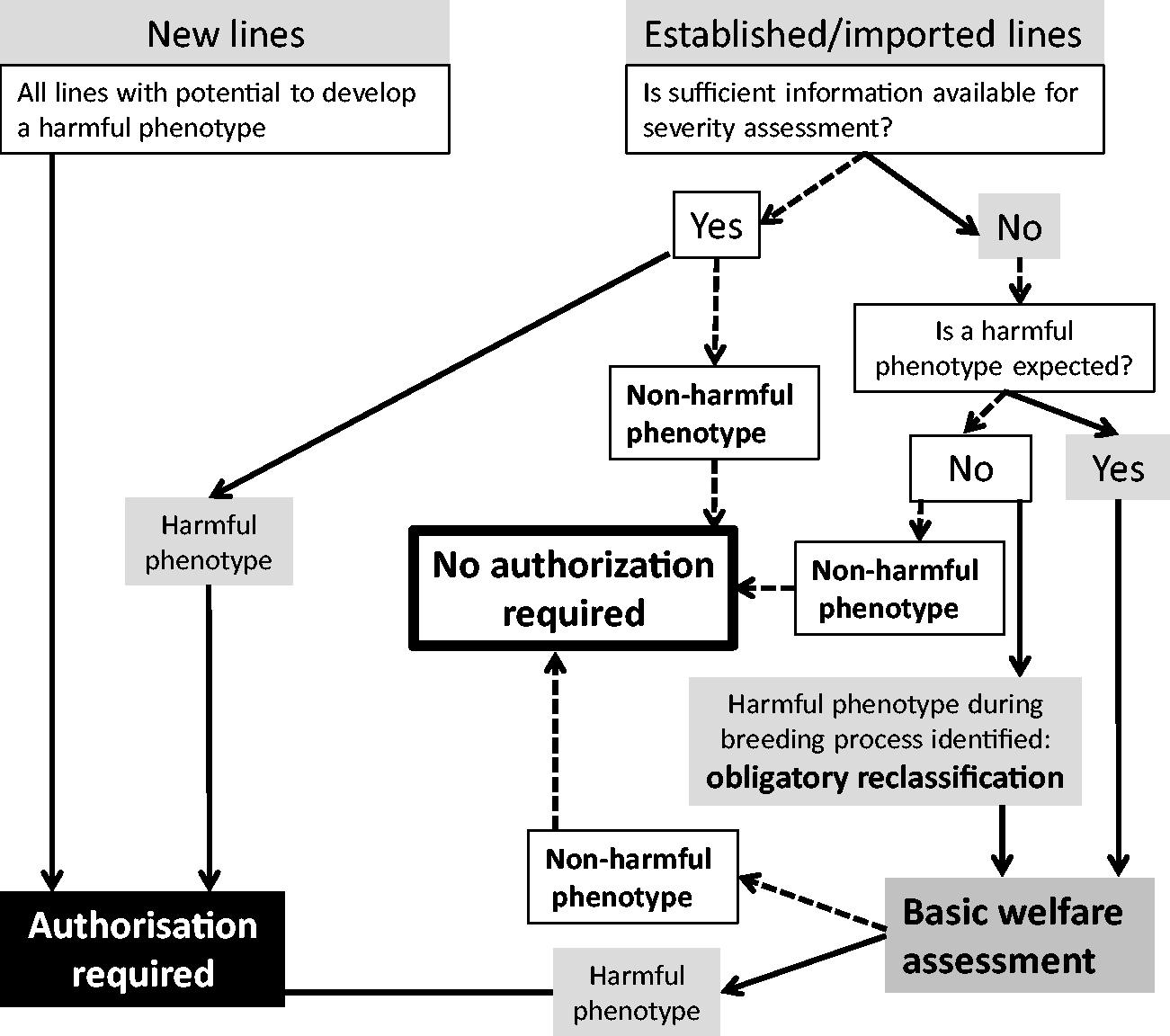
In order to assess applicability of the guidelines, 26 out of 35 participants classified 1 to 10 of their own mouse or rat lines. Forty-nine percent of the testers fully agreed with the resulting classification, whereas 34% only partially agreed. Seventeen percent ended the test without any classification. The main reason for the disagreement was that no applicable symptoms or diseases were included in the guidelines.
Conclusions made from the survey
In summary, the survey served as a valuable review tool for improving the guidelines. The participants represented all areas of animal welfare and laboratory animal science across Europe, therefore covering all stakeholder groups.
Statements on the current management of severity assessment clearly showed that there were substantial imbalances in the practice of severity classification. The elimination of these insecurities was required in order to ensure comparability and harmonization in the field.
All feedback has been intensively reviewed and integrated into the guidelines, supporting the initial idea of an open document that will undergo continuous extensions and improvements. The guidelines are now available as version 1.1.
Guidelines on severity assessment and classification of GA mouse and rat lines
Implementation of legal requirements for severity assessment and classification in Germany
According to current regulations, all facilities have to (I) collect phenotypic information of established lines, (II) assess new lines that are likely to develop a harmful phenotype, and (III) apply for authorization to breed and maintain such lines that show any compromise of wellbeing or have the potential to develop a harmful phenotype. An overview of the legal requirements regarding basic welfare assessment and authorization is shown in Figure 1.
Criteria for assessing severity and selecting a degree of severity
Clinical signs indicative of health or welfare problems may be used to assess severity at the cage level. In addition to observable (behaviour, grimace scale,14,15 gross morphological abnormalities), and measurable (body weight, reproductive performance, life expectancy) parameters, it may be necessary to use analytical methods, e.g. level of stress hormone metabolites in urine or faeces to specify the animals' state of wellbeing and characterize the severity of possible suffering. Any application of stressful or invasive test methods should preferably be avoided for basic welfare assessment unless detailed characterization is necessary in order to establish an effective therapy for affected animal models (refinement). It is especially important to keep in mind that in cases of mild or moderate phenotypes, suffering and distress are difficult to detect in mice and rats, as these species are naturally prey animals and will hide their discomfort for as long as possible. 16
The penetrance, expressivity and duration of the expression of a genetic alteration in the phenotype should be considered, including cumulative phenotypic effects as well as appropriate refinement strategies to relieve symptoms, when defining a total degree of severity. Since the duration of a burden is linked with severity, 11 the early use of animals with progressive harmful phenotypes verifiably mitigates any negative impact on the animals' wellbeing. Time periods such as short-term or long-lasting cannot be defined in a general manner but have to be addressed on a case-by-case basis. Factors such as the intensity and nature of symptoms should also be considered when selecting a severity degree.
The illustrative list in the guidelines consists of clinical signs and line-specific parameters to characterize a stage of disease.
Severity classification in GA animals – illustrative list of symptoms and severity classification
A catalogue of criteria and examples is helpful for selecting a degree of severity for each individual case. Directive 2010/63/EU comprises general definitions and some examples laid out in supplement VIII. In addition EU guidance documents are available.10,17 Nevertheless, examples for classifying genetically induced phenotypes are scarce. In order to fill this gap, the illustrative list provided by our working group is intended to serve as a guide and to offer field-tested examples for severity classifications of phenotypic characteristics of GA lines.
Since the category of severity might be species-specific due to differences in the experience of pain, suffering, distress, and intrinsic behaviour, this list only applies to mice and rats. For example, a blind mouse will experience less distress than a blind non-human primate that mostly uses its visual sense for orientation.
Major categories of systemic and behavioural disorders and diseases.

Examples of the illustrative list.
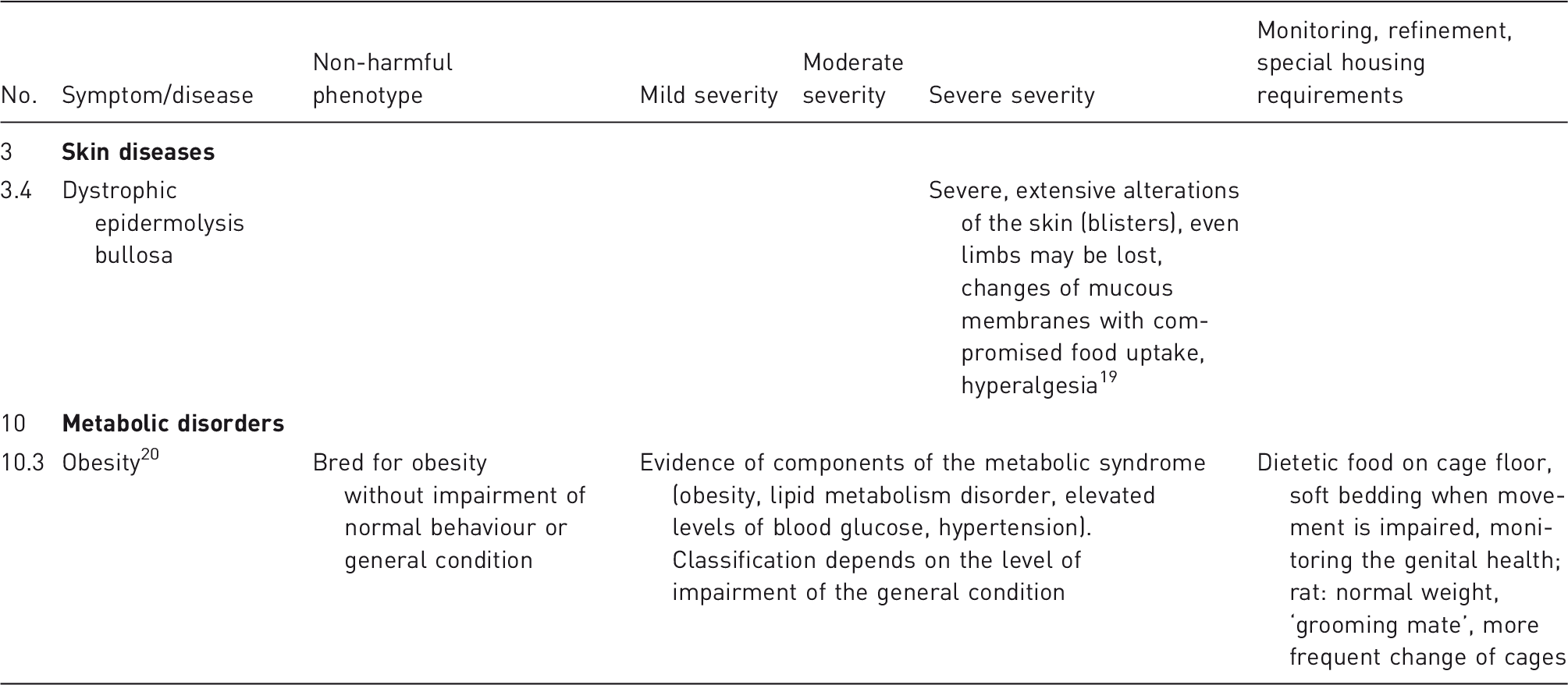
The list is divided into different categories of organ systems and respective diseases or disorders. A symptom, syndrome or disease including examples of each severity category is shown. The list contains references for diseases, line-specific phenotypes as well as recommendations for monitoring, refinement or specific housing conditions. Examples can be used to compare phenotypes and to classify lines.
These classifications are based on a consensus reached by experts in veterinary medicine, laboratory animal science, and animal welfare. Additionally, feedback from the testing period of version 1.0 of the guidelines have been reviewed and implemented. Recommendations of the European Commission's Expert Working Group on severity classification, 17 the British Home Office, 18 as well as selected scientific publications on distinct phenotypes and refinement have also been taken into account.
Supplement on selecting appropriate animal numbers for welfare assessment
An appropriate number of animals should be assessed to identify line-specific welfare issues. In order to characterize phenotypes of new GA lines, the European Mouse Disease Clinic (EUMODIC) uses a cohort sample size of seven females and seven males. The same sample size can also be found in the recommendations by an EU working group 3 and the BfR. 6
However, to our knowledge, this recommended and commonly accepted number of animals to be examined is not based on statistical analysis for detecting a harmful phenotype.
Therefore, taking into consideration the expected frequency of genotypes as well as the presumable penetrance and expression of the corresponding phenotype, we decided to perform a statistical analysis. Fisher's exact conditional test has been used to calculate the number of animals necessary to detect a state of higher severity for two distinct probabilities (Appendix A of the presented guidelines). This shows that an analysis of 10 animals is sufficient to document a state of higher severity with a power of 80%. A higher number of animals might be necessary in exceptional cases (e.g. the low penetrance of a specific phenotype).
Animals of corresponding genetic backgrounds or target strains usually serve as controls. During the establishment of a line, wild-type littermates are particularly suitable if the genetic alteration concerns an undefined genetic background and the generation of a congenic strain is not yet completed.
Discussion and further action
Guidance on severity classification of GA animals in Europe
Directive 2010/63/EU includes examples for assigning severity categories for procedures in Annex VIII, but no specific guidance is available for the severity classification of the phenotypes of GA animals.
Following the FELASA/ECLAM/ESLAV working group on severity assessment, consistency in the assignment of severity categories across Member States is a key requirement. Consequently, a guidance document 19 has been developed. This EU working document on severity classification in scientific procedures performed on animals may be considered as a severity assessment framework. It defines the assignment of pain, suffering, distress and lasting harm into the severity categories of mild, moderate and severe.
A first step towards classifying GA animals has been made by the EU working group on severity assessment. The example-driven guidelines published in 2013 include three examples on how to assess the severity of new and established GA lines. 17
Several guidance documents have been published in the past to classify common scientific procedures, e.g. in Switzerland20,21 and by the WG Berlin AWO. 22
In 2014, the British Home Office issued national guidance documents to help comply with the severity classifications and reporting requirements for GA animals.18,23 Examples and criteria for severity categories are included.
The guidelines of the WG Berlin AWO will contribute to the selection of severity assessment resources by giving more comprehensive guidance on the classification of GA animals.
Guidance and example-driven compilations on severity classification
Catalogues for severity classifications are beneficial for a transparent and consistent approach to selecting an appropriate degree of severity for a procedure, and for the impact of an experimental intervention. Criteria are set to guide less experienced personnel in the determination of an adequate degree of severity and to raise awareness of the impact of the procedure on the animals' wellbeing. In a broad sense, these compilations and criteria enable scientists and competent authorities to fulfil legal requirements on the classification of experimental procedures into severity categories on a reliable basis. 16
Critics of listing examples of degrees of severity argue that there is a risk of using the guidance document in a rigid and unquestioning manner. In this case, local conditions might be neglected and severity might be judged according to a fixed system instead of discussing the conditions on a case-by-case basis. Schilling 17 has commented that well educated persons in laboratory animal science, e.g. AWOs, provide better advice than a catalogue does. Consequently, the assessment is more flexible and adjusted to the individual conditions.
Taking these concerns into account, the guidelines of the WG Berlin AWO on severity assessment of GA animals was compiled to provide examples of symptoms and syndromes. It will serve as a dynamic document that will be constantly reviewed by the authors. Annual updates are planned to add more examples, to consider recent scientific progress, and to integrate feedback on their practicality from experts from European countries who actively use the suggested guidelines. For this purpose, the updated version will be permanently published in English and German on the WG Berlin AWO web page. 24 The guidelines should be used as a source of help for anyone involved in the assessment of GA rodents in a consistent manner, 17 but are not intended to replace professional judgement of any individual case. 13
Team approach and communication
Efficient communication strategies should ensure the exchange of information so that welfare implications can be considered immediately. At the institutional level, information has to be shared among animal care staff, scientists and the responsible veterinarian and/or animal welfare body. Especially in light of the restrictions on access to animal facilities, a format has to be established to guarantee a quick and efficient dissemination and translation of available data. The severity assessment will help to collect data with standardized procedures and criteria.
Creation of a passport, communication and databases
A so-called animal passport could serve for the collection of all important data. This information should accompany the animals, particularly when lines are exchanged between different facilities. The transfer of important information regarding scientific and welfare characteristics is needed to ensure the quality of scientific research, and to comply with the demand for providing appropriate care to animals with impaired welfare. A documentation system should be established to record the results of welfare assessments within each facility. 4 The passport should provide details on the genetic modification as well as information on severity assessments and husbandry. The accurate dissemination of data will enhance science and animal welfare. 25
Furthermore, funding opportunities are needed to establish and manage appropriate databases in order to make data on welfare assessments readily available which can support severity classification of harmful phenotypes.
Conclusion
A systematic approach in assessing phenotypical changes in GA animals and classification of severity promotes transparency and consistency. A shared sense for classifications of pain, suffering, distress and lasting harm into categories should be developed. Severity assessments also form the basis of ethical considerations when designing, reviewing and conducting studies on experimental animals. 26 Exchanging line-specific information will help to comply with the 3Rs and ensures the quality of both science and animal welfare.
This collection of severity classifications is a starting point for addressing severity assessments and classifications in GA mice and rats. AWBs and researchers throughout Europe are encouraged to contribute information on phenotypes which will be incorporated into the collection. The guidelines will be reviewed periodically in order to provide a large collection of different syndromes and clinical symptoms that are relevant to animal models used in biomedical research. The list will serve as a guide in the complex process of determining degrees of severity, and as a way of further harmonizing the approaches of diverse research institutions in the European Union.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
