Abstract
Surgical transfer of embryos is carried out daily in animal facilities worldwide for the rederivation of mouse strains/lines, among other purposes. Current protocols described in laboratory manuals recommend using a high number of embryos during transfer, typically in the range of 15 up to 25. To optimize the use of resources it is necessary to estimate and relate the effort required and the yield obtained. Here, we analyse the balance between the number of embryos transferred (the effort), and the yield as the number of born pups obtained from surgical embryo transfer. To accomplish this, we have analyzed data obtained during rederivation of nearly one hundred lines of mice to a new animal facility. Our results confirm that the use of increasing numbers of embryos per transfer increases the yields of born pups, as has been described previously in the literature, but they also highlight the disproportionate effort required, i.e. in the number of embryos that needed to be transferred. An estimate of the mean expected yields of surgical transfers and their comparison with the actual observed yields indicated that the balance between effort and yield is optimized when using lower numbers of embryos than in currently used protocols, in the range of 8 to 12. Given the heterogeneous nature of the data presented and analyzed here, which is from a population of mice that may be considered as representative of any animal facility, our optimization approach should help save resources in similar facilities and improve the yields of embryo transfer procedures.
Surgical transfer of mouse embryos to the oviduct or uterine horn of a pseudopregnant mouse is a commonly used technique that has proven to be useful in a number of applications in animal facilities worldwide, such as the production of transgenic mice and of specific pathogen-free (SPF) animals by rederivation.1–3 With this technique a variable number of embryos from a donor strain/line are typically transferred to a pseudopregnant recipient female housed under SPF conditions. As a result, the number of embryos produced and handled for embryo transfer in a conventional facility may be very large. Apart from the number of animals sacrificed as embryo donors, the technique requires a considerable investment in time and economic resources, as well as diverse technical skills. Good examples of these are superovulation through injection of gonadotrophins in donor females to improve embryo production, maintenance of the vasectomized male stock that will be used to induce pseudopregnancy of foster mothers, or the need to ensure proper synchronization between the estrous cycles of the recipient females and their donor counterparts.4–6 Fine-tuning of all these processes is crucial to optimize resource investment in day-to-day embryo transfer procedures.
One of the factors that is pertinent to the efficient production of live offspring in an animal facility through routine embryo transfer is the number of embryos that need to be transferred in these procedures. It was shown previously that the yield of a transfer, i.e. the number of born pups, increases proportionally with the number of fertilized eggs 7 or embryos transferred. 8 With the intention of achieving the maximal uterine capacity of a foster mother it is usual to have large numbers of embryos during a transfer, typically in the range of 15 up to 25.5,8–12 However, the use of such a high number of embryos may increase the effort, considered here as the number of embryos invested per transfer to obtain a single pup. In this study we have revisited this issue with the aim of fine-tuning and improving the number of embryos that need to be transferred by conventional unilateral surgery, using the experience gathered in our facility. With this goal in mind we have analyzed data accumulated during the rederivation of 93 lines of mice to a new facility during a period of 10 months, in the years 2013–2014. Analysis of data from transfers that used between 5 and 25 embryos per transfer focused on finding the optimal balance between the effort provided and the yield obtained. Our analyses show that this balance was optimal using lower numbers of embryos than those that are currently used. Data presented and analyzed here is from the heterogeneous population of mice housed in our facility, which may be considered as representative of any other animal facility. Therefore, we believe our approach and findings can help to improve the effectiveness of surgical embryo transfer procedures in similar facilities.
Materials and methods
Animals
The Centro de Investigaciones Biológicas (CIB) animal facility is a state-of-the-art center (http://www.cib.csic.es/facilities/scientific-facilities/animalarium). Humane care and use of mice in the rederivation program in this facility were approved by the relevant local ethics committees for animal experimentation, and procedures were carried out in accordance with European Union guidelines and Spanish legislation. The set of donor lines to be rederived consisted of a diversity of genetically-modified mice from various sources, generated on a C57BL/6 genetic background. Lines are owned by the different CIB scientists who use the facility. All lines have the C57BL/6 genetic background in common, and they differ only in the transgenic construction they carry. SPF Crl:CD-1 (ICR) mice, first purchased from Charles River, Saint-Germain-Nuelles, France and then generated in-house, were used as either recipient females or vasectomized males. The microbiological status was evaluated for the list of agents recommended by the Federation of European Laboratory Animal Science Associations (FELASA), 13 and included in the mouse FELASA complete PCR rodent infectious agent (PRIA) panel in CRL Diagnostic Services (Wilmington, MA, USA) on a sample from the purchased males and females. Subsequently a standard program based on sentinel screening, two mice housed per cage, one cage per single rack, was performed quarterly (FELASA basic PRIA panel) and yearly (FELASA complete PRIA panel), respectively. From the beginning to the end of the rederivation program all sampled animals, whether purchased or sentinels, tested negative. A diurnal rhythm was maintained with a 12/12 h light–dark cycle and with artificial light from 07:00 h. Animals were housed at 21–22℃ with a relative humidity of 50 ± 10%. Mice were fed with a standard autoclavable mouse diet supplied ad libitum (D40, SAFE, Augy, France) and with sterilized tap water. Vasectomized and breeding males were individually caged. Females were housed in type 3 cages (Tecniplast, Buguggiate, Italy) in groups of 8–10 animals from weaning until they were coupled with males. Cages were provided with sterilized Lignocel Select Fine (Rettenmaier Iberica, Barcelona, Spain) bedding material and environmental enrichment, according to standard welfare recommendations.
Embryo transfer
Collection of embryos and their surgical transfer were performed according to standard procedures. 10 Superovulation was induced in 4–5-week-old donor females by an intraperitoneal injection of 5 IU of pregnant mare serum gonadotropin (PMSG) (Sigma-Aldrich, St Louis, MO, USA), followed 47 h later by 5 IU of human chorionic gonadotropin (hCG) (Sigma-Aldrich). Female mice were immediately singly-mated with the breeding males. The presence of a vaginal plug was determined the following morning before 10:00 h. Females with a vaginal plug were sacrificed 1.5 days post-coitum; their oviducts were isolated and the embryos were obtained by flushing the oviducts with 100 µL of phosphate-buffered saline (PBS) and 0.5% bovine serum albumin (BSA) buffer kept at 37℃. The morphological quality of the two-cell stage embryos to be transferred was routinely assessed by visual inspection under binocular lens (Leica M80; Leica, Wetzlar, Germany) with a magnification of 60×, based on the experience of qualified technical staff from the animal facility. After 10 washes, embryos were surgically transferred to pseudopregnant females. For this purpose, 6–8-week-old nulliparous CD-1 recipients mated the previous night with vasectomized males were used. Transfers were planned according to the availability of embryos. Embryos (from a minimum number of 5 and a maximum of 25 per transfer) were transferred unilaterally to the left oviduct through the infundibulum. Surgical procedures were performed specifically by two highly-skilled technicians from the animal facility team. Both of them were well-trained and achieved comparable results performing embryo transfer. Surgery was performed after induction of anaesthesia with 5% isoflurane (Abbvie Farmacética SL, Madrid, Spain) in oxygen and then 2% for maintenance. The mouse eyes were protected with an ophthalmic lubricating ointment (Lipolac; Angelini Pharmaceutical, Barcelona, Spain) to prevent corneal drying and damage during surgery. Each surgery typically lasted 10–15 min. Before recovery from anaesthesia the animals were treated with 100 µL buprenorphine (0.1 mg/kg) (Buprex; Reckitt Benckiser Pharmaceuticals Ltd, Berkshire, UK) by intraperitoneal injection, and then transferred to a peaceful environment for 30 min. Housing in standard conditions was subsequently maintained until offspring birth.
Statistical analyses
All the analyses were performed by grouping the data into five categories, according to the number of embryos employed for each embryo transfer (1st: 5–8 embryos; 2nd: 9–12; 3rd: 13–16; 4th: 17–20; 5th: 20–25). Grouping the data increased sample size within categories and corrected for potential bias due to data heterogeneity. Pregnancy rates among categories were compared by the chi-square test of association. Subsequent analyses of birth rates, and effort–yield comparisons were carried out after removing the incidence of cannibalism and births by Caesarean section from the pool of data. Comparison of yield (number of pups born per single transfer) and effort (number of embryos invested per transfer to obtain each single pup) among categories was performed using both analysis of variance (ANOVA) and the non-parametric Kruskal–Wallis tests. Although raw data within two out of the five categories did not meet the criteria for normality and homoscedasticity, similar results were essentially obtained, independently of the test used. Post hoc pairwise by the least significant difference (LSD) method was used to compare yield and effort from each category with the remaining categories. In addition, Pearson correlation coefficient was used to measure the linear dependence between effort and yield. To estimate expected yields the ratio between the mean value of embryos used for a transfer in each category and that of the 1st category was first obtained. Then, the mean expected yield was estimated as the product of multiplying this ratio by the mean observed yield in the 1st category. Comparison of the observed yield values with the estimated expected value for each category was performed using the Student’s t-test against the null hypothesis of equal means. Statistical analyses were performed using Stata v14.0 software (StataCorp, College Station, TX, USA).
Results
Comparison of pregnancy rates in the five categories in which transfers were grouped, according to the number of embryos used.
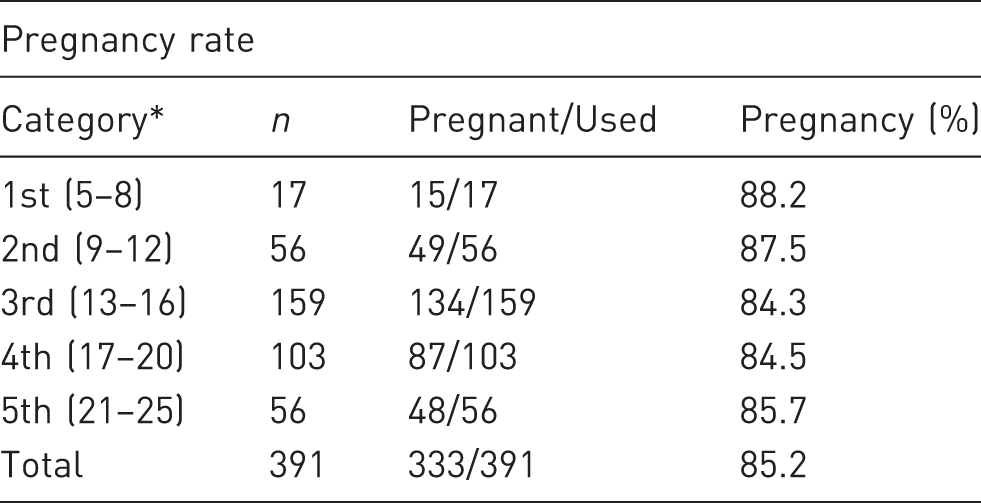
Numbers of embryos transferred per transfer were grouped into five categories. Numbers within parentheses are the numbers of embryos transferred in each category.
Birth rates in the five categories in which transfers were grouped, according to the number of embryos used.
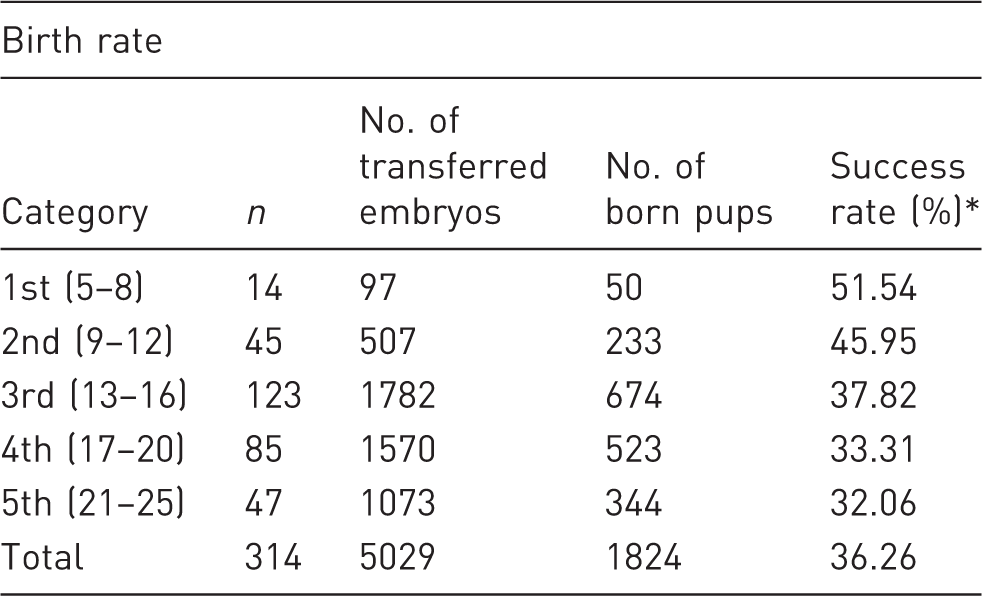
Here success rate is expressed as the ratio between the absolute number of born pups and the absolute number of transferred embryos within the category.
Comparison of observed effort, and observed and expected yield, in the five categories in which transfers were grouped, according to the number of embryos used.
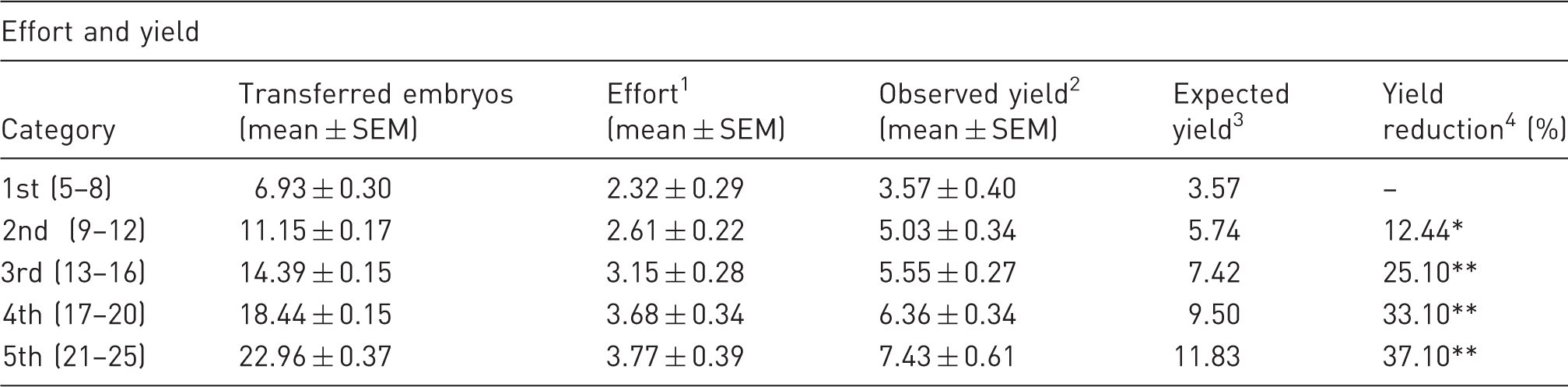
Average number of embryos invested per transfer to obtain a single pup (number of embryos transferred/number of pups obtained). 2Average number of pups obtained per transfer. 3An estimate of the average number of pups that would be expected to be obtained for each category, taking into account the mean number of transferred embryos and the yield observed in the 1st category: the ratio between the mean number of transferred embryos used in each category and that of the 1st category was first obtained. Then, this ratio was multiplied by the mean observed yield in the 1st category. 4The proportion that the observed yield decreases with regard to the estimated expected yield *t = −2.13; P ≤ 0.035 **t ≤ −6.46; P ≤ 10−3.
Discussion
Our analyses clearly show that increasing the number of embryos used for a transfer did not just result in an increase in the yield of live born pups, as has already been shown, 8 but also entailed a disproportionately larger increase in the effort made, as seen by the number of embryos invested to obtain a single pup (see the percentage of observed versus expected yield reduction in last row of Table 3). It is interesting to note that a similar result to this was observed for unilateral transfers performed with high numbers of embryos (15 and 25) when mean yield data in Table 2 from the paper by Johnson et al. 8 were used to estimate expected yields. Since the experiments were performed using the more productive ICR strain, both as a donor and receptor of embryos, a general conclusion seems to be that for resource optimization the use of lower numbers of embryos than those commonly used is recommended in unilateral surgical transfer. The idea that surgical transfer should be performed using a high number of embryos originates from works published a couple of decades ago, when the technique was routinely performed with up to 25 unmanipulated embryos. 9 Even though more recent protocols advise the use of smaller numbers of embryos per transfer, approximately15, 10 the idea of using high embryo numbers is still widespread. 5 The results presented here demonstrate that a reduction in the number of embryos used for unilateral embryo transfers improves resource investment.
Current protocols suggest that more than 50–75% of unmanipulated embryos should develop into normal fetuses when transferred into the oviducts,9–10 although this claim seems to apply only to transfers performed with small numbers of fertilized eggs/embryos (see7,8), and to do so unilaterally. Our success rates were lower than the described range limits. Differences in success rates may be related, among other factors, to the genetic background of the strains employed as donor and receptor and to their compatibility interactions. In our case the donor lines were a heterogeneous set of transgenic lines engineered in a C57BL/6 genetic background, while the recipient females were of the CD-1 strain. To rule out the possibility that the births rates of some transgenic lines when they are rederived could be lower than those in natural births, we compared litter sizes of natural births with those obtained after rederivation in a subset of 24 of the transgenic lines used in this study, and found no correlation (data not shown). In our facility, natural births from wild-type C57BL/6J give on average litters of 7.16 ± 1.28 (mean ± SD) pups against 13.16 ± 2.13 (mean ± SD) in the case of the CD-1 strain. Taking into account a 75% success rate, a transfer of 15 embryos should render a number of pups that is still far below the uterine capacity of foster CD-1 females. Furthermore, transferring 25 embryos, the maximal number transferred in this study, should at a theoretical success rate of 75%, render a number of pups that is close to the maximal uterine capacity of one of the naturally pregnant CD-1 females seen in our colony, 18–20 pups per single litter. However, despite some litters numbering up to 12 pups for the 4th and 5th categories of transferred embryos, the average yield of transfers was far below the theoretically expected value. Taking into account that no transuterine migration of embryos has been demonstrated for unilateral transfer in mice, 12 it is likely that by performing surgical transfer bilaterally yields closer to the maximum uterine capacity of CD-1 fosters may be achieved.
Although a more economical and non-invasive method, non-surgical embryo transfer (NSET), has been described in the mouse,14,15 conventional surgery remains the option of choice for performing embryo transfer. The low apparent success rate of NSET, as compared with conventional surgery, may dissuade researchers from using this method 16 (but see also 17 ). In addition, despite the usefulness of NSET for the transcervical transfer of blastocysts, embryos at earlier stages of development may only be transferred to oviducts by conventional surgery. Development of pre-implanted embryos from the two-cell stage to the blastocyst stage entails a marked reduction in the number of embryos obtained from the donor female. Therefore, even at equal efficiency, NSET still involves the use of a higher initial number of embryo donors than does surgical transfer.
In conclusion, our results show that the pregnancy rate is not dependent on the number of embryos transferred, while the success rate is indicating that the yield maximizes when using a low number of embryos per unilateral transfer, and the ideal number of embryos to transfer is somewhere between the 1st (5–8) and the 2nd (9–12) categories. Reducing the number of embryos used per transfer should reduce the number of females used as embryo donors. By performing the transfer with 10 embryos we reduced the number of donors by an estimated 1.5–2.5 times. Reduction of embryos transferred may also be useful when strains undergoing rederivation produce low numbers of embryos, or in cases where the available number of donors is scarce. A 1.5–2.5 time reduction of the number of donors together with the repeated use of the surrogate mothers as described by Kolbe et al. 18 should reduce the number of animals needed for surgical transfer procedures, thus fulfilling the 3R principle (replacement, reduction and refinement). As the set of transgenic lines presented here may be considered as representative of a mouse population kept in a standard animal facility dedicated to biomedical research, our approach may be extended to similar facilities.
Footnotes
Acknowledgements
The authors wish to thank Pablo Díaz, Francisco Casado, Carlos Escobar, Esther Sánchez, Olivia Barcón, María Herrera and Andrés Rodríguez for technical assistance. We would also like to thank Dr Jesús del Mazo for his research support, and Dr Tomás Canto for critical reading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was sponsored with core funding derived from the user fees of the CIB animal facility.
