Abstract
The temporomandibular joint (TMJ) is a condylar synovial joint that, together with the masticatory muscles, controls mandibular movement during mastication. The rabbit is often used as a model species for studying the mechanisms of TMJ diseases, and in regenerative research. However, there are significant differences between rabbit and human TMJs that should be taken into account before using this model for experimental research. Here, we use several analytical approaches (radiography, computed tomography and magnetic resonance imaging) to enable a detailed description and analysis of the rabbit TMJ morphology. Moreover, possible surgical approaches have been introduced with a focus on available access into the rabbit TMJ cavity, which relate our findings to clinical usage.
The temporomandibular joint (TMJ, articulatio temporomandibularis) is a multi-component structure that, together with the masticatory muscles, controls mandibular movement during mastication. It consists of the condylar process of the mandibular ramus (ramus mandibulae), mandibular fossa of the temporal bone (fossa mandibularis ossis temporalis), a thin articular disc (discus articularis), and a loose joint capsule reinforced with fibrous lateral ligaments. 1 The structure and function of this incongruent, condylar synovial joint (hinge sliding joint) are unique among the diarthrodial joints. 2
There are many diseases and traumas affecting the TMJ. While many studies have provided detailed information about the development, anatomy and aetiopathogenesis of TMJ diseases, there is a lack of comprehensive understanding of the mechanism initiating these diseases. Many of the symptoms are difficult or impossible to treat using current methods, highlighting the need for extensive research in this field. The use of animal models for such research is indispensable. 3 Due to physiological and anatomical differences between the human TMJ and those of experimental animals, no one animal model can provide a full understanding that could be directly related to humans. 4 In addition, many other variables, such as gender, age, size and depth/age of the defect, postoperative treatment, etc., need to be considered when designing experiments. Therefore, an investigator faces the challenging task of choosing the most suitable model and treatment protocol in order to answer a specific question. Discrepancies in the results of clinical studies focused on treatment modalities for TMJ dysfunction make it obvious that animal models are needed to define and control experimental variables. 5 Although no one animal model can exactly duplicate the human condition, a number of authors have argued that, despite its prognathism, the pig, Sus scrofa, is the best non-primate model for modelling the human TMJ form and function.6–9Although large animal models such as the pig may resemble humans more closely than smaller animals, they are usually neither practical nor economically feasible for conducting initial experiments in large animals. 10
One of the smaller animal models frequently utilized in research is the rabbit. Rabbits are easy to handle and inexpensive to maintain in cages, making them convenient animals for use in experimental studies. Rabbits are often used in studies to test dental implants,11–14 to test bone grafts following the extraction of cheek teeth, 15 and to investigate the influence of biomaterials on the bone healing process around the tooth sockets.16,17 Studies on the effect of tooth loss on the histochemical composition of TMJ cartilage and disc 18 and on the histology of the condyle 19 have been performed. Surgical alterations of the rabbit TMJ for investigation of discectomy effects20,21 and implantation of disc replacements have also been reported. 22 However, although the rabbit has been routinely used as an experimental animal model, very little information is available about the anatomy of its TMJ, and its coverage in veterinary textbooks is even more cursory. 23 The best description of the rabbit TMJ 24 is unfortunately not easily accessible to English-speaking researchers; and other descriptions of rabbit TMJs are contradictory, particularly with respect to their nomenclature. 25
Considering the lack of complete information about the anatomical and histopathological characteristics of the rabbit TMJ, the purpose of this investigation was to (1) provide an overall description of a rabbit TMJ with an emphasis on gross anatomy and its comparison to a human TMJ; (2) describe the appearance of the anatomical structures on radiographs, computed tomography (CT) and magnetic resonance imaging (MRI) scans, and discuss the advantages and disadvantages of individual techniques for TMJ studies; and (3) relate our findings to clinical usage and describe achievable surgical approaches into a rabbit TMJ.
Materials and methods
Animals
Twelve clinically normal eight-month-old outbred male New Zealand white specific pathogen-free rabbits (Oryctolagus cuniculus, strain Hsdlf:NZW; Harlan Laboratories Inc, Belton, UK) were used in this study. The weight of the animals ranged from 2.92 to 3.15 kg (mean 3.03 ± 0.2 kg). The animals were fully developed and were free of any pathological processes. All the rabbits were housed individually in an animal care facility under controlled conditions, and were handled and euthanized according to the agreement of the Branch Commission for Animal Welfare of the Ministry of Agriculture of the Czech Republic (project PP52-2013 UVPS).
Diagnostic imaging
The rabbit heads were radiographed using a Gierth X-ray machine (Gierth HF 200A; Gierth X-Ray International GmbH, Riesa, Germany). Radiographs were captured on computed radiography cassettes. The focal length was set to 100 cm and the exposure values were 50 kV, 200 mA, and 150 ms (30 mAs). The images were stored in a digital imaging and communications in medicine (DICOM) format using a computed radiography system (FCR Capsula XL; Fuji, Tokyo, Japan). The image resolution was 1760 × 2140 pixels.
Native transverse CT scans of each rabbit’s head were obtained using a multidetector CT scanner (LightSpeed 16; GE Medical Systems, Milwaukee, WI, USA). The scanning protocol was 80 kV, with an automatic mA setting, a tube rotation time of one second, a slice thickness of 1.25 mm, a spiral pitch factor of 0.9 and a high frequency convolution kernel (proprietary name: bone). All the scans were obtained using the helical mode. Radiographic and CT analyses were performed at the Department of Diagnostic Imaging, Small Animals Clinic, Faculty of Veterinary Medicine (Brno, Czech Republic).
MRI analysis (Bruker Avance 9.4T, Bruker BioSpec; Bruker, Ettlingen, Germany) of the rabbit heads was conducted at the Institute of Scientific Instruments of the Czech Academy of Sciences (Brno, Czech Republic). Images were acquired in sagittal and transverse planes with fast spin echo (FSE) sequences. The heads were positioned in ventral recumbency during the scanning procedure. FSE T1-weighted transverse MR images were obtained with the following parameters: FLASH, AVG = 6, TE = 3.7 ms, TR = 369 ms, matrices = 512 × 512. Acquisition time 14.5 min and RARE, AVG = 6, TE = 25 ms, TR = 3500 ms, matrices = 512 × 256, RARE factor = 8.
Dissection procedure
Palpable anatomical landmarks were identified prior to dissection using a rabbit skull model as a reference. Anatomical dissections of the TMJ were performed using a stratigraphic approach, and each dissected layer was photographed using a Nikon D 5100 camera (Nikon Europe BV, Amsterdam, The Netherlands). The key anatomical structures were identified and indicated on the photographs.
Histological evaluation
The harvested mandibular condyles (condyles mandibulares), including the articular cartilage, were fixed for three days in 10% buffered formaldehyde and decalcified in 12.5% EDTA solution (pH = 8), which was prepared as a mixture of 0.5 mmol/L EDTA, 4% buffered formaldehyde and PBS buffer (pH = 7,4). The tissue samples were left in this decalcification solution in an incubator at a temperature of 37℃ for 11 months. Following the decalcification, the samples were processed for histological analysis using a standard procedure – they were washed in water (overnight) and dehydrated in an increasing alcohol series (30% for 3 h; 50% for 3 h; 70% overnight; 80% for 2 h; 95% for 2 h; 100% for 1 h). Then, the samples were soaked in xylene for 5 h, run through three paraffin baths (for 1 h in each of them) and embedded in paraffin blocks.
Five micrometre paraffin sections were stained with haematoxylin and eosin (H&E), Alcian blue (AB) and Masson’s trichrome. 26 The staining protocols used in this study were modified in the following steps: after staining in Mayer’s haematoxylin, staining in acid–alcohol was not differentiated but was left in warm water for 5 min. In the case of the AB staining, H&E was used for a counterstaining. In the case of the Masson’s trichrome, Weigert’s haematoxylin (Diapath SpA, Martinengo BG, Italy) was used, then the sections were differentiated in acid–alcohol, stained with Fuchsin Ponceau acc. Masson (Diapath SpA), differentiated in 1% phosphotungstic acid and stained with a light green (Diapath SpA).
Morphometric assessment
Morphometric assessment was carried out on the dissected and macerated skulls (Figure 1). TMJ width (TMJW) was determined by measuring the distance between the medial aspect of the zygomatic process of the temporal bone (processus zygomaticus ossis temporalis) and the squama of the temporal bone. TMJ length (TMJL) was measured as the distance between the rostral aspect of the zygomatic process of the temporal bone and the most caudal projection of the zygomatic arch (arcus zygomaticus). Condylar width (CW) was measured at its widest point.
Mandible of the rabbit. (a) Lateral view of the rabbit mandible. (b) Medial view of the rabbit mandible. (c) Detail of lateral aspect of the mandibular caput (arrow) (d) Detail of dorsal aspect of the mandibular caput. (e) Detail of medial aspect of the mandibular caput. (f, g) Radiographs of rabbit skull. 1: cranial aspect of the ramus mandibulae, 2: processus zygomaticus ossis temporalis, 3: processus condylaris mandibulae, 4: meatus acusticus externus, 5: bulla tympani, 6: caudal aspect of the ramus mandibulae, 7: processus angularis, 8: ramus mandibulae, 9: corpus mandibualae, 10: arcus zygomaticus, 11: spatium articulare of the temporomandibular joint (TMJ), 12: condylus mandibulae, 13: processus caudalis arcus zygomatici, 14: os temporale, 15: os prespheonidale, w: TMJ width (TMJW).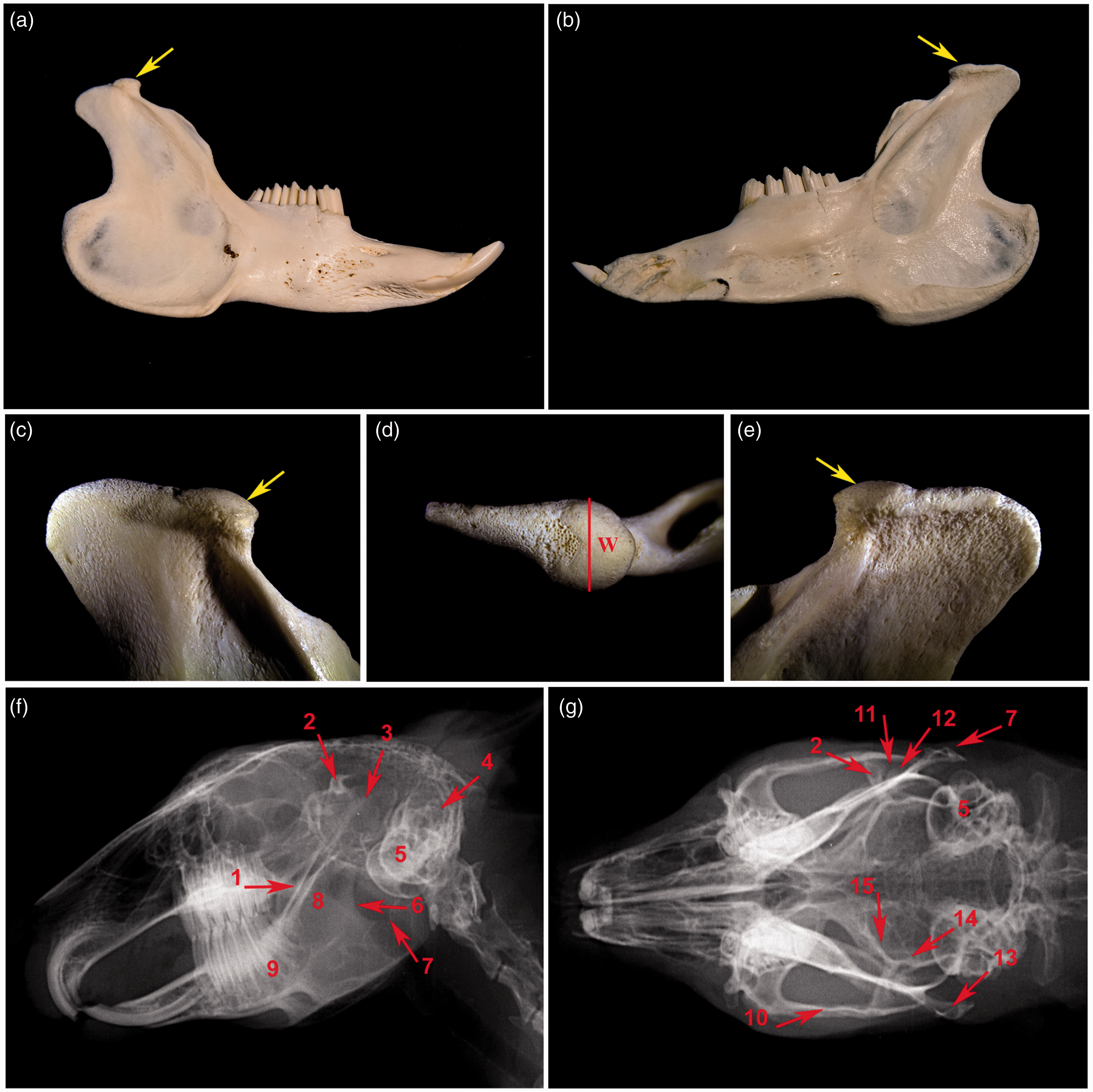
Furthermore, a surface projection of the TMJ on the rabbit skull was established by using an axis of the zygomatic arch and the line connecting the caudal aspect of the zygomatic process of the frontal bone (processus zygomaticus ossis frontalis) and ear canal (meatus acusticus externus).
Histological samples of the head of the mandible (caput mandibulae) were stained using AB and were used for measurements of the cartilage thickness. Photodocumentation was taken using a DMC2900 camera (Leica, Wetzlar, Germany), and the thickness of the hyaline cartilage was measured using the AxioVision 40 version 4.8.2.0 software (Carl Zeiss MicroImaging GmbH, Munich, Germany). The acquired data were processed using the Statistica software (StatSoft, Tulsa, OK, USA).
Results
The TMJ is a complex congruent joint in a rabbit (Figure 1), in which the convex surface of the mandibular condyle articulates with the concave articular surface of the temporal bone (facies articularis squama ossis temporalis) located ventrally on the zygomatic process of the temporal bone. The articular disc is formed within the joint despite the joint being congruent in rabbits.
Radiography
Lateral skull radiographs clearly displayed the bony structures of the facial area. It was not possible to visualize the bones of the TMJ in detail due to the superimposition of the adjacent bones and masticatory muscles, therefore only some of the structures were identified (Figure 1). Radiographs gave a clear picture of the location of the zygomatic process of the temporal bone overlying the less discernible mandibular condyle followed by an overlap of the caudal process of the zygomatic arch (processus caudalis arcus zygomaticus). The bony part of the ear external opening and tympanic bulla (bulla tympani) were located caudally (Figure 1f).
A dorsoventral radiographic view of the rabbit skull showed the TMJ in even greater detail, allowing easy identification of the TMJ articular space (spatium articulare), the coronoid process of the mandible (processus coronoideus mandibulae), the mandibular ramus and the wall of the temporal bone (squama ossis temporalis) with the tympanic bulla (Figure 1g).
The cross-section of the line connecting the ear canal and caudal process of the supraorbital margin (processus caudalis margo supraorbitalis) and the line connecting the caudal processes of the zygomatic arch was located above the joint space of the TMJ in all the skulls under investigation.
Computed tomography
All TMJ structures identified on the radiographs were identifiable on the CT images; however, a more detailed morphology was visible in the transverse or sagittal CT slices (Figure 2). The articular surface of the temporal bone, which superimposed with the zygomatic arch in the lateral view on the radiograph, was discernible on the CT images (Figure 2). This surface was composed of a transversally elongated shallow articular tubercle of the temporal bone (tuberculum articulare ossis temporalis) and a shallow triangular mandibular fossa of the temporal bone, which was positioned sagittally and caudally. In the dorsal view, the articular surface of the mandibular condyle descended in a dorsolateral to ventromedial direction. The articular surface was also located rostrally on the mandibular condyle in the shape of a convex triangle, with the base situated rostrally and the apex caudally (Figure 2). Apart from a small flat and indistinct mastoid eminence (processus mastoideus), there were no caudal borders to the mandibular fossa. The retroarticular process (processus retroarticularis) was missing in the rabbits. Only the anterior–superior aspect of the mandibular condyle was in contact with the articular surface of the temporal bone, interposing the central part of the articular disc (Figure 2). Most of the muscular tissues were recognizable on the CT images, including the temporal, masseter (musculus masseter) and pterygoid (musculus pterygoideus) muscles (Figure 2).
(a–f) Transverse and three-dimensional computed tomography images of the rabbit skull with focus on TMJ. 1: ramus mandibulae, 2: corpus mandibulae, 3: collum mandibulae, 4: arcus zygomaticus – processus temporalis ossis zygomatici, 5: arcus zygomaticus – processus zygomaticus ossis temporalis, 6: arcus zygomaticus, 7: os temporale, 8: os frontale, 9: musculus massetericus, 10: musculus pterygoideus medialis, 11: musculus temporalis superficialis, 12: sulcus muscularis (musculus temporalis), 13: tongue, 14: condylus mandibulae, 15: condylus mandibulae, 16: porus acusticus externus, 17: processus caudalis arcus zygomatici, 18: processus zygomaticus ossis temporalis, 19: processus condylaris, 20: margo supraorbitalis, 21: processus caudalis margo supraorbitalis, 22: processus rostralis margo supraorbitalis, 23: processus angularis mandibulae, 24: bulla tympani, 25: processus coronoideus mandibulae, 26: incisura supraorbitalis rostralis, 27: incisura supraorbitalis caudalis, 28: processus mastoideus.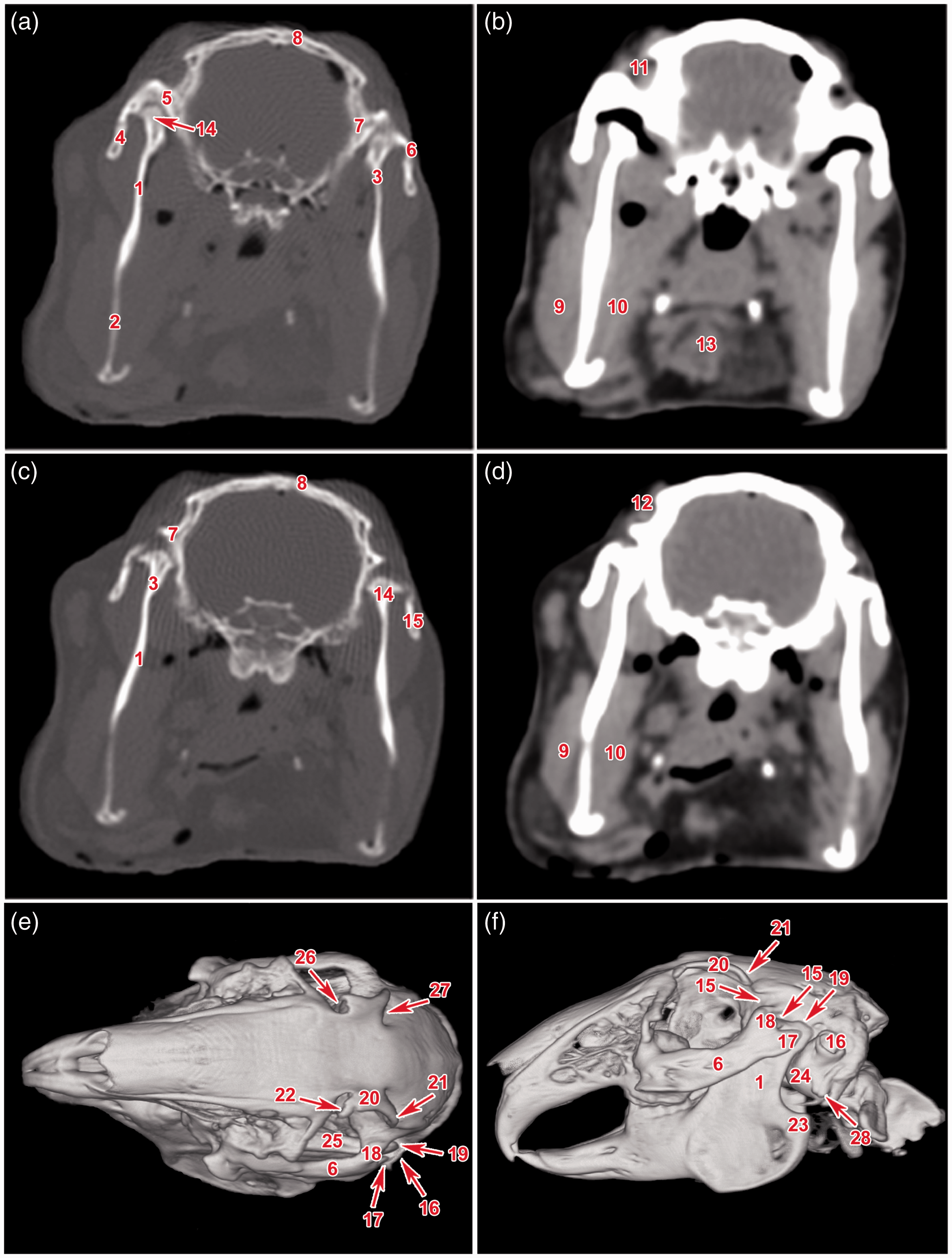
Magnetic resonance imaging
The soft tissues of the TMJ, including the surrounding muscles, were well delineated on the MRI scans. The oval articular disc was transversally positioned (Figure 3). The TMJ capsule was attached to the borders of the articular surfaces. The articular disc was connected to the medial and lateral aspects of the joint capsule.
(a–d) Magnetic resonance imaging of the rabbit head in a transversal plane with focus on TMJ. 1: ramus mandibulae, 2: condylus mandibulae, 3: processus zygomaticus ossis temporalis, 4: os temporale, 5: os preshenoidale, 6: ala ossis presphenoidalis, 7: auris externa, 8: auris media, 9: bulbus oculi, 10: discus articularis, 11: bulla tympani, 12: musculus temporalis, 13: processus pterygoideus, 14: foramen mandibulae, 15: musculus masseter, 16: musculus zygomaticomandibularis, 17: musculus pterygoideus medialis.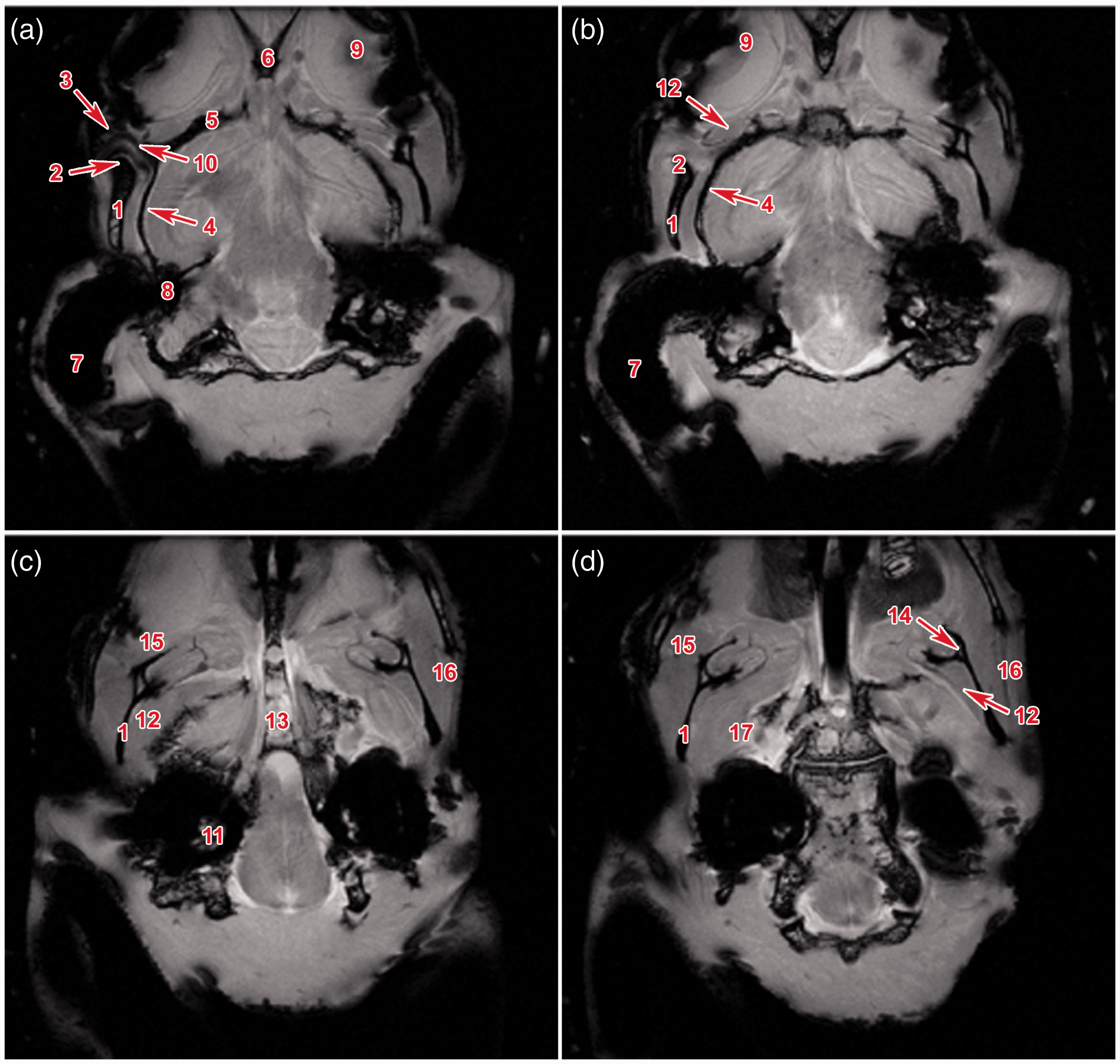
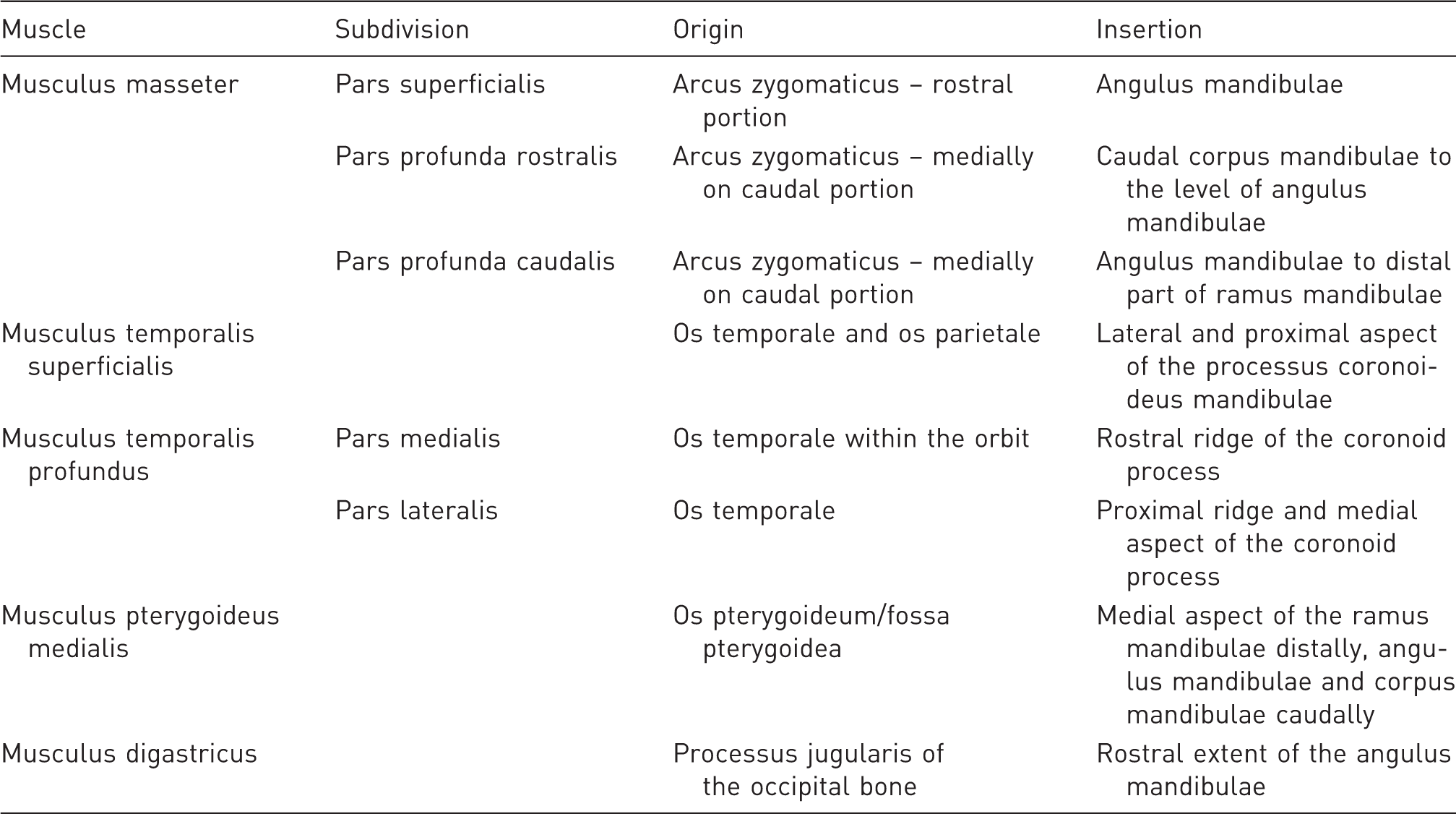
There are five main masticatory muscles in a rabbit (Table 1);24,27 however, only four were identified on the MR images. The masseter muscle consisted of three heads (Figure 4). The superficial head, or pars superficialis, originated from a point two-thirds of the way along the anterior portion of the lower border of the zygomatic arch (Figure 4), and was inserted into the mandibular angle (angulus mandibulae). The deep head, or pars profunda, originated one-third of the way along the medial–posterior part as well as from the medial portion of the zygomatic arch (Figure 4). It was inserted into the masseteric fossa (fossa masseterica) and subdivided into a rostral and a caudal part, which were indistinct on the MR images (Figure 3). During mastication, these two heads differed in function.
Masticatory muscle attachments in the rabbit. 1: musculus temporalis superficialis, 2: musculus masseter pars profunda caudalis, 3: musculus masseter pars superficialis (part 1), 4: musculus masseter pars superficialis (part 2), 5: musculus masseter pars profunda rostralis.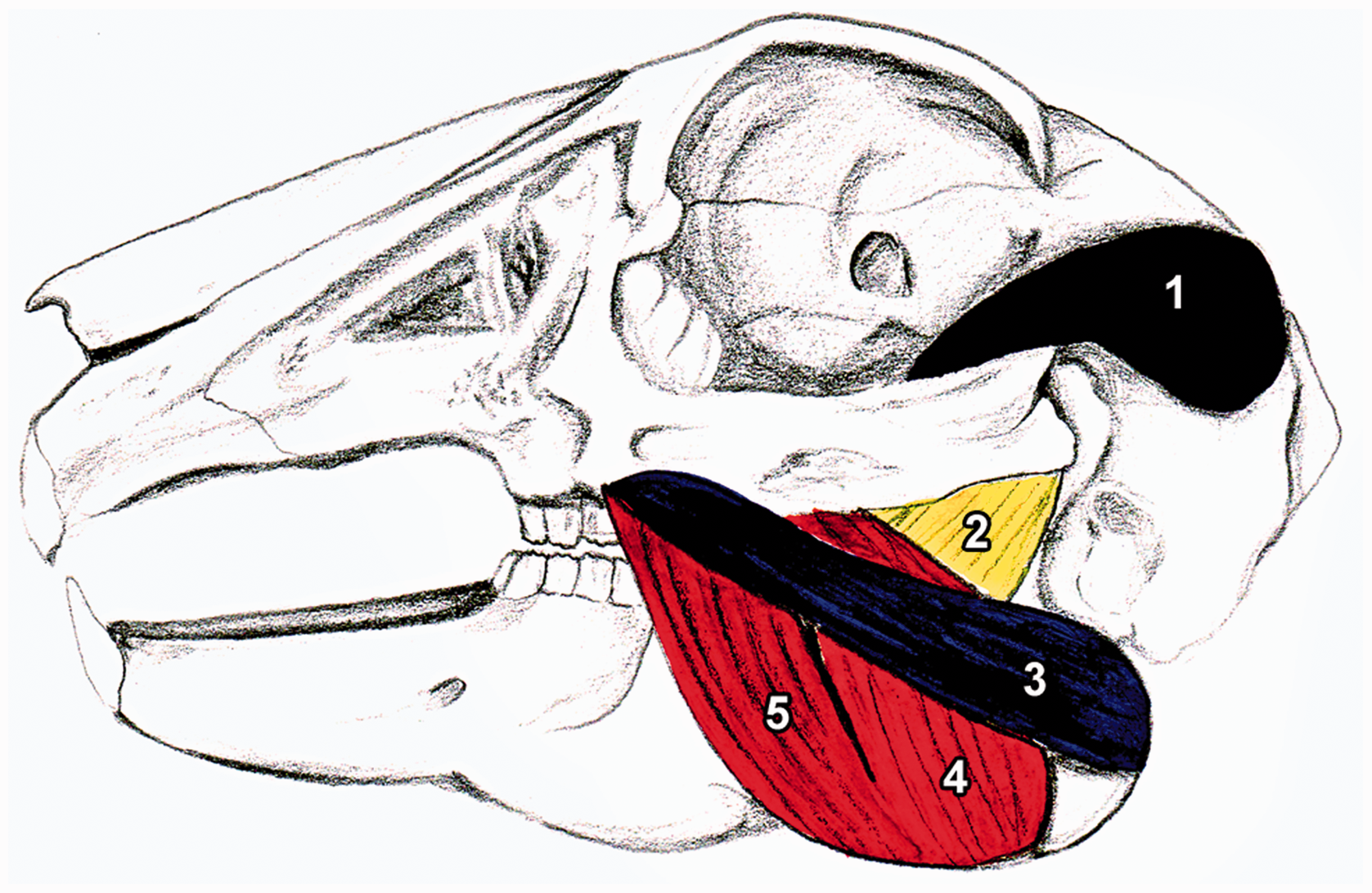
The superficial temporal muscle (musculus temporalis superficialis) tendon slided in the muscular groove (sulcus muscularis) dorsally over the TMJ. The medial aspect of the condylar process of the mandible (processus condylaris ossis mandibularis) and the mandibular ramus were the sites of attachment of the deep temporal muscle (musculus temporalis profundus) and both the pterygoid muscles (Figure 3, Table 1). The deep temporal muscle in the rabbits was further divided into the medial and lateral heads, which however were not discernible on the MR images.
Surgical anatomy
The superficial projection of the TMJ could be located by palpating the osseous part of the ear canal and caudal process of the supraorbital margin as well as the laterally located zygomatic arch (Figure 5). The cross-section of the axis of the zygomatic arch and the line connecting the supraorbital margin and the ear canal was located just above the TMJ space (Figure 5).
(a–f) Dissection of the temporomandibular joint in the rabbit. 1: margo supraorbitalis, 2: processus caudalis margo supraorbitalis, 3: ligamentum orbitale, 4: processus caudalis arcus zygomatici, 5: meatus acusticus externus, 6: arcus zygomaticus, 7: line connecting processus caudalis margo supraorbitalis and meatus acusticus externus, 8: line connecting contralateral processus caudalis arcus zygomatici, 9: intersection of the lines defines surface projection of the temporomandibular joint, 10: incision connecting margo supraorbitalis and meatus acusticus externus, 11: musculus temporalis, 12: capsula articularis of the temporomandibular joint, 13: processus zygomaticus ossis temporalis, 14: processus condylaris mandibulae, 15: ligamentum orbitale transacted, 16: sulcus musculi temporalis, 17: condylus mandibulae/facies articularis, 18: removed processus zygomaticus ossis temporalis, 19: discus articularis, 20: cavum articulare after the removal of the discus articularis.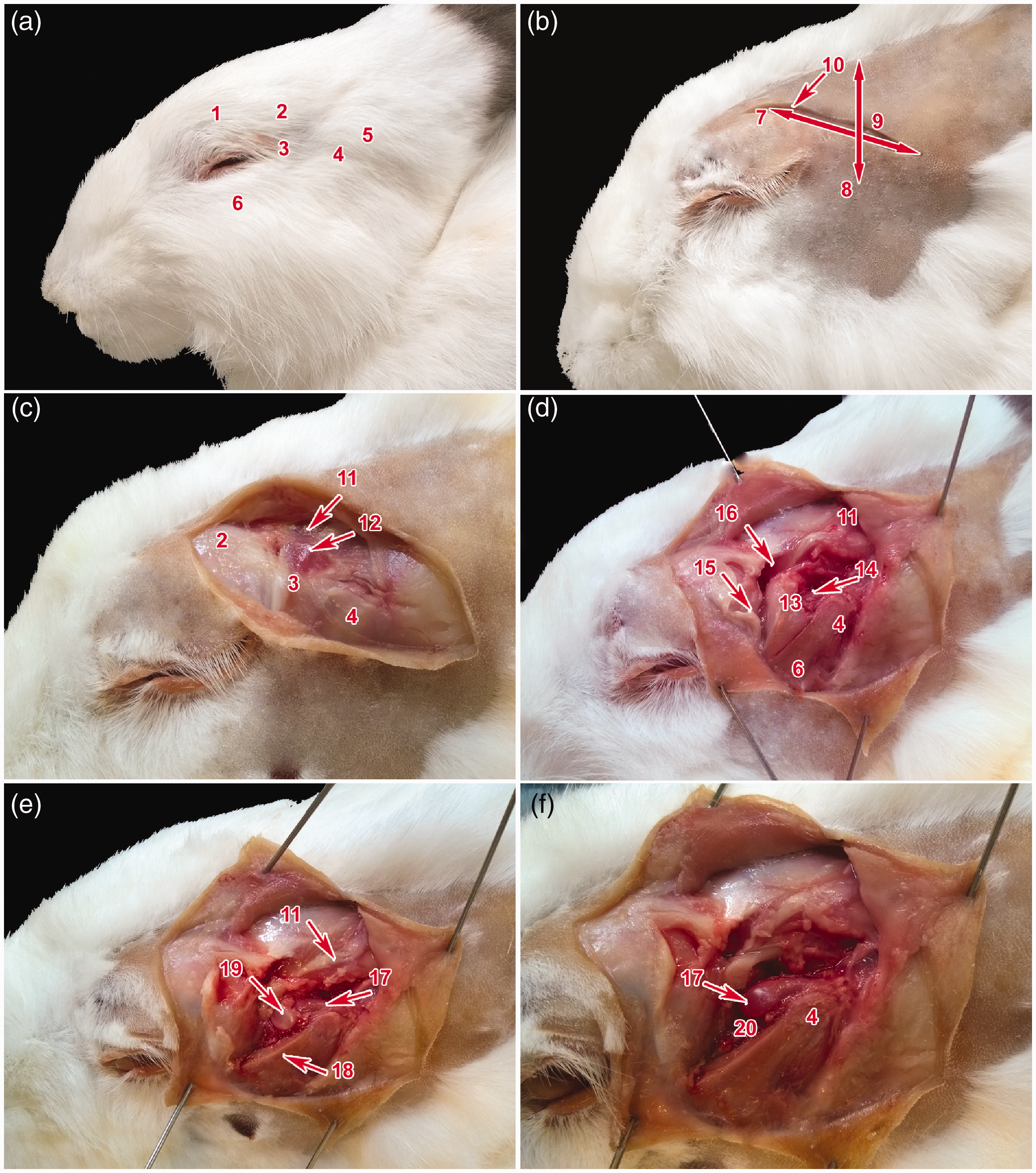
The TMJ was bordered dorsally by the zygomatic process of the temporal bone, medially by the squamous part of the temporal bone, and laterally by the zygomatic arch composed of the zygomatic process of the temporal bone and the temporal process of the zygomatic bone, which was projecting caudally into the caudal process of the zygomatic arch beyond the borders of the mandibular ramus. The TMJ was caudally and ventrally opened and surrounded by soft tissues. The dorsal aspect of the TMJ was covered by the musculotendinous origin of the superficial temporal muscle, which was directed rostrally over the zygomatic process of the temporal bone in the muscular groove (Figure 5) and attached rostrally from the TMJ on the indistinct coronoid process (processus coronoideus). The TMJ was bounded medially by the deep temporal muscle, which originated from the squamous part of the temporal bone and sphenoid bone (ossis sphenoidalis) and attached medially on the coronoid process and mandibular ramus. The condylar process of the mandible was covered caudolaterally only by the superficial fascia and by the skin, and was therefore easily palpable (Figure 5). The rostrolateral aspect of the zygomatic arch served as the origin of the masseter muscle – its superficial part originated from the base of the zygomatic arch and its deep part originated from the caudal process of the zygomatic arch (Figures 4 and 5).
The TMJ was surrounded by a thin capsule consisting of fibrous tissue and synovial lining. The capsule stretched from the edge of the mandibular fossa to the neck of the mandible (collum mandibulae) and as far caudally as the caudal process of the zygomatic arch. The articular space was divided into superior discotemporal and inferior discomandibular spaces. No large vessels or nerves were located in close proximity to the rabbit TMJ. The lacrimal gland was located rostrally and dorsally adjacent to the TMJ.
The interposed articular disc exhibited a biconcave morphology and concave surface proximally and distally (Figure 5). Its central area was thinner and the disc was attached to the joint capsule medially and laterally.
Histological analysis of the TMJ
Individual areas of the TMJ, such as the mandibular head, mandibular fossa and fibrocartilaginous disc, exhibited differences in microscopic structure (Figure 6). The mandibular head was composed of two types of cartilage – hyaline cartilage and fibrocartilage. Collagen fibres of the fibrocartilage were arranged parallel to the surface. The fibrocartilage passed into the hyaline cartilage in a highly cellular zone of small flattened chondrocytes, which represents the resting zone of the cartilage. Chondrocytes in the hypertrophic zone were arranged in columns which were transversally orientated to the surface (Figure 6). The calcified cartilage zone was wide, especially in younger animals, and passed into the subchondral bone at a depth of about 200 µm under the surface. Trabecular bone contained a large amount of chondro-osseous tissue (Figure 6).
Microscopic analysis of the rabbit temporomandibular joint (TMJ). (a–e) Sagittal sections of caput mandibulae in the rabbit. Alcian blue, haematoxylin and eosin stainings of an eight-month-old animal. (b) Graph of hyaline cartilage thickness. We observed statistically significant differences in cartilage thickness between individual areas of caput mandibulae. *P < 0.05, **0.00 < P < 0.01. (c) Detail view of the cranial part of caput mandibulae. (d) Detail of the middle part of caput mandibulae. (e) Detail of the caudal part of caput mandibulae. (f–n) Horizontal sections of articular disc. (f–h) Transitional area from disc to the capsule. (i–k) Most of the disc is formed by fibrocartilage. (l–n) Central area of the disc is mostly formed by cartilage. Scale bar in Figure A = 500 µm, scale bars in Figures B–N = 100 µm.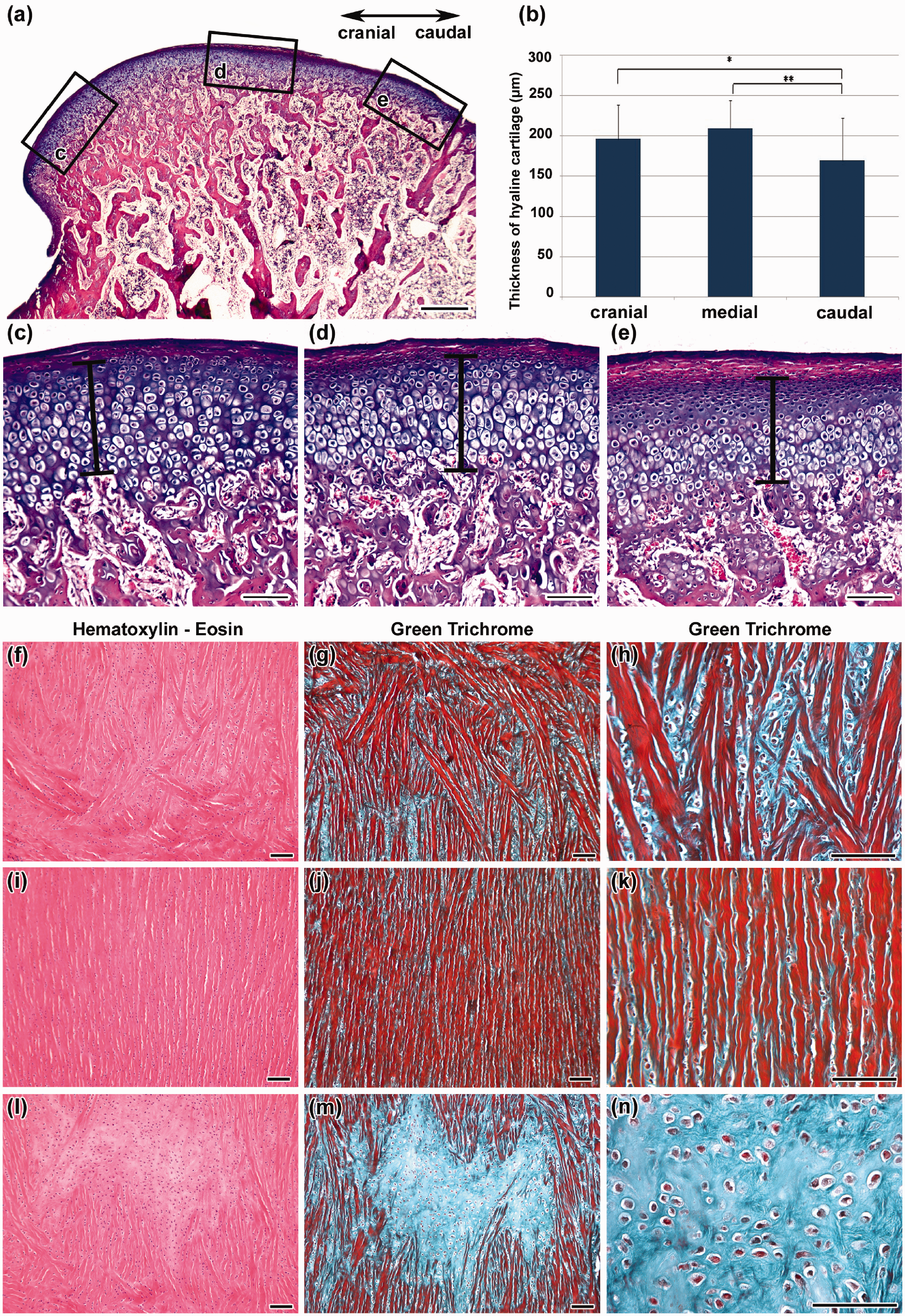
The articular disc was fibrocartilaginous (Figure 6) and revealed dense interwoven collagen fibres oriented predominantly mediolaterally on the periphery of the disc, whereas the crimped collagen fibres of the intermediate zone were directed rostrocaudally (Figure 6).
Morphometric assessment of rabbit TMJ
The cross-section of the line tangential to the dorsal aspect of the zygomatic arch and the line connecting the ear canal and the caudal process of the supraorbital margin was located above the TMJ in all the rabbits under investigation. The TMJ was roughly oval in shape, with an average width of 5 ± 0.02 mm and an average length of 15 ± 0.22 mm. The width of the mandibular condyle was 3 ± 0.09 mm.
Morphometric analysis of the cartilage thickness in the mandibular head area was carried out on histological sections, revealing differences in cartilage thickness in the rostral and caudal areas. The average thickness of the hyaline component was 196 µm rostrally, 209 µm medially and 169 µm caudally (Figure 6a,b). The thickness of the fibrous component also varied significantly – it was the thickest caudally and the thinnest rostrally.
Discussion
The TMJ is unique to mammals, but its morphology and function vary enormously among orders and species. Currently, the most commonly used model species for studying TMJ diseases are rats, rabbits, pigs, and ruminant ungulates. Each of these animal groups has distinctive TMJ adaptations. With the exception of pigs, these species show less loading of the jaw joints during chewing than humans. 28
Origins and insertions of muscles connected with temporomandibular joint (TMJ) function.
The anatomy and location of the rabbit TMJ make its evaluation by radiography a challenge due to the limited visualization of craniofacial structures. The superimposition of several bones and some soft tissues disguises the clear pattern of the delicate structures of the skull. Only frontal sinuses and the facial skeleton are clearly discernible due to their superficial character. Different radiographic views have been suggested to improve visualization of the rabbit TMJ.25,27 Another study that suggested the use of different angles of projection for the visualization of the rabbit TMJ have reported a satisfactory assessment of the joint components and space in the rabbit when using a rostrocaudal view and a 10–20 degree rotation in a lateral direction. 27
Any mini-invasive treatment (injection or arthrocentesis) requiring TMJ access must always take into account the surrounding anatomical structures. In humans, these include the ear canal, facial nerve, skull base, superficial temporal vein, maxillary artery and parotid glands. In rabbits, structures such as the facial nerve, maxillary artery and parotid gland, do not need to be considered due to the different anatomical location of its TMJ. To perform a mini-invasive procedure in a rabbit, open surgery exposing the joint capsule is necessary and special care has to be taken with respect to the joint position. Whereas the joint is located posteriorly to the muscular prominence in humans, it is positioned anteriorly in rabbits. 30 Therefore, the best approach for any mini-invasive surgery in a rabbit TMJ should be from a dorsal aspect of the TMJ. An incision should be made halfway through the line connecting the caudal process of the supraorbital margin and the base of the ear.
The condyle of a rabbit is covered by secondary cartilage and fibrous tissue as in humans. 31 It is also important to consider the depth of experimental defects created in the condylar cartilage before using the rabbit as an animal model. We proposed a depth of less than 150 µm for a cartilage defect and more than 220 µm for a subchondral defect. Unlike humans, the rabbit cartilage is thickest in the middle area and not in the posterosuperior region. The densest part of the human TMJ cartilage is 480 µm. 32 This fact should be taken into consideration when selecting tools for the preparation of experimental defects. With regard to the arrangement of cartilaginous cells, there are no significant differences between human and rabbit TMJ cartilages. 33 However, there are significant differences between human and rabbit TMJ discs. Whereas the human disc is described as dense fibrous tissue, or fibrocartilaginous plate, where the chondrocyte-like cells should not be referred to as chondrocytes, 34 the rabbit disc contains well-differentiated cartilaginous tissue. These chondrocytes are arranged between fibrous stripes, and islets of this cartilaginous tissue are present in the central area of the disc. It is worth noting that the cartilaginous tissue is found in young animals. Nevertheless, for a deeper understanding of the origin of these cells further immunohistochemical investigation will be necessary.
To identify the exact area for local injection into the rabbit TMJ it is possible to use the connection of the palpable reference points of the bony ear canal and caudal process of the supraorbital margin and the intersection of that line with a line tangential to the dorsal aspect of the zygomatic arch. The needle should be inserted at this point with its bevel pointing towards and parallel with the zygomatic arch. However, this approach may penetrate the tendon of the temporal muscle overlying the joint, which can cause sample contamination or possible tendinitis. Therefore, in cases in which an non-contaminated sample has to be collected, an open surgical approach above the TMJ is the only viable option. The incision should be performed sagittally above the TMJ along the line connecting the caudal process of the supraorbital margin and the base of the ear and by retracting the tendon of the temporal muscle laterally. A practical execution is however relatively difficult due to the size of the joint and also to the risk of cartilage damage. Unlike in humans, it is not possible to rely on the ‘clap’ of the needle on the fossa during its insertion. From our point of view, tilting the needle perpendicularly to the joint surface and performing a lateral perforation as in humans seems to be the best solution. The maximum dip of the needle insertion should be about 2–3 mm.
An arthrocentesis in a rabbit model is obviously even more difficult to perform and may also require open surgery. Due to the differences between rabbit and human TMJs, the rabbit TMJ is not really a suitable educational model for practicing arthroscopic surgery. Rabbit TMJ can however be used as a research model: while performing a surgical intervention, anatomical differences should be taken into account. Moreover, due to the very small size of the articular disc, the rabbit TMJ is not suitable for performing procedures such as arthroscopies, discopexis or discectomy. 35
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Grant Agency of the Czech Republic (14-37368G to MB lab, 14-29273P to JS) and the Ministry of Health, Czech Republic (MH CZ-DRO-FNOs/2013).
