Abstract
Postoperative analgesia in rodent sepsis models has been considerably neglected in the past. However, intentions to model clinical practice, increasing awareness of animal ethics, efforts to apply the 3Rs (replacement, reduction, refinement), and stricter legislation argue for a change in this respect. In this review, we describe different concepts of analgesia in rodent models of sepsis focusing on opioid agonists as well as non-opioid analgesics. Advantages and pitfalls in study design and side-effects are discussed. Score sheets should be used to adapt analgesia or to terminate experiments using humane endpoints. Further research is needed to differentiate behavioral changes caused by sepsis and pain or as a consequence of analgesia. Information on the efficacy of analgesia in sepsis models is scarce. Hence, studies are needed to identify the best ways to reduce suffering of research animals and thereby optimize the clinically relevant rodent models of sepsis.
Animal model
Over recent decades, animal models of sepsis have been refined continuously. Among the wide range of existing models, the majority are focused on abdominal sepsis in rodents, as this has been shown to be both valid and reproducible.1,2 Advantages and pitfalls of rodent sepsis models have been discussed extensively.1–4 One main issue is the difficult translation from rodent to human studies, which partly results from the complexity of human sepsis and the interplay of an unknown number of confounders. The traditional model of fecal peritonitis is cecal ligation and puncture (CLP), which causes subacute abdominal sepsis. Mice are the predominant species used in sepsis research.
5
However, the use of mouse models has been criticized mainly due to the pathobiological differences between mouse and human sepsis.
6
By contrast to humans and rats, sepsis in mice lowers body temperature and oxygen consumption.
7
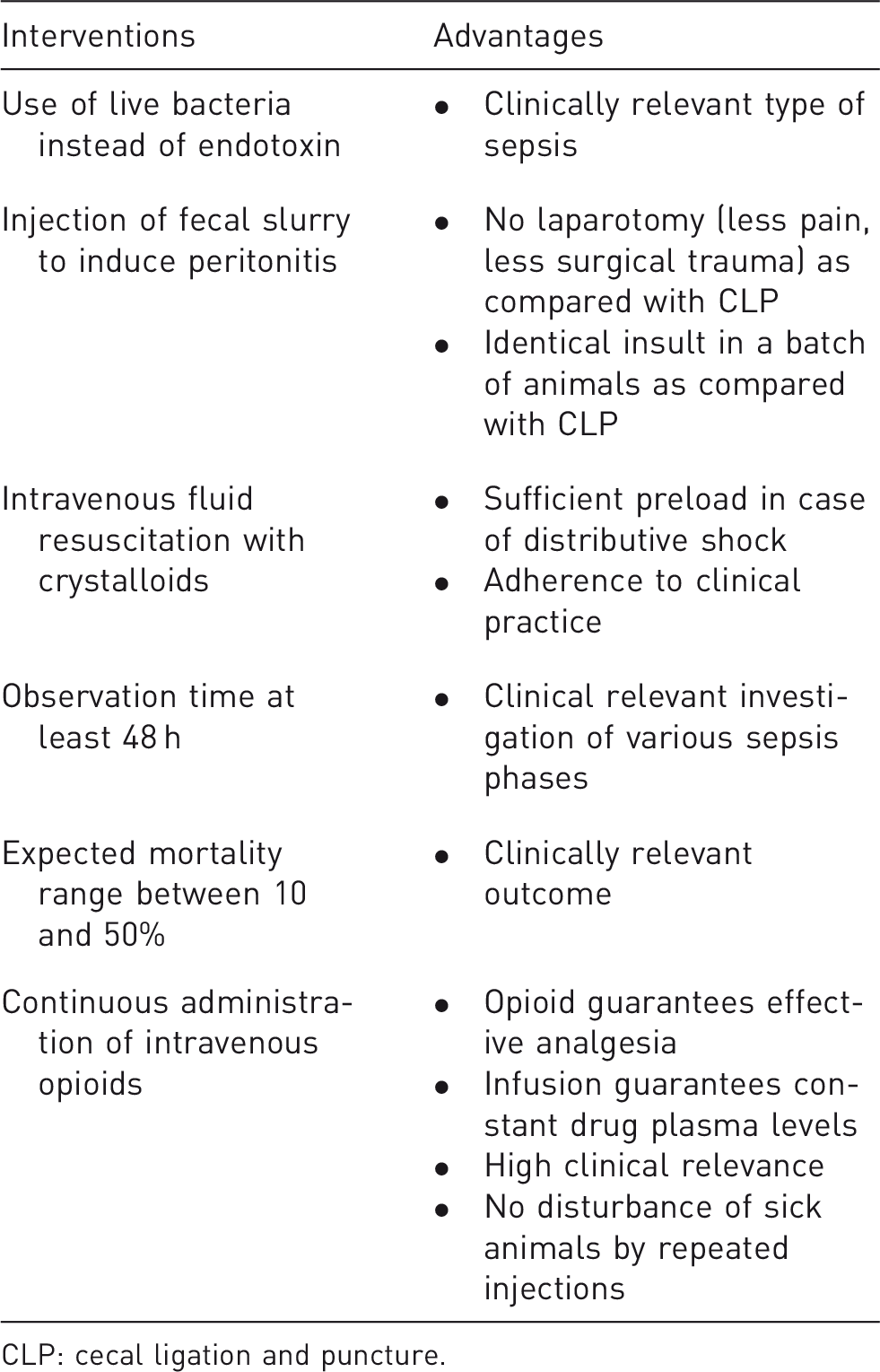
The debate about the usefulness of murine sepsis models is still ongoing. In this current review the focus is on rat sepsis models as we believe that they resemble human patients more closely. Characteristics of a clinically relevant rat model of sepsis are given in Table 1 and Figure 1.
Representative picture of a rat equipped with a central venous line on a swivel–tether system. Suggestions for a clinically relevant rat model of fecal peritonitis. CLP: cecal ligation and puncture.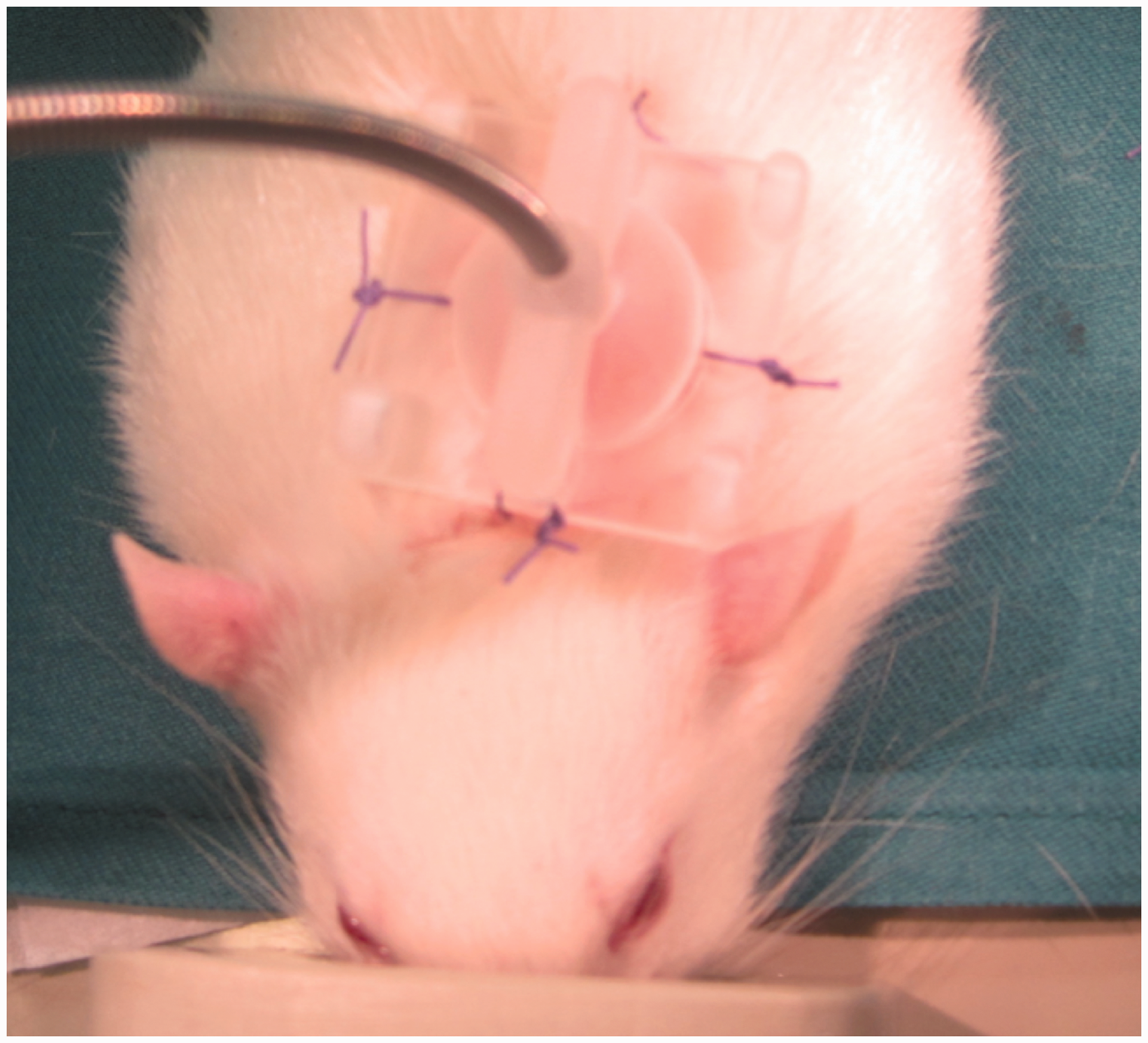
After induction of polymicrobial sepsis (by CLP or intraperitoneal injection of fecal slurry), animals are usually awake during the experiment. In general, anesthesia (e.g. isoflurane) is only applied during instrumentation and induction of sepsis. 1 These experiments are highest in the severity grade (in Switzerland class III, European severity category ‘severe’) and require appropriate animal ethics permission, which may differ according to the legislation of the country where the experiments are performed. In Switzerland, the regulation of ‘animal’s dignity’ in legislation influences the design of animal models even more strictly, which has been discussed elsewhere. 8
In the early days of sepsis research, the aim was to observe the effect of the infection itself, therefore neither fluids nor antibiotics were administered. During the last decade, there has been a paradigm shift to model clinical practice of human sepsis in animals instead of reproducing animal sepsis. 1 Therefore the use of fluid resuscitation during septic shock is mandatory and the use of antibiotics is widely applied.1,2,9 By contrast to the clinical reality of intensive care units, the application of analgesia or even analgosedation has been neglected for a long time in animal research. In comprehensive reviews analgesia is barely mentioned.1,2 In 2012, only a few research groups (15%, 7 out of 45 publications) used analgesia, although pain and distress were likely in all animals undergoing experimentally-induced sepsis. 10 Marshall et al. have expressed concern about the interaction between analgesia and the pathobiology of sepsis, especially with opioid-induced immunosuppression. 3
Assessment of welfare and pain
As indicated above, animal welfare in sepsis studies has not been a major topic of concern in sepsis research. 10 However, pain and distress are important confounders in animal models and may influence behavior and the animal’s immune system. 10 The induction of peritonitis may cause pain and will influence the behavior of animals.11,12 To improve animal welfare, researchers have to identify animal distress and should therefore know the behavior of healthy animals beforehand. Behavior changes and distress can be quantified using clinical scores.13–16 Scoring may then be used to either refine the animal model by using adequate analgesia or to implement termination criteria (also called humane endpoints) to the sepsis model. Huet et al. have described and validated a clinical scoring system for a murine sepsis model. 13 It was based on behavioral signs (activity, posture, reaction to stimulation), fur aspect, respiration and chest sounds as well as body weight. The authors have shown that severe clinical scores corresponded to high cytokine and reactive oxygen species levels. 13
In order to improve animal welfare, termination criteria should be designed such that not only moribund animals close to death are recognized. 17 Early clinical signs with a high sensitivity and specificity to predict the animals’ demise should be defined so that experiments can be terminated early. Good predictors will also allow comparisons between potential survivors and non-survivors, thereby enabling comparisons between adaptive mechanisms of survival and maladaptive mechanisms of death.
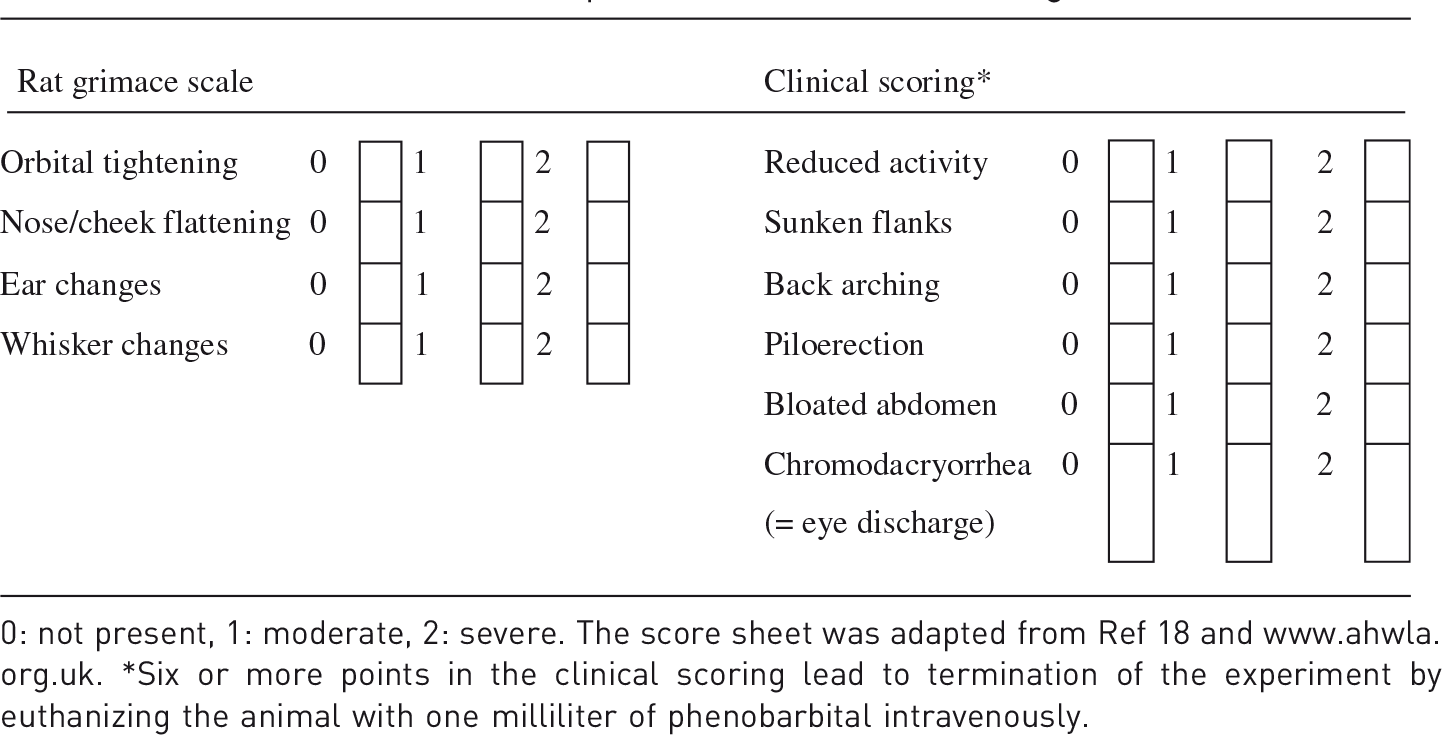
Even more subtle indicators may be used to assess pain in animals, as applied in the rat grimace scale validated for acute pain. 18 There is no report of its use in acute/subacute sepsis models. Kawano et al. have recently applied the rat grimace scale using video stills, and have used a blinded evaluator to explore the role of lipopolysaccharide (LPS) on an incisional pain model retrospectively. 14 However, sepsis models should be evaluated in real-time and not retrospectively in order to identify relevant termination criteria. Furthermore, the rat grimace scale has not yet been validated to differentiate sepsis-induced distress from acute pain. Our experience in a rodent model of fecal peritonitis reveals that orbital tightening and ear changes are obvious. However, identification and quantification of nose/cheek flattening and whisker changes are difficult if not impossible even for experienced investigators. Other clinical signs such as inactivity, piloerection, eye discharge, sunken flanks or back aching have also been used, 19 but it is unclear how they might accurately predict severity of illness in general or abdominal pain in particular.
The implementation of physiological parameters such as changes in water intake, body weight, body temperature or heart rate may be more reliable, and therefore better discriminators for the outcome.12,17,20 We have described that clinical scores during early sepsis might not prognosticate outcome, whereas echocardiography-derived stroke–volume and heart rate can predict death with a good sensitivity and specificity as early as 6 h after the septic insult,
21
and which can therefore lead to shorter observation times and robust and early termination criteria. The use of implantable and wireless telemetry devices may further help to determine quantitative cut-off values, first described as sickness behavior by Bauhofer et al.
12
Figure 2 shows an example of telemetry recordings in two septic Wistar rats. In a recent murine sepsis model, predefined deterioration thresholds of heart rate and body temperature assessed by telemetry, independent of the delay since sepsis induction, were superior to data sampling at predefined time points. This led to more homogenous study groups.
22
The authors stated that it might be possible to predict the outcome by defining adequate thresholds based on telemetry data.
22
Although not mentioned by them, these thresholds may be used to define termination criteria. However, pilot studies are needed for each species and sepsis model to define thresholds based on vital signs.
Example of a rat ECG recording based on the implanted telemeter electrode. ECG: electrocardiogram; mV: amplitude in mV; °C: body temperature in degree Celsius; BPM: beats per minute.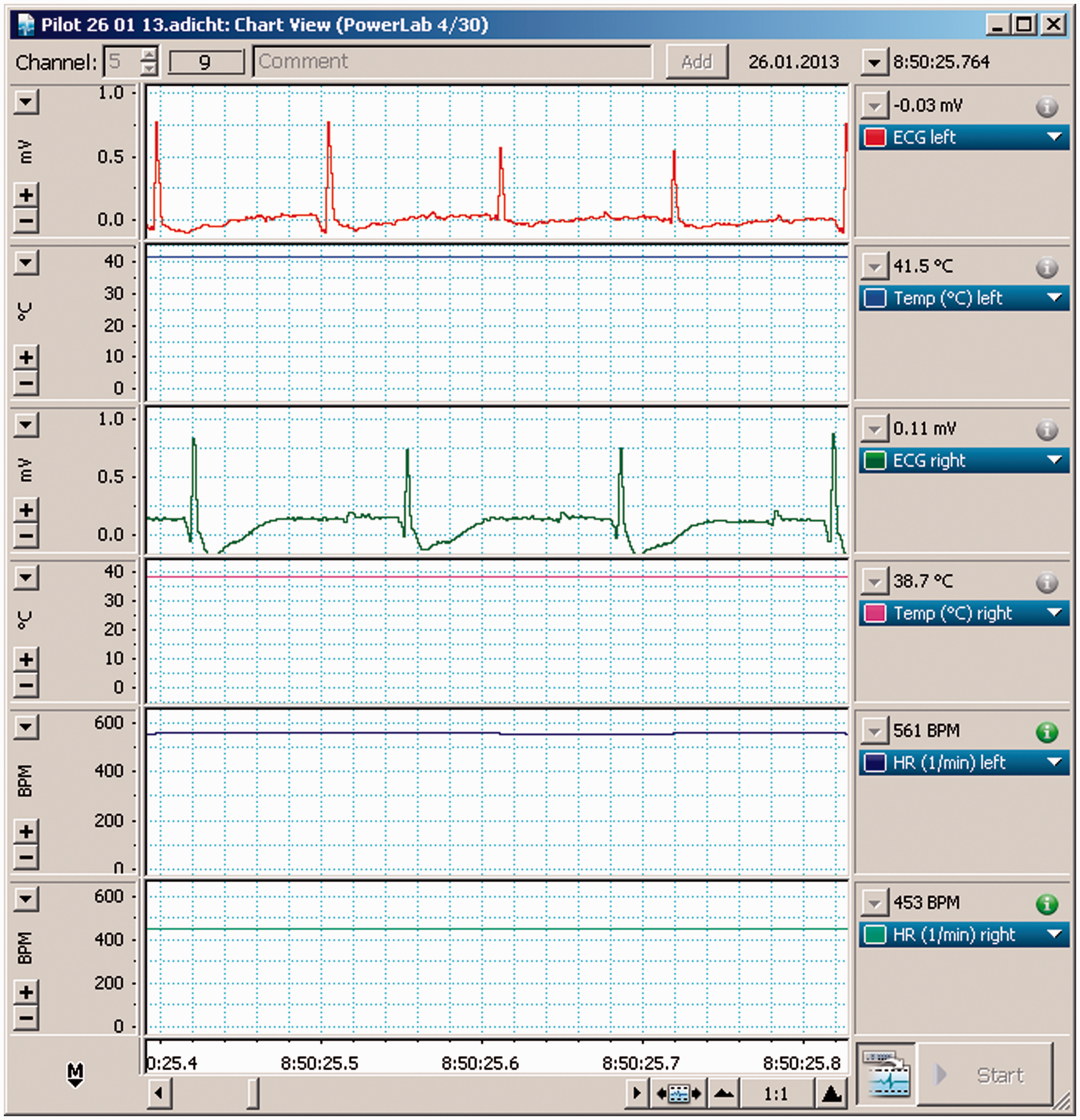
Analgesia should reduce stress levels and thereby increase reliability of the model. One important difficulty is that sepsis and analgosedation may cause similar behavior changes in rats. 23 In particular inactivity and apathy may be caused by the disease as well as by (over-)sedation caused by opioid analgesia. Reduction of vertical movement of rats – as a marker of behavior change – in response to pain was similarly observed in healthy animals receiving high doses of morphine. 23 Therefore behavior changes induced by analgesia have to be distinguished from sepsis-induced distress and pain in further studies.
Refinement opportunities – analgesics in sepsis models
Opioid agonists/antagonists
Publications on nalbuphine in pain studies and postoperative settings.
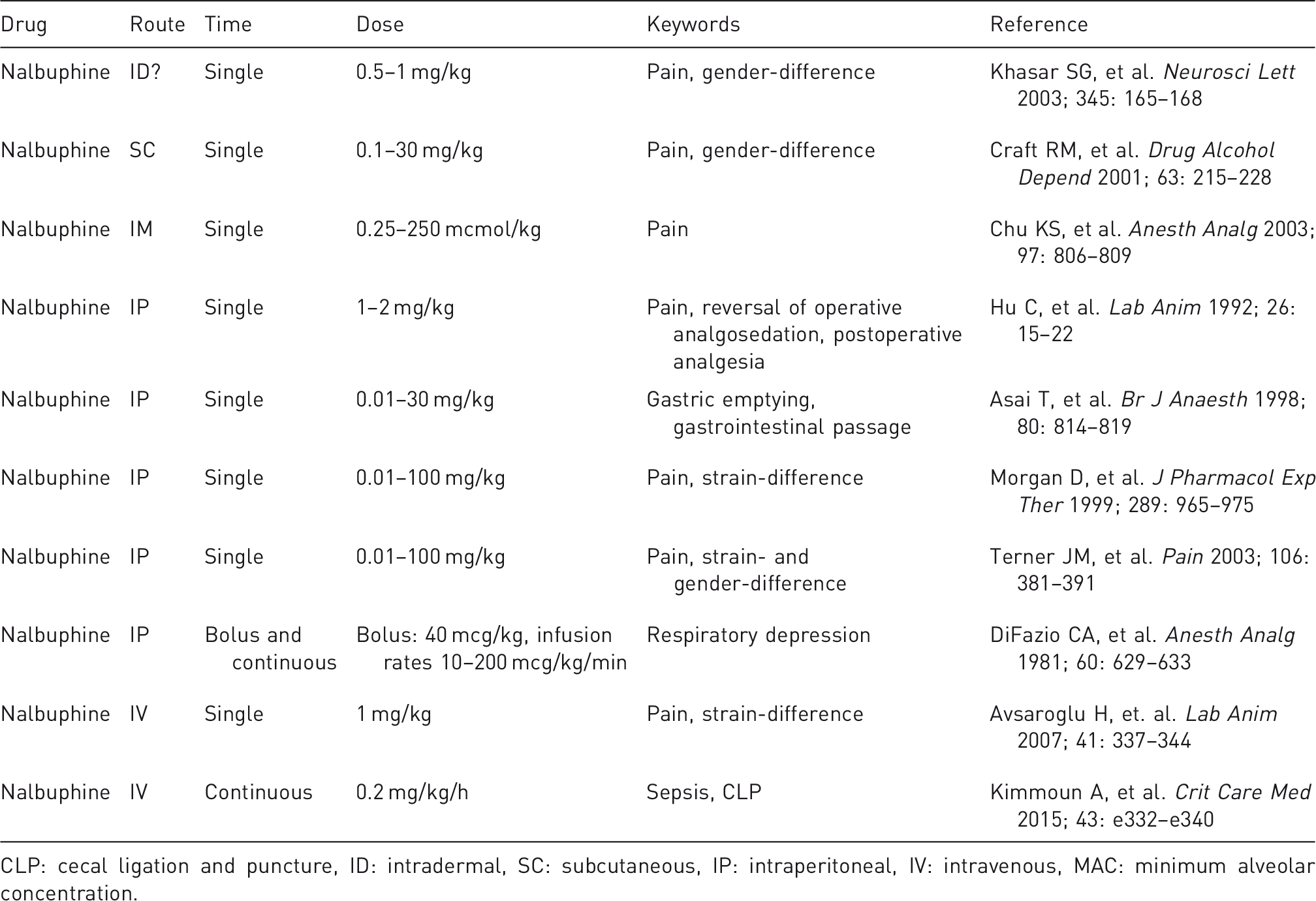
CLP: cecal ligation and puncture, ID: intradermal, SC: subcutaneous, IP: intraperitoneal, IV: intravenous, MAC: minimum alveolar concentration.
There are two major advantages of nalbuphine compared with pure mu-opioids: it only weakly inhibits gastric emptying and gastrointestinal passage, 34 and does not cause marked respiratory depression.35,36 Nalbuphine also has an effect on hemodynamics. 37 These side-effects may interfere with sepsis-induced hypotension or tachycardia, which has to be taken into account in an experimental design.
Publications on buprenorphine in pain studies and postoperative settings.
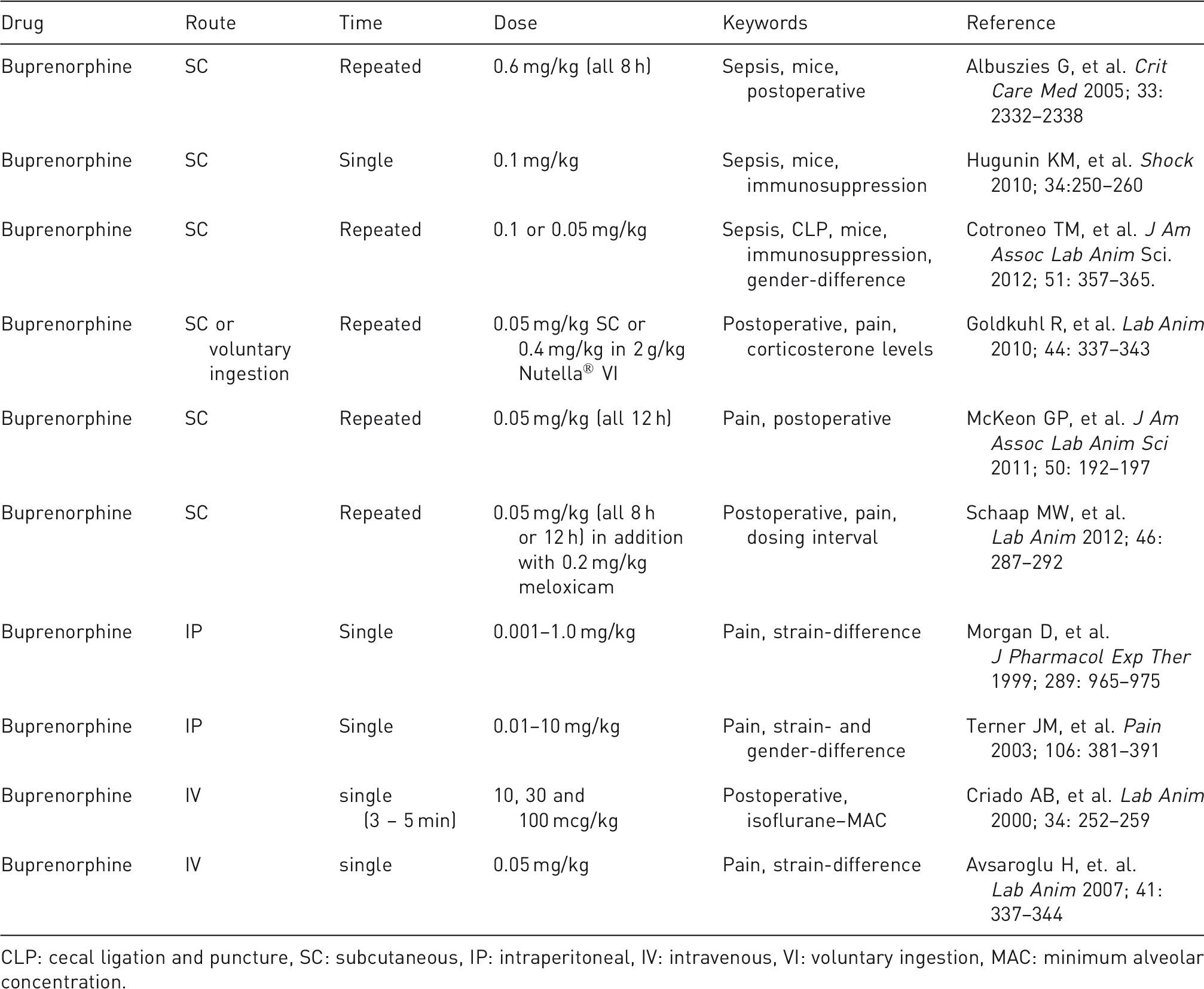
CLP: cecal ligation and puncture, SC: subcutaneous, IP: intraperitoneal, IV: intravenous, VI: voluntary ingestion, MAC: minimum alveolar concentration.
Butorphanol may be an alternative partial agonist/antagonist and has been used in rat postoperative pain models.48,49 However, there are no data so far regarding its use in sepsis.
Pure mu-agonists
Morphine and fentanyl are potent mu-agonists, which are used in adult intensive care every day. In larger animals, where a safe airway and controlled ventilation can be achieved, fentanyl may be the favorite choice. 50 In rodents, where long-term (>6 h) controlled ventilation is difficult, if not impossible, continuous fentanyl administration has to be evaluated carefully due to respiratory depression.51,52 Other strategies may be the use of epidural analgesia 53 which again may be easier to apply in larger animals than in rodents. Of note, the immune-modulating effects of morphine affect mechanistic studies in sepsis models. 54
Non-opioid analgesia
Non-steroidal anti-inflammatory drugs (NSAIDs) have been used in postoperative analgesia in rats.55,56 The use of NSAIDs in sepsis has been discussed, and a trend might exist towards reduced acute lung injury. 57 In clinical practice with septic patients, the use of NSAIDs is avoided due to the risk of renal impairment and bleeding. Furthermore their anti-inflammatory effect, mainly on prostaglandin synthesis, 25 may interfere with the sepsis model itself and could thereby influence the pathobiology of the disease.
Local anesthetics such as lidocaine and ropivacaine might be used as further adjuncts to reduce intra- and postoperative pain.58–60 We use local anesthesia in addition to isoflurane for surgical instrumentation. So far no data exist on whether a prolonged or repeated application of local anesthetics may improve analgesia in sepsis models.
Score sheet to assess sepsis-induced behavior changes.
As discussed above, assessment of pain and distress may be challenging in sepsis models. To measure the effectiveness of analgesia may be even more difficult as analgesics themselves interfere with normal animal behavior. 23 In a postoperative laparotomy rat model, efficacy of analgesia was assessed by changes in food intake, body weight, and by measurement of pain threshold using a paw-flick latency test. 62 However, sepsis itself may have an effect on these parameters. 61 Furthermore LPS increases hyperalgesia in incisional pain models 14 but hyperalgesia in polymicrobial sepsis models has not been investigated so far. Assessment of pain thresholds together with measurements of plasma levels may improve our understanding of pain therapy in sepsis models. Plasma drug levels from human studies could serve as a reference for animals. 46 To our knowledge, there are no reports on plasma concentrations of analgesics in sepsis models. In addition, sepsis-induced organ dysfunctions might influence drug metabolism and could lead to accumulation of active compounds and metabolites.
Further refinement opportunities other than analgesia could be achieved using less invasive sepsis models and/or different housing conditions. For example, peritoneal injection of fecal slurry does not require painful laparotomy, which is necessary in CLP models. Furthermore, animals have to be singly-housed if a swivel–tether system is used. Lilley and colleagues from an expert working group on 3Rs in sepsis models have mentioned the idea of co-housing healthy, non-instrumented animals with instrumented animals. 63 This may be an advantage in controlling body temperature in these social animals. 63 However, co-housing could also induce stress to the healthy animals due to the presence of their sick cage mates, as it is well known that housing and experimentation within the same room may cause stress to the untreated animals. 64
Recommendations and further 3R research
Recently, recommendations on applying the 3Rs in sepsis research have been published, providing an excellent overview of potential refinement of sepsis models. 63 The authors conclude that ‘applying the “R” in Refinement of animal studies can be a highly effective way to reduce suffering and improve scientific quality’. 63 Osuchowski et al. have mentioned in their editorial that the 3Rs should also be interpreted as ‘relevance, robustness and reproducibility’. 65 Humane endpoints were proposed more than a decade ago in sepsis research,12,16 however we still lack preclinical studies with the primary aim of assessing and improving analgesia and termination criteria in sepsis. More research should therefore focus on the translational approach of sepsis models and the ‘refinement’ aspect. More studies are also needed to investigate the pharmacokinetics and pharmacodynamics of different types of analgesia, behavior and physiological alterations in sick animals, and the implementation of humane endpoints to reduce unnecessary suffering.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article..
