Abstract
A disease affecting guinea pigs called ‘guinea pig lameness’ characterized by clinical signs of depression, lameness of limbs, flaccid paralysis, weight loss and death within a few weeks was first described by Römer in 1911. After a research group in our facility kept laboratory guinea pigs from two different origins together in one room, lameness was observed in two animals. Further investigations revealed a serological immune response against Theiler’s murine encephalomyelitis virus (TMEV; GDVII strain) in these animals. Histopathology of the lumbar spinal cord of these animals showed mononuclear cell infiltration and necrotic neurons in the anterior horn. Therefore, all guinea pigs from this contaminated animal unit, from other units in our facility, as well as from different European institutions and breeding centres were screened for antibodies directed against GDVII. Our investigations showed that approximately 80% of all guinea pigs from the contaminated animal unit were seropositive for GDVII, whereas animals from other separate units were completely negative. In addition, 43% of tested sera from the different European institutions and breeding centres contained antibodies against GDVII. The present data confirm that an unknown viral infection causes an immune response in experimental guinea pigs leading to seroconversion against GDVII and that guinea pigs from a commercial breeder are the source of the infection.
Laboratory animals should be free of infectious agents, which ultimately pose a risk to the animal’s well-being and the research outcome. Paul Römer first described a disease affecting laboratory guinea pigs called ‘guinea pig lameness’ in 1911. 1 While searching for new animal models for human poliomyelitis he randomly observed paralysis in the hind part of guinea pigs due to an infection of the central nervous system (CNS) accompanied by infiltrating lymphocytes and hypotrophic ganglia, leading to cachexia and death within a period of up to four weeks after onset of the symptom. According to Koch’s postulates, Römer tried to identify the causative agent and described a filterable virus comparable to the poliovirus that causes poliomyelitis virus infection in humans. In 1997, Hansen and colleagues reported on pet shop guinea pigs suffering from lameness, and that they were seropositive for Theiler's murine encephalomyelitis virus (TMEV; GDVII strain). The agent causing encephalomyelitis in guinea pigs could not be isolated so far but a viral origin has been considered. 2
TMEV is a member of the genus Cardiovirus in the family Picornaviridae. Under natural conditions, mice usually develop asymptomatic infections of the gastrointestinal tract. Infections seldom spread to the CNS and cause neurological diseases, which can occur especially in immune-deficient mice. Experimental intracerebral or intranasal infection serves as a model for poliomyelitis and induces demyelinating diseases. 3 Factors modifying infection outcomes include route of inoculation, host genotype, age as well as virus strain (see overview 3 ). For example, GDVII represents a highly virulent strain causing acute and severe infection of the CNS, whereas the original isolate, Theiler's original (TO), represents a group of strains with low virulence causing a persistent infection of the CNS in mice. It has been shown that both hamsters at younger ages and laboratory rats are susceptible to intracerebral infections. 3 As a result of polymerase chain reaction (PCR) analyses of cerebrospinal fluid from guinea pigs suffering from lameness, a 231 bp sequence was found. Further analysis revealed sequence homologies to the internal ribosomal entry site of the encephalomyocarditis virus (EMCV), which is also a member of the genus Cardiovirus. 4
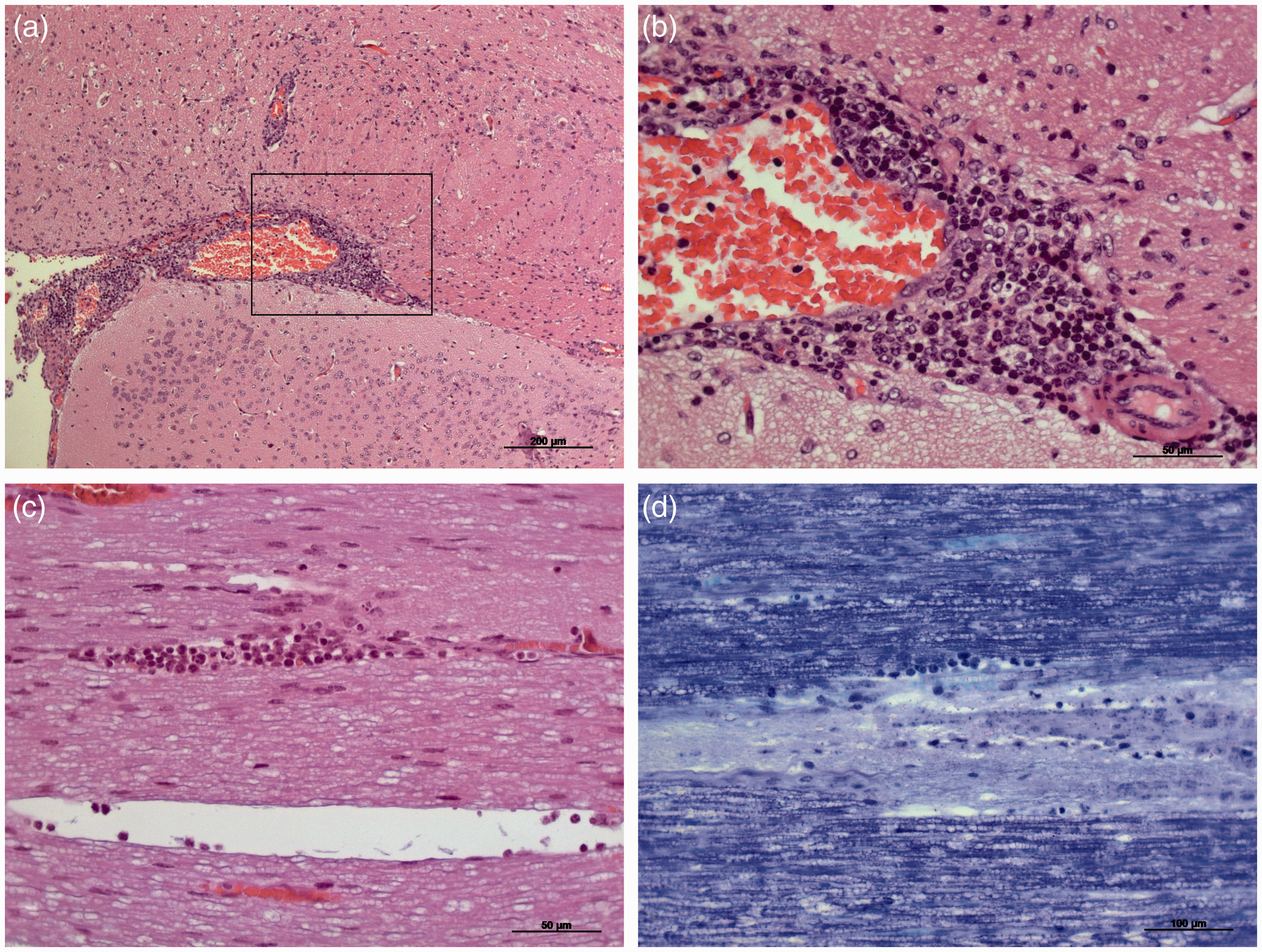
We have routinely maintained experimental guinea pigs over several decades at the Central Animal Facility of the Hannover Medical School (Ztm). The animals are generally kept separated according to their origins. Because of a specific research project, guinea pigs from a commercial breeder (CB1) had to be exceptionally maintained together with animals from another supplier (CB2) in one room. After obtaining guinea pigs from CB2 we observed lameness and death in two animals from the first supplier CB1. Histopathological examinations of the diseased guinea pigs revealed no pathological changes in their brains (data not shown), but motor neuron damage and diffuse mild infiltration of the lumbar spinal cord were observed (Figures 1a–c). By comparison, GDVII (ATCC VR-57, BHK-21 cell supernatant; provided by BioDoc, Hannover, Germany) infection via oral gavage in three of the mice (female BALB/cNAnHanZtm, 6–8 weeks of age) caused circling behaviour in one animal 10 days after infection. Histopathological analysis of the respective mouse revealed inflammatory perivascular mononuclear cell infiltration in the brain (Figures 2a and b) and patchy demyelination associated with leukocyte infiltration in the spinal cord (Figures 2c and d).
Overview of the lumbar spinal cord of the diseased guinea pig stained by Klüver-Barrera (a) and haematoxylin and eosin (H&E) (b). (c) Detail of the anterior horn with necrotic neurons, indicated by pyknotic nuclei, eosinophilic cytoplasm and loss of cytoplasmic structures (marked by black arrows). Histopathology of the brain and lumbar spinal cord of a Theiler’s murine encephalomyelitis virus (TMEV)-infected mouse. (a and b) Haematoxylin and eosin (H&E)-stained sections of the brain with mononuclear perivascular cell infiltration in the pons. (c) H&E-stained section of the lumbar spinal cord with inflammatory cell infiltration. (d) Klüver-Barrera-stained section of the lumbar spinal cord with patchy demyelination of neurons.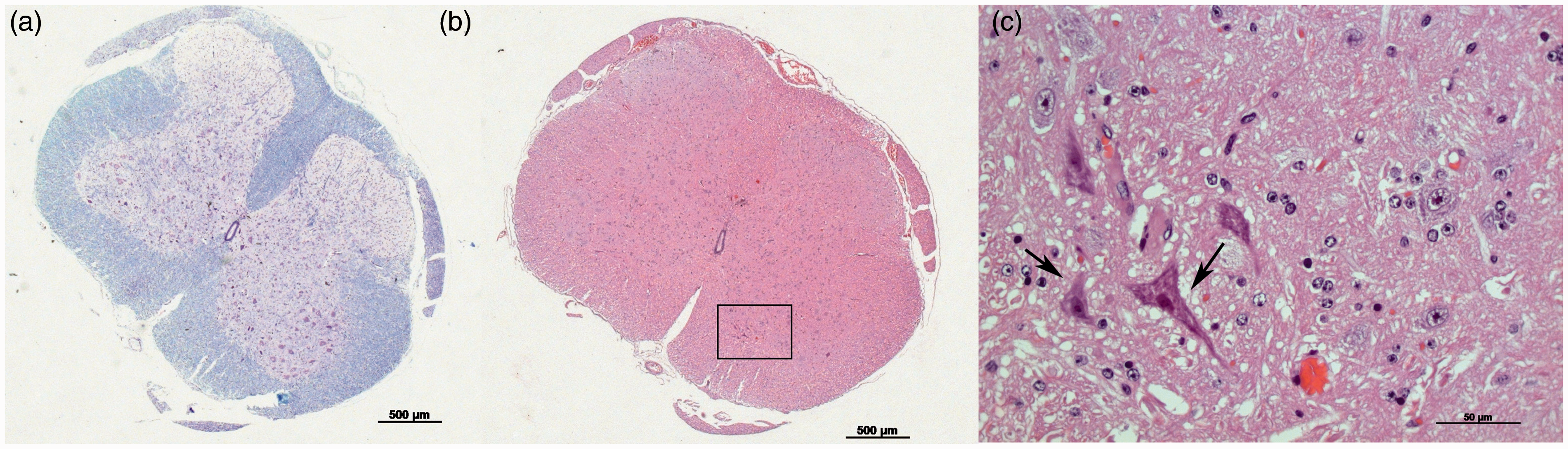
Routine microbiological monitoring (according to the Federation of European Laboratory Animal Science Associations [FELASA] recommendations) did not reveal the presence of any pathogens in the affected guinea pigs; however, antibodies binding to TMEV GDVII virions were detected. In an extended screen, all guinea pigs maintained in the affected unit purchased from breeders CB1 and CB2 (n = 66 in total) and guinea pigs in a separate unit purchased from CB2 (n = 13) as well as animals from our own stock maintained in a separate breeding unit (n = 18) were investigated by serology for antibodies against GDVII. In addition, retired breeders (n = 10) from CB1 directly imported from its breeding unit as well as freshly imported animals (n = 10) from CB2 were tested. GDVII seroconversion was tested in four guinea pigs by repeated testing.
The presence of antibodies was determined by BioDoc using a validated in-house immunofluorescence assay (IFA, specificity 95%) using BHK-21 cells infected with GDVII (ATCC–VR57), performed as described previously. 5 In addition, a commercially available enzyme-linked immunosorbent assay (ELISA; Charles River Laboratories, Wilmington, MA, USA) was used for the detection of antibodies against GDVII. ELISA was carried out according to the manufacturer’s standard protocol. Additionally, serum samples from guinea pigs that were sent to BioDoc from customers for routine monitoring in 2011 were surveyed retrospectively. In total, 506 serum samples provided by 21 different institutions, including universities, research centres, industry facilities, breeding companies, and other diagnostic laboratories were tested.
Ztm has the permission of the local government (Bezirksregierung Hannover) to maintain and breed laboratory animals. Animals are maintained in accordance with current regulations (German Animal Welfare Law, ETS123, Directive 2010/63/EU). All experiments are conducted in line with these regulations, are approved by the local Institutional Animal Care and Research Advisory Committee and permitted by the local authority (Lower Saxony State Office for Consumer Protection, Food Safety, and Animal Welfare Service).
Examination of the affected animal unit in our facility with animals from CB1 and CB2 showed 53 seropositive results out of 66 animals (≈80%) tested by IFA. All 13 animals from CB2 maintained in a separate area were seropositive. To exclude an in-house transmission, 18 animals maintained in a breeding unit from our institution (barrier maintained, no exchange from outside) were also investigated. All tested guinea pigs were seronegative for GDVII. Notably, routine surveillance over several decades of rodents other than guinea pigs at our institution had revealed no indications of TMEV infection. Additionally, IFA using sera from the 10 animals originating from the breeding unit of CB1 revealed no positive results, while all 10 animals from CB2 were strongly positive. To examine seroconversion, guinea pigs from CB1 that were proven to be seronegative by IFA were maintained together with seropositive guinea pigs from CB2. All previously negative animals showed seroconversion four weeks after co-housing. Interestingly, after sanitization of the unit (decontamination using hydrogen peroxide), animals from CB1 transferred to this unit remained seronegative. Retrospective analyses of sera from the German and other European BioDoc customers revealed, together with samples from our institution, a total of 216 out of 506 guinea pig sera (≈43%) to be positive by IFA. In cases where it was possible, positive sera could be traced back to breeder CB2 but not to CB1 in the retrospective analyses. Selected sera that tested positive by IFA were also screened for the presence of anti-GDVII antibodies using ELISA, but no positive results could be detected. We suggest that differences in the composition of antigens used for IFA and ELISA could be the reason for these contradictory results. IFA relies on expression of a variety of antigens during the amplification of virions in permissive cells, making binding of cross-reacting antibodies more likely than the usage of partially purified antigens such as in the ELISA used here. Likewise, we were able to detect seroresponse to rat theilovirus using the GDVII IFA, but not the GDVII ELISA, described above. Notably, Hansen et al. 2 were able to detect antibodies using a commercial ELISA; however, the antigens were not specified. As more detailed information cannot be obtained by the ELISA used here, the reasons for the discrepancies between the two studies remain open to speculation.
Taken together, the results of the present study show a high prevalence of antibodies directed against GDVII in experimentally used guinea pigs, likely caused by viral infections. Additional analyses, including a retrospective study of a large number of sera, underlined the assumption that CB2 animals are the cause of these infections. Though a direct link between serological findings and disease expression in two of the animals was not shown in this study, the high prevalence of GDVII detecting antibodies poses a risk of interference in experimental research. Since serological tests are available, institutional health monitoring programmes can easily be adapted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
