Abstract
Transcranial direct current stimulation (tDCS) constitutes a promising approach for promoting recovery of function after stroke, although the underlying neurobiological mechanisms are unclear. To conduct translational research in animal models, stimulation parameters should not lead to neuronal lesions. Liebetanz et al. recommend charge densities for cathodal stimulation in rats, but parameters for mice are not established. We established tDCS in the wild-type mouse, enabling studies with genetically-engineered mice (GEM). tDCS equipment was adapted to fit the mouse skull. Using different polarities and charge densities, tDCS was safe to apply in the mouse where the charge density was below 198 kC/m2 for single or repeated stimulations. These findings are crucial for future investigations of the neurobiological mechanisms underlying tDCS using GEM.
Transcranial direct current stimulation (tDCS) is a non-invasive method of neurostimulation which is easy to apply. 1 Human clinical studies have shown that tDCS may promote recovery of function after stroke. 2 However, the neurobiological mechanisms underlying this effect are still unclear, preventing its routine use in stroke patients. To elucidate the mechanisms mediating tDCS effects, animal studies are urgently required.
Liebetanz et al. have established tDCS in the rat and have determined parameters that prevent brain lesions due to stimulation. 3 Using this model, we demonstrated the effects of tDCS on neural stem cells and microglia,4,5 both factors representing a promising therapeutic concept for stroke recovery. Future studies with genetically-engineered mice (GEM) will allow further investigation of tDCS. However, standardized surgical protocols and optimal stimulation parameters have not been established in the mouse. In this study, we adapted the tDCS equipment from rats to mice and evaluated its specific safety features. The proposed standardized application protocol is crucial for a reduction in animal numbers as well as for a refinement for future experiments.
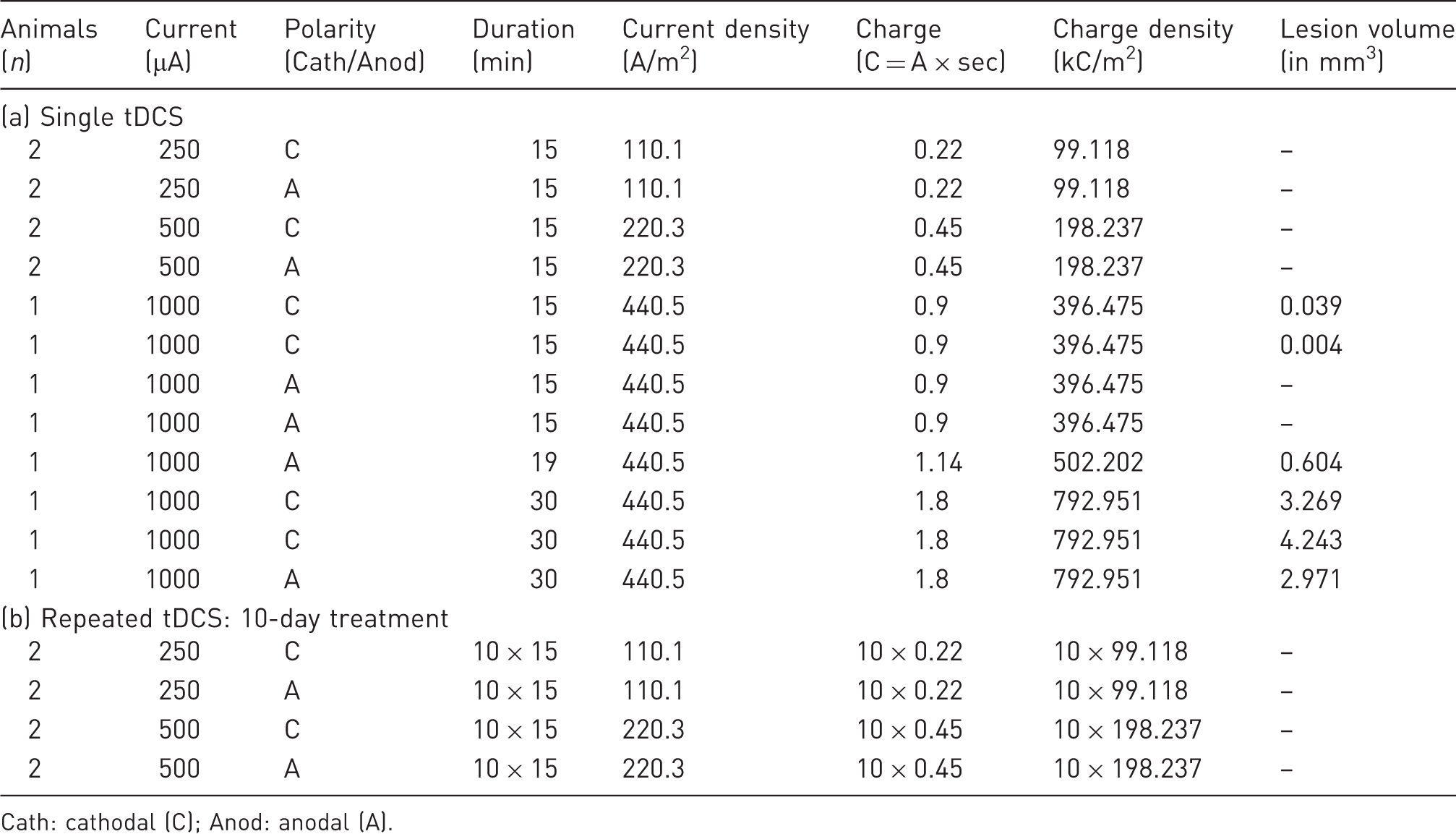
All animal procedures were approved by the local animal care and use committee and governmental authorities (LANUV, #84-02.04.2013.A068). To exclude the influence of hormonal changes on the results, while limiting the number of experimental animals, only male mice were used. Surgery was performed on 24 male C57BL/6JRj mice, aged 10–12 weeks old (Janvier Labs, Le Genest St Isle, France) and weighing 28–35 g, under isoflurane anaesthesia (4% induction; 1–1.5% maintenance) applied via a face mask; and local anaesthesia using bupivacaine hydrochloride subcutaneously (0.25%; Actavis, München, Germany). Carprofen (4 mg/kg; Pfizer, Berlin, Germany) was applied intraperitoneally for post-surgical analgesia. To ensure identical electrode placement for tDCS, custom-made polycarbonate tubes (Medres Medical Research, Cologne, Germany) were stereotactically placed on the skull two days prior to the first stimulation to later fix the electrodes (Figures 1a and b). The implants were similar to those previously used in rats3,4 albeit smaller so as to fit the skull of a mouse with a radius of 0.85 mm, resulting in a contact area of 2.27 mm2 (0.852 × π) between electrode and skull (Figure 1a). The polycarbonate tube was stereotactically placed on the intact skull at bregma AP –1.5 mm, ML + 1.5 mm, and attached to the bone surface via a thin layer of non-toxic dental cement (Super-Bond C&B; Sun Medical, Shiga, Japan). To avoid debris accumulation, a custom-made screw cap sealed the device when it was not being used. After surgery, the mice were transferred to a heated recovery box. For daily tDCS sessions, the polycarbonate tube was filled with saline, and a silver-coated tDCS electrode was inserted. As a counter electrode, a rectangular 1.5 × 2 cm silver-coated sensor electrode (#DENIS01526; Spes Medica, Genova, Italy) was placed under the shaved thorax using electrode contact gel (Spectra®360; Parker, Fairfield, NJ, USA). tDCS was performed under 1% isoflurane using a constant current stimulator (CX-6650; Schneider Electronics, Gleichen, Germany) with a ramp time of 6 s. Mice were randomized to receive either cathodal or anodal stimulation for either 15 or 30 min at various charge densities (σ [C/m2], calculated as charge Q per area A: σ = Q/A) either as a single or repeated treatment (Table 1).
(a) Technical drawing of the polycarbonate tube for transcranial direct current stimulation (tDCS) (in mm). (b) The polycarbonate tube ensured identical placement of the electrode during tDCS. Overview of stimulation parameters and polarity during single (a) and repeated (b) transcranial direct current stimulation (tDCS). Cath: cathodal (C); Anod: anodal (A).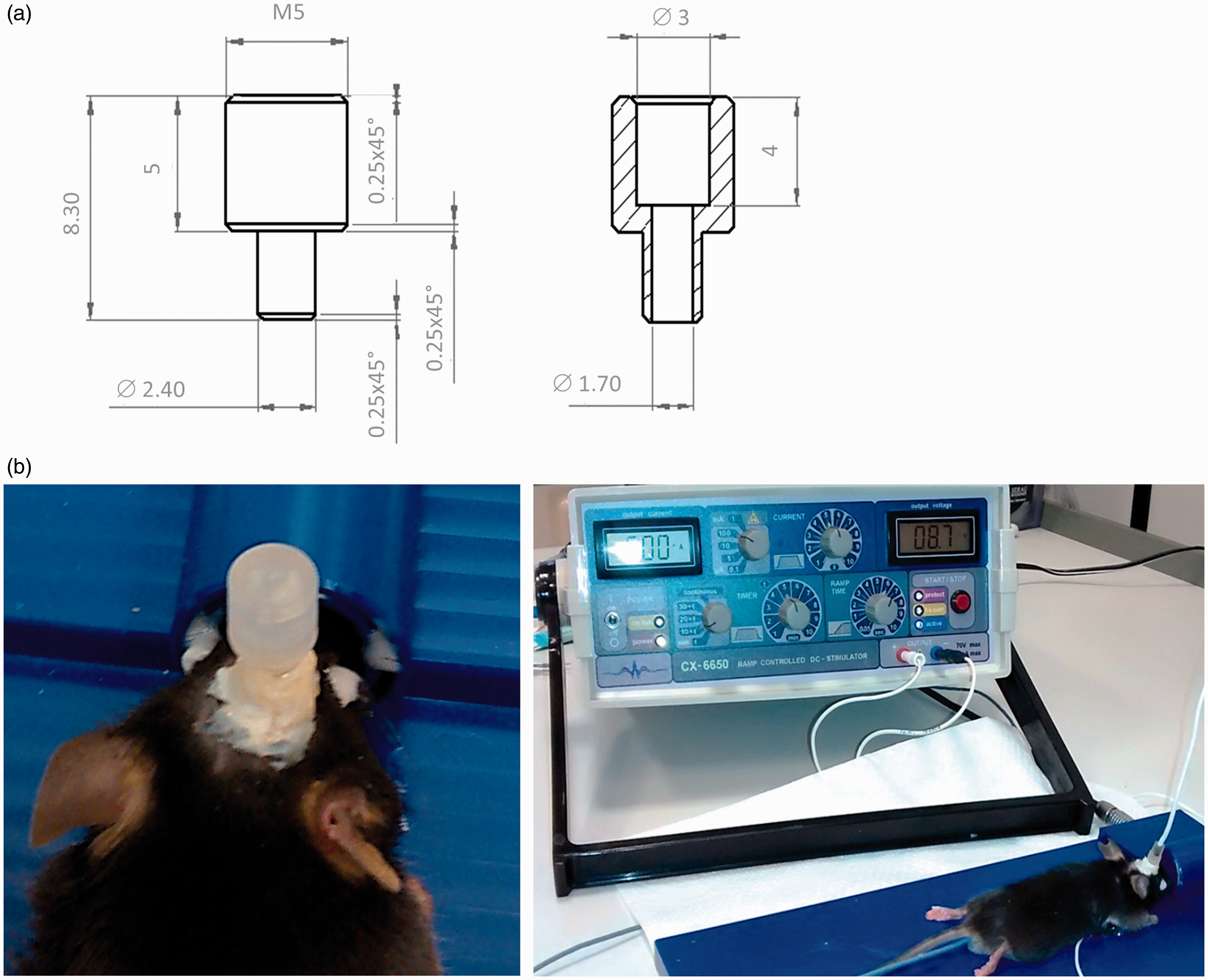
For repeated tDCS, the mice were stimulated daily for five consecutive days, followed by a two-day break, and then for another five consecutive days. This experimental design was adapted from clinical studies with stroke patients. After recovery from anaesthesia the mice were observed for any deficits in motor function. After single tDCS sessions (n = 18), the mice were euthanized by decapitation on the day following. After repeated tDCS (n = 4), the mice were euthanized two days after the last session. Brains were frozen at −80 ℃, cut in 10 µm coronal sections and fixed with 4% paraformaldehyde. Haematoxylin staining was performed to evaluate tissue integrity. To assess neuronal integrity, neurons were stained for NeuN (1:2000; Millipore, Schwalbach, Germany) using an avidin–biotin kit (Vector Labs, Burlingame, CA, USA). Lesion volumes were quantified on NeuN staining using Image J (NIH, Bethesda, MD, USA). The lesion area of each affected slide was measured in µm2. Lesion volumes were calculated as [1/3* (a2 + ab + b2)*h], h being the distance between two measured slides (a and b).
The mice were randomly selected to receive either cathodal (n = 12) or anodal (n = 12) stimulation with charge densities of between 99 and 793 kC/m2 (Table 1). Eight cathodally and eight anodally stimulated animals were sacrificed on the day following a single tDCS session. At a charge density of 99 or 198 kC/m2, no lesions were found after tDCS of either polarity (Figure 2a, Table 1). Increasing the cathodal charge density to 396 kC/m2 resulted in focal neuronal loss (0.039 mm3 and 0.004 mm3) directly underneath the epicranial electrode (Figure 2b). Charge densities of 502 to 793 kC/m2 led to even larger necrotic lesions (0.604 mm3 to 4.243 mm3), partly spreading to the contralateral hemisphere (Figures 2c and d). To assess the effects of repeated tDCS using non-lesional charge densities, four cathodally and four anodally stimulated mice were subjected to 10 sessions of tDCS with 99 or 198 kC/m2, simulating application in a rehabilitation environment. These repeated stimulations over 10 days evoked neither neuronal lesions nor neurological deficits.
(a) Neuronal staining (NeuN) demonstrates neuronal integrity after single transcranial direct current stimulation (tDCS) with 198 kC/m2 (arrowhead at stimulation tube’s position). (b) Single tDCS at 396 kC/m2 led to selective neuronal loss underneath the electrode. (c) At 792 kC/m2, haematoxylin staining displayed large coagulation necrosis extending to the contralateral cortex. (d) Neuronal staining of (c) demonstrates extensive neuronal loss in the same region. Scale bars represent 300 µm.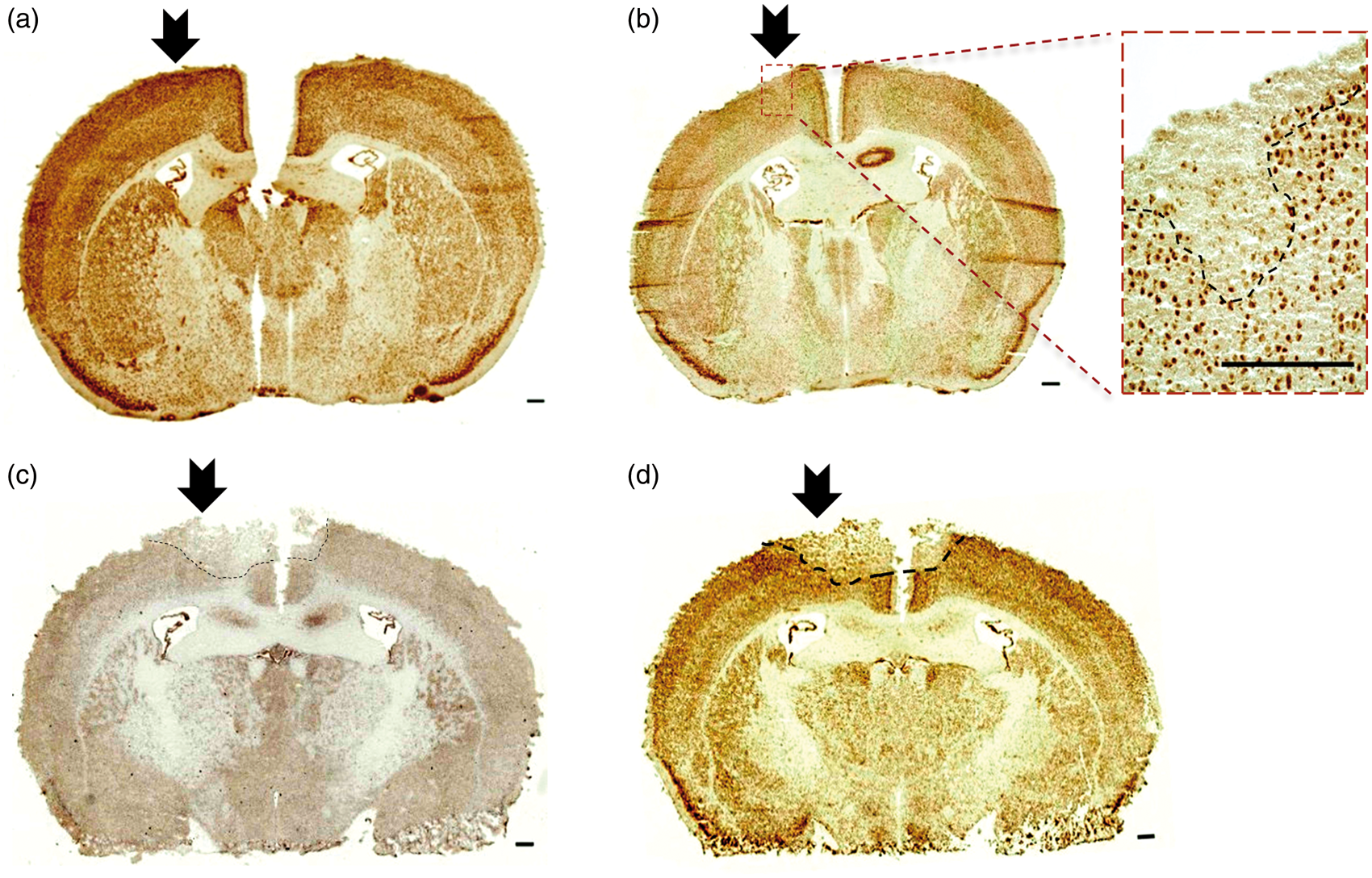
We established non-lesional tDCS in mice by repeatedly applying a maximum charge density of 198 kC/m2 tDCS, which simulates rehabilitation in a clinical setting.
Liebetanz et al. have identified a lesion threshold for rats at 52.4 kC/m2, while in humans, lower charge densities of 0.5 kC/m2 are typically used. 3 We applied comparatively high charge density in mice because the clinical effects of tDCS depend on both the polarity and the charge density applied, 6 with tDCS effects being more pronounced at higher charge densities.4,5 The main factor contributing to neuronal loss above the respective threshold is the development of heat between the electrode and extracranial tissue. Thus, it is conceivable that different species possess different lesion thresholds due to differences in skull anatomy. Factors such as tissue perfusion or skull temperature were recently confirmed to have major effects on scalp conductivity and intracranial current density during tDCS. 7
Aiming for a reduction in animal numbers, we did not explicitly establish a formal lesion threshold since this would have required numerous additional animals. This is the first study to report a standardized protocol for tDCS in the mouse using commercially available equipment, enabling international standardization and comparison, thereby presenting a significant refinement to this methodology. Additionally, the protocol allows intra-individual and longitudinal studies, which serves to reduce animal numbers in future studies, thus improving animal welfare.
Footnotes
Acknowledgements
We thank Claudia Drapatz and Wilfried Haider for excellent technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the ‘Marga und Walter Boll-Stiftung’ (#210-12-12).
