Abstract
Sustained-release formulations of analgesic drugs are promising alternatives to repeated drug injections. Here, we compared a sustained-release formulation of buprenorphine (SB, 2.2 mg/kg) with a standard protocol of three injections of buprenorphine (Temgesic, 0.1 mg/kg/8 h) in mice. Buprenorphine serum concentration and analgesic action (thermal sensitivity) were determined in healthy mice. Additionally, the pain relief properties of both protocols were assessed after laparotomy using physiological and ethological measures of pain and recovery. Serum concentrations and thermal sensitivity tests indicated duration of action of at least 4 h (but less than 8 h) with the Temgesic protocol, and 24–48 h with SB. Behavioural and clinical parameters indicated at least partial pain relief after surgery for both protocols. Observed side-effects of buprenorphine independent of the protocol were increased activity, disturbed circadian rhythm and several abnormal behaviours. A tendency for decreased food and water intake as well as body weight reduction was also seen. Body weight decreased significantly in animals that received three injections of Temgesic, regardless of whether surgery was performed or not (P = 0.015; P = 0.023), hinting at a stress response towards this repeated intervention. In conclusion, an application interval of 8 h (Temgesic) appears too long and might lead to repeated periods with insufficient analgesia in animals undergoing lasting and/or substantial pain after surgery. In comparison to the standard protocol, SB provided a long-lasting, assured analgesia without possible stressful repeated injections in a standard surgical model, with only limited and acceptable behavioural side-effects.
Reliable alleviation of pain is essential to assure the humane treatment of laboratory animals in invasive experimental procedures and to ensure high-quality scientific outcomes. Pre-requirements of effective pain management are sufficient intrinsic activity and an effective application system for the analgesic drug of choice. Buprenorphine – a fast acting and potent opioid – has been used in humans to treat moderate to severe pain. 1 Also very popular in veterinary medicine, buprenorphine has become one of the analgesic drugs most commonly used nowadays in post-surgical treatment of laboratory rodents.1–4
A typical buprenorphine application regimen for the treatment of post-surgical pain in rodents involves two or three subcutaneous injections a day, while the duration of treatment depends on the type and severity of surgery. 5 Nevertheless, application doses and reported duration of analgesic activity for buprenorphine in rodents published in the recent literature vary considerably. In mice, doses of 0.05–0.75 mg/kg or higher are reported to deliver efficient analgesia for 3–7 h,6–9 and in rats 0.05–0.5 mg/kg appears to have an analgesic effect for 5–8 h.7,10–13 It is therefore to assume that if the drug is injected only every 12 h, a fall of drug blood concentration below optimal therapeutic values between two applications is possible.
Additionally, repeated post-surgical injections of analgesic drugs have been criticised as potentially stressful procedures in laboratory rodents. Indeed, the necessary restraint is known to have a distinct impact on physiological and behavioural parameters that might hint at stress even in healthy animals14–16 and might also evoke additional, or increase existing, pain in animals with fresh surgical wounds and tissue trauma. As additional stressors can have detrimental effects on animal well-being and recovery from procedures,15,17,18 alternative application methods in laboratory animal medicine would be of considerable value.
Several attempts have been made to overcome these problems and assure the continuous and stress-free administration of buprenorphine analgesia in laboratory rodents. Oral administration, for example via drinking water,19,20 medicated food pellets 21 or Nutella paste,11,22 as well as sustained release of the drug via osmotic pumps 23 or a solvent injectable system containing the drug, have been presented recently using rats and mice.6,24–26 These application routes appear as promising alternatives to repeated injections, and sustained-release formulations, with slow and long-lasting continuous release, promise controlled delivery and assure a therapeutically effective blood concentration of the drug.
Recently, sustained-release formulations of buprenorphine have become commercially available on the US market (Animalgesics® for Mice, Animalgesic Labs Inc, Millersville, MD, USA; Buprenorphine HCl CIII SR, Wildlife Pharmaceuticals Inc, Windsor, CO, USA). The efficiency and duration of the analgesic activity of these products have been confirmed in mechanical and thermal nociceptive tests as well as in two surgical models in rats.24,25 However, only two studies have analysed analgesic efficiency and duration with a thermal nociception test in healthy mice.6,26 To date, European customers have no access to these products. Therefore we compared a sustained release formulation of buprenorphine manufactured in-house using a drug carrier developed by a Swiss producer (HumanVet, Lausanne, Switzerland) with a standard repeated injection protocol of buprenorphine after laparotomy in female mice using physiologically and ethologically relevant measures of pain, well-being and recovery. Bioavailability during the observation period was assured with measurements of serum concentrations of the drug. Additionally, to allow comparison with other studies we assessed effective analgesic action with a thermal nociception test in healthy, naïve mice.
Materials and methods
Ethics statement
The animal housing and experimental protocols were approved by the Cantonal Veterinary Office, Zurich, Switzerland, under licence No. ZH 181/2012, and were in accordance with Swiss Animal Protection Law and conform to European Directive 2010/63/EU of the European Parliament and of the Council on the Protection of Animals used for Scientific Purposes and to the Guide for the Care and Use of Laboratory Animals. 27
Animals and standard housing conditions
The animals were 112 female C57BL/6 J mice obtained at the age of 6–8 weeks (Charles River, Sulzfeld, Germany). The animals’ health status was monitored by a health surveillance programme according to the Federation of European Laboratory Animal Science Associations (FELASA) guidelines. The mice were free of all viral, bacterial, and parasitic pathogens listed in the FELASA recommendations, except for Helicobacter species. 28
All the animals were housed in groups of three to six animals for three weeks prior to testing in our animal room. The animals were kept in Eurotype III clear-transparent plastic cages (Techniplast, Hohenpeißenberg, Germany) with autoclaved dust-free sawdust bedding and hay. They were fed a pelleted and extruded mouse diet (Kliba No. 3436, Provimi Kliba, Kaiseraugst, Switzerland) ad libitum and had unrestricted access to drinking water. The light/dark cycle in the room consisted of 12/12 h with artificial light (approximately 40 lux in the cage). The temperature was 21 ± 1℃, with a relative humidity of 55 ± 10%.
Analgesic protocols
As representative of a commonly accepted analgesic protocol, Temgesic (Temgesic® solution, 0.3 mg/mL; Reckitt Benckiser, Wallisellen, Switzerland), a widely used water-soluble buprenorphine medicinal product, was injected subcutaneously at a dose of 0.1 mg/kg body weight buprenorphine once (T1) or every 8 h for 3 times (T3). Shortly before injection, Temgesic was diluted in sterile NaCl (0.9%) so that the injection volume was 2 µL/g body weight.
A sustained-release drug carrier, composed of different pharmaceutically approved excipients (TempoVet; HumanVet), was mixed in-house with a buprenorphine base (Johnson Matthey, Royston, UK), and was injected once subcutaneously at a dose of 2.2 mg/kg body weight buprenorphine (SB). Sterile injection solutions were prepared freshly to be injected with a volume of 20 µL per mouse, which was approximately 1 µL/g body weight.
Experimental design, experimental animal housing conditions and data acquisition
Animals were used in several experimental set-ups if scientifically justified. Treatment allocation was random. Mice were assigned either (a) to behavioural assessment after surgery or respective control procedures (e.g. analgesic treatment only) and then to analgesiometric testing, or (b) first to analgesiometric testing followed by behavioural assessment. Animals were allowed to recover between both experiments for at least two weeks in standard group housing. Two weeks after the second experiment, mice that did not receive any analgesic treatment till this time point were used to determine the serum concentration of the two different buprenorphine formulations (c).
Behavioural assessment
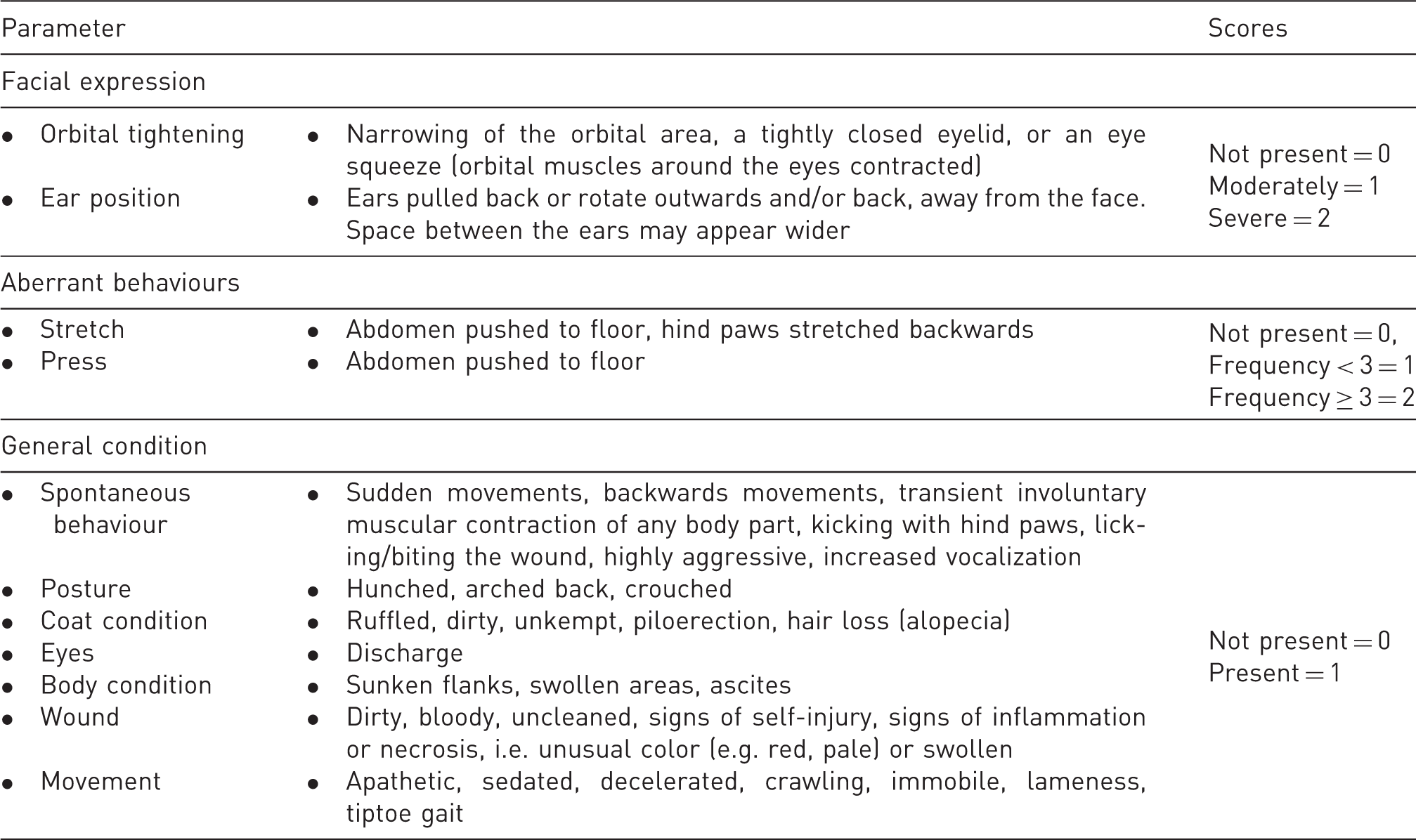
Scoring system for clinical investigation and behaviour-based pain assessment. In total a maximum score of 15 can be reached. Observation time per mouse was 3 min.
For each experimental housing condition (burrowing set-up or observation cage), eight mice were allocated to one of six experimental groups: control (no manipulation); T3 only (three Temgesic injections only, observation cage) or T1 only (one Temgesic injection only, burrowing set-up); SB only (one injection of sustained-release buprenorphine only); OP: surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); OPT3 or OPT1: surgery + anaesthesia + T3/T1 (mice underwent anaesthesia and surgery with three/one Temgesic injections) or OPSB: surgery + anaesthesia + SB (mice underwent anaesthesia and surgery with one injection of sustained-release buprenorphine).
Surgical procedure: The experiment began with a subcutaneous injection of the respective analgesic or saline. Two hours after injection, the animals were transferred to an operating theatre and anaesthetized with sevoflurane (Sevorane; Abbott, Baar, Switzerland). Mice in surgery groups underwent a one-side sham embryo transfer. Surgery was completed within 3–4 min in the surgery groups. Anaesthesia lasted 6–8 min in all groups. Animals were allowed to recover fully from anaesthesia for one hour before subsequent behavioural observation (see below) was undertaken.
Clinical investigation
Clinical investigation and behaviour-based pain assessment in a small observation chamber lasted 3 min per animal and was performed at one hour after the animal had undergone surgery and 12 and 24 h after first investigation. Scoring was performed according to a system documenting the general condition,
30
indicators of abdominal pain, and changes in facial expression. Symptoms were converted into scores according to a scoring system (Table 1) and summarized to a composite score. Additionally, urination and number of faeces droppings were assessed.
Burrowing test
The burrowing test determines burrowing performance and can be used as a simple method to assess post-surgical impairment in mice.
29
Cages were video-recorded from above using infrared sensitive cameras (Ikegami, Tokyo, Japan). The latency of each animal to burrow was measured by analysis of video recordings taken during the 12 h following the return of the animal to its home cage.
Activity analyses
Observation cages were video-recorded from above using infrared sensitive cameras for home cage activity analyses with automated tracking software (EthoVision XT 7; Noldus Information Technology, Wageningen, The Netherlands). The distance (in cm) moved by the centre point of the animal was assessed to measure static behaviours as well as horizontal locomotion.
Body weight, and food and water intake
Body weight progression as well as daily food and water intake was measured 24 h before and 24 h after the procedures in all the animals housed in special observation cages.
Analgesiometric testing
Mice were housed in groups of four in standard cages as described above. Cages were assigned randomly to one of three groups: saline: one saline injection; T1 or SB. The experiment began with a subcutaneous injection of the respective analgesia or saline at 11:00 h (first half of the light phase). The animals were then returned to their home cages and four animals from each experimental group were tested at one of four time points after injection (2, 4, 8, 24 h). Nociceptive thresholds were measured by the hot-plate method.
31
The mice were placed individually on the heated surface of a hot-plate analgesic meter (Columbus Instruments, Columbus, OH, USA), maintained at 55℃ and latency was recorded from the start to the endpoint of jumping, licking or shaking hind paws (cut-off time 40 s).
Serum concentration
The mice were housed in groups of four in standard cages as described above. Cages were assigned randomly to one of three groups: T1, T3 or SB. Subcutaneous injections of the respective analgesia started at 11:00 h. The animals were then returned to their home cages and four animals from each experimental group were bled at one or two of the time points (T1: 2, 4, 6, 8, 10, 24 h; T3: 2, 4, 6, 8, 10, 16, 24 h; SB: 2, 4, 6, 24, 48 h). Blood was sampled by sublingual vein puncture under sevoflurane anaesthesia. Blood was centrifuged and the serum was stored at −20℃ until further analysis. Buprenorphine serum concentrations were determined by ultra-performance liquid chromatography tandem mass-spectrometry by a commercial laboratory (MVZ Labor Dessau GmbH, Dessau-Roßlau, Germany).
Investigators performing behavioural testing were blinded for all treatment groups. For a detailed description of the experiments performed see Supplementary data (all supplementary material can be found online with this article at http://lan.sagepub.com).
Statistical analysis
Statistical analyses were performed with SPSS 22 software (IBM, Armonk, NY, USA). All data were tested for normal distribution and homogeneity of variance (Shapiro–Wilks, Levene’s test).
Mean and standard error of the mean were calculated for all parameters. The Kruskal–Wallis H-test was performed for composite scores to reveal significant differences in means. One way analyses of variance (ANOVA) were performed for burrowing latencies, % change body weight (% change compared with baseline measurements two days before experiments), weight of food pellets and water bottles, as well as for centre point distance moved during 24 h of activity analyses, followed by post hoc tests (Bonferroni) to show significant differences between groups. Absolute body, food and bottle weights, taken over 24 h measurements, were additionally compared with baseline measurements with a paired t-test.
Mean withdrawal responses from hot-plate tests were analysed using a univariate general linear model with time and group as fixed factors, and a Bonferroni post hoc test was used to analyse significant differences between groups.
Significance for all statistical tests was established at P ≤ 0.05.
Results
Behavioural assessment Clinical investigation
Observation of the animals’ outer appearance revealed no physical complications from the surgical procedures performed or of the analgesic treatment, e.g. skin irritation at the injection site. In general, quantities of urine and faeces excreted during the short observation bouts were relatively low at the first observation after recovery from anaesthesia (0 h) but increased 12 h and 24 h later.
Behavioural signs of pain and distress were observed frequently in animals undergoing surgery with or without analgesic treatment, which led to increased composite scores compared with control animals, especially at time point 0 h. Compared with animals that underwent surgery without pain treatment, composite scores were reduced when buprenorphine was given (Figure 1).
Means (±SEM) of composite scores taken after recovery from anaesthetic procedures or at a comparable time point in control and analgesia-only groups, as well as 12 h and 24 h later. Kruskal–Wallis H-test revealed significant differences in means at 0 h (P = 0.0001), but not at 12 h and 24 h (P = 0.053; P = 0.168). *Significant (P ≤ 0.05) differences between experimental groups. Group n = 8. T3: three Temgesic injections only; SB: one injection of sustained-release buprenorphine only; OP: surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); OPT3: surgery + anaesthesia + T3; OPSB: surgery + anaesthesia + SB.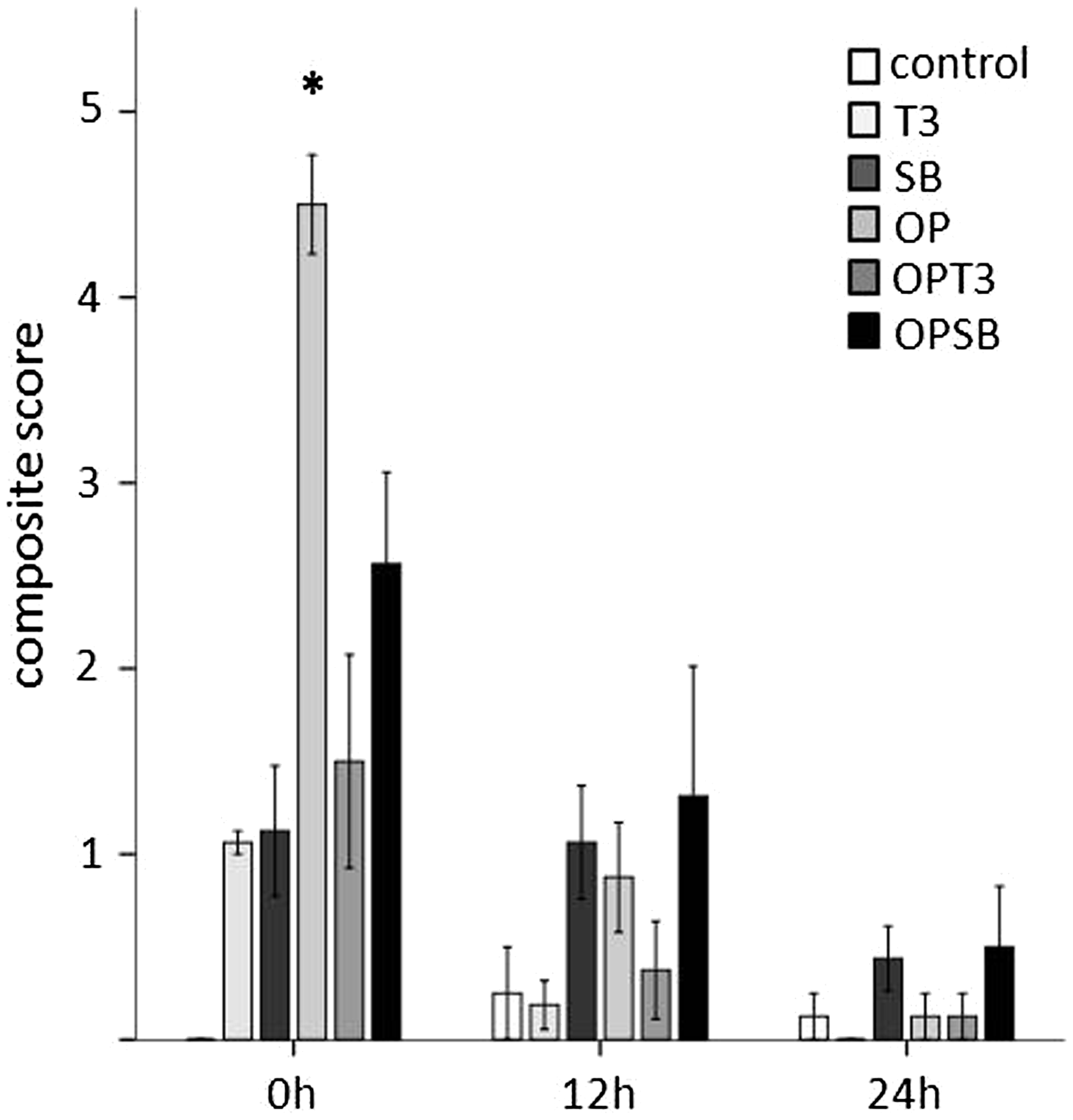
While narrowing of the orbital area or tightly closed eyelids, hunched posture, and immobility were observed mainly in those of the groups that underwent surgery, several behavioural and postural changes occurred in groups that received buprenorphine injections only. Frequently observed effects were hyperlocomotion, circling and tiptoe gait (walking on the tip of the toes instead of the pad), as well as a bent tail, curved over the back of the animal. Rarely were animals observed biting or manipulating their incision wounds. Additionally, muscle contractions of back and flank muscles, rounded backs and instances of a behaviour we named ‘lay down’, i.e. a short bout where mice pressed their thorax and abdomen gently to the floor without additional muscle contractions, were displayed.
Burrowing test
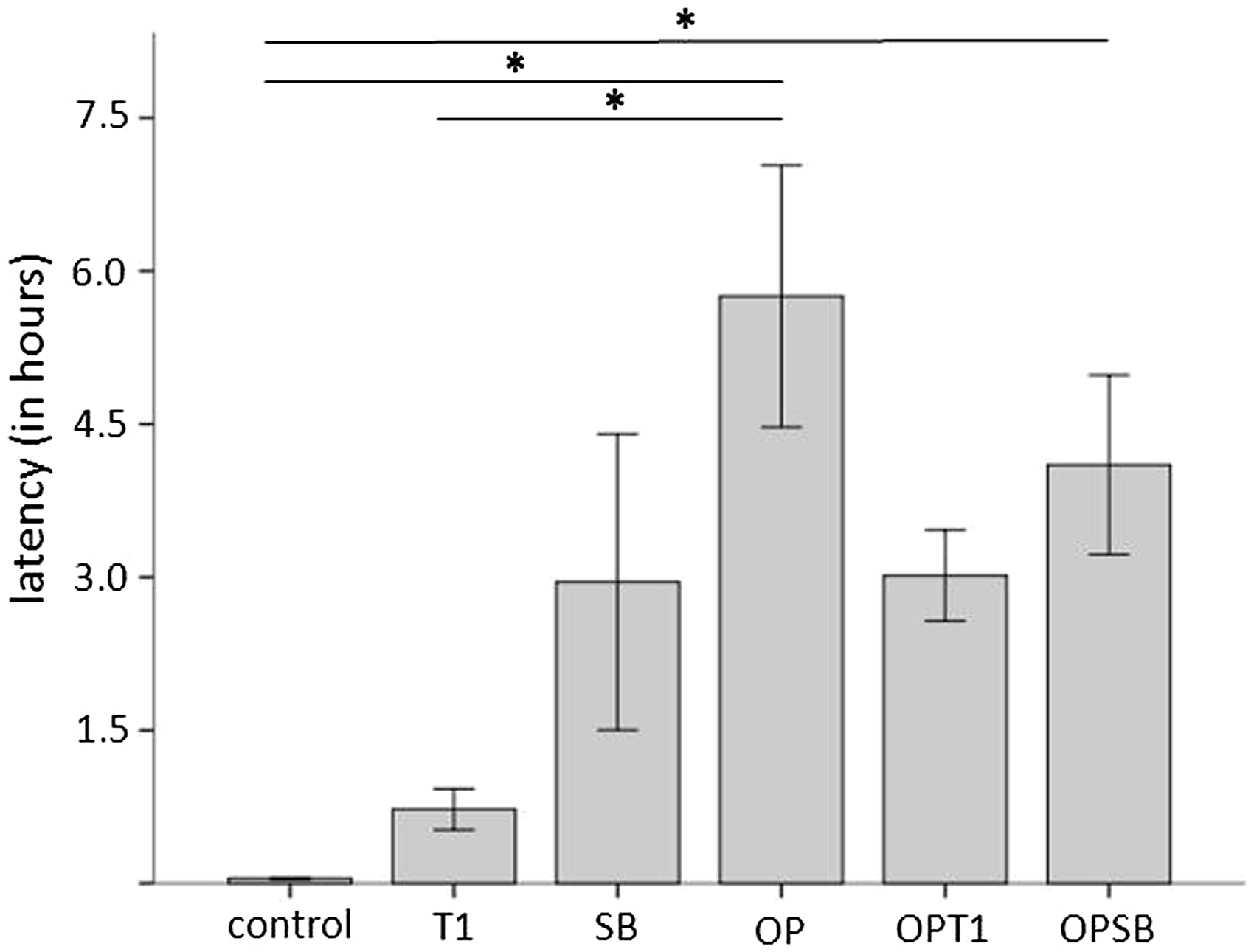
Burrowing latencies in untreated control animals were short, with a mean of 2.7 ± 0.5 min, and were prolonged significantly after surgery without pain treatment (5.3 ± 1.2 h; P = 0.001). Buprenorphine treatment alone increased latency to burrow compared with control animals. Animals that underwent surgery with buprenorphine treatment showed latencies comparable with buprenorphine-only treatments, which were shorter in duration than those of animals that underwent surgery without pain treatment (Figure 2).
Activity analyses Means (±SEM) of latencies to burrow. Mean latency to burrow was prolonged significantly in OP and OPSB groups compared with control animals (P = 0.001; P = 0.033). T1 animals showed significantly shorter latencies than OP animals (P = 0.002). *Significant (P ≤ 0.05) differences between experimental groups. Group n = 8. T1: one Temgesic injection only; SB: one injection of sustained-release buprenorphine only; OP: surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); OPT1: surgery + anaesthesia + T1; OPSB: surgery + anaesthesia + SB.
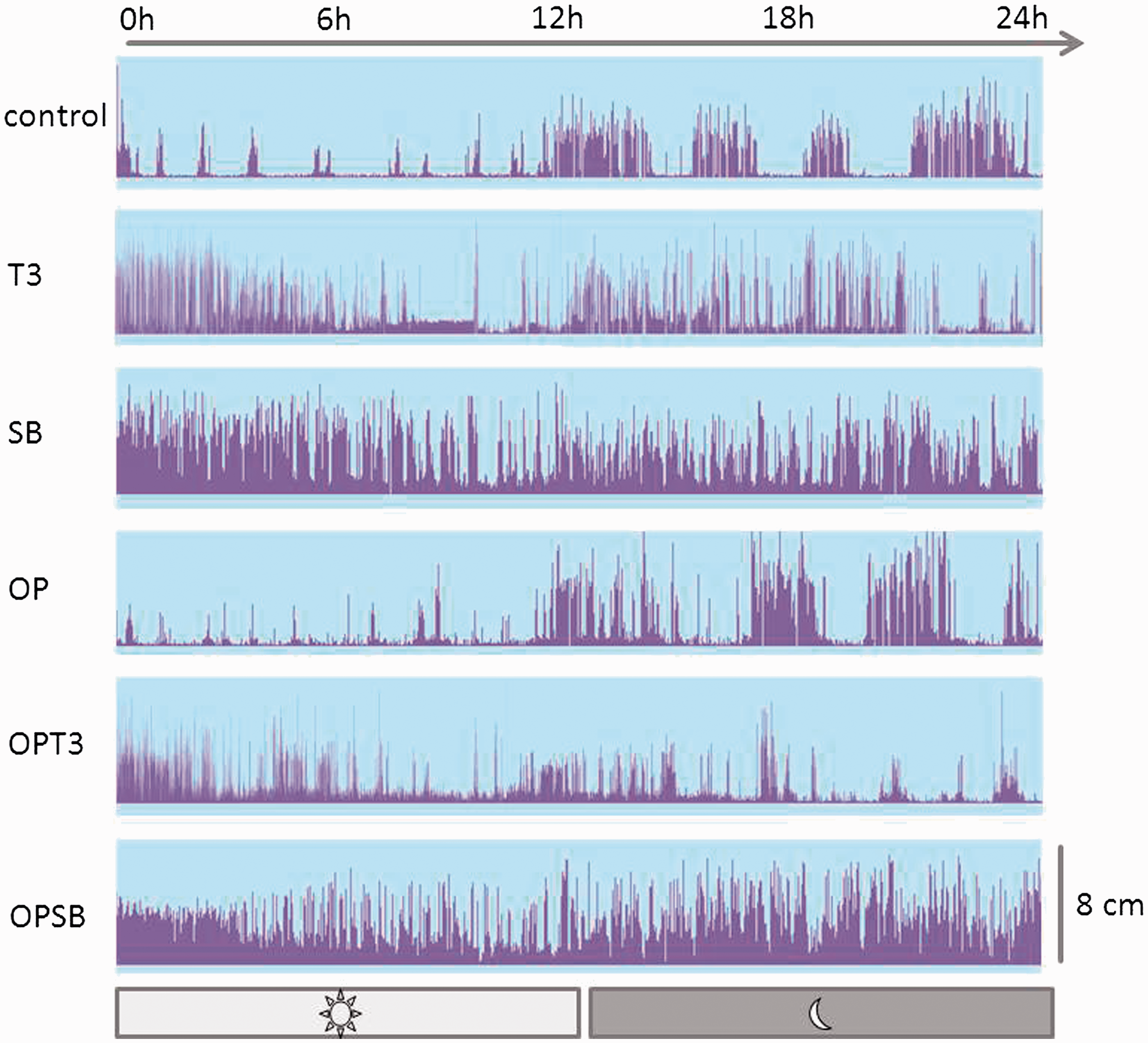
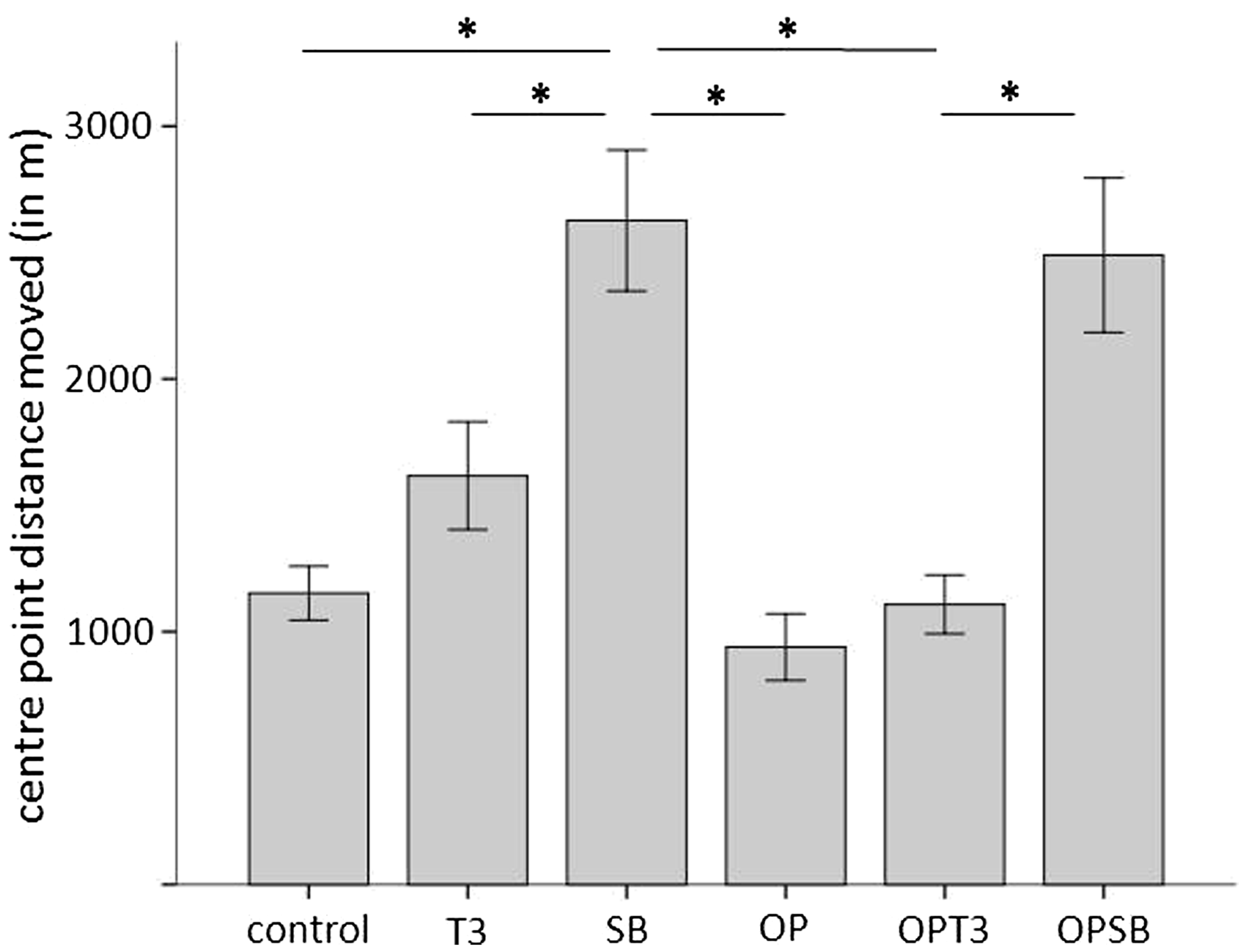
While non-treated control animals showed a mean distance of centre point movement of 1152 ± 107 m, distance moved was increased by buprenorphine to a maximum of 2626 ± 278 m in SB animals. This increase was due mainly to bouts of high mobility during the first 6 h, while activity levels approximated those of control animals during the following 18 h. Surgery decreased overall distances and led to high levels of immobility – a trend that was absent with SB treatment (Figures 3 and 4).
Body weight, food and water intake Visualization of activity. Representative data of individual animals. Distance moved between different samples (in cm) is shown for 24 h of observation. While circadian rhythm were flattened but remained after surgery, activity was slightly decreased. After buprenorphine injection, circadian activity was lost and activity increased distinctly. T3: three Temgesic injections only; SB: one injection of sustained-release buprenorphine only; OP: surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); OPT3: surgery + anaesthesia + T3; OPSB: surgery + anaesthesia + SB. Means (±SEM) of distance moved during 24 h of automated analysis. Post hoc tests revealed several significant differences between treatment groups: distance moved in SB animals was increased significantly compared with control (P = 0.0001), T3 (P = 0.021), OP (P = 0.0001) and OPT3 (P = 0.0001); OPSB animals moved significantly longer distances compared with control (P = 0.001) and OPT3 (P = 0.0001) animals. *Significant (P ≤ 0.05) differences between experimental groups. Group n = 8. T3: three Temgesic injections only; SB: one injection of sustained-release buprenorphine only; OP: surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); OPT3: surgery + anaesthesia + T3; OPSB: surgery + anaesthesia + SB.
Body weight: Mice weighed 20–24 g at 24 h before the experiments started. Compared with baseline measurements, all experimentally-treated groups showed decreases in body weight at 24 h after experiments, while non-treated control animals showed small increases in body weight (Figure 5a). Compared with their absolute baseline weight 24 h before experiments, the absolute weight of animals receiving three injections of buprenorphine (Temgesic), with or without surgery, was decreased significantly; OPT3 (P = 0.015) and T3 (P = 0.023).
(a) Means (±SEM) of percentage body weight change at 24 h: compared with control animals, OPT3 animals showed distinctly decreased weights (P = 0.012). Compared with their baseline weight 24 h before experiments, weights of T3 and OPT3 animals were significantly decreased (P = 0.023; P = 0.015). (b) Means ( ± SEM) of food intake at 24 h: animals that received three injections of buprenorphine for surgery (OPT3) consumed significantly less food compared with control (P = 0.011) and OP animals (P = 0.026). Compared with their absolute food intake 24 h before experiments, absolute food intake of OPT3 animals was decreased significantly (P = 0.002). (c) Means ( ± SEM) of water intake at 24 h: no significant differences occurred between groups (P = 0.208). Compared with the absolute water intake 24 h before experiments, absolute water intake of OPSB animals was decreased significantly (P = 0.033). *Significant (P ≤ 0.05) differences between experimental groups. #Significant (P ≤ 0.05) differences between baseline and after experiment. Group n = 8. T3: three Temgesic injections only; SB: one injection of sustained-release buprenorphine only; OP: surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); OPT3: surgery + anaesthesia + T3; OPSB: surgery + anaesthesia + SB.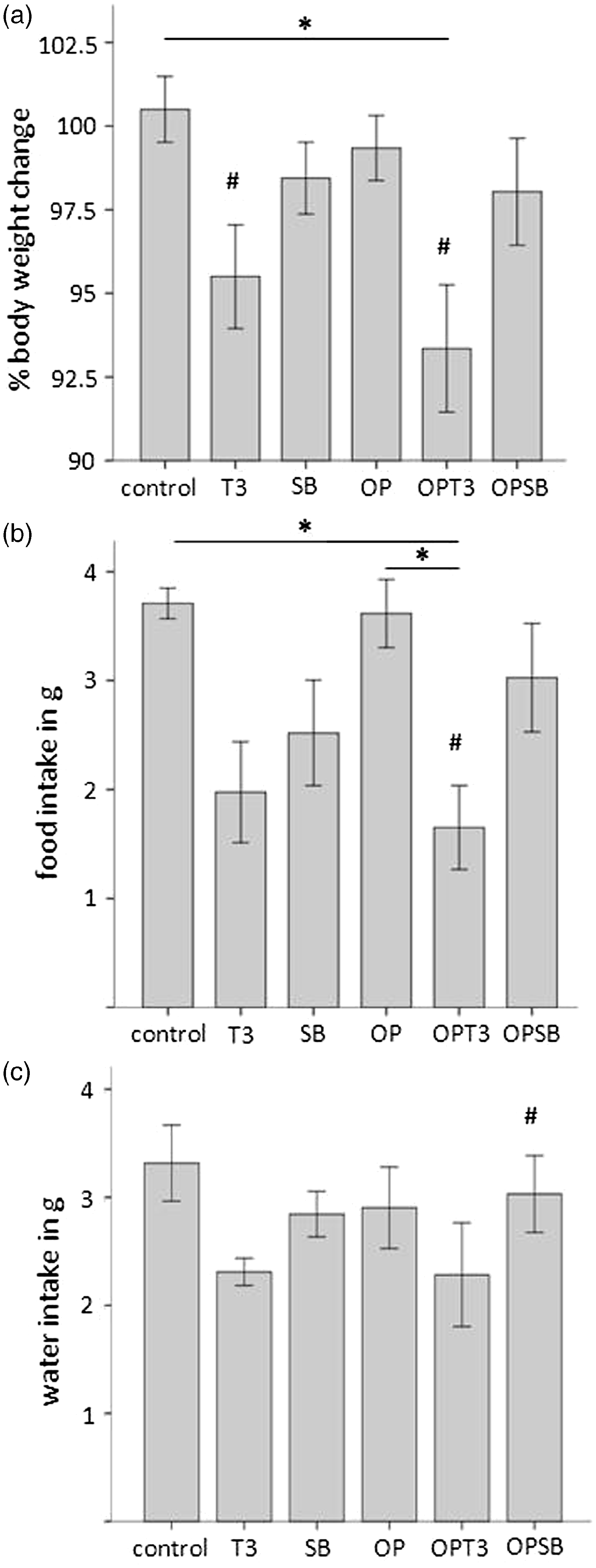
Food intake: Control mice ate in general 3.7 ± 0.3 g a day. Food intake was decreased after buprenorphine injection, regardless of protocol or whether surgery was performed. T3 and OPT3 animals showed the largest decreases (Figure 5b). Compared with their absolute food intake 24 h before experiments, absolute food intake of animals receiving three injections of buprenorphine (Temgesic) after surgery (OPT3) was decreased significantly (P = 0.002).
Water intake: Control mice drank in general 3.1 ± 0.2 g (excluding a mean evaporation/loss of 1.9 g) a day. Water intake decreased after all treatments compared with baseline, but this tendency was not significant (P = 0.208, Figure 5c). Compared with the absolute water intake 24 h before experiments, absolute water intake of OPSB animals was decreased significantly (P = 0.033).
Analgesiometric testing
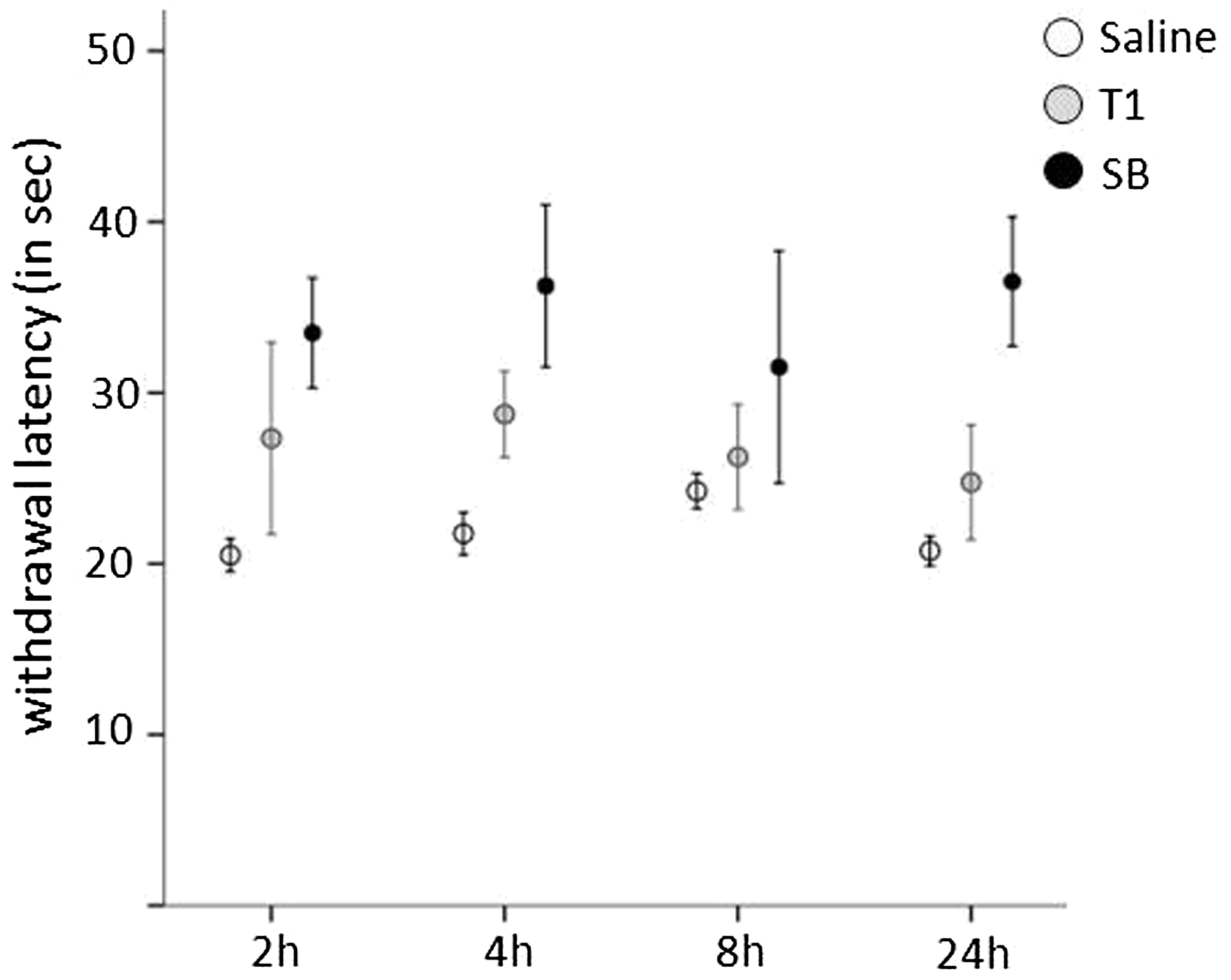
SB-treated animals showed long latencies throughout the observation period of 24 h, while T1 animals returned to latencies comparable with those of saline-injected mice within 4–8 h (Figure 6). No main effect of the ‘Time’ factor could be shown (P = 0.915, F = 0.171), while the ‘Treatment group’ factor had a significant effect on withdrawal latency (P = 0.0001, F = 13.428). No significant interactions between the two main factors, Time and Treatment, were found (time * group: P = 0.884, F = 0.385). Post hoc tests revealed a significant difference between the saline and SB groups (P = 0.0001) as well as between the T1 and SB groups (P = 0.012).
Serum concentration Mean (±SEM) withdrawal response latency to thermal stimulus in mice treated with either saline, sustained-release buprenorphine (SB) or Temgesic (T1) at the time points shown. SB-treated animals showed long latencies throughout the observation period of 24 h, while T1 animals returned to latencies comparable with those of saline injected mice within 4–8 h. No main effect of the ‘Time’ factor could be shown (P = 0.915, F = 0.171), while the ‘Treatment group’ factor had a significant effect on withdrawal latency (P = 0.0001, F = 13.428). No significant interactions between the two main Time and Treatment factors were found (time * group: P = 0.884, F = 0.385). Post hoc test revealed a significant difference between the saline and SB groups (P = 0.0001) as well as between the T1 and SB groups (P = 0.012). Group n = 4.
Serum concentrations of buprenorphine after injection of the sustained-release formulation were high compared with concentrations after single or repeated injections of Temgesic (T1 and T3); these high concentrations could be seen up to 24 h after injection. In T1 and T3 animals, serum concentrations decreased rapidly from 1.5 ± 0.3 ng/mL or 1.8 ± 0.1 ng/mL at 2 h after first injection or second injection to 0.4 ± 0.1 ng/mL at 4 h after injection; a second decrease was seen at 8 h to 0.2 ± 0.1 ng/mL (Figure 7).
Mean (±SEM) serum concentration of buprenorphine in mice given either Temgesic (T1) or sustained-release buprenorphine (SB) at the time points shown. T3 animals were injected with Temgesic every 8 h. In humans, the target blood concentration for effective buprenorphine treatment is 0.5–0.7 ng/mL; in rodents levels of 1 ng/mL have been proven to be effective. These values are indicated by grey dotted lines. Group n = 4.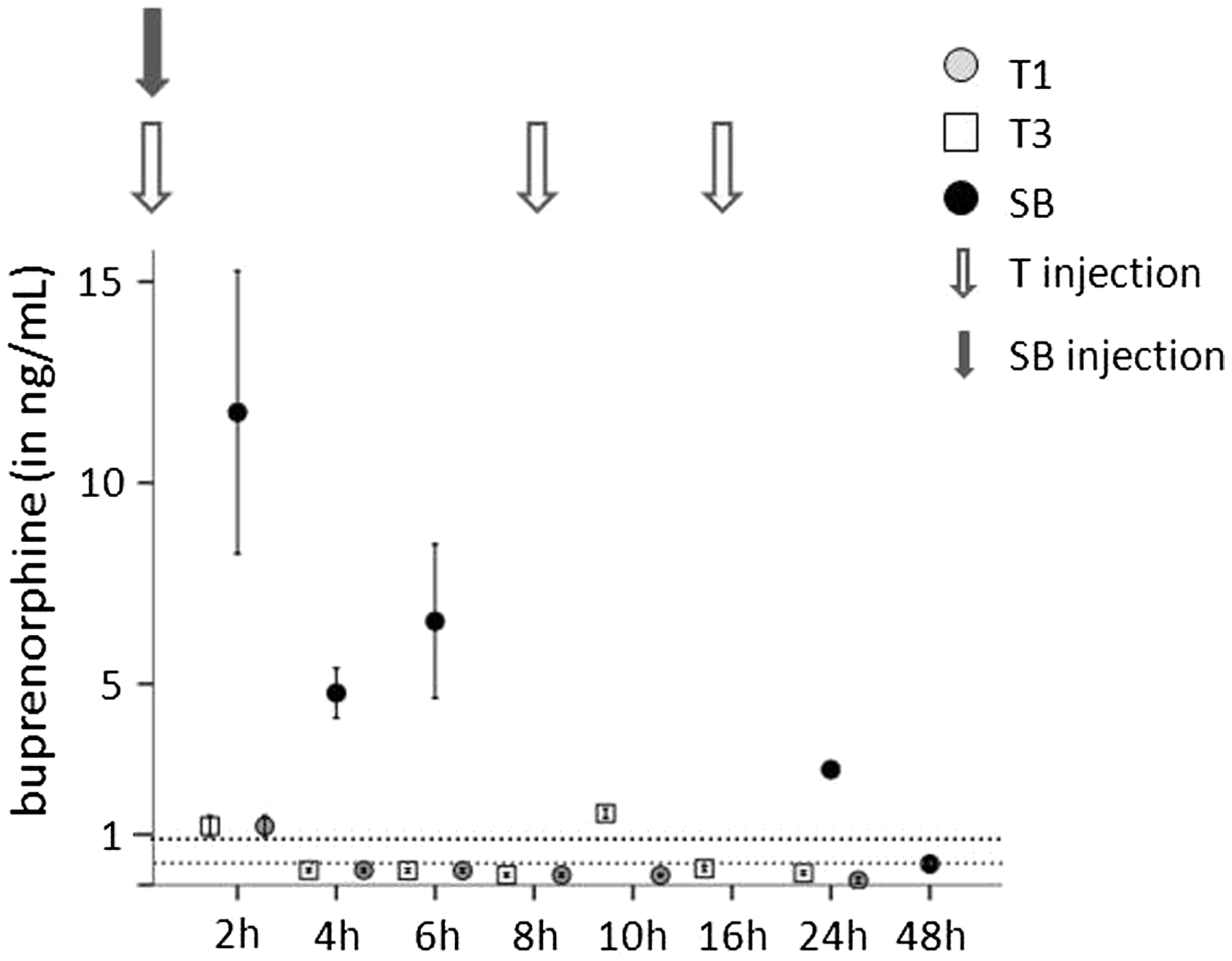
Discussion
The aim of this study was to characterize a new buprenorphine sustained-release formulation for laboratory mice with a targeted duration of action of 24 h. Analgesic efficiency, duration of action and side-effects were analysed in a clinically relevant surgical model and compared with a standard protocol of repeated subcutaneous buprenorphine (Temgesic) injections. Both protocols proved to have an analgesic effect in a classical thermal sensitivity test and resulted in drug serum concentrations assumed to be therapeutically effective, even though the intensity and duration of the effect clearly differed. Both analgesic protocols seem to relieve pain, at least partially, also in the surgical model used. Nevertheless, side-effects of different intensity occurred which have to be considered when choosing an appropriate analgesic protocol.
Buprenorphine is the opioid analgesic most widely used in laboratory rodent post-surgical treatment, although large variabilities in dosage and means of application can be found in the literature. In the present study, we used a protocol that is applied routinely, consisting of three subcutaneous injections of 0.1 mg/kg Temgesic in 8 h intervals or one single subcutaneous injection of 0.1 mg/kg Temgesic.
In humans, the target blood concentration of buprenorphine leading to effective analgesia is considered to be approximately 0.5 ng/mL or higher. 32 Determination of the serum concentration of buprenorphine in our mice at 2 h after injection of 0.1 mg/kg Temgesic resulted in values exceeding this concentration. This was true regardless of the injection scheme, as this result was seen at the 2 h time point in Temgesic single injection (T1), and also 2 h after the second Temgesic injection, which is at the 10 h time point in the repeated injection group (T3). Four hours after Temgesic injection, serum concentrations decreased to just beneath the target value of 0.5 ng/mL. These serum concentrations proved to be still sufficient to induce a distinct analgesic effect, as hot-plate withdrawal latencies were prolonged compared with saline-treated animals at 4 h after injection. However, we observed a drop in serum concentration to 0.2 ng/mL at the 8 h sample time point, accompanied with a decrease of withdrawal latency in the hot-plate test, with values moving toward those of saline-treated animals. We therefore assume that the time frame between 4 and 8 h marks the probable end of the analgesic action of Temgesic at the dose used. These conclusions are in line with data of Gades and colleagues, 7 who concluded a duration of action of up to 5 h after injection of 2.0 mg/kg buprenorphine in mice. In light of this evidence, an application interval of 8 h appears to be too long and might lead to repeated periods without, or with insufficient, analgesia in animals undergoing lasting pain after surgery, a concern also expressed by others (see examples6,7).
Support for a general analgesic effect from the subcutaneous injection of 0.1 mg/kg Temgesic and the sustained-release formulation without any indication of duration of action is also given by the results of the burrowing test and the visual clinical investigation and behaviour-based pain assessment performed.
Along with the behavioural side-effects of buprenorphine such as circling, tiptoe gate and bent tail, a slight and non-significant increase of activity was observed in animals receiving repeated injections of 0.1 mg/kg Temgesic only – a widely known side-effect of buprenorphine.33–36 While activity after surgery without pain treatment decreased as was expected, 37 activity increased slightly when animals received Temgesic after surgery. Nevertheless, it cannot be determined whether this was due to the analgesic action or was a side-effect of Temgesic. Increased activity also affected behavioural circadian rhythm. A flattened circadian rhythm could be interpreted as a sign of pain or reduced well-being,17,18,38 but in our experiment it was more likely to be a side-effect of buprenorphine.
Interestingly, we observed marked decreases in body weight and correlated food intake, compared with baseline values as well as with values of control animals, in animals that received three injections of 0.1 mg/kg Temgesic with or without surgery (OPT3 and T3). While general decreases of body weight and food intake are expected after surgery, and reductions of food intake and body weight gain are well-known side-effects of buprenorphine,3,26,33,39,40 the decrease of these parameters was distinctly stronger than in those animals that received only one injection of buprenorphine sustained-release formulation (SB and OPSB). Keeping in mind that the dose of buprenorphine given was much higher in the latter animals, resulting in substantially higher serum concentrations, this effect was probably induced by the repeated application of Temgesic. It is known that routine procedures like restraint or intraperitoneal injections may induce stress in mice, as indicated by, e.g. increased urinary cortisone, heart rate or body temperature for 1–2 h following manipulation.14–16 Repeated restraint and subcutaneous injection of animals suffering from tissue trauma due to surgery might have had detrimental effects on recovery in our study.
The sustained-release buprenorphine protocol consisted of one pre-surgery injection and proved to be a very convenient protocol offering reliable long-term analgesic action. Even though we caught and observed the animals frequently for monitoring purposes no repeated restraint and injections as in the Temgesic protocol were needed.
As we assumed a slightly delayed onset of effect in the sustained release protocol, the formulation was given 2 h before surgery was performed. This waiting time was shown to be sufficient as hot-plate withdrawal latencies 2 h after injection were distinctly prolonged compared with saline-injected animals. Additionally, at 2 h, the mean value of serum concentration of animals that received sustained-release buprenorphine was more than 10 times higher than in Temgesic-treated animals. After a rapid decrease, serum concentrations remained substantially higher compared with Temgesic-treated animals, reaching concentrations of over 1 ng/mL at 24 h and still maintaining 0.5 ng/mL at 48 h, thereby exceeding our targeted serum concentration until the end of the observation period. In consideration of these high concentrations, an assured and continuous analgesic effect for up to 48 h could be assumed. By implication, these serum concentrations proved to be effective, and hot-plate withdrawal latencies were distinctly increased compared with values of saline- and Temgesic-treated animals for up to 24 h. These long-acting times were indeed expected as other studies have found comparable durations of action with even lower doses of buprenorphine in sustained-release formulations in thermal and mechanical sensitivity tests.6,24–26
In addition to the observed circling behaviour, and the noticeably erected tail, a behavioural abnormality frequently observed in animals that received sustained-release buprenorphine was a behaviour we termed ‘lay down’. This behaviour occurred also in animals without pain, and therefore has to be carefully separated from pain behaviours like ‘press’ or ‘stretch’, which are indicative of abdominal pain and, in contrast to ‘lay down’, are accompanied by slight muscle contractions.
Compared with all other experimental groups, including Temgesic-treated animals, activity after injection of sustained-release formulation increased significantly and persistently for 24 h, resulting in a disruption of circadian rhythm. An increase in activity has been described in mice receiving sustained-release buprenorphine. 26 It can be assumed that the marked intensity of this side-effect is due to the high dosage of buprenorphine in the sustained-release formulation used in our study.
As described above, a well-known side-effect of buprenorphine, also given in sustained-release formulations,24–26 is a decrease in body weight and food/water intake. But the observed decrease in these parameters in SB animals compared with baseline values was small compared with the pronounced reduction in T3 and OPT3 animals. Given that the buprenorphine dosage and serum concentrations were even higher in the SB and OPSB animals than in the animals that received repeated injections of buprenorphine, we suggest that the reduction in application frequency in the SB protocol minimized handling stress and could therefore be considered to be a methodological refinement.
Other than the above described effects, side-effects such as gross pathological changes, skin irritation at the injection site or encapsulations were not observed upon standard gross macroscopic evaluation during necropsy (data not shown).
Conclusions
Our results suggest that three injections of Temgesic to treat post-surgical pain might be insufficient, as the duration of action after each injection of Temgesic lasts from 4 to less than 8 h. Thus, when injections are performed at 8 h intervals, animals might suffer periods without, or with insufficient, pain relief. Nevertheless, the substantial impact of repeated injection on recovery and well-being, as evidenced by the marked body weight decrease in animals that received three injections of Temgesic, suggests that even more frequent injections are not to be recommended. By contrast, the sustained-release formulation offers a long-lasting, assured blood concentration of drug, resulting in a sound antinociceptive effect and a suggested relief of post-surgical pain for 24–48 h without causing additional stress to the animal. As mouse strains may differ in respect to nociception as well as in the response to different analgesics these results have yet to be verified for other strains.
In conclusion, the side-effects of buprenorphine seen in this and other studies might be acceptable when pain relief in experiments with more than mild pain has to be assured. Therefore, besides intensity and duration of analgesic action, stress induced by the application method and possible behavioural and physiological side-effects should be taken into account when choosing the appropriate analgesia protocol for mice. A careful balancing of the impacts of possible side-effects against the expected pain impact, has to be a mandatory component of animal experiment design.
Footnotes
Acknowledgements
We thank Professor Ulrich Zeilhofer and William Ralvenius for generously providing advice and resources.
Funding
This work was sponsored by grants from the Federal Food Safety and Veterinary Office (Bern, Switzerland) and the Swiss Laboratory Animal Science Association.
Declaration of conflicting interests
Arnaud Tourvieille is an employee of HumanVet, Lausanne, Switzerland. The authors, Paulin Jirkof, Paolo Cinelli and Margarete Arras, declare that they have no conflicts of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
