Abstract
Small bowel motility analyses using magnetic resonance imaging (MRI) could reduce current invasive techniques in animal studies and comply with the ‘three Rs’ rule for human animal experimentation. Thus we investigated the feasibility of in vivo small bowel motility analyses in mice using dynamic MRI acquisitions. All experimental procedures were approved by the institutional animal care committee. Six C57BL/6 mice underwent MRI without additional preparation after isoflurane anaesthetization in the prone position on a 4.7 T small animal imager equipped with a linear polarized hydrogen birdcage whole-body mouse coil. Motility was assessed using a true fast imaging in a steady precession sequence in the coronal orientation (acquisition time per slice 512 ms, in-plane resolution 234 × 234 µm, matrix size 128 × 128, slice thickness 1 mm) over 30 s corresponding to 60 acquisitions. Motility was manually assessed measuring the small bowel diameter change over time. The resulting motility curves were analysed for the following parameters: contraction frequency per minute (cpm), maximal contraction amplitude (maximum to minimum [mm]), luminal diameter (mm) and luminal occlusion rate. Small bowel motility quantification was found to be possible in all animals with a mean small bowel contraction frequency of 10.67 cpm (SD ± 3.84), a mean amplitude of the contractions of 1.33 mm (SD ± 0.43) and a mean luminal diameter of 1.37 mm (SD ± 0.42). The mean luminal occlusion rate was 1.044 (SD ± 0.45%/100). The mean duration needed for a single motility assessment was 185 s (SD ± 54.02). Thus our study demonstrated the feasibility of an easy and time-sparing functional assessment for in vivo small bowel motility analyses in mice. This could improve the development of small animal models of intestinal diseases and provide a method similar to clinical MR examinations that is in concordance with the ‘three Rs’ for humane animal experimentation.
Small animal imaging has gained an important role in basic research used in non-invasive in vivo monitoring of the animals in ongoing studies, providing fast, secure and reliable information and thereby reducing the number of animals necessary for obtaining statistically significant information.1–3 The most commonly applied small animal imaging modalities are microcomputed tomography (microCT) and small animal magnetic resonance imaging (MRI). Compared with computed tomography in humans, even the most modern microCT devices need to apply relatively high doses of ionizing radiation. 4 Moreover MRI provides a much higher soft tissue contrast which is of special importance in investigations of the intestinal tract. Therefore, in longitudinal studies small animal MRI seems to be the method of choice.
Most commonly, mice are used as a small animal model; less common models include the use of rats, hamsters and non-small animals. 5 Yet MRI investigations especially of the intestinal tract in mice remain challenging. The technique has proved its clinical feasibility and reliability in humans where it typically depends on ultrafast sequences such as fast spin echo or single-shot fast spin echo sequences in a coronal orientation, repetitively acquired over the same location and thus displaying motility.1,6–12 Motility analyses in humans have been demonstrated to constitute a reliable method for investigations in patients suffering from inflammatory bowel diseases (IBDs). These measurements seem to serve as a biomarker of disease activity.7,8,11,12. Yet to the best of our knowledge no study has investigated whether the concept established in humans could be used as well for functional analyses of the intestinal tract in mice and provide corresponding information. In consequence animal models today rely on invasive techniques used to estimate small bowel motility. 12 Similar to the invasive techniques previously used in humans, these methods commonly include manometric approaches to monitor intraluminal pressure or methods using exteriorization of (parts of) the animal’s small bowel, such as the Trendelenburg method which becomes more challenging with smaller animals. In this method the small intestine needs to be fixed outside the animal’s body to acquire video imaging and electromyographs (EMGs) of the small intestine.12–15 These invasive techniques should be avoided by application of non- or less-invasive radiological examinations in compliance with the ‘three Rs’ (reduction, refinement, and replacement) rule for humane animal experimentation. Radiological techniques have so far remained limited to the use of 2D fluoroscopy and complex mathematical models of small bowel motility used in experimental MRI modalities, as has been proposed recently. 16
Translational research providing a fast transition from preclinical testing into clinical studies on humans is an essential part of today’s clinical research. The translation in this case might not only be limited to one way. Just as they are widely established in clinical research protocols, small bowel motility analyses could also use small animal imaging of mice, thus helping to improve preclinical research. In this study, we investigated the feasibility of a small bowel motility examination in mice which has proved feasible and established in humans11,17 for examining intestinal physiology.
Animals
For the purpose of this feasibility study a total number of six C57BL/6 mice (one year old) were used (Harlan Laboratories, Ittingen, Switzerland). No particular intestinal preparation of the animals for the experiment was needed. Animals received their regular food and water supply prior to the experiment which was supplied to them at a 24 h access point. All experimental procedures were approved by the institutional animal care committee and all procedures were performed in strict accordance with the National Institute of Health instructions for the Guide to Care and Use of Laboratory Animals.
Materials and methods
MRI
During MRI all the animals were anaesthetized using isoflurane (Attane; Minrad Inc, Buffalo, NY, USA) with a concentration of 1–1.5% isoflurane in 100% oxygen. A MR compatible physiology unit (respiratory monitoring system [Small Animal Instruments Inc, Stony Brook, NY, USA]) was applied to monitor anaesthesia of the animals which conformed with the standard operating procedure (SOP) for MRI examinations of mice at our institution. The mice were placed in a prone position on the scanner bed and kept warm using a pad with a continuous warm water supply. The prone position was chosen to reduce potential dislocation out of the plane of the small bowel. Imaging was performed using a Bruker 4.7 T PharmaScan 47/16 US (Bruker Biospin GmbH, Ettlingen, Germany) with a maximum gradient strength of 375 mT/m, a maximum slew rate of 3375 T/(ms), and a linear polarized hydrogen whole-body mouse radiofrequency coil. After gradient echo localizers in three spatial directions, small bowel motility was assessed with a balanced steady-state free precession sequence (true fast imaging in steady precession [trueFISP]) in coronal orientation using the following parameters: TE 2 ms, TR 4 ms, flip angle 60°, field of view 30 × 30 mm2, matrix size 128 × 128, slices 5, slice thickness 1 mm, spacing 0 mm, 1 average. The stack of slices was placed to cover the abdomen caudal to the liver reaching to the pelvic floor and covering the abdomen ventrally to dorsally. For each slice 60 frames with a time resolution of 0.5 s were acquired. The scan time in total accumulated to approximately 4 min per mouse.
Image analysis
The cine MRE image sequences were transferred to a picture archiving and communication station (PACS) for analyses of small bowel motility (Osirix, Version 5.02; Osirix Imaging Software, Geneva, Switzerland). 6 Image quality was assessed for depiction of the small bowel without relevant breathing artefacts, banding artefacts or image distortions. Small bowel motility was evaluated in two different small bowel segments per mouse, randomly covering the different abdominal quadrants. To assure randomization, small bowel segments were depicted and chosen indeterminately of the anatomical origin (ileum or jejunum). Small bowel motility was then assessed as previously described by one reader (SB >2 years experience in dedicated small bowel motility research), measuring the small bowel diameter change of the chosen segments over time, thus displaying motility.
Initially out of the five spatial coronal image sequences covering the abdomen ventrally to dorsally, the spatial image sequence was selected which displayed the chosen small bowel loop best in its centre (highest luminal diameter). Using this technique we were able to ensure that the measurement was always performed as closely to the center of the lumen as was technically possible. Measurement of the small bowel diameter was then done orthogonally to the long axis of the small bowel, resulting in cross-sectional diameters of the small bowel over time. The linear regions of interest (ROIs) were defined on the first slice, then propagated through the complete stack and adjusted for diameter on each slice in order to depict the lumen diameter exactly. In particular, as the cine sequences were not acquired in a breath hold but in mice with free breathing, the measured location shifted slightly from slice to slice according to the breathing-induced motion of the diaphragm and they had to be manually adjusted for each single image. As a consequence of the high frequency rate of the mice, the breathing-related motion resulted in some scintillation that had to be corrected using the described manual adaptation of the measurement lines in each slice. The distances obtained were plotted over time to undergo further analyses of small bowel motility parameters. The resulting curves were analysed for the following parameters, as has been previously described:10,12 contraction frequency per minute (cpm), maximal contraction amplitude (maximum to minimum [mm]), luminal diameter (mm). Furthermore, the amplitude–diameter ratio was calculated. This derived measure describes the individual quotient of the contraction amplitude divided by the mean luminal diameter measured in the identical segment. It can be useful for assessing the relative luminal occlusion which occurred during contractions in coronal orientation. 12 The time needed to perform the small bowel motility analyses was noted.
Statistical analysis
Quantitative variables were expressed as the mean ± standard deviation (SD). To provide an overview of the results box-and-whisker plots marking the upper and lower 25th quartiles and the mean were plotted out of the individual results of each mouse. The commercially available GraphPad Prism (version 5.0; GraphPad Software Inc, LA Jolla, CA, USA) statistical software was used for all analyses.
Results
Image quality
The MRI scans were successfully completed in all the animals. All trueFISP datasets provided excellent depictions of the small bowel without relevant breathing artefacts, banding artefacts or image distortions. Slight opposed-phase signal loss was visible at the interface between the small bowel wall and peritoneal fat, and this provided an excellent delineation of the intestinal structures with a typical size of the small bowel wall ranging between 250 and 400 µm (Figures 1a–f). The in-plane resolution of the sequence was 234 × 234 µm. Therefore, a reasonable evaluation of bowel wall motility was easily possible. However, we observed the typical banding artefacts near to the tissue air interface at the diaphragm, which is a well known feature of trueFISP sequences applied in humans and is caused by magnetic field inhomogeneity between the lungs and abdomen.
Example of a segmental small bowel motility analysis: (a) Magnification of a chosen small bowel segment for motility analyses. (b–e) 2D T2w-balanced steady-state free precession sequence (trueFISP) in coronal orientation (TE 2 ms, TR 4 ms, flip angle 60°, field of view 30 × 30 mm2, matrix size 128 × 128, slices 5, slice thickness 1 mm) of a representative small bowel motility analysis performed in a single segment of one of the mice. The white arrow marks the segment used for the analysis and its diameter change over time in different slices, the white lines show the measurement of the small bowel diameter. (f) The diagram displays three exemplary motility curves/contraction patterns measured using MRI motility analyses as the diameter change plotted over time. The sinusoidal temporal behaviour of the bowel contractions is mostly visible depending on the frequency and height of the spontaneous contractions in the evaluated small bowel segments (mouse 1–3).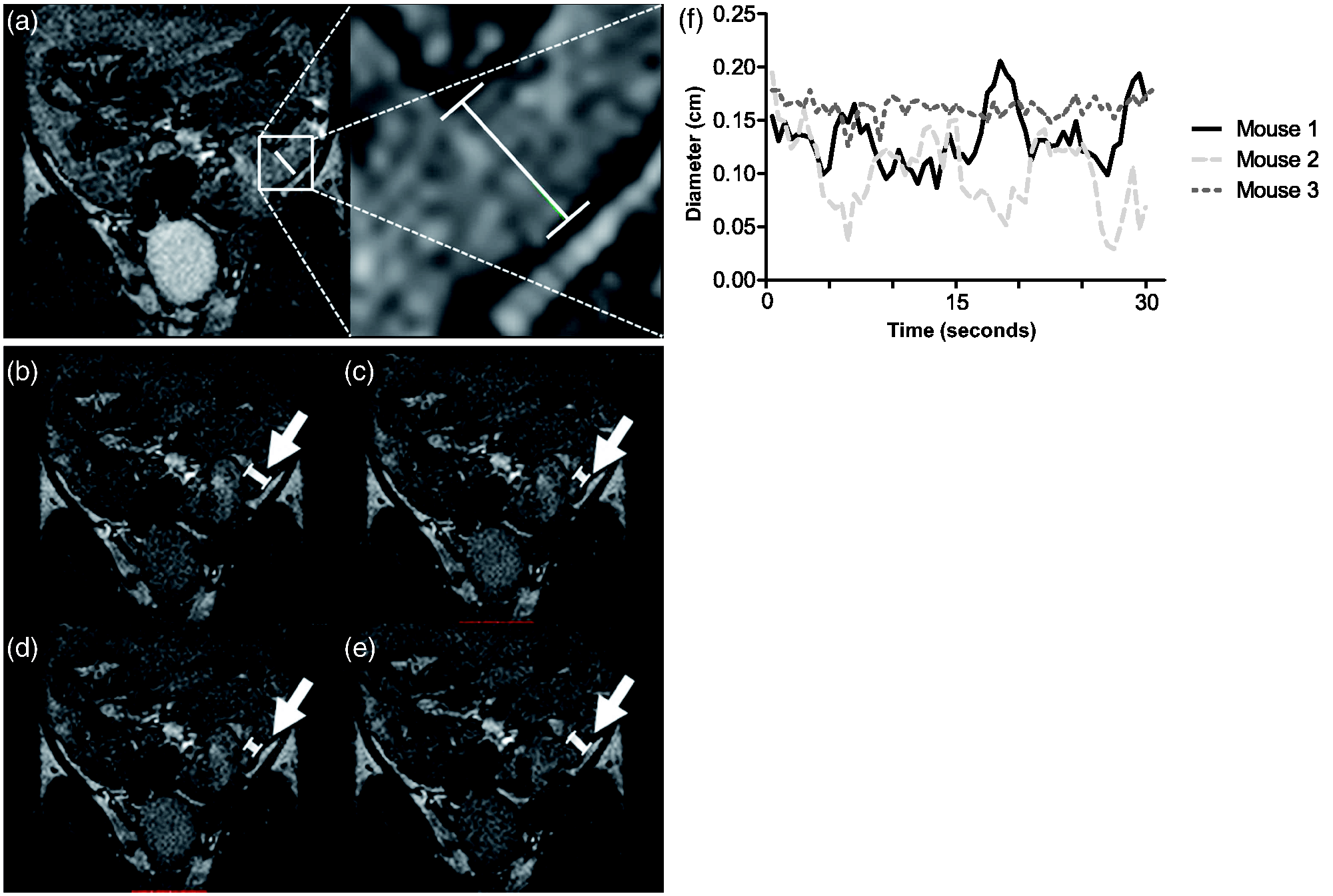
Small bowel motility parameters
The overall mean small bowel contraction frequency of the C57/BL6 mice was 10.67 cpm (SD ± 3.84, range 4.00–16.00) (Figures 2a and 2b, Table 1).
Results of quantitative small bowel motility parameters. SD: standard deviation, cpm: contractions per minute, mm: millimetre.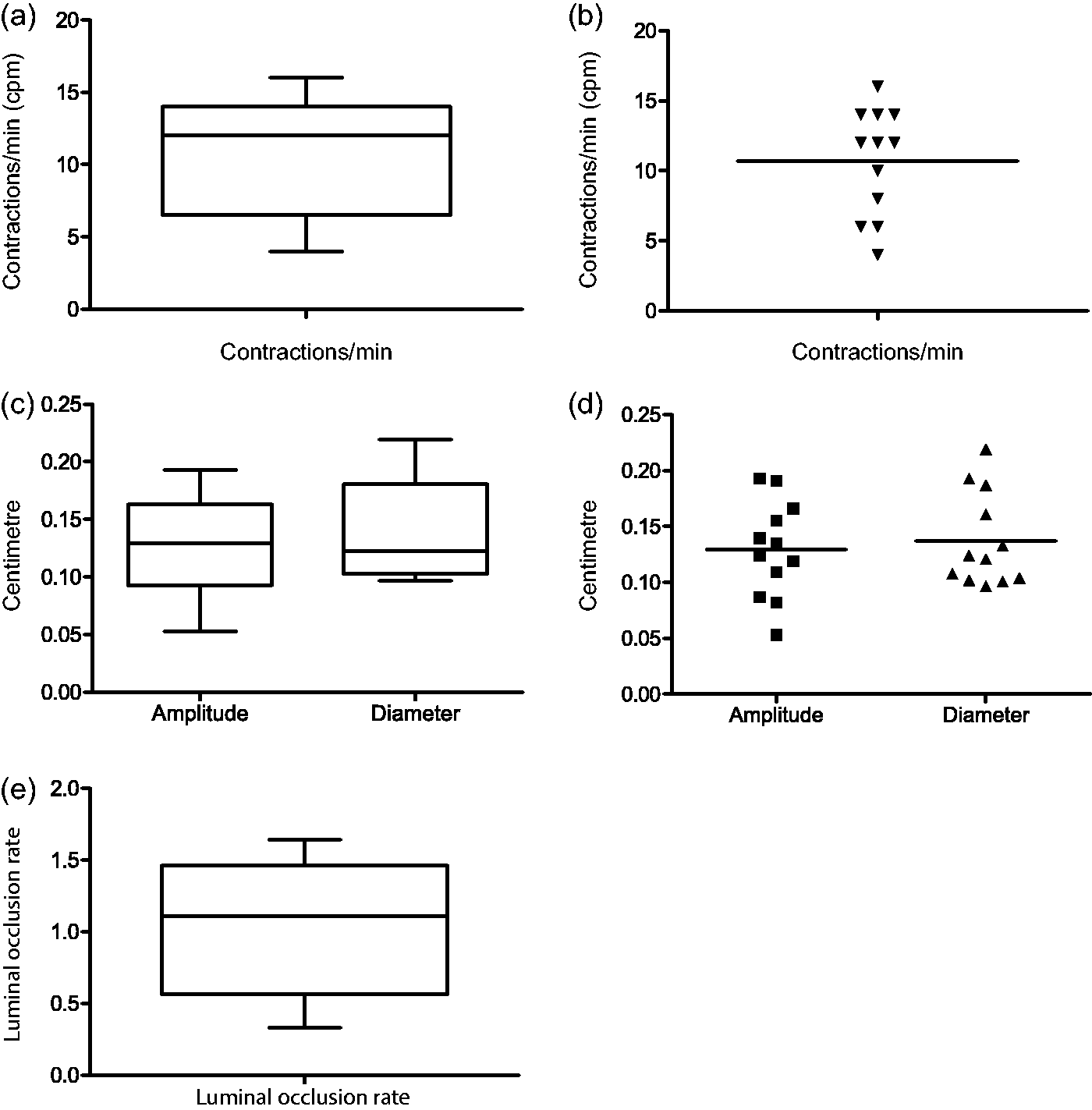
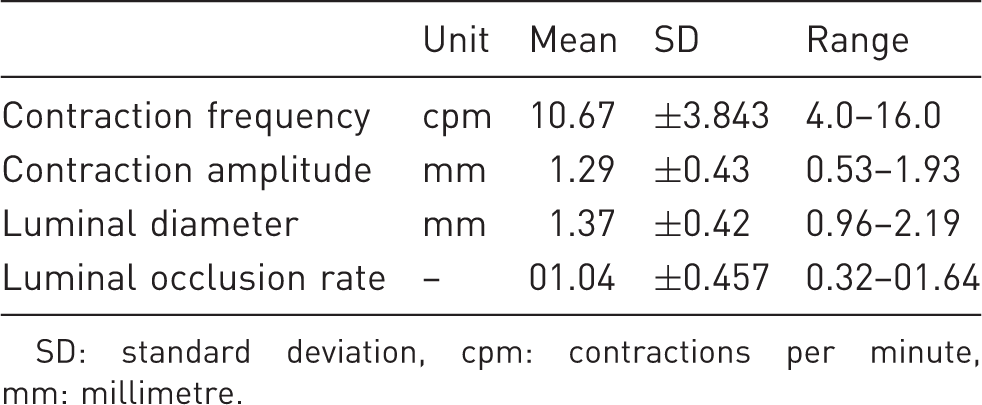
As further important parameters of small bowel motility contraction amplitudes and mean luminal diameters were assessed, the overall mean maximal amplitude of the contractions was 1.29 mm (SD ± 0.43, range 0.53–1.93) with a mean luminal diameter of 1.37 mm (SD ± 0.42, range 0.96–2.19) (Table 1, Figures 2c and 2d).
The luminal occlusion rate as a derived parameter was 1.04%/100 (SD ± 0.45, range 0.33–1.64) in our group of mice (Figure 2e, Table 1).
Duration of single small bowel segment analysis
The mean duration needed for a single small bowel motility assessment was 185 s (SD ± 54 s, range 60–240 s).
Discussion
Small animal imaging is an essential part of preclinical studies and the use of small animal MRI to investigate and monitor the animals is widely established. 5 Yet for studies examining both morphological and functional changes of the intestinal tract in mice, one of the most commonly used animal models, invasive techniques have been unavoidable due to the lack of options for non-invasive in vivo examination of small bowel functions.5,12,14,15 In humans small bowel motility analyses are increasingly used for investigating the intestinal function. They have been demonstrated to provide reliable data in good accordance with histopathological evaluations of alterations of the intestine.6–8
Here we show that small bowel motility analyses using MRI are feasible in small animals such as mice, and provide a fast method for investigation. The motility patterns could be reliably detected even without the administration of dedicated contrast agents. In all the mice measurements could be successfully performed, and the results between the clonogenic mice presented a relatively homogenous pattern of observed spontaneous contractions, with a high level of inter-individual reproducibility.
Investigations of small bowel motility remain a challenging part in small animal studies. One of the most commonly used animal models is the mouse model and current methods of measuring intestinal physiology are mainly invasive and use manometric approaches or the exteriorization of the small intestine to perform video analyses and EMGs, which might be of limited usability, especially in cases where repetitive examinations of intestinal motility are required.13–15 Radiological examinations of intestinal motility in animals has so far been limited to the use of 2D radiographies and MRI examinations in rats to provide the basis of complex mathematical models for investigating motility. 16
In this study we were able to translate an established method which is increasingly being used in humans into a mouse model. 6 High-resolution ultrafast cine acquisitions are the basis of small bowel motility analyses in humans and have been shown to provide clinically relevant and reliable additional information in pathophysiological investigations in humans suffering from IBDs.6,8 The successful translation of the model into the mouse promises well for further translational research, e.g. in models of Crohn’s disease. We were able to reliably measure small bowel motility without special food preparation or invasive procedures; thereby minimizing the invasiveness of the proposed approach and benefitting animal welfare. The mean observed contractions per minute was significantly lower than that described in the literature for small bowel segments (about 30–40/min) which are cut out of sacrificed animals and prepared in Krebs–Ringer solution or exteriorized.18–21 As our method analysed small bowel motility in living animals with healthy bowel loops that are part of a physiologically intact, functioning organism it can be assumed that the method probably more accurately describes the natural contraction patterns of the regular digestive process than those methods which used cut-out segments of sacrificed animals or exteriorization, which might be a benefit with our method. Thus the different contraction frequencies might be more closely related to the complex physiology which influences motility in the living, in contrast to the experimental investigation of motility which uses cut-out small bowel segments. This is important as motility in animals is influenced by a variety of intrinsic factors (interaction between enteric neuronal system [ENS], spontaneous smooth muscle activity, and the interstitial cells of Cajal [ICC]) and extrinsic factors which result in different motility patterns of static, propelling and spontaneous contractions.15,22 This is of special interest in comparing results between different studies as it has been shown that a variety of factors might influence motility on a periprocedural level, such as day-and-night rhythms, the age of the mice, hormone status and the kind of food formula used for the animals; as different ingested substances have already been shown to have influence in animals and humans, for example for high caffeine intake and a high-fat diet.12,23,24 Yet future studies are needed to evaluate the detection and differentiation of the origins of different contractions in the method presented here.
As shown by Ailiani et al., MRI can be used in rats to monitor complex patterns of motility to provide different 3D and non-3D models of intestinal motility. 16 In concordance with their work we now present an additional simple and fast method to reliably and non-invasively determine small bowel motility in mice. Moreover this method could easily be transferred to rats and other small animals. Because there is no need for additional preparation and and the short prolongation of the MRI examination time (typically less than 5 min, depending on the length of the acquired cine sequence) the method presented here might easily be included in abdominal MR examinations and provide additional information on intestinal motility. 5
The imaging protocol chosen was designed as a translational approach and it provided a high image quality without significant image distortions or/and artefacts. This was achieved by using a trueFISP MRI sequence, as previously described for humans, which ensured clear detection of the bowel walls necessary to reliably measure motility parameters. One reason for the relatively low sensitivity to magnetic field distortions is the choice of TR = 2*TE in the trueFISP sequence. For this choice of sequence parameters, the trueFISP sequence exhibits a nearly complete refocusing of the transverse magnetization (acting very similarly to a spin echo sequence). 25 However, near to the tissue air interface at the diaphragm we observed the typical banding artefacts which are well known from trueFISP sequences applied in humans and are caused by the magnetic field inhomogeneity between the lungs and abdomen but which do not limit the evaluation of motility parameters. The in-plane resolution of the sequence was 234 × 234 µm. Therefore, a reasonable evaluation of bowel wall motility was easily possible. Yet, even higher image resolution might be possible in future studies using acceleration techniques such as parallel acquisition or partial Fourier acquisition. Such techniques may, however, result in notably lower signal-to-noise ratios and additional image artefacts. The better soft tissue contrast and the lack of ionizing radiation in MRI examinations in comparison to microCT is a further important beneficial aspect of the method presented here and one of the main reasons why small bowel examinations are performed using MRI. This is especially important in investigations of IBDs of the intestinal tract – one of the main reasons for small bowel motility analysis in humans where the many necessary repetitive examinations of the small bowel using CT in relatively young patients with a livelong chronic disease results in a very high cumulative dose of ionizing radiation which can become critical. This issue becomes even more pronounced in animal experimentation, as the current microCT imagers require relatively high doses of ionizing irradiation compared with CT scanners built for humans. Tissue damage and even death of animals have been reported where longitudinal studies using microCT have been performed. 4
Yet there are several limitations in our study. First the number of investigated animals needs to be increased, although the chosen animal model consists of identical clonogenic mice and in the standardized setting of the investigation one would not expect different results in bigger cohorts. Another limitation is found in the lack of comparisons between the method presented here and manometric, histopathological and other invasive methods of assessing small bowel motility in mice (as previously shown in the method for humans). Out of plane movement of the small bowel segments resulting from breathing (the breathing rate in mice is about 40–60/min during anaesthesia and about 200/min for mice that are awake) or moving of the anaesthetized animals might have further influenced the motility measurements. Using Fourier analyses of the contraction curves (data not shown), we were able to demonstrate that the influence of breathing on the contraction curves by craniocaudal or ventrodorsal displacement of the bowel loops is negligible. We did not investigate the potential influence of breathing motion on bowel deformation, as this was beyond the scope of this study. Furthermore, isoflurane, applied as anaesthesia in our study, has been shown to potentially reduce intestinal motility in animals. 26 Yet isoflurane is considered to be the standard mouse anaesthetization, in accordance with the approved animal anaesthesia SOP, at our institution. In general it more likely reduces intestinal motility, rather than promotes it. Other anaesthetic substances might have affected bowel motility to a lesser degree (ketamine) or even to a much stronger degree (opiate drugs such as fentanyl). However, all of these drugs would have required intraperitoneal or subcutaneous injection of the substance, which is notably more invasive than inhalation anaesthesia. Thus further studies investigating the periprocedural influencing factors, such as meal examination intervals, influence of different food and liquid supply, weight and age of the animals, are necessary to establish reliable reference values. There is also concern for the lack of a current external reference standard, due to the pilot design of this study. One way to address this issue might be to compare the results (if considered comparable) to invasive measurements, or to assess the intra-individual reproducibility of the measurements in identical segments and identical animals, and this should be included in future studies.
Still we think that our results might provide an important new tool for translational research in the investigation of intestinal diseases. Small bowel motility measurements using the method presented can be applied easily and rapidly and the examination does not need invasive procedures, which are important benefits in animal research compared with other recently used invasive methods. It serves alongside limited, but relevant, information on small intestine physiology in humans. 6 Applications of this method might be found in different fields of preclinical research that either directly or indirectly investigate intestinal motility. Direct investigations of intestinal motility might be of interest in the fields of preclinical gastroenterology and the investigation of inflammatory and non-inflammatory bowel diseases, or useful for visceral surgeons investigating the influence of different kinds of bowel anastomoses in, for example, food propelling and small intestinal bacterial overgrowth which might be related to motility disturbance.27,28 Indirect research applications could be found in investigations on the influence of therapeutic agents on small bowel motility both regarding therapeutic effects and adverse effects.
Future studies might also further focus on increasing the usability of the method in order to investigate the use of semi-automated small bowel motility measurements on a segmental or a global level, as well as develop feasibility and usability with humans.8,11,29
In conclusion we demonstrated that MRI examinations are a feasible and fast method for assessing small bowel motility in mice using ultrafast MRI sequences and a dedicated small animal imager. Our results support further investigations in small bowel motility in mouse models of intestinal diseases.
Footnotes
Funding
This work was supported by the Clinical Research Priority Program of the University of Zurich on Molecular Imaging.
