Abstract
The study of factors responsible for eliciting ovulation in rabbits has been hampered by the lack of a suitable method of monitoring the ovaries in vivo. Ovarian imaging by ultrasound biomicroscopy was used in two experiments designed to determine the effects of seminal plasma on the ovulatory response in rabbits. In Experiment 1, female rabbits were group-housed and treated intramuscularly with saline, gonadotropin releasing hormone (GnRH), or seminal plasma of llamas or rabbits (n = 4 to 6 per group). Rabbits were euthanized eight days later to evaluate the ovarian response by ultrasound biomicroscopy ex situ. No differences among groups were detected in the proportion of rabbits that ovulated or in the number and size of corpora lutea. The high incidence of ovulation in the negative control group was unexpected, and confounded determination of an ovulation-inducing effect of seminal plasma. In Experiment 2, female rabbits were caged individually, and treated as in Experiment 1 (n = 5 to 7 per group). The ovarian response was evaluated in vivo by transcutaneous ultrasound biomicroscopy. Ovulation and formation of corpora lutea were detected only in rabbits given GnRH. A preovulatory surge in plasma luteinizing hormone concentration and a post-ovulatory rise in plasma progesterone concentration were detected only in rabbits treated with GnRH. Surgical translocation of the ovaries to a subcutaneous position enabled longitudinal assessment of the ovulatory response by ultrasound biomicroscopy. Results clearly documented the effect of physical/social interaction on ovulation in rabbits, and did not support the hypothesis that seminal plasma elicits ovulation in rabbits.
In rabbits, coitus has been reported to be the main factor in inducing ovulation. 1 Authors of an early study 2 reported that artificial insemination followed by sterile mating resulted in a higher pregnancy rate than artificial insemination alone (20/22 [91%] versus 1/28 [4%]). However, authors of a later study concluded that mechanical stimulus of the penis may not be the main stimulus for ovulation. 3 Although the primary trigger for inducing ovulation in rabbits is not well understood, it is clear that a coitus-related stimulus of the pituitary gland4,5 results in a rise in circulating luteinizing hormone (LH) concentration1,6 and causes ovulation.
In other induced ovulators, such as alpacas and llamas, results of recent studies have challenged the traditional idea that physical stimulation of the genital tract during coitus triggers ovulation in these species and have shown that seminal plasma plays a pivotal role in induction of ovulation.7,8 Intramuscular administration of seminal plasma in alpacas and llamas resulted in a surge release of LH followed by ovulation.7–9 Intramuscular or intravaginal administration of seminal plasma in bactrian camels10,11 and koalas 12 induced ovulation in the corresponding females. Observations in these species of induced ovulators suggest that a factor present in the seminal plasma is responsible for eliciting the ovulatory cascade.
Ovulation-inducing factor (OIF) is a seminal plasma protein that has been found in the ejaculates of alpacas and llamas.7,8,13,14 The protein was initially isolated from llama seminal plasma as a molecule with a molecular mass of about 30 kDa. 14 However, the same researchers reported more recently that OIF is a 14 kDa protein molecule responsible for eliciting a preovulatory LH surge and ovulation when administered in llamas. 9 Additionally, a dose-dependent effect of purified OIF on ovulation and corpus luteum (CL) form and function in llamas was documented. 15 Recently, the OIF protein isolated from seminal plasma was found to be identical in sequence and structure to beta-nerve growth factor (β-NGF), 16 and has been detected in the ejaculates of camels, cattle, horses, and pigs.10,11,13,17 Furthermore, the biological activity of OIF has been reported in seminal plasma from different species by using the llama as an in vivo ovulation bioassay, suggesting that seminal OIF and its functions are conserved among species.13,17
Use of a prepubertal mouse model documented the ovulation-inducing effect of seminal plasma in a common laboratory species, 17 but the model is terminal; i.e. it does not permit serial/longitudinal evaluation. Rabbits represent an attractive animal model for studies of ovulation because they are induced ovulators, and they are small and easy to handle. The presence of OIF/NGF in the seminal plasma of rabbits was reported recently, but no effect of seminal plasma on ovulation in female rabbits was detected. 18 However, the ovarian status of the females was not taken into account before treatment, and the rabbit semen was diluted in saline such that the dose of OIF/NGF was unknown. Whether these factors were involved in the failure of ovulation after treatment remains unclear. Studies in llamas and alpacas confirmed that ovulatory capability depends on follicular maturity at the time of mating; 19 hence, the effectiveness of an ovulation-inducing treatment may be assessed only when a sufficiently mature follicle is present at the time of treatment. The lack of suitable methods for monitoring the ovaries in live rabbits has provided little or no opportunity for assessing the size of follicles in the ovaries at the time of treatment. Indeed, ovarian characteristics are usually evaluated only after treatment using either terminal20–22 or invasive procedures.23–25
Ultrasonography has been useful for in vivo detection of ovulation in llamas and alpacas treated with seminal plasma and/or purified OIF.7–9,14,15,17,18 However, conventional ultrasonography does not provide sufficient resolution to reliably detect ovarian structures in small species. Ultrasound biomicroscopy (UBM) involves the use of high frequency ultrasound waves (e.g. ≥25 MHz) to provide image resolution of structures smaller than 100 µm. We have recently established and validated the UBM technique as a reliable method of assessing ovarian structures in rabbits. 26 This technology enables imaging of the same animal repeatedly, thus permitting detection of dynamic processes (cause-and-effect) and decreasing the number of animals required per experiment.
The objective of the present study was to develop an in vivo rabbit model for determining factors responsible for eliciting ovulation. Experiments were designed specifically to determine the effects of seminal plasma (llama and rabbit) on the ovulatory response in rabbits, in vivo, using UBM.
Materials and methods
Animals
New Zealand White rabbits (Charles River Canada, Pointe Claire, Quebec, Canada) were housed under controlled environmental conditions (20.2 ± 2℃, 14:10 h light:dark cycle) and were allowed ad libitum access to food and water. Llamas were maintained on pasture with free access to supplemental hay and fresh water. The experimental protocol was approved by the University of Saskatchewan's Animal Research Ethics Board and procedures were done in accordance with guidelines of the Canadian Council on Animal Care.
Experiment 1
Llama semen was collected from five adult males once a week for three months by artificial vagina. 27 Ejaculates were diluted 1:1 (v:v) with phosphate buffered saline (PBS; Invitrogen, Grand Island, NY, USA), drawn back-and-forth through an 18-guage needle attached to a 10 mL syringe as previously described. 7 Seminal plasma was separated from the cellular components of the ejaculate by double centrifugation at 1500 × g (Sorvall Legend RT; Mandel Scientific Co Ltd, Guelph, Ontario, Canada) for 30 min, and sperm-free samples were stored at −80℃. Upon thawing, the diluted seminal plasma samples from all five males were pooled. Rabbit semen was collected from two adult males twice on each of two days per week for four months by artificial vagina (adapted from 28 ). An interval of 10–15 min between the first and the second ejaculates per day was permitted. Undiluted semen was centrifuged as described above and seminal plasma was separated from the sperm and gel of the ejaculate, and was stored at −80℃. Upon thawing, the seminal plasma samples from both males were pooled. For both species, 1% penicillin/streptomycin (v/v; 10,000 units/mL penicillin and 10 mg/mL streptomycin; Sigma-Aldrich, St Louis, MO, USA) was added to the pooled seminal plasma.
Mature female rabbits (5.5 months old) were housed together in a single room. An acclimation period of seven days was allowed before ovarian examinations were initiated. Imaging of the ovaries was performed daily using transabdominal ultrasonography (MyLab 5; Esaote, Ajax, Ontario, Canada) with a 12 MHz linear-array probe to ensure the presence of follicles ≥1.5 mm in diameter prior to treatment. The hair over both flanks of each rabbit was shaved before each ultrasound examination using soap and a razor. A towel was wrapped around the forelimbs and the head of the rabbit for restraint during examination. Rabbits were placed in lateral recumbency and remained calm without the need for sedation or anesthesia. Acoustic gel (Eco Gel 200; Eco-Med Pharmaceutical Inc, Mississauga, Ontario, Canada) was applied to the skin over the area to be scanned.
When one or more follicles ≥1.5 mm was detected, each rabbit was assigned randomly (in block) into one of four groups and given an intramuscular dose of (1) 1.5 mL of saline (negative control, n = 6); (2) 0.02 mg gonadotropin releasing hormone (GnRH) (Fertagyl; Intervet, Whitby, Ontario, Canada; positive control, n = 4); (3) 2 mL of llama seminal plasma (diluted 1:1 v:v with PBS, n = 6); or (4) 1.5 mL of rabbit seminal plasma (undiluted, n = 6). The dose of llama and rabbit seminal plasma was adapted from previous studies.7,13,18 The day of treatment was designated Day 0. On Day 8, rabbits were euthanized with an overdose of sodium pentobarbital, and the ovaries were removed for evaluation ex situ. Each ovary was imaged in a saline bath using a high-resolution ultrasound biomicroscope (UBM, Vevo 660 Imaging System; Visual Sonics Inc, Toronto, Ontario, Canada) equipped with a 25 MHz oscillating sector transducer (RMV 704), as described previously. 26 Ovarian images were recorded in cine-loops and individual CL were identified, counted and measured by scrolling through the cine-loops frame-by-frame.
Experiment 2
Given the unexpected results of group-housed rabbits in Experiment 1, female rabbits (5.5 months old, n = 13) were housed individually in stainless steel cages (Lab Products Inc., Maywood, NJ, USA) for Experiment 2. Surgical translocation of both ovaries of each rabbit to a subcutaneous position in the flank region was performed to permit in vivo ovarian imaging by transcutaneous UBM, as previously described. 26 Starting three weeks after surgery, transcutaneous UBM (Vevo 660; 25 MHz probe) of the ovaries was done daily for at least four days before treatment to confirm the presence of follicles ≥1.8 mm in diameter (i.e. sufficiently large to respond to an ovulatory stimulus 29 ). Rabbits were restrained and prepared for ultrasound examination as in Experiment 1. Follicles were more clearly distinguished in vivo by UBM compared with the conventional ultrasonography used in Experiment 1, and greater precision was possible in assessing follicle diameter before treatment.
When 6 to 10 ovarian follicles ≥1.8 mm were detected, each rabbit was assigned randomly (in block) to one of four groups and given an intramuscular dose of (1) 1.5 mL of saline (negative control, n = 5); (2) 0.02 mg GnRH (Fertagyl; positive control, n = 5); (3) 2 mL of llama seminal plasma (diluted 1:1 v:v with PBS, n = 6); or (4) 1.5 mL of rabbit seminal plasma (undiluted, n = 7). The same pool of llama/rabbit seminal plasma collected during Experiment 1 was used in Experiment 2. The day of treatment was designated Day 0. Rabbits were examined daily by transcutaneous UBM from Days 0 to 8 to determine ovulation and CL development. The occurrence of ovulation was determined on the basis of detection of CL. Individual CL were counted and measured, as described in Experiment 1.
For measurement of plasma progesterone concentration, blood samples were collected from the marginal ear vein or the lateral saphenous vein every other day from Day 0 to Day 8. A topical lidocaine–prilocaine 5% cream (EMLA; AstraZeneca Canada Inc, Mississauga, Ontario, Canada) was applied over the vein at least 15 min before blood sampling. Blood samples (approximately 2 mL each) were centrifuged at 1700 × g for 30 min and the plasma was stored at −20℃. Plasma progesterone concentrations were determined using a commercial, double-antibody radioimmunoassay kit (Coat-a-Count progesterone; Siemens Medical Solutions Diagnostics, Los Angeles, CA, USA). All samples and standards (100 uL) were assayed in duplicate in a single assay. The intra-assay coefficients of variations were 9.7%, 6.5% and 6.3%, respectively, for reference plasma progesterone concentrations of 1.99, 3.47, and 17.06 ng/mL.
For measurement of plasma LH concentration, blood samples (approximately 2 mL each) were taken at 0, 1, 2, 3 and 6 h after treatment. Topical lidocaine–prilocaine 5% cream was applied over the vein before blood sampling and the samples were centrifuged and stored as described above. Plasma LH concentrations were determined using a specific homologous double-antibody radioimmunoassay for rabbit LH (AF Parlow, National Hormone and Peptide Program, Harbor-UCLA Medical Center, Torrance, CA, USA). 30 Concentrations of LH are expressed in terms of NIDDK–RbLH (AFP-7818C). The standard curve ranged from 0.5 ng/mL (90% ligand labelled LH) to 16.0 ng/mL (20% ligand labelled LH). The primary antibody (AFP3120489) was raised in guinea pigs, and the anti-guinea pig secondary antibody was raised in sheep (kindly provided by Susan Cook and Dr B Laarveld, Animal and Poultry Science, University of Saskatchewan, Saskatoon, SK, Canada). All samples and LH standards (50 uL) were assayed in duplicate in a single assay. The intra-assay coefficient of variation was 3.9% for the high reference plasma LH concentration (6.33 ng/mL) and 10.9% for the low reference plasma LH concentration (0.83 ng/mL). Values that exceeded more than three standard deviations from the mean for the respective group were considered outliers and removed from further analysis.
Statistical analysis
Analysis of variance was used to compare non-serial data (i.e. number of follicles at the time of treatment, number and size of CL on Day 8) among groups. Analysis of variance for repeated measures (Proc-mixed in SAS, Enterprise Guide 4.2; Statistical Analysis System Institute Inc, Cary, NC, USA) was used to compare serial data (i.e. plasma progesterone and LH concentrations) to determine the effects of treatment, day, and treatment-by-day interaction. Tukey’s multiple comparison was used as a post-hoc test when a main effect of treatment or a treatment-by-day interaction was detected. Ovulation rates were compared among groups by Fisher's exact test. P < 0.05 was considered statistically significant. For the purposes of analysis and illustration, hormone data were centralized to Day 0 (day of treatment).
Results
Experiment 1
No swelling or redness was noted at the injection site in any of the rabbits, although a transient limp was clinically apparent in all rabbits (6/6) treated with rabbit seminal plasma. The limp disappeared and no untoward effects were apparent 30 min after treatment.
The mean number of follicles ≥1.5 mm at the time of treatment did not differ among groups (P = 0.25), nor did the proportion of rabbits that ovulated (P = 1.0; Table 1). Conventional transabdominal ultrasonography was inadequate for evaluating the ovarian response to treatment because CL were not clearly differentiated from the surrounding tissue. CL were clearly distinguishable in the ovaries ex situ by UBM. In rabbits that ovulated, no differences were detected among groups in the number (P = 0.51) or size (P = 0.82) of CL (Table 1, Figure 1).
Rabbit ovaries imaged in vivo by conventional ultrasonography (a), ex situ by ultrasound bio-microscopy (b) and in vivo by transcutaneous ultrasound bio-microscopy (c, d). Figures show follicles (arrows) and corpora lutea (arrowheads). Note cumulus-oocyte complexes (COC) within the follicular antrum. Scale marks are spaced at 5 mm in (a), and at 0.1 mm in (b), (c) and (d). Number and diameter (mean ± SEM) of follicles and corpora lutea (CL) in rabbits treated with saline, gonadotropin releasing hormone (GnRH), or seminal plasma (SP) of llamas or rabbits (Day 0 = day of treatment). Ovarian structures were assessed in vivo prior to treatment by transabdominal ultrasonography (12 MHz) and ex vivo after treatment by ultrasound biomicroscopy (25 MHz) – Experiment 1. No significant differences among groups for any endpoint.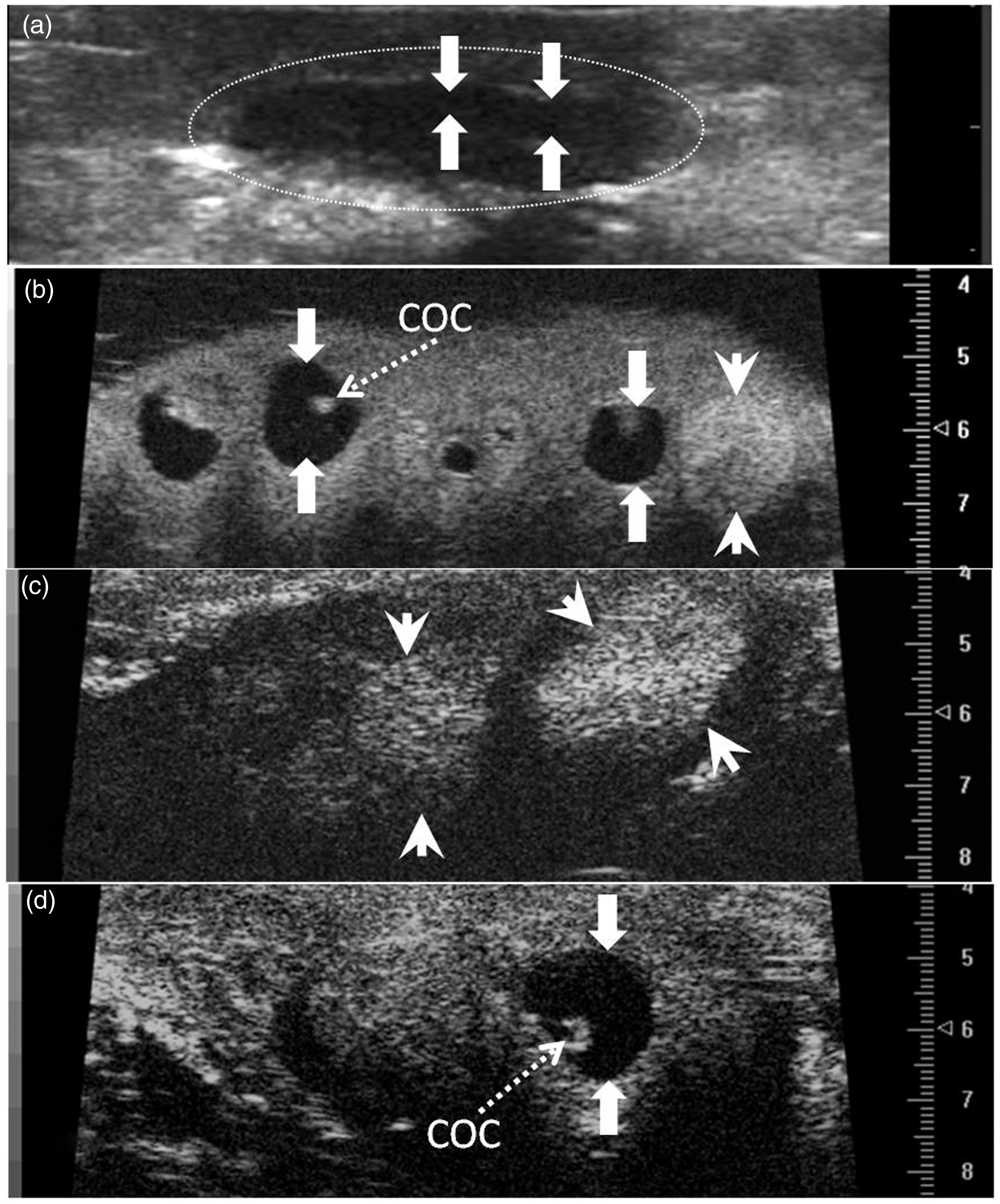

Experiment 2
Number and diameter (mean ± SEM) of follicles and corpora lutea (CL) in rabbits treated with saline, gonadotropin releasing hormone (GnRH), or seminal plasma (SP) of llamas or rabbits (Day 0 = day of treatment).
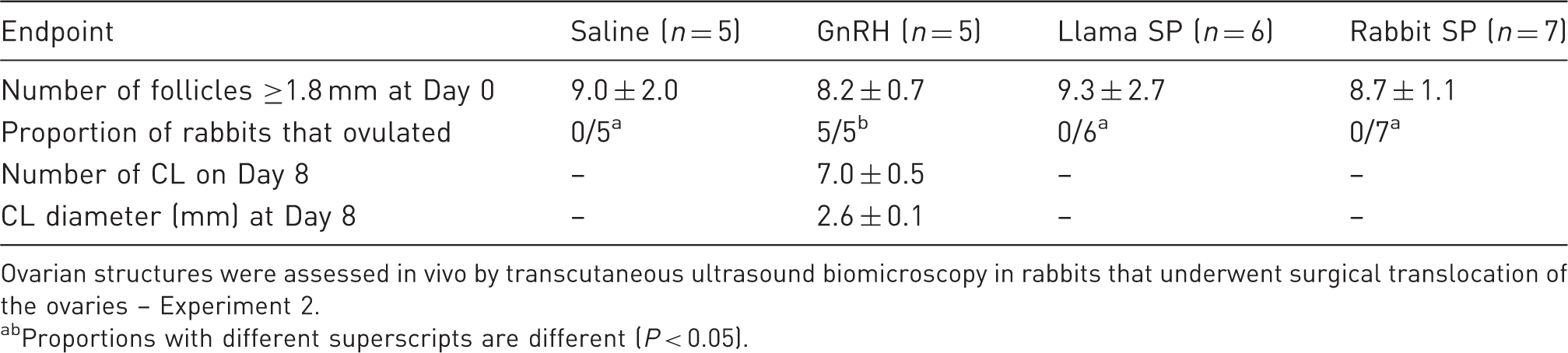
Ovarian structures were assessed in vivo by transcutaneous ultrasound biomicroscopy in rabbits that underwent surgical translocation of the ovaries – Experiment 2.
Proportions with different superscripts are different (P < 0.05).
Plasma LH concentration increased only in the GnRH group (positive control), and remained basal in all other groups (treatment-by-time interaction, P < 0.01; Figure 1). Plasma LH concentrations in the GnRH group were maximal at 1 h after treatment, began to decrease at 3 h, and were basal at 6 h after treatment (Figure 2).
Plasma luteinizing hormone (LH) concentrations (mean ± SEM) in female rabbits after intramuscular treatment with saline, gonadotropin releasing hormone (GnRH), or seminal plasma (SP) of llamas or rabbits. abAt a given time-point, values in the GnRH group were greater than in other groups (P < 0.05).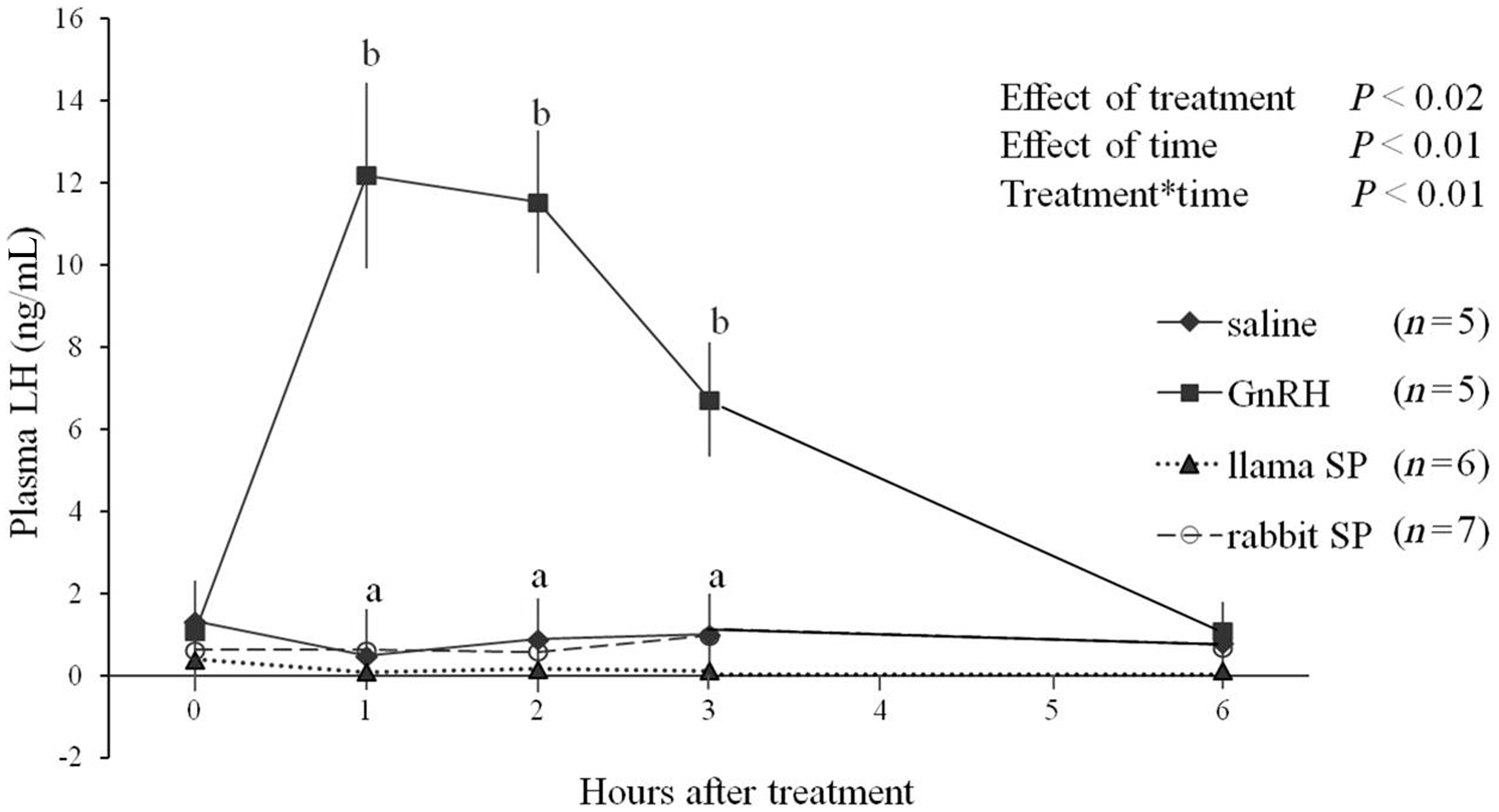
Plasma progesterone concentrations increased only in the GnRH group and remained basal in the other groups (treatment-by-day interaction, P < 0.001; Figure 3). In the GnRH group, progesterone concentrations were elevated by Day 2 and increased linearly to the last day of sampling (Day 8; Figure 3).
Plasma progesterone concentrations (mean ± SEM) in female rabbits after intramuscular treatment with saline, gonadotropin releasing hormone (GnRH), or seminal plasma (SP) of llamas or rabbits. abAt a given time-point, values in the GnRH group were greater than in other groups (P < 0.05).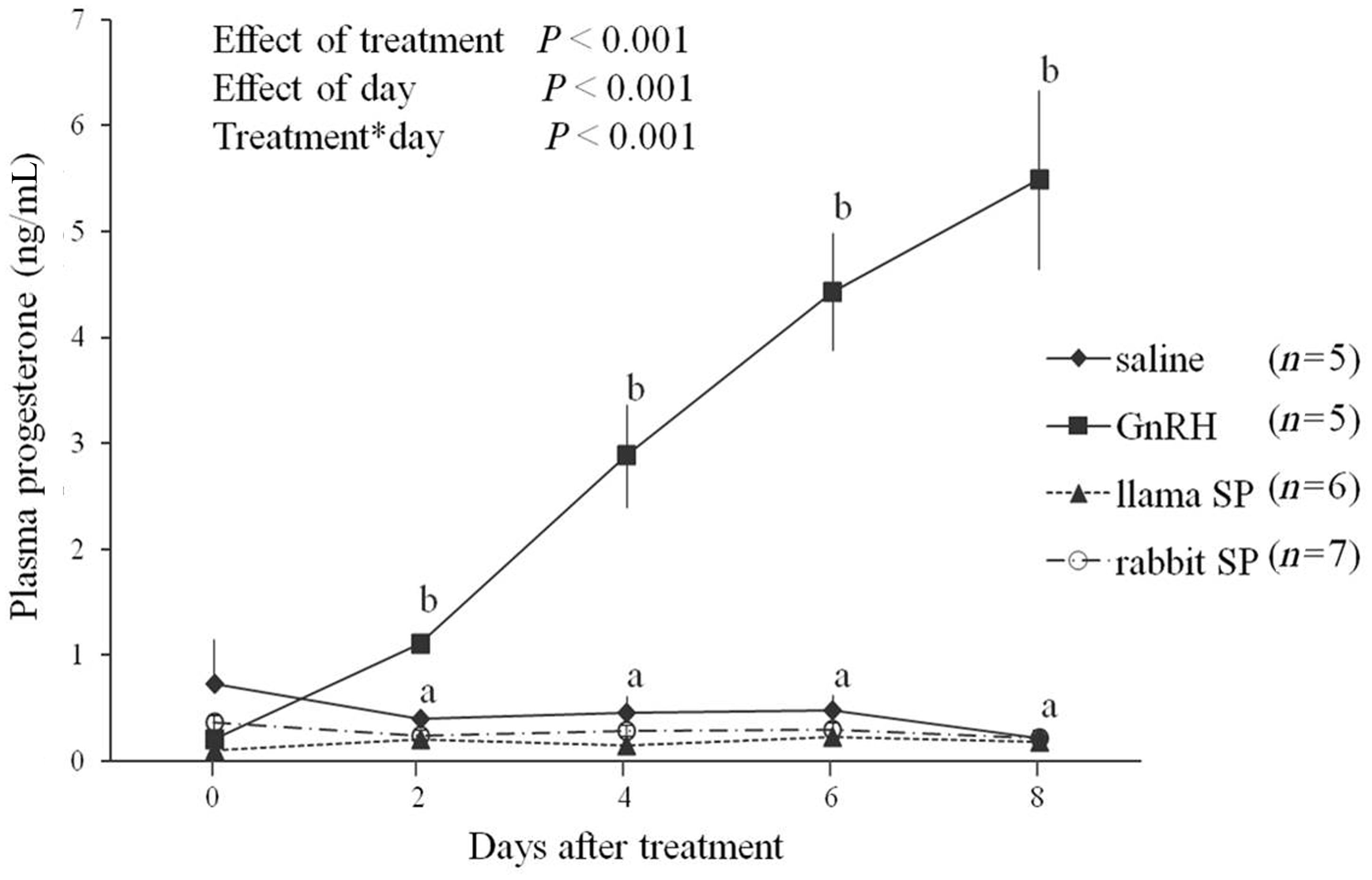
Discussion
The existence of OIF/NGF in seminal plasma and its biological effects (i.e. LH release and ovulation) have been well documented using the female llama and alpaca as animal models.3,7,8 Although OIF/NGF has recently been found in seminal plasma of rabbits, 18 no effect on ovarian function in rabbits has been reported. The present study was conducted in two experiments to determine the effect of OIF/NGF (seminal plasma of llamas and rabbits) on the release of LH and ovulation in female rabbits. The administration of seminal plasma as a single intramuscular dose was chosen based on results reported in alpacas. 8
In Experiment 1, the occurrence of ovulation in the saline-treated group (negative control) was unexpected and confounded interpretation of treatment effects. That is, continuous rabbit-to-rabbit interaction in group-housed females in Experiment 1 was associated with a high incidence of ‘spontaneous ovulations’ in all treatment groups, even in the negative control group. To address the possibility that behavioral stimuli in group-housed females contributed to spontaneous ovulations, Experiment 2 involved individual housing of females in separate cages – and no spontaneous ovulations were detected. A constant rabbit-to-rabbit interaction in Experiment 1 may have produced the ovulation of follicles that would not have ovulated with only the treatment given. In this sense, it is likely that follicles that reached a certain diameter at a certain time ovulated independently of the treatment given but induced by some unknown factors related to the rabbit-to-rabbit interaction during the duration of the experimental period. Behavior-related induction of ovulation was reported in an early study when female rabbits caged in pairs repeatedly mounted one another. 31 Mounting behavior among females was observed occasionally in Experiment 1, but the study was not designed to test behavior and no further notation of the frequency or the identification of the individuals involved was recorded. However, the occurrence of pseudopregnancy in group-housed rabbits was reported even when only occasional mountings were observed. 32 In ewes (i.e. spontaneous ovulators) and musk shrews (i.e. induced ovulators), olfactory cues (pheromones) were thought to participate in the hypothalamic control of pituitary secretions and ovulation.33,34 In rabbits, however, olfactory bulbectomy did not block mating-induced ovulation. 35 Researchers proposed that visual stimuli, in conjunction with acoustic and olfactory stimuli, with an intact male may play a role in ovulation in rabbits. 36 In our study, female rabbits were in complete isolation from male rabbits throughout both experiments. Whether pheromones play a role in the induction of ovulation in rabbits remains unclear.
In rabbits housed individually (Experiment 2), ovulation occurred only in the positive control group (GnRH) and, similar to the negative controls, treatment with seminal plasma did not elicit LH release or ovulation. The ovulatory mechanism in this species of induced ovulator may be different from that of camelids. In a recent study, 37 the ovulation rate after artificial insemination of rabbits with raw semen, saline, or raw semen after lumbar epidural anesthesia resulted in ovulation in 8/8, 3/8, and 0/8 rabbits, suggesting that semen may augment a primary somatosensory stimulus to trigger LH release and ovulation in rabbits. Failure of seminal plasma to induce ovulation in rabbits in the present study may also be related to the dose and route of administration. In this regard, the effect of OIF in other species was dose-dependent.15,38 An intramuscular route of administration was used in the present study, but an intravenous route was used in a recent study in llamas using pure ß-NGF, 16 and may have provided a more immediate and pronounced spike in circulating concentrations. Conversely, pituitary release of LH in rabbits may require a more prolonged period of stimulation that a single dose of seminal plasma did not provide.
Seminal plasma is divergent in protein composition among species; however, ß-NGF was found to be conserved in different mammalian species such as alpacas, llamas, camels, cattle, sheep, pigs and horses.17,39 Whether ß-NGF in seminal plasma induced an antigenic response in rabbits in the present study is unknown, but components of the seminal plasma have the capacity for inducing and suppressing the immune response in mice. 40 A weak antigenic response was observed when semen was given intramuscularly to female rabbits. 41 The female rabbits used in the present study were entirely naïve to seminal plasma antigens and treatment with llama or rabbit seminal plasma was given only once. While an antibody response to seminal plasma may be expected to be mild, if any, the neutralizing effects of such antibodies would have blunted ovulatory response in the present study.
Since a preovulatory peak of LH and ovulation in Experiment 2 was elicited in the GnRH group, ovulation failure in the other treatment groups appears to be due to insufficient stimulation to provoke the LH surge rather than to a lack of mature follicles in the ovaries, as stated by others. 42 Ovarian follicular size at the time of treatment is an important determinant of the ovarian response in induced ovulators and must be taken into account in studies involving ovulatory response.7–9,13,15,17,19,43,44 In this regard, ultrasonography was used in the present study to detect the presence of large follicles in similar numbers among rabbits at the moment of treatment. In rabbits, there is no clear understanding of the relationship between follicle diameter and ovulatory capability, but preovulatory follicles have been variously referred to as those between >800 to 900 µm, >1.5 mm, >1.8 mm, or >2 mm in diameter.22,29,45–47 In Experiment 1, follicle numbers and diameters were monitored in vivo using conventional transabdominal ultrasonography (12 MHz linear-array probe), but images lacked the clarity and precision achieved by transcutaneous UBM. 26 The ability to accurately detect ovulation and to distinguish the CL in live rabbits by conventional transabdominal ultrasonography in Experiment 1 was deemed unsatisfactory; hence, the rabbits were euthanized and the ovaries were evaluated ex situ by UBM, as recently reported. 26 This approach did not permit serial observations in the same rabbit and it is, therefore, subject to the same limitations as studies involving laparotomy23–25,37 or post-mortem histological examination.18,20,22,25,37
To permit repeated examination of the ovaries in vivo in Experiment 2, we employed a recently developed technique of transcutaneous UBM. 26 The approach involved surgical translocation of the ovaries to a subcutaneous position in the flank without interrupting the vascular supply and ovarian function. This technique enabled serial monitoring of follicles ≥0.6 mm and CL development in vivo without sedation or anesthesia. The number of CL detected per rabbit after the GnRH treatment was similar to that reported in previous studies based on post-mortem or laparotomy examination.26,42
Ovarian responses following different ovulatory treatments were clearly distinguished using transcutaneous UBM in rabbits. Unlike the effects previously reported in llamas and alpacas,7,8,13 data from the present study do not support the hypothesis that llama or rabbit seminal plasma, given intramuscularly, induces a preovulatory surge of LH and ovulation in rabbits. Although results suggest that OIF/NGF in seminal plasma may not play a pivotal role in ovulation in rabbits, there is a need for further studies to determine whether a minimum effective dose is required to stimulate ovulation in rabbits, or if this factor acts differently in rabbits than in other induced ovulators. Differences in results between Experiments 1 and 2 clearly demonstrate the influence of direct rabbit-to-rabbit contact in group-housed females on ovulation. With this knowledge, and the UBM technique, future experiments may be designed specifically to examine the relative contributions of social, physical and chemical factors involved in eliciting ovulation in rabbits. Such knowledge may be particularly important for improving breeding efficiency and reproductive management practices for rabbit farms.
Footnotes
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This research was supported by the Natural Sciences and Engineering Research Council of Canada [grant number 122236], and the Alpaca Research Foundation.
Acknowledgements
The authors gratefully acknowledge Susan Cook for assistance with hormone assays, and Monique Burmester and her staff in the Animal Care Unit at the Western College of Veterinary Medicine for the care and maintenance of the rabbits.
MPC participated in study design, acquisition, analysis and interpretation of data, and in preparation and revision of the manuscript. JMP participated in semen collection, acquisition and interpretation of the data, and helped to draft the manuscript. As principal investigator, GPA was involved with the original concept of the study, experimental design, the acquisition, analysis and interpretation of data, as well as writing and revising the manuscript. All authors read and approved the final manuscript.
