Abstract
Laboratory rodents are commonly euthanized by exposure to gradually increasing concentrations of carbon dioxide (CO2). Current recommended flow rates range between 10 and 30% chamber vol/min and result in insensibility before exposure to painful concentrations (<40%). However, this method causes dyspnea, indicated by deep, rapid breathing. In humans dyspnea is associated with a negative affective experience. Sensations of dyspnea may explain why rodents find CO2 concentrations >3% aversive. This study aimed to assess the effect of CO2 flow rates on time between the onset of dyspnea and various measures of insensibility (recumbency, loss of the righting reflex and loss of the pedal withdrawal reflex) to identify flow rates that minimize the potential experience of dyspnea. The results of this study indicate that a flow rate of 50% chamber vol/min, while holding the CO2 cage concentration just below 40%, minimizes the interval between the onset of labored breathing and recumbency. Using a 50% flow rate this interval averaged (±SE) 30.3 ± 2.9 s versus 49.7 ± 2.9 s at 20% chamber vol/min (F3,22 = 7.83, P = 0.0013). Similarily, the interval between the onset of labored breathing and loss of the righting reflex averaged 38.2 ± 2.4 s at a flow rate of 50% versus 59.2 ± 2.4 s at 20% chamber vol/min of CO2 (F3,22 = 13.62, P < 0.0001). We conclude that higher flow rates reduce the duration of dyspnea, but even at the highest flow rate mice experience more than 30 s between the onset of dyspnea and the most conservative estimate of insensibility.
Carbon dioxide (CO2) gas is commonly used to kill laboratory rodents. Current guidelines1,2 suggest that the chamber should be filled gradually using a flow rate between 10 and 30% chamber vol/min of CO2. Use of flow rates lower than 30% chamber vol/min are thought to reduce the likelihood that CO2 concentration in the chamber will exceed painful levels (>40%) before insensibility is reached. 3
Unfortunately, pain is not the only welfare concern associated with exposure to CO2. Humans report sensations of dyspnea, defined by the American Thoracic Society as ‘a subjective experience of breathing discomfort that consists of qualitatively distinct sensations that vary in intensity’. 4 Humans describe the experience of dyspnea as distressing5–8 and this sensation has been used to induce fear and panic in humans using CO2 concentrations between 7.5 and 35%.9–12 Different types of dyspneic sensations have been identified resulting from pathological breathlessness, including air hunger, tightness and work. 13 In the human literature, the term dyspnea contains both an affective and behavioral response, as the negative emotional response resulting from dyspneic experiences results in aversion.13,14 In the veterinary literature, dyspnea typically refers to labored breathing in animals, as currently labored breathing is associated with a negative affective experience in mice or rats. However, previous studies in mice and rats have shown that CO2 concentrations ranging from 3–20% are aversive,15–21 concentrations between 10 and 35% have been shown to cause fear responses,22–24 and 10% CO2 may be used as an unconditioned fear stimulus in mice. 24 In the current study, labored breathing was directly assessed but we cannot be certain that this behavior was associated with a negative affect. However, on the basis of human evidence and the rodent research on aversion and fear responses, we posit that this autonomic response is accompanied by a negative affective experience. On this basis, we argue that euthanasia methods that minimize the duration of labored breathing should be considered more humane. When the term dyspnea is used in this paper, we refer to the veterinary definition meaning ‘labored breathing’, while acknowledging that the known rodent aversion to CO2 (as referenced above) is likely accompanied by a negative affective experience.
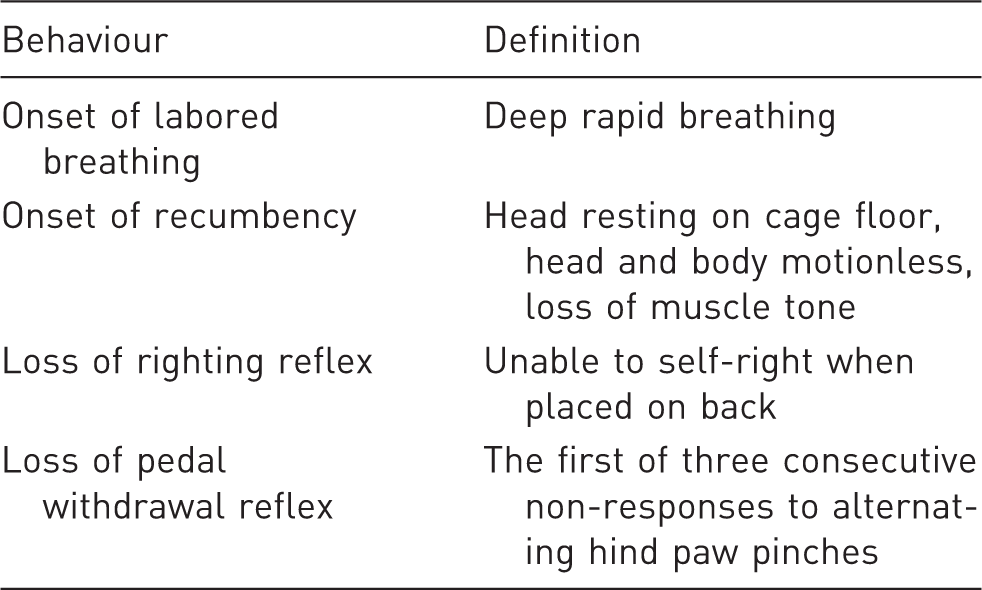
Definitions of mouse behaviors used in this study to assess dyspnea and various levels of insensibility.
We hypothesized that of the flow rates tested in this study (20, 30, 40 and 50%), the higher flow rates would minimize the experience of dyspnea, as measured from the onset of labored breathing to recumbency, loss of the righting reflex, and loss of the pedal withdrawal reflex, for mice euthanized using the gradual-fill method of CO2 euthanasia. The 20 and 30% flow rates were chosen because they are commonly recommended, and the 40 and 50% rates were chosen based on the prediction that these would shorten the interval between the onset of dyspnea and insensibility. Throughout the study, cage concentration was not allowed to exceed painful levels (>40%) via a gas holding technique, until all three measures of insensibility had been satisfied.
Materials and methods
Animals and housing
We used 23 surplus naive female albino C57BL/6J-Tyr mice (British Columbia Cancer Research Centre, Vancouver, Canada) at the University of British Columbia’s Centre for Comparative Medicine, Vancouver, Canada. Mice were five months old during testing and weighed 21.6–28.4 g. Mice were group-housed in an OptiMICE® (Animal Care Systems, Centennial, CO, USA) cage system with autoclaved clean polycarbonate cages (Makrolon®, Animal Care Systems) with dimensions 31.8 cm long × 27.9 cm wide front × 8.9 cm wide rear × 12.9 cm height. Each cage contained autoclaved ECOfresh™ (Absorption Corporation, Ferndale, WA, USA) bedding, a nest box, a cotton nesting square (Ancare, Bellmore, NY, USA), brown crinkle paper (Enviro-dri®; Shepherd Specialty Papers Inc, Richland, MI, USA) and free access to food (5001 PMI Lab Diet; Harlan Laboratories Inc, Indianapolis, IN, USA) and filtered water. The average humidity and temperature during testing were 48% and 23℃, respectively. Mice were kept under a 12 h light:12 h dark cycle; testing took place during the light phase (between 08:00 and 10:00 h) on two consecutive days. The University of British Columbia’s Animal Care Committee approved all the procedures used in this study.
Experimental apparatus
An Innocage® mouse disposable individual ventilated cage (IVC) transparent mouse cage (Innocage®, Universal Euro Type II Long, Innovive Inc, San Diego, CA, USA; 37.3 cm L × 23.4 cm W × 14.0 cm H, with 205 cm2 floor space) was used as the test cage (Figure 1). On one side of the cage a hole was cut to project a powdered surgical latex glove (Perry® Style 42®, Ansell, size 7, Ansell, New Jersey, USA) sealed with tape. This allowed one hand to be placed into the cage during testing. A non-slip pad made from wood flooring underlayment (Shaw Floors; Shaw Industries, Dalton, GA, USA) was cut to fit the bottom of the test cage to minimize slipping. A clear Plexiglas lid with a small hole in the middle was placed on top of the cage to allow insertion of a tube to deliver the CO2. Before each trial, 500 mL of aspen-chip bedding was added to the test cage and removed after each euthanasia procedure. The tube delivering gas to the test cage was connected to a CO2 tank (Praxair, Delta, BC, Canada) and the flow was measured with a CO2 flow meter (Western Medica, Westlake, OH, USA). A small hole near the base of the cage in the center of the anterior wall of the test cage allowed insertion of the sampling tube connected to an O2 analyzer (Series 2000, Percent Oxygen Analyzer; Alpha Omega Instrument Corporation, Cumberland, RI, USA). The sampling tube was placed at the base of the cage such that gas samples within the test cage were taken at a similar height to that of the mouse exposure.
Diagram of experimental set-up.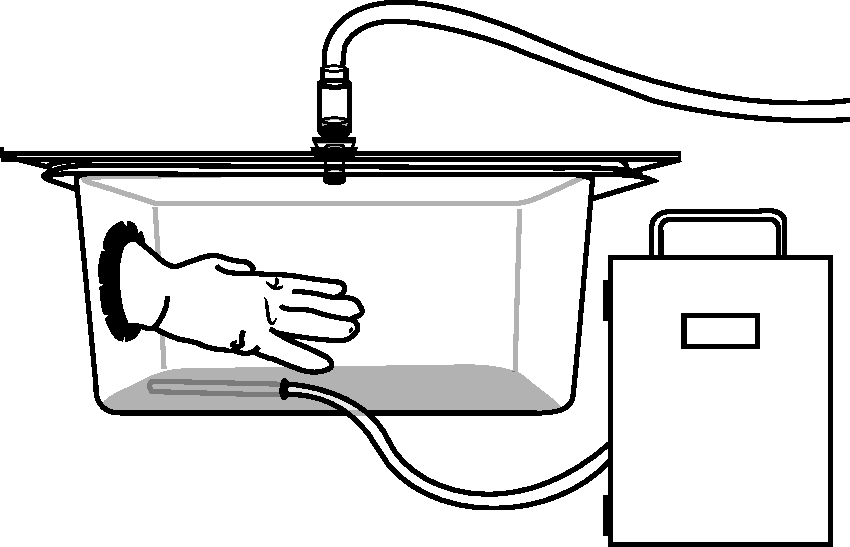
Oxygen analyzer testing
Prior to experimental testing, the lag time of the O2 analyzer was measured as the time from insertion of an anoxic sample until the time the reading on the analyzer began to decline. Repeat testing showed this delay to be 10 s. When euthanizing mice with gradual-fill CO2, an uneven distribution of CO2 occurs within the euthanasia chamber due to flow rate turbulence and CO2 heaviness in comparison to air. To assess variability in CO2 concentration within the test cage, the sampling tube independently measured seven different areas (all four corners, the center, and the two ends of the test cage with placement in the middle at each end), one area at a time, with a CO2 flow rate of 20% cage vol/min. O2 concentrations were recorded every 15 s for 5 min. CO2 concentration in the test cage was calculated: [CO2(t=x)] = 100 – (100 × ([O2(t=x)]/[O2(t=0)])). Values for the anterior left corner were found to be most similar to the average of the readings for the test cage (see Figure 1 for placement of the O2 analyzer tube).
Experimental procedure
One researcher retrieved a mouse from the housing room while another cleaned the apparatus, added 500 mL of bedding, and a stainless steel 3.5 inch straight mosquito hemostat (Lawton, Fridingen, Germany), and placed the O2 sampling tube through the cage hole into the left anterior corner of the test box. Higher flow rates result in a faster accumulation of CO2 within the test box. During each trial the O2 analyzer readings were monitored and the gas flow was altered via manual adjustment of the CO2 flow meter such that CO2 cage concentration was held just below 40%. In this way conscious mice were never subjected to CO2 concentrations associated with pain.
At the beginning of each trial a mouse was placed into the test cage. The Plexiglas lid was placed on top of the cage and the CO2 tube was placed into the centrally placed lid hole. A researcher then placed one hand into the test cage glove and kept the hand motionless on the floor of the cage. Trials began with an onset of CO2 into the cage; mice were randomly assigned to one of four flow rates: 20 (n = 6), 30 (n = 6), 40 (n = 6), or 50 (n = 6) % cage vol/min. Once the mouse was recumbent the experimenter (blind to treatment) tested for loss of the righting reflex by placing the mouse on its back. Three mice attempted to escape the approaching hand and in these cases the researcher waited until the animal was recumbent again for 3 s, before re-testing. Immediately after failure to self-right, loss of the pedal withdrawal reflex was tested, and then re-tested every 10 s using the hemostat (first notch) to pinch alternating hind paw interdigital webbing. Loss of the pedal withdrawal reflex was signified by the absence of a response to three consecutive pinches. After loss of this reflex, gas flow was increased to 60% cage vol/min until the mouse was no longer breathing. The gas was then turned off and cervical dislocation was used to ensure death.
Data collection
Each trial was recorded using a high definition camera (Model TM41P, Panasonic Corporation, Osaka, Japan). Videos were scored for: onset of labored breathing, onset of recumbency, loss of the righting reflex, and loss of the pedal withdrawal reflex (Table 1), with observers blind to treatment. We calculated the interval between onset of labored breathing–onset of recumbency, onset of labored breathing–loss of the righting reflex, and onset of labored breathing–loss of the pedal withdrawal reflex. Before the experiment, all behavioral scoring was practiced to establish inter-observer reliability. Video recordings of previous mouse euthanasia procedures were viewed and scored independently by two observers; all score times were consistent (±2 s) between observers. One observation from the 30% treatment group was identified as an extreme outlier (more than three standard deviations above the mean) for loss of the pedal withdrawal reflex and was removed from the analysis, resulting in five subjects for this treatment.
Statistical analysis
The effect of flow rate (three degrees of freedom) on behavioral responses was tested with a general linear model (Proc GLM in SAS v. 9.2; SAS Institute Inc, Cary, NC, USA) that included the home cage as a block (with eight degrees of freedom) and mouse body weight as a covariate (one degree of freedom). Least-square means ± one standard error are reported below.
Results
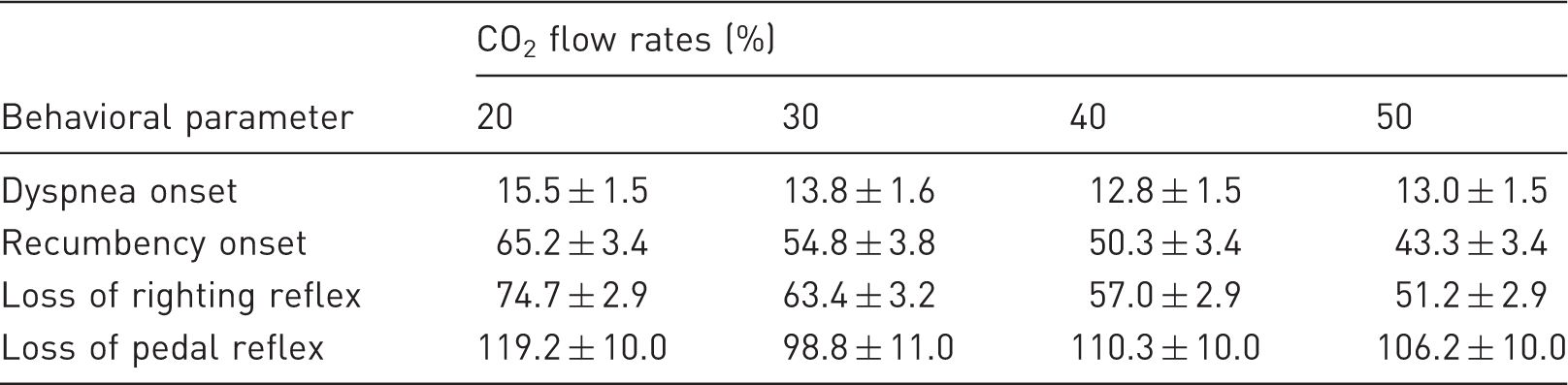
The onset of dyspnea (i.e. the onset of labored breathing) occurred approximately 14 s after the start of gas flow; onset and dyspnea did not vary with gas flow (F3,22 = 0.70, P = 0.56; Table 2). Our most conservative estimate of insensibility (loss of the pedal withdrawal reflex) occurred approximately 109 s after CO2 began to flow into the cage, again with no effect of flow rate (F3,22 = 0.66, P = 0.59), but with considerable between-subject variation. The interval between the onset of labored breathing and the pedal withdrawal reflex averaged approximately 95 s, and was also not affected by gas flow (F3,22 = 0.57, P = 0.64). Less conservative estimates of insensibility (recumbency and loss of the righting reflex) did vary with gas flow (F3,22 = 7.12, P = 0.0021 and F3,22 = 11.68, P = 0.0001, respectively). Mice became recumbent approximately 22 s sooner when exposed to a 50% versus 20% flow rate of CO2. Similarly, loss of the righting reflex occurred at approximately 51 s at the highest flow rate versus 75 s at the lowest flow rate. The interval between onset of labored breathing and onset of recumbency was reduced with increasing flow rate (F3,22 = 7.83, P = 0.0013, Figure 2a), as was the interval between onset of labored breathing and loss of the righting reflex (F3,22 = 13.62, P < 0.0001, Figure 2b).
Mean (±SE) interval in which mice exposed to a 20, 30, 40 or 50% flow rate of gradual-fill CO2 may have experienced dyspnea. Two periods are shown: (a) onset of labored breathing until recumbency, and (b) onset of labored breathing until loss of the righting reflex. Mean (±SE) time of first sign of dyspnea and three measures of insensibility in mice euthanized using gradual-fill CO2 at flow rates of 20, 30, 40, and 50% chamber vol/min.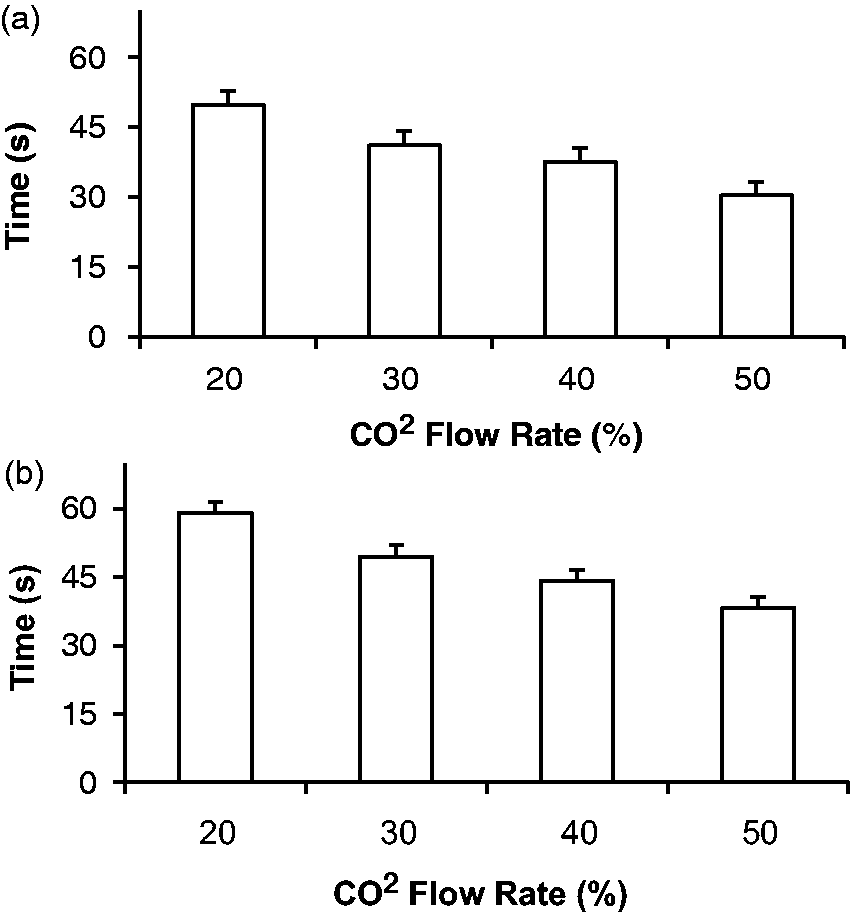
Discussion
The onset of labored breathing occurred at a similar time for all the flow rates tested, suggesting that dyspnea may begin soon after exposure to even low concentrations of CO2. The gas flow rate had a stronger effect on the time until mice became recumbent and lost their righting reflex (and also on the intervals between the onset of labored breathing and these measures). These results are consistent with the idea that loss of sensibility will occur more quickly when animals are exposed to a higher cumulative dose of anesthetic. 29
Loss of the pedal withdrawal reflex did not vary with flow rate, and this measure showed considerable variability among mice. It is possible that some variation may be due to mice spending more or less time in areas of the cage with higher CO2 concentrations. Past studies30,31 suggest that use of a hemostat to assess loss of the pedal withdrawal reflex is the best indicator of a surgical depth of anesthesia in mice. However, these previous studies used a locking pin on the hemostat whereas we used the first notch on the hemostat. The variation in this measure may indicate difficulty in applying the hemostat in a consistent manner or differences among animals in their response to the pinch. We suggest that further work is needed to assess the repeatability of methods used to assess loss of the pedal withdrawal reflex.
A study by Coenen et al. 32 suggested that when euthanizing rats with CO2, loss of posture and muscle tone (i.e. recumbency) was correlated with the onset of an abnormal electroencephalogram pattern, and the authors suggest this indicates loss of consciousness. However, in many animal species, nociceptive reflexes such as loss of righting and loss of pedal withdrawal are commonly used to assess insensibility.25,33 It has been suggested that failure to respond to a verbal command in humans is correlated with loss of the righting reflex in rodents, both signifying loss of consciousness. 33 The correlation between loss of consciousness and lack of ability to respond to a verbal command in humans has been recognized since the introduction of anesthesia. 34 However, this definition may be problematic; awareness (appropriate response to a command) and memory may be lost at anesthetic concentrations below 50% of those needed to abolish movement. 25 Given the lack of knowledge in this area, several indicators should be used when assessing insensibility. It is possible that indicators used to assess insensibility may vary with strain. Minimum alveolar concentrations (that prevent purposeful movement) differ across strains, and depend upon the type of insensibility indicator and the type of anesthetic used. 35
We used the interval from onset of labored breathing to: (1) recumbency, (2) loss of the righting reflex, and (3) loss of the pedal withdrawal reflex, to assess the time when mice may experience unpleasant sensations associated with dyspnea. The most relevant of the three intervals assessed in this study depends upon when mice undergoing gradual-fill CO2 euthanasia are no longer able to experience a negative affect. From a welfare perspective, the best case would be that mice are unresponsive after the onset of recumbency, suggesting that they consciously experience between 30 and 50 s of dyspnea, depending on the flow rate. The worst case is that they are able to experience a negative affect associated with dyspnea up until loss of the pedal withdrawal reflex (i.e. 90 s or more) with no benefit from a faster flow rate. Severity of any negative affect should be considered in addition to the duration of the experience to assess the welfare effects of different procedures.
In humans, CO2-induced air hunger results in a conscious awareness of the urge to breath, evoked by hypercapnia. 13 CO2 causes hypercapnia and a reduction in blood pH; 36 increased respiration is the body’s attempt to eliminate excess CO2. The inability of the body to overcome this excess results in secondary effects including the physical and emotional components of dyspnea. In a future study, it would be interesting to examine when each of the three insensibility parameters occurred in relation to partial pressure of CO2 in the blood of mice being euthanized with CO2.
A study by Ziemann et al. 24 examined four paradigms to assess CO2 as a fear-inducing stimulus in mice, by examining CO2 and: (1) freezing behavior, (2) open-field test, (3) aversion, and (4) fear conditioning. A 10% concentration of CO2 was found to cause more freezing behavior and reduced time in an open-field test. Also mice with the choice between a chamber with <2% CO2 or 15% CO2 spent >90% of their time in the chamber with the lower CO2 concentration. In the fear conditioning test, mice subjected to 10% CO2 before and while receiving foot shocks showed more freezing behavior than those not subjected to CO2 while receiving the foot shocks. When re-tested the following day without CO2, mice again showed more freezing behavior if they had previously been subjected to the CO2. These results indicate that CO2 gas is both fear inducing and aversive in mice, even at the relatively low concentration of 10%. In addition, a series of studies have now shown that rodents do not willingly tolerate exposure to similar CO2 concentrations.15–21 Therefore, if CO2 is used to kill rodents, refinements to minimize distress during this procedure are important.
Our study results indicate that a gradual-fill CO2 flow rate of 50% cage vol/min reduced the period from onset of labored breathing until onset of recumbency and loss of the righting reflex. When using this flow rate, a gas holding technique should be used to ensure that painful CO2 concentrations (>40%) are not reached until after insensibility occurs. This can be achieved by manually controlling the flow meter or by using a programmable automated euthanasia machine. Once a mouse has become insensible, it is up to the user how to proceed. The animal may remain in the test cage at a concentration of just below 40% CO2 until death, or the flow rate of CO2 may be turned up to decrease time to death, or the animal may be taken out and subjected to a secondary method of euthanasia such as cervical dislocation.
Conclusion
The results of this study indicate that the time until recumbency and loss of the righting reflex in mice can be reduced using higher flow rates of gradual-fill CO2 (e.g. 50% versus 20% chamber vol/min). When using higher flow rates, a gas holding technique should be used to ensure that painful CO2 concentrations (>40%) are not reached until after insensibility occurs. Even when using this refinement, mice likely experience more than 30 s of dyspnea before insensibility occurs.
Footnotes
Acknowledgements
We thank the British Columbia Cancer Research Centre for donating the surplus animals used in this study and the staff at the Centre for Comparative Medicine for taking care of the mice for the duration of the study. This study could not have been conducted without the support of Chris Harvey-Clark and technical help from Gordon Gray. We would also like to thank Joanna Makowska for reviewing this manuscript, as well as Andre Vits and Giovanni Mancini for many conversations regarding mouse euthanasia. We are grateful to Warren Riley (Innovive Inc) for donating the test cage used in this study. This work was supported by an NSERC Discovery grant to D M Weary.
