Abstract
The rabbit is one of the most widely used models for studying bone remodeling or dental implant osseointegration but very few data are available about the rabbit’s mandible. The aim of this work was to describe the anatomy of the rabbit mandible and to estimate the available bone volume for experimental studies. First, with a dissection, the morphology of the mandible was described and the mental foramen, the position of the main salivary glands and muscular insertions were located. Then, by X-ray imaging, the position of the inferior alveolar canal, the dental root courses and volume and bone density were described. Finally, with frontal sections of the mandible body, the rabbit’s dental and alveolar bone histological structure were assessed. Thus, the relevance of the rabbit mandible as an experimental model for wound healing or surgical therapies was discussed.
Animal experimentation is often a necessary step for developing therapies applicable to man. Various animals are used as experimental models. The rabbit is one of the most widely used models for studying bone remodeling or dental implant osseointegration. According to Neyt et al., 1 it is used for approximately 35% of musculoskeletal studies. However, most of the experiments are performed on rabbit’s legs and are not directly on mandibles.2,3 There are very few data available about the rabbit’s mandible.
El Bialy et al. 4 have chosen the New Zealand rabbit to show the therapeutic effects of ultrasounds on mandibular growth, for the treatment of Class II malocclusion. Indeed they have revealed that ultrasounds enhance mandibular growth by condylar endochondral bone growth and consequently mandibular ramus growth. La Scala et al. 5 described growth alterations of the orbitozygomatic complex after a 25 or 35 Gy irradiation.
To our knowledge, there has been no anatomical or histological study of the rabbit’s normal mandible. The aim of this work was to describe the anatomy of the rabbit mandible, specifically the position of the inferior alveolar canal and the mental foramen, as well as the dental root courses to estimate the available bone volume for experimental studies. The volume and location of the main salivary glands were also studied.
A histological study aimed at determining its relevance as an experimental model, especially after treatment induced alterations through the description of the structure of the rabbit’s mandible, was also performed.
Animals, materials and methods
This work has been performed at the Institut de Chirurgie Expérimentale (ICE) of the Centre Léon Bérard, Lyon and approved by the internal ethic review committee (registered at the CNREEA No. 10; internal study number: AVI069). Four adult female New Zealand white rabbits, weighing 3.5 kg were used. These animals came from Eurolap (Le Moulin aux Moines, Argentre du Plessis, France). They were housed in individual cages with a lighting period of 12 h per day. There was an acclimatizing period of seven days before any operation. The intramuscular premedication of the animals was provided with a blend of ketamine (1 to 6 mL of Imalgene 1000™; Merial, Lyon, France) and xylazine (0.6 mL of Rompun™; Bayer AG, Leverkusen, Germany). Euthanasia was performed by an intravascular injection of 4 mL of dolethal (Vetoquinol France, Paris, France).
Scalpels, tissue retractors, tissue forceps, syringes of physiological serum, and scissors were used for the dissection. Photographs were taken using a digital camera. Measures were executed with a Dermotrace® ruler (Novatech SA, La Ciotat, France) and Image J® software (National Institutes of Health, Bethesda, MD, USA).
Two rabbits were used for the evaluation of bone volume via intraoral access. The intraoral access was made by an incision on the alveolar crest in order to visualize the available bone volume between the incisors and molars and to locate the mental foramen. The cutaneous plane was then removed in a posterior direction in order to visualize the mandible angle with the insertion of the masseter muscle, and to locate the parotid gland.
For two other rabbits, a submandibular access was performed to visualize, if present, other salivary glands as well as muscular insertions. The temporomandibular joint was then dissected, and the muscular and cutaneous planes connecting the mandible to the skull were lifted in order to dislocate the mandible and to remove it. The right half mandible was imaged with a conventional X-ray on an occlusal film to locate the inferior alveolar canal and dental root courses and to estimate the available bone volume in the various areas of the mandible. The X-ray system used was a Gendex® 765DC (65 kV/7mA; Dentsply International, Des Plaines, IL, USA), with a dose from 0.025 to 0.040 mSv.
The histological study was based on rabbit mandibular sections made with two other rabbits. The mandible samples were embedded in paraffin, and demineralized using formic acid. The sections were processed with a hematoxylin, eosin, phloxine (HEP) coloration.
Results
Rabbit teeth consist of a heterodont dentition with incisors, premolars and molars, but no canines. They have 28 teeth: four incisors, six premolars and six molars on the upper maxillary, and two incisors, four premolars and six molars on the mandible. There is a relatively large diastema between the incisors and premolars, mainly due to the lack of canines. Premolars and molars have approximately the same morphology. Their teeth have a continuous mode of growth. The root is not an actual root, but rather a continuation of the crown in the gum and alveolar bone, which defines an elodont dentition. 6
With an intraoral access it is possible to have an overview of the mandibular corpus (Figure 1). The diastema between the incisors and premolars is about 19 mm long. The presence on the corpus of the mental foramen can be seen at the posterior 2/3 of the diastema. The mental nerve, which is a branch of the inferior alveolar nerve, comes out through this foramen. The rabbit’s mental foramen is located 0.8 mm under the bone crest, and 4.6 mm above the mandibular basilar edge. The total bone height in this area is about 6.5 mm.
Mandible’s corpus lateral view by intraoral access. In a posterior direction, it can be seen that the masseter muscle has been cut.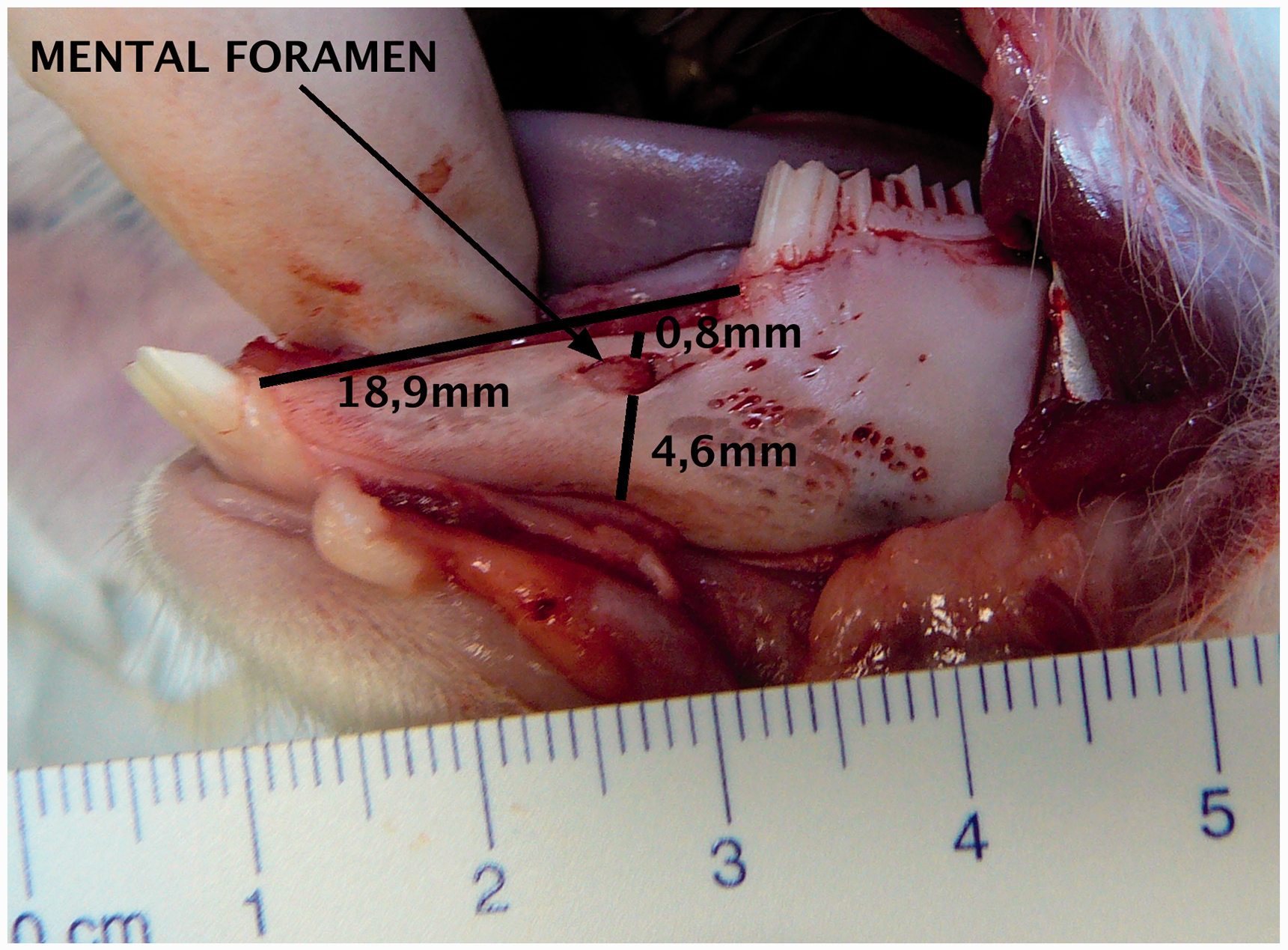
The resection of one-half of the mandible allows the description of the morphology of this bone (Figure 2). On the lateral view, it has a global triangular shape. The corpus is narrow and elongated in an anterior direction. The bone volume is important (43 to 96 mm3) in this area, with the presence of the trabecular bone. The ramus is wider, with a very developed condylar process. It is marked with voluminous muscular insertions: the masseter muscle on the lateral face, and the medial pterygoid muscle on the medial face. These are both chewing muscles, elevators of the mandible. Despite these voluminous chewing muscles, it must be specified that the rabbit is not a rodent because rodents only have one pair of incisors per maxillary. As a result of these wide muscular insertions there is a much reduced bone volume of the ramus and the mandibular angle. Indeed, we observe an apposition of two corticals composed of compact bone. Light can be seen by transparence through the bone. It can be seen that the mandibular angle is reinforced with a voluminous bone crest that ends in a posterior direction with an angular process. Moreover, on the medial face of the mandible, the mandibular foramen can be observed, whereby the inferior alveolar nerve penetrates. Then, the nerve has an intraosseous course.
Medial view of the right half of the mandible showing the mandibular foramen, and the mandibular part of the temporomandibular joint.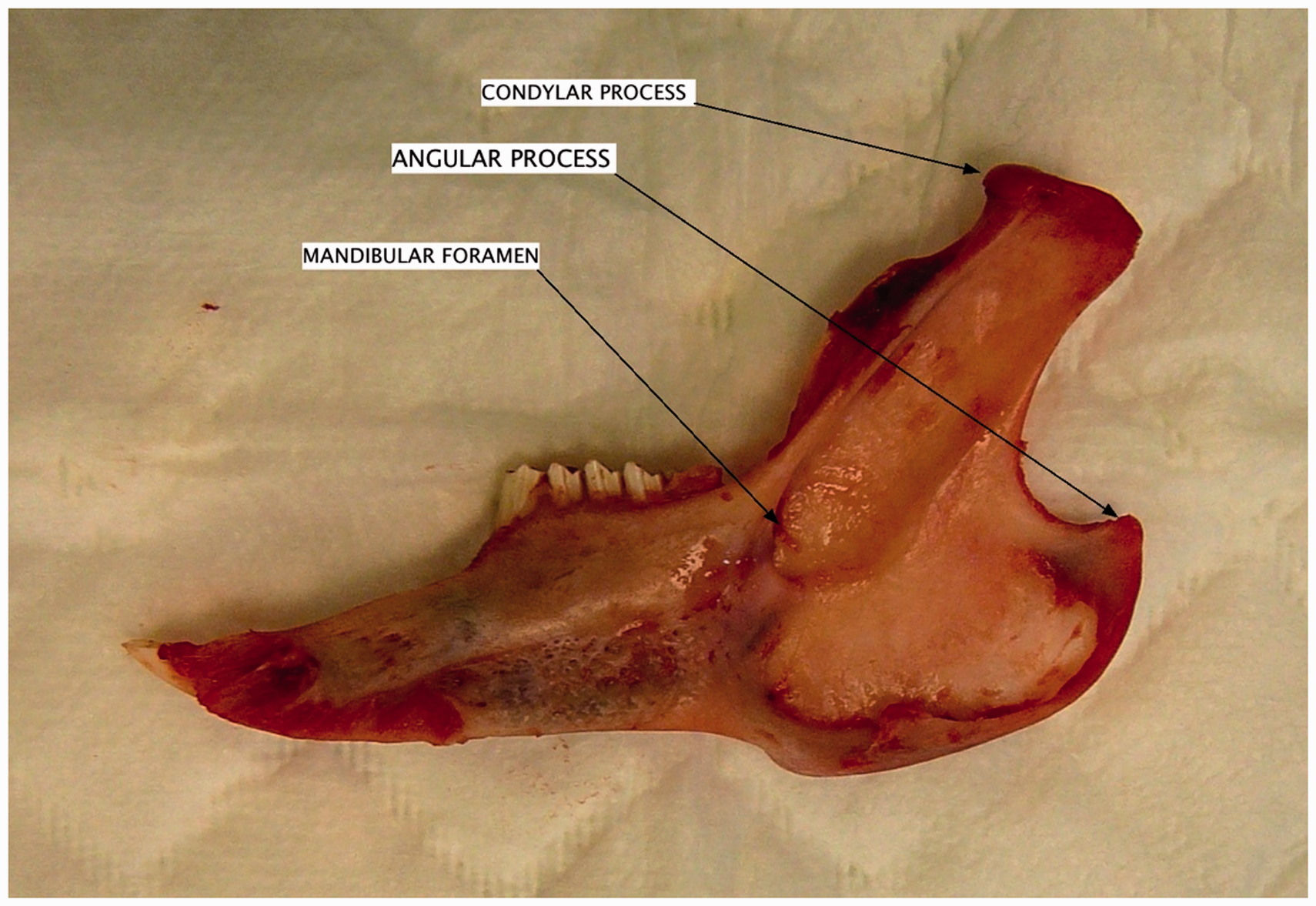
A transversal section of the mandible shows a thickness of 3 to 6 mm in diameter in the anterior part of the mandible and 2 to 4 mm in the posterior part. The average available bone volume between the incisors and molars ranges from 43 to 96 mm3. The inter-incisive stitch appears to be very weak and could be described more as a gliding joint.
After the resection of cutaneous planes in a posterior direction from the ramus, the parotid gland can be observed. This gland is large (2 to 3 cm2) and is located above and behind the formation of the external jugular vein by the merging of the retromandibular and posterior auricular veins. The parotid duct runs laterally in front of the masseter muscle and then goes round its anterior side (Figure 3).
Parotid gland.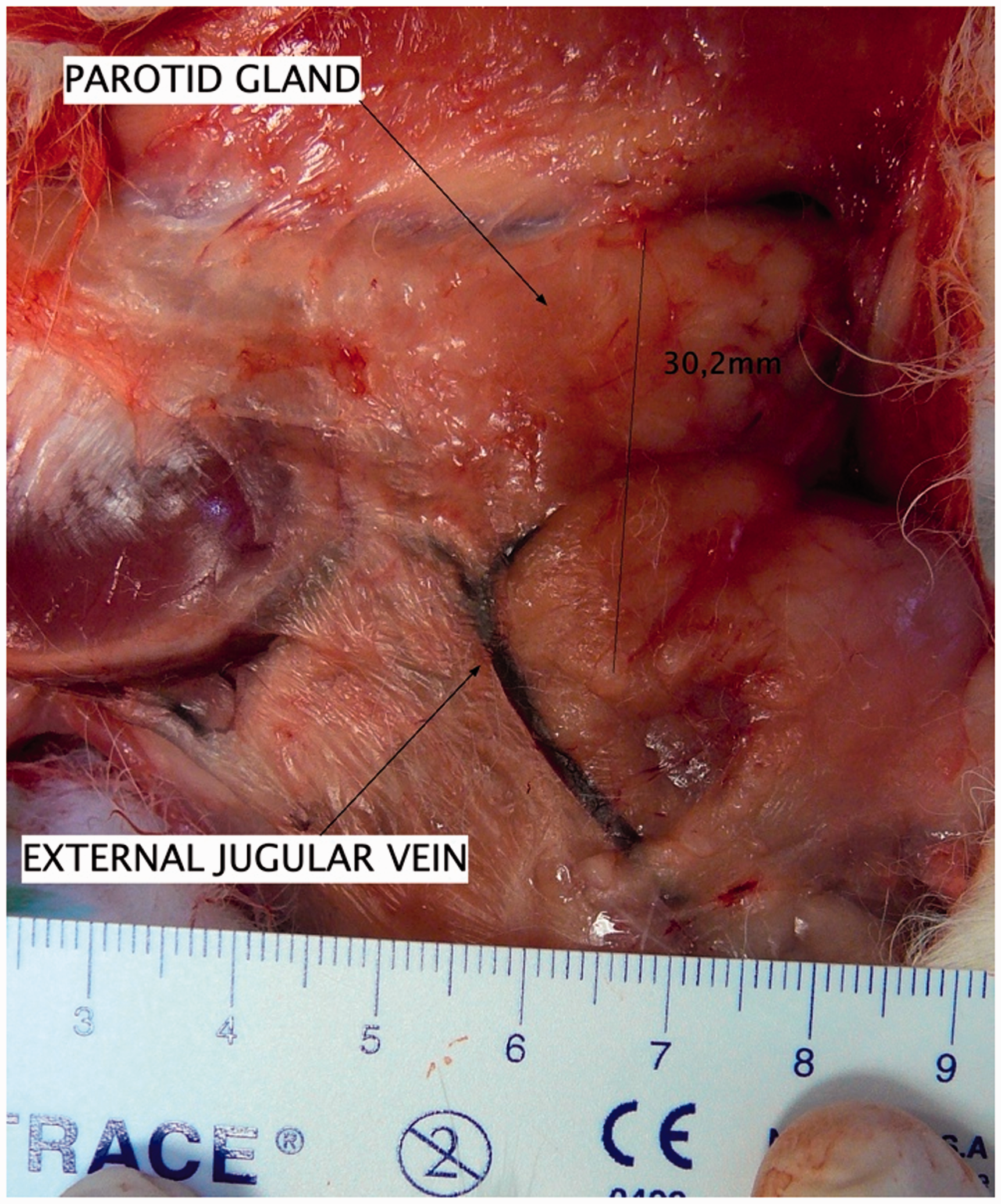
With the submandibular access dissection, the submandibular glands can also be located. The sublingual salivary glands, as described by Barone, 7 appeared to be difficult to differentiate from the submandibular glands.
A conventional X-ray radiography was performed on an occlusal film, to describe the mandible’s radiographic anatomy (Figure 4). Thus, the premolar’s and molar’s root courses could be observed.
X-ray radiography of a right half mandible.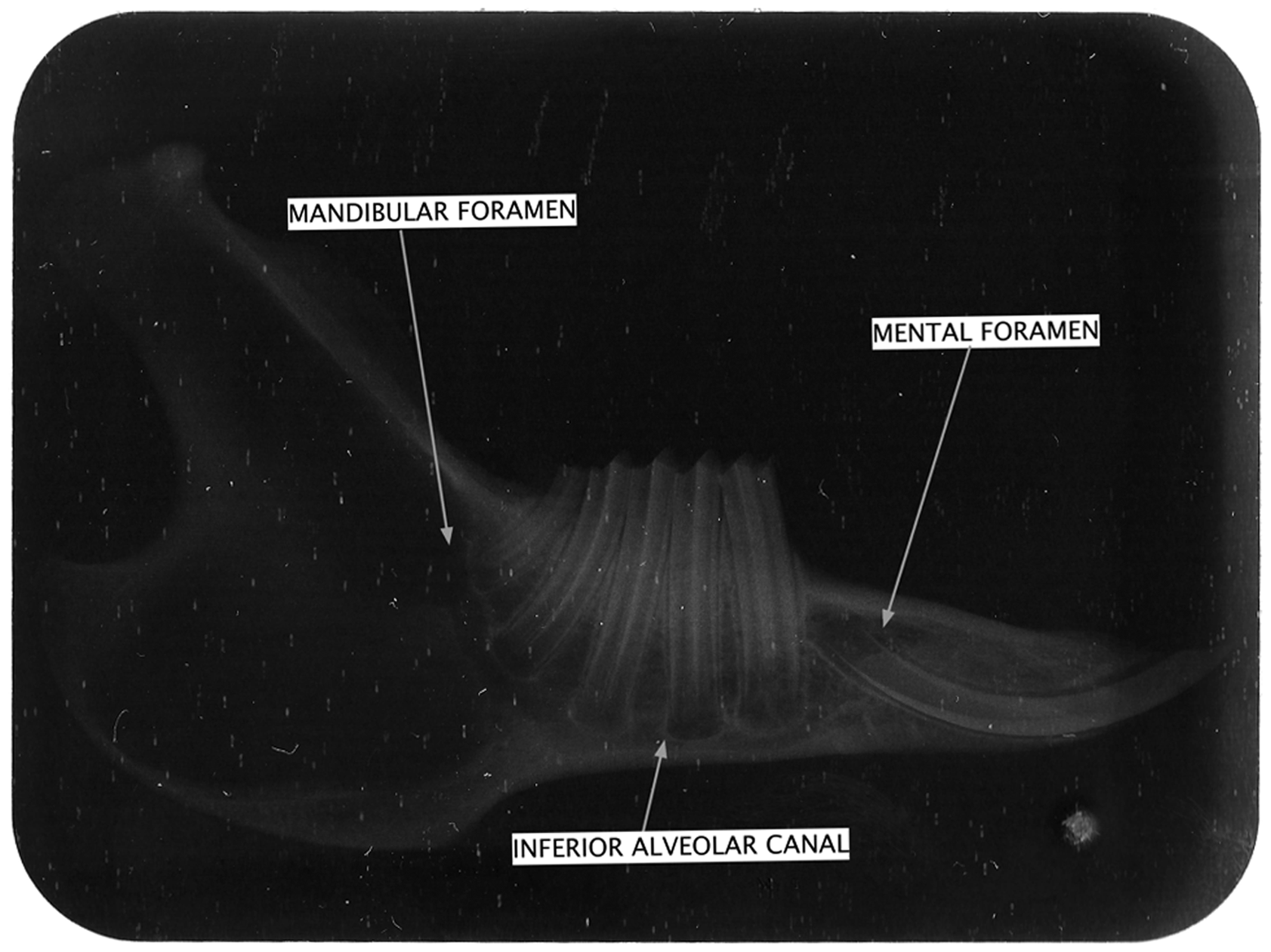
The roots of the premolars are more elongated than those of the molars. The apexes appear to be widely opened whereas it is an adult animal. The inferior alveolar canal, in which the alveolar nerve runs, has a course passing by the teeth’s apexes. Consequently the alveolar canal is located in a much inferior position in the body of the mandible. This implies that bone volume is reduced in the area around the posterior teeth. The dental roots occupy almost the entire mandible height in the teeth area, and the part of the bone located underneath the apexes is much less.
The incisors have an elongated and curved root, in the posterior and upper directions. The mental foramen is located slightly anterior and above the incisor apex.
Furthermore, using radiography, bone thickness can be estimated in various areas of the mandible. The ramus appears to be very radiolucent because it is very thin and it absorbs little of the X-rays. However, the basilar and anterior sides of the ramus appear to be more radiopaque, which corresponds to the basal cortical bone.
The mandibular body has a more radiopaque aspect in its anterior area, facing the diastema between the premolars and incisors. This corresponds to a higher bone volume and the presence of cancellous bone.
Frontal sections of the mandible body were performed for the histological study (Figure 5). According to these sections, the global dental structure can be described, with a dental root with a pulp space delimited by dentine, which is fixed by conjonctival fibers. As for bone, large amounts of spongy bone with quite expanded medullar spaces containing adipose tissue can be observed. The cortical bone, which is compact, and the Haversian bone can also be observed.
Histological section of mandible with hematoxylin, eosin, phloxine (HEP) coloration.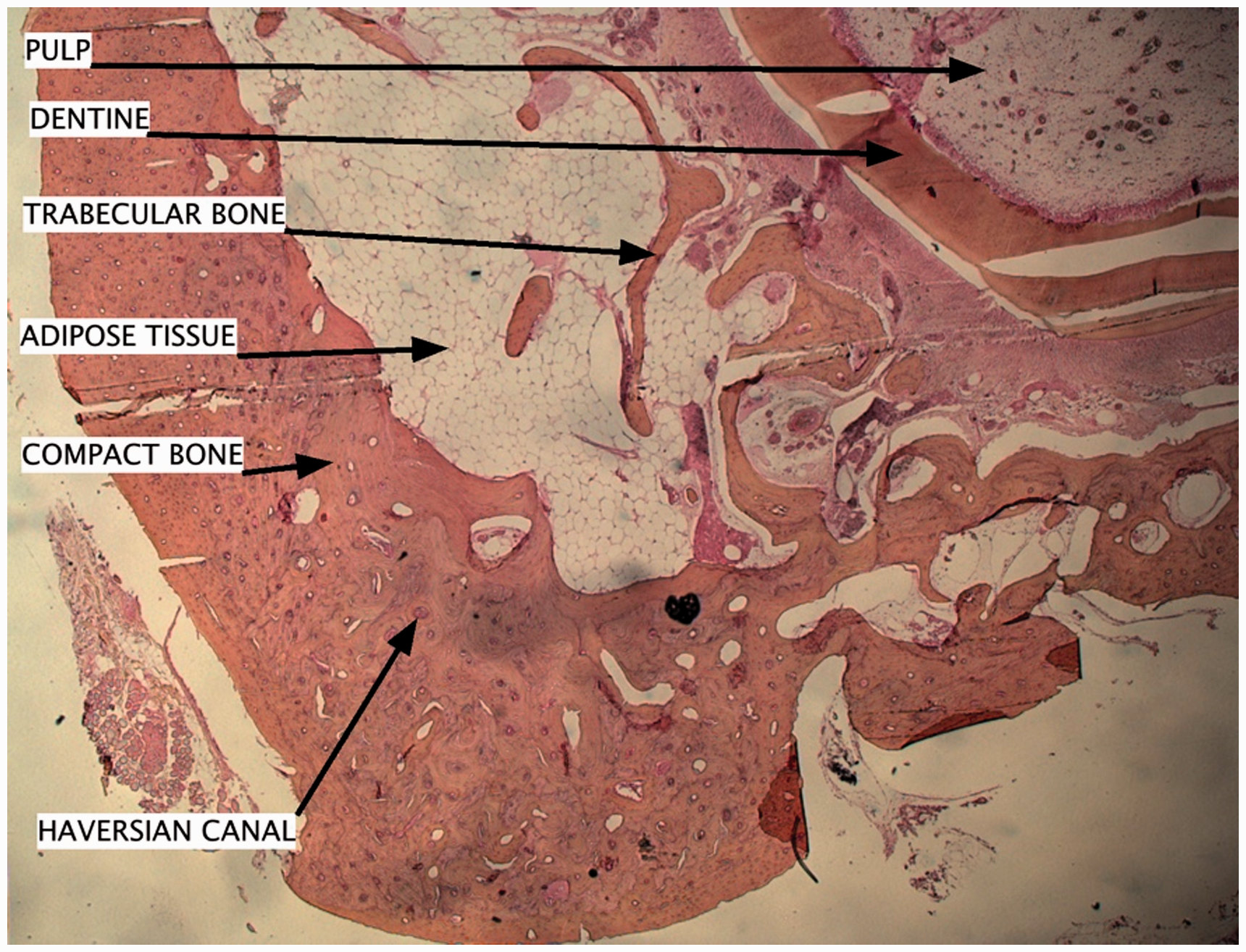
Discussion
The rabbit is a widely used experimental model for oral surgery and implantology tests. It has a convenient size, is inexpensive, and in some respects has a physiology comparable to humans. One of the rabbit’s main advantages is that it reaches its skeletal maturity shortly after reaching its sexual maturity, which is at 6 months. 8
In this study, two different accesses have been used. The intraoral access aimed to evaluate the mandible as an experimental model for intraoral surgery, and the submandibular access aimed to evaluate the size and location of the parotid gland.
A detailed observation shows bone morphology to be very different from the human one. The mandible corpus is elongated and has a low bone height. The ramus is thin and marked with important muscular insertions, which is not surprising, considering the well-developed chewing function of the rabbit. The mental foramen is located 4.6 mm above the basilar edge at 2/3 of the diastema between the incisors and molars, which allows a 43 to 96 mm3 bone volume to be available for experimentation. By contrast, the human mental foramen is located in an area facing the first or second premolar’s apex.
The tooth morphology is also very different, with the absence of a distinction between the crown and root due to the rabbit’s continuous tooth growth. The apexes appear to be opened, which could be due to the continuous mode of growth.
In comparison with the human parotid gland, the gland is relatively larger and is located in a position more posterior from the masseter muscle in the rabbit.
According to the radiographic study, the anterior area of the mandible body, facing the diastema between the incisors and premolars, seems to be the more appropriate area for bone experimentation on the mandible, because of the presence of cancellous bone.
Histologically, the distinction between alveolar and basal bones, described in the human mandible, does not seem appropriate for the rabbit. The main difference with the human mandible’s bone is given by the richness of medullar spaces with large amounts of adipose tissue. Human bone marrow contains more hematopoietic cells and less adipocytes than the rabbit’s bone marrow. One may wonder how this tissular composition affects bone healing, considering that adipose tissue contains higher densities of mesenchymal stem cells than bone marrow. 9 It is known that adipose tissue is used for autologous grafts to repair tissue defects after radiotherapy or surgical tumor resection. Furthermore, Lim et al. have shown a protective effect of human adipose tissue-derived mesenchymal stem cell transplantation against irradiation-induced cell damage in the salivary glands. 10 Moreover, according to Castañeda et al., 11 rabbit bone turnover is faster than that of primates or rodents.
All of these differences require careful consideration before extrapolation to humans of any experimental results performed using rabbit’s bones. Aerssens et al. 12 have noticed marked inter-species differences regarding bone composition, bone density and bone mechanical competence. They have concluded that the dog bone gives the best approximation to human bone characteristics.
Using the rabbit as an experimental model has several advantages such as cost and size; but one should be aware of differences that may have an impact on experimental results.
Footnotes
Declaration of conflicting interest
None.
