Abstract
Eighteen female New Zealand White rabbits (3.9 ± 0.4 kg) were anaesthetized with sufentanil–midazolam by intravenous infusion (SUF–MID, n = 9) or isoflurane (ISO, n = 9) for bilateral creation of an osteochondral defect in the medial femur condyle. Subcutaneous premedication with 0.1 mg/kg medetomidine and anaesthesia induction by intravenous infusion of 1.1 µg/kg sufentanil and 0.2 mg/kg midazolam were identical in both groups. During surgery (60 min), the effects on respiratory and circulatory variables serum lactate, total protein and blood glucose were examined. Intermittent positive pressure ventilation (IPPV) was initiated if apnoea lasted>30 s or if end-tidal CO2 ≥8 kPa. The righting reflex was lost in 3 min. IPPV was necessary during most of the anaesthesia for most of the rabbits. Maintenance doses during surgery were 2.0 µg/kg/h sufentanil and 0.4 mg/kg/h midazolam, and 1.4% isoflurane, respectively. Mean arterial blood pressure (MAP) was higher in group SUF–MID than group ISO during surgery (63 ± 12 vs 50 ± 8 mmHg). In group ISO the heart rate was higher during surgery than before anaesthesia (197 ± 26 vs 158 ± 40 bpm) as was blood glucose (9 ± 2 vs 12 ± 3 mmol/L). Serum lactate levels remained unchanged whereas total protein decreased in both groups. Time to recover from anaesthesia did not differ between groups (20 min). Intravenous sufentanil–midazolam infusion provided surgical anaesthesia with a higher MAP than isoflurane anaesthesia. The protocol can be useful in situations in which gas anaesthesia cannot be used or in animals with limited cardiovascular reserves. However, IPPV is necessary.
Keywords
The rabbit (Oryctolagus cuniculus) is the third most commonly used laboratory animal species, and is frequently used in orthopaedic research.1,2 Anaesthesia in rabbits is associated with a high mortality rate, in healthy as well as sick animals.3,4 The reasons are largely unknown; possible causes are high stress levels, difficulty of intubation and failure to intubate the airways, and neglect in monitoring during and after anaesthesia. Several rabbit anaesthesia studies report a high prevalence of hypotension5–7 which may contribute to the risk if it passes unnoticed and is not treated.
The use of rabbits in medical research can be very costly, especially if the animals undergo surgery followed by a long postoperative recovery and rehabilitation phase, which is common in orthopaedic research. Thus, every type of refinement that may reduce anaesthesia-related risks is an economic as well as ethical improvement. Moreover, with some research techniques, such as magnetic resonance imaging, positron emission tomography or extracorporeal studies (e.g. in a heart–lung machine) or during transportation of anaesthetized animals, there is a need for alternatives to inhalation anaesthesia. Advantages of total intravenous anaesthesia (TIVA) protocols are the avoidance of environmental pollution and prevention of exposure of staff to volatile anaesthetics. The need for endotracheal intubation and provision of oxygen is however independent of the anaesthetic protocol used.
Few protocols for intravenous infusion anaesthesia have been described in rabbits, one being fentanyl–fluanisone–midazolam. 8 Fentanyl is, however, more slowly metabolized compared with the opioid sufentanil, which therefore is potentially more suitable for use in rabbits which will recover from anaesthesia. Other reported intravenous protocols only provide a light level of anaesthesia, insufficient for surgery, such as propofol or alphaxalone–alphadolone, 9 and include the use of neuromuscular blocking agents, which makes the evaluation of anaesthetic depth difficult 10 or are non-recovery studies. 11
Intravenous infusion anaesthesia based on rapidly metabolized opioids have been described in dogs, swine, primates and humans.12–16 Opioids provide an excellent intraoperative analgesia, with little or no reduction of cardiac contractility or cardiac output, and an effective protection from surgical stress by the inhibition of sympathetic activity. 16 In higher doses opioids often cause bradycardia, which can be reversed with anticholinergic drugs. 17 Short-acting opioids have been shown to provide haemodynamic stability during as well as after surgery.12,18 In comparison with volatile anaesthetics, opioids produce less severe hypotension and protect the heart from the arrhythmogenic effects of epinephrine. 19
The use of sufentanil–midazolam for intravenous infusion anaesthesia in medetomidine-premedicated female New Zealand White (NZW) rabbits was recently evaluated. 20 The conclusion was that sufentanil–midazolam provided anaesthesia with a rapid and smooth induction and recovery, marked depression of nociceptive reflexes, good muscle relaxation and an increase in cardiac output, whereas blood pressure and respiratory rate were markedly decreased. In that study, there was no control group and no surgery was performed. In the present study, the combination was further evaluated, by measuring cardiorespiratory variables and serum lactate and total protein, and blood glucose in medetomidine-premedicated NZW rabbits undergoing stifle joint surgery for the creation of cartilage defects. 21 For comparison, surgery was performed under maintenance with either sufentanil–midazolam or isoflurane anaesthesia.
Materials and methods
Animals and housing
Twenty female NZW rabbits, with a mean age of 7 months (range 6.5–8.3) and a mean body weight of 3.9 kg (range 3.5–4.4) were obtained from a specific pathogen free breeding colony (Lidköpings kaninfarm, Lidköping, Sweden). According to health monitoring by the Federation of European Laboratory Animal Science Associations (FELASA) recommendations 22 the colony was free from RDHV, rabbit rotavirus/rabbit coronavirus, Bordetella bronchiseptica, Clostridium piliforme, Pasteurella multocida, Salmonella spp., endoparasites, ectoparasites and dermatophytes.
The rabbits were housed in twos or threes in home-made floor pens with an area of 1.4–2.0 m2 per rabbit. The pens were equipped with shelters and autoclaved pine wood shavings and cleaned weekly. The animals were fed a restricted standard pelleted rabbit diet (Lactamin K3; Lantmännen, Stockholm, Sweden) and autoclaved hay and had access to water ad libitum. For enrichment, the rabbits were given aspen gnawing blocks and small bits of washed apple or pear. The light–dark-cycle was 12:12 h with lights on at 06:00 h. Room temperature was 20 ± 2℃ and humidity 55 ± 10%. The animals were acclimatized to the new environment for three weeks before study start. Body weights were recorded daily starting five days before surgery. The experiments were approved by the ethics committee for animal experiments in Uppsala, Sweden (C158/10).
On the day before anaesthesia and surgery, the rabbits were clinically examined and a blood sample (1 mL) was taken from the ear artery after application of a local anaesthetic cream (EMLA®; AstraZeneca, Södertälje, Sweden). Haematology and complete blood cell counts (CBC) were performed with ADVIA® Automated Solutions (Siemens Health Care Diagnostics, Erlangen, Germany); and analyses of serum amyloid A (SAA) with an ELISA kit (Tridelta Development Ltd, Maynooth, Ireland), serum lactate with a GM7 analyser (Analox Instruments Ltd, London, UK), and total serum protein with a refractometer (Master, Atago, Kruuse Svenska AB Uppsala, Sweden).
Experimental design and surgical procedure
Medetomidine-sedated rabbits were randomly allocated with a random number generator (Microsoft Office Excel 2007, Microsoft, Kista, Sweden) to receive either isoflurane by inhalation (group ISO, n = 10) or a mixture of sufentanil–midazolam by intravenous infusion (group SUF–MID, n = 10) for the maintenance of anaesthesia. In both groups anaesthesia was induced with sufentanil–midazolam intravenously. This part of the study was not blinded. During anaesthesia orthopedic surgery was performed. Surgery included the drilling of a bilateral full thickness osteochondral defect of 4 mm in diameter in the femoral medial condyle. Unilaterally, a hyaluronic hydrogel system, with or without bone morphogenetic protein-2 (BMP-2), was delivered intra-articularly. 21 The contralateral joint defect was treated with saline as a control. The rabbits were randomly allocated into the cartilage treatment groups using a random number generator (Microsoft Excel 2007). The surgical procedure lasted approximately one hour. The joint was accessed by a medial longitudinal parapatellar incision, the patella was laterally dislocated, and the defect was created by drilling in the medial femur condyle down to the subchondral bone. The patella was relocated, the joint capsule tightly sutured, and a volume of 0.4 mL of the hyaluronan hydrogel (with or without BMP-2) or saline was injected into the joint, before the skin wound was closed.
Premedication and preparation for anaesthesia and surgery
The rabbits were premedicated in the home pen with medetomidine (Domitor®, 1 mg/ mL; Orion Corporation, Espoo, Finland) 0.1 mg/kg subcutaneously in the scruff. At the same time local anaesthesia cream (EMLA, AstraZeneca) was applied to the outside skin of both ears. Thirty minutes later the rabbits were transported (∼5 min) in a cage to the surgical preparation area in an adjacent building. The rabbits were left in the cage for at least 10 min in the preparation area. The respiratory rate was then counted by observing abdominal movements, and the heart rate counted by auscultation using a stethoscope. To avoid infection the antibiotic ceftiofur (Excenel® vet, Orion Pharma Health, Sollentuna, Sweden) was administered intramuscularly with a dose of 10 mg/kg. An intravenous catheter (Neoflon 24 G; BD Medical, Stockholm, Sweden) was placed in the lateral ear vein for the administration of Ringer acetate solution (10 mL/kg/h) during surgery. The rabbits in group SUF–MID received a second intravenous catheter in the other ear, for separate delivery of Ringer acetate and sufentanil–midazolam solutions. An intra-arterial catheter (Insyte-W catheter, 22 G; BD Medical) was placed in the central ear artery for the measurement of blood pressure and blood sampling. One millilitre of arterial blood was collected at each time-point for measurements of blood gas, serum lactate, total protein, and blood glucose. A total of 3.5 mL blood/kg body weight was collected and replaced with Ringer acetate infusion. The fur of the tail was clipped for placement of a pulse oximeter probe for measuring oxygen saturation (SpO2).
Induction of anaesthesia
Before induction, baseline values (SEDATION) of SpO2, arterial blood gases, serum lactate and total protein and blood glucose were recorded in the sedated rabbits. Anaesthesia was induced with a mixture of sufentanil–midazolam intravenously in all the rabbits. The solution was prepared by mixing 50 µg sufentanil (Sufenta®, 50 µg/mL; Jansen-Cilag, Sollentuna, Sweden) with 10 mg midazolam (Midazolam®, 1 mg/mL; Actavis AB, Stockholm, Sweden) and physiological saline to a final volume of 22 mL, giving a concentration of 2.3 µg/mL of sufentanil and 0.45 mg/mL of midazolam. The mixture was delivered by an infusion pump (Braun Compact S; B Braun Melsungen AG, Melsungen, Germany) at a flow rate of 0.3–1.8 mL/kg/h. One minute after the start of the infusion, boluses of 0.1 mL of the sufentanil–midazolam solution were additionally injected intravenously every 20 s until the induction was complete, as indicated by a loss of righting reflex and reduction of muscle tone. During induction 100% oxygen was delivered via a face mask at a flow rate of 3 L/min. Bolus volume and time to induction were noted and the larynx was sprayed with 0.5 mL of lidocaine (Xylocaine® 10 mg/ mL; AstraZeneca). After one minute, endotracheal intubation of the airways was attempted using an uncuffed PVC endotracheal tube of 3.0–3.5 mm outer diameter (Rusch; Swevet, Sjöbo, Sweden) after visualization of the larynx by a small animal laryngoscope (Flecknell®; Alstoe Ltd, York, UK). The tube was connected to the anaesthesia machine (Anmedic Q-Circle System; Anmedic AB, Stockholm, Sweden) via a pediatric circular breathing system (Intersurgical Ltd, Wokingham, UK) providing a mixture of 1.5 L oxygen + 0.5 L air/min. Following endotracheal intubation, the rabbit was placed in dorsal recumbency on an electric heating pad, the allocated anaesthesia maintenance protocol started and the rabbits were prepared for surgery. Preparation for surgery consisted of the clipping of fur, washing and disinfecting of the skin.
Maintenance of anaesthesia
Anaesthesia was continued and maintained with either an infusion of sufentanil–midazolam (group SUF–MID) or inhalation (group ISO) of isoflurane (Isoba® vet; Intervet, Sollentuna, Sweden) from a precision vaporizer (Tech 5; Datex Ohmeda GE Health Care, Stockholm, Sweden).
During preparation for surgery the same infusion rate as during induction was continued in group SUF–MID. In group ISO, the initial isoflurane vaporizer setting was 1%. If apnoea occurred (>30 s), a reservoir bag (0.5 L) was carefully compressed every 30 s until spontaneous breathing was resumed or the rabbit had been moved into the surgical theatre where IPPV was initiated (see below).
After preparation for surgery, the rabbits were disconnected from the vaporizer or the sufentanil–midazolam infusion pump and carried into the adjacent surgery room and anaesthesia delivery was resumed. The start of surgery was recorded. During surgery the sufentanil–midazolam infusion rate or isoflurane concentration were adjusted to maintain an adequate level of anaesthesia, i.e. preventing a nociceptive response in the form of spontaneous movements or marked changes (>10%) in cardiorespiratory variables.
In the surgery room an electrical heating blanket was used to maintain body temperature and the Ringer acetate infusion was started (10 mL/kg/h). The arterial catheter was connected to a pressure transducer (Gabartith; BD Medical) which was calibrated to air at the level of the heart. Self-adhesive electrocardiography (ECG) electrodes (GM Health Care, Stockholm, Sweden) were placed laterally on the elbows and the left knee (lead II registration). A human pulse oximeter finger clip sensor (Datex-Ohmeda GE Health Care) was placed on the tail of the rabbits. The ECG leads, the pulse oximeter probe and a rectal thermometer were connected to a monitor (CS/ 3; Datex-Engstrom, Datex Ohmeda, Bromma, Sweden) for continuous recording and storage on a PC (software: Datex-Ohmeda S/ 5 Collect; GE Healthcare Sverige AB, Stockholm Sweden). Heart rate was deduced from the ECG or pulse oximeter during anaesthesia. End-tidal CO2 (ETCO2) was monitored using sidestream capnometry. Respiratory rate was obtained from the capnometry readings of the Datex monitor.
During surgery, the rabbits were allowed to breathe spontaneously, unless apnoea (30 s without a breath) occurred or ETCO2 was ≥8 kPa, in which case IPPV was performed, using a ventilator (Anmedic vent JB1; Anmedic AB). The ventilator was used at a frequency of 5–20/min, as needed to maintain ETCO2 at 5–8 kPa, a maximum peak inspiratory pressure of 20 cmH2O and an inspiration:expiration ratio of 1:3.
Monitoring and sampling during anaesthesia and surgery
The following parameters were recorded every 10 min from the start of preparation in the anaesthetized rabbits to the end of surgery: SpO2, heart and respiratory rates, end-tidal isoflurane concentration (ETiso) and sufentanil–midazolam infusion rate. Body temperature, mean, systolic and diastolic blood pressures and ETCO2 were recorded at the same time intervals during surgery. Arterial blood samples (1 mL) were taken anaerobically into non-heparinized syringes at 15, 30, 60 and 90 min after the start of preparation as well as 30 min after the end of surgery (POST-OP). A portable analyser (CG8 + cartridges, i-STAT®1 Portable Clinical Analyzer; Abbott Laboratories, Abbott Scandinavia AB, Solna, Sweden) was used for immediate analyses of haematocrit, pH, PaCO2 and PaO2. Blood analyses included concentrations of serum lactate and total protein, and blood glucose. Arterial blood gas and pH values were corrected to body temperature.
Recovery and postoperative care
After the completion of surgery, anaesthesia was discontinued and buprenorphine (Temgesic®, 0.3 mg/mL; RB Pharmaceuticals Ltd, Slough, Berkshire, UK) was administered intravenously (0.03 mg/kg) and subcutaneously (0.02 mg/kg). Delivery of oxygen and air was continued until endotracheal extubation. Time to extubation and recovery of the righting reflex were recorded. Following extubation, the rabbits were placed in a cage with a temperature of 26℃ for recovery. Once fully recovered, the rabbits were returned to their home pens.
The rabbits that underwent surgery in the morning received an additional dose of buprenorphine subcutaneously (0.05 mg/kg) the same evening (POST-OP day 0). On postoperative day 1, buprenorphine was administered twice subcutaneously at a dose of 0.05 mg/kg, and on postoperative days 2 and 3 twice subcutaneously at a dose of 0.03 mg/kg. Rabbits that were suspected to be in pain after that (limping, unwilling to walk or to eat, swelling of the stifle or reaction at palpation) received buprenorphine for a further two days. According to the study plan no adjunct non-steroidal anti-inflammatory drug was allowed to be administered due to potential interference with the orthopedic research objectives.
The rabbits were examined clinically daily for three weeks after surgery. Any rabbit that did not eat within 24 h, or lost more than 10% body weight compared with the day of surgery, was hand fed with critical care (Oxbow; Oxbow Animal Health, Murdoch, NE, USA) twice a day, until its body weight started to increase.
Rabbits that showed signs of infection (fever, swelling of stifle, pain on palpation, limping, unwillingness to move), received 5 mg/kg of enrofloxacin (Baytril® vet 25 mg/ mL; Bayer Animal Health, Copenhagen, Denmark) subcutaneously twice daily for up to three weeks.
The rabbits were euthanized with an intravenous overdose of pentobarbitone (Allfatal vet, 100 mg/mL; Omnidea, Stockholm, Sweden) at 12 or 24 weeks after surgery and healing of the cartilage defects were evaluated. The results are published elsewhere. 21
During acclimatization, one rabbit was isolated due to minor fight wounds. Haematological examination revealed moderate heterophilia (5 × 109/L), and antibiotic treatment was initiated with 10 mg/kg ceftiofur (Excenel® vet; Orion Pharma Animal Health) intramuscularly, every 24 h. The rabbit’s clinical condition seemingly improved, but a week later the rabbit was found dead. Necropsy revealed excessive fat depots and severe liver lipidosis. Thus only 19 rabbits were entered for the study.
Also one rabbit in group ISO was found dead on the day after surgery. This rabbit was the first to be anaesthetized and unintentionally received 2–3% of isoflurane for the first 60 min, which was higher than for the subsequent rabbits. When MAP, O2-sat and PaO2 decreased, the isoflurane concentration was reduced and the variables rapidly improved. The remaining anaesthesia period and the recovery were unremarkable and the rabbit was returned to its home pen. Autopsy at the Swedish National Veterinary Institute, Uppsala, revealed circulatory failure in the form of acute haemorrhages in the right brain temple lobe and the lungs and acute lung oedema. Data from this rabbit were excluded from statistical evaluation, leaving nine rabbits in group ISO.
Statistical analyses
Sigma Plot (Systat Software Inc Analysis, Chicago, IL, USA) was used for data analysis. Data are presented as mean ± SD or median (range). Unless indicated, n = 9. Student’s t-test or Mann-Whitney rank sum test was used to compare measures between groups. For a comparison of repeated measures, mean values were calculated for each rabbit during preparation (PREPARATION) and during surgery (SURGERY), respectively for the following parameters: ETiso, sufentanil–midazolam infusion rate (including boluses), heart rate, MAP, serum lactate and total protein, and blood glucose levels. For comparisons within groups, one-way repeated measures analysis of variance (ANOVA) or repeated measures ANOVA on ranks, paired t-test or Kruskal Wallis Wilcoxon matched pairs signed-ranks test were used. A P value < 0.05 was considered significant.
Results
Preoperative haematology and CBC results were within the normal range. 21 The serum levels of amyloid A showed a pronounced increase three days after surgery and had decreased to preoperative levels four weeks after surgery.
Data from medetomidine-premedicated New Zealand White rabbits undergoing surgery during sufentanil–midazolam (SUF–MID) or isoflurane (ISO) anaesthesia.
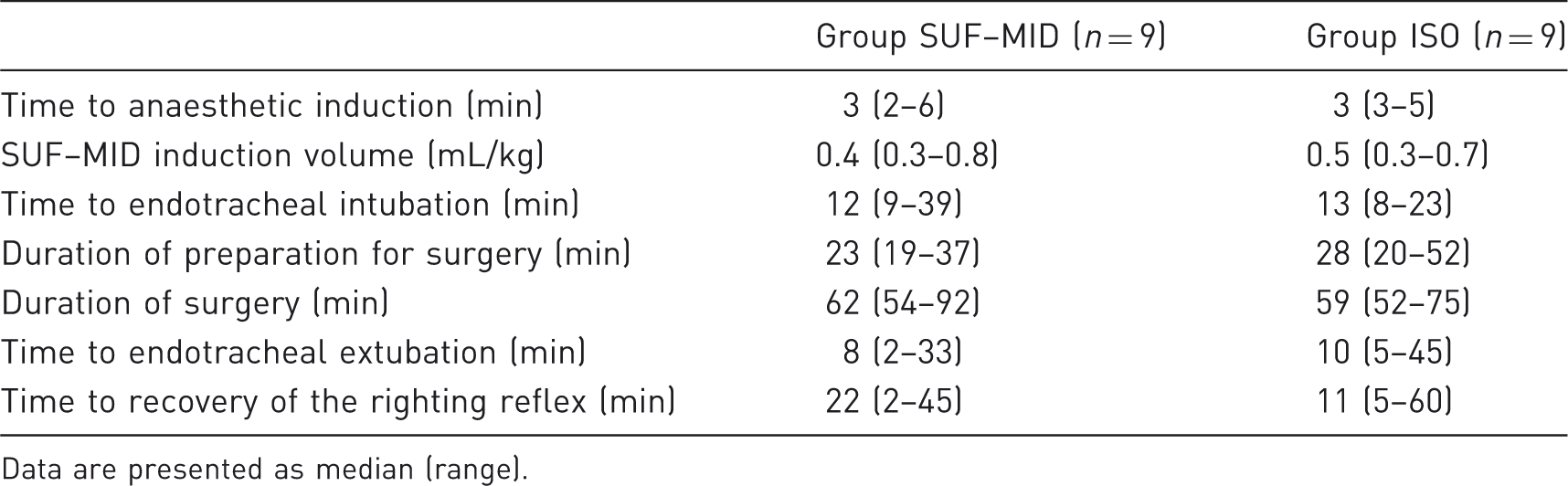
Data are presented as median (range).
In group SUF–MID, six rabbits needed boluses of sufentanil–midazolam (0.34 ± 0.23 mL/kg) because of reactions to surgery at 34 (1–63) min after the start of surgery. Boluses included, the infusion rate did not change during anaesthesia (Figure 1). The end tidal concentration of isoflurane increased from 1.1 ± 0.2 to 1.4 ± 0.3% during surgery (compared with during preparation), due to responses to surgery in five rabbits at 33 (5–47) min after the start of surgery.
Infusion rate of sufentanil–midazolam (SUF–MID, n = 9) and end-tidal concentration of isoflurane (ISO, n = 9) in medetomidine-premedicated New Zealand White rabbits undergoing surgery. ETiso: end-tidal isoflurane concentration.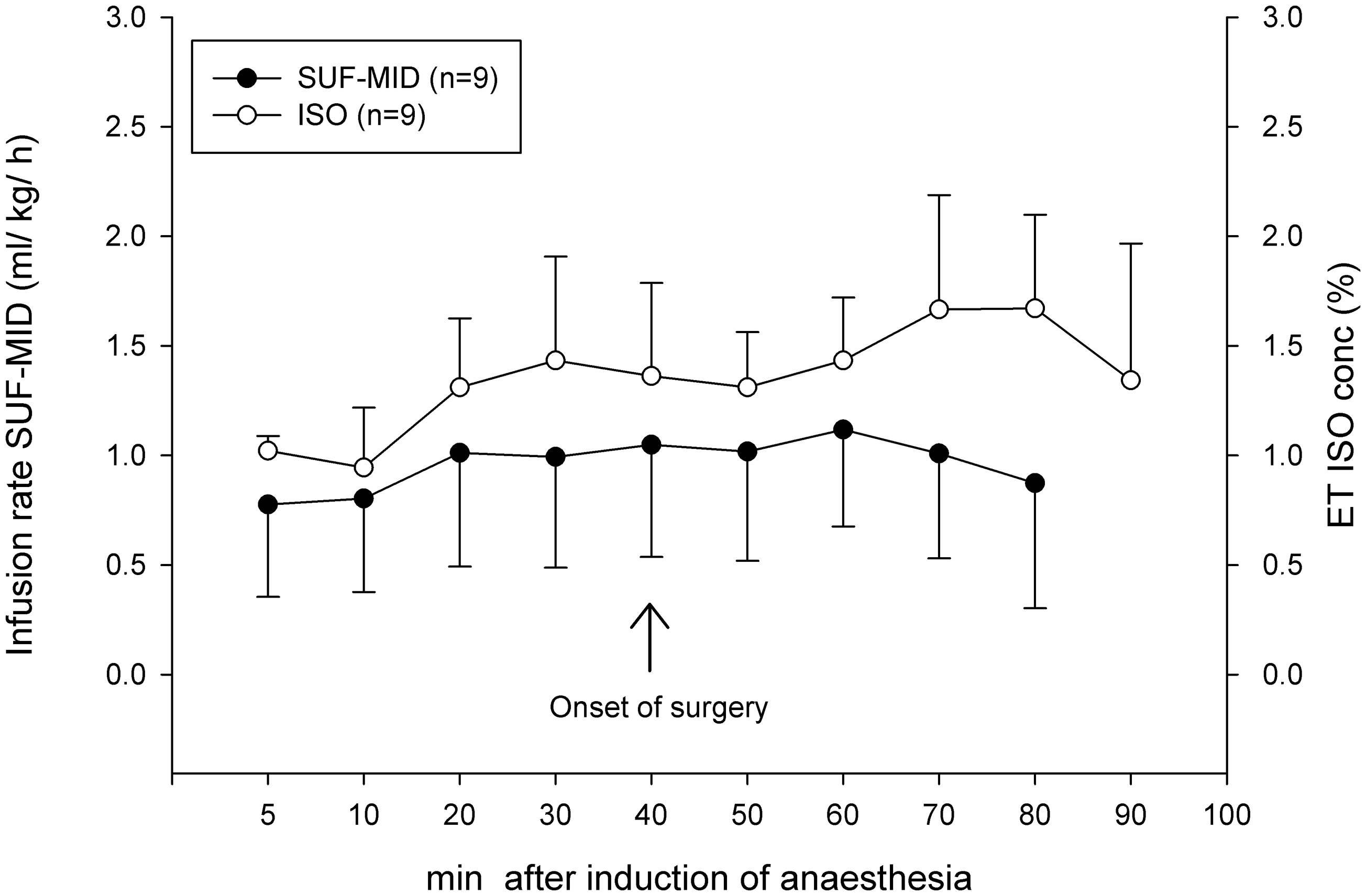
The initial anaesthesia infusion rate of 0.3 mL/kg/h that was used during induction and preparation turned out to be too low, since the first rabbit in group SUF–MID regained consciousness during transfer to the surgery room. The induction infusion rate was therefore increased to 0.6 mL/kg/h in both groups, which was sufficient to keep rabbits in group SUF–MID unconscious during transfer. Because 3/5 rabbits that followed in group SUF–MID needed boluses of sufentanil–midazolam during surgery, the induction infusion rate was increased to 1.2 mL/kg/h in both groups. At this induction rate, two rabbits showed muscle rigidity in the front legs, and the rate was decreased to 0.9 mL/kg/h in the last four rabbits. No muscle rigidity was seen after that. The muscle rigidity occurred during induction and lasted for a few minutes.
IPPV was required in eight rabbits in group SUF–MID and in six rabbits in group ISO at some time point during anaesthesia. Of these, seven in group SUF–MID and three in group ISO needed IPPV during most of the anaesthesia, starting at 5–10 min after induction. The ventilator was used with a respiration frequency of 11–18/min, a peak inspiratory pressure of 16 cmH2O and a positive end expiratory pressure of 1–2 cmH2O.
Comparison of PaO2, PaCO2 and pH in medetomidine-premedicated New Zealand White rabbits undergoing surgery during sufentanil–midazolam (SUF–MID) or isoflurane (ISO) anaesthesia.
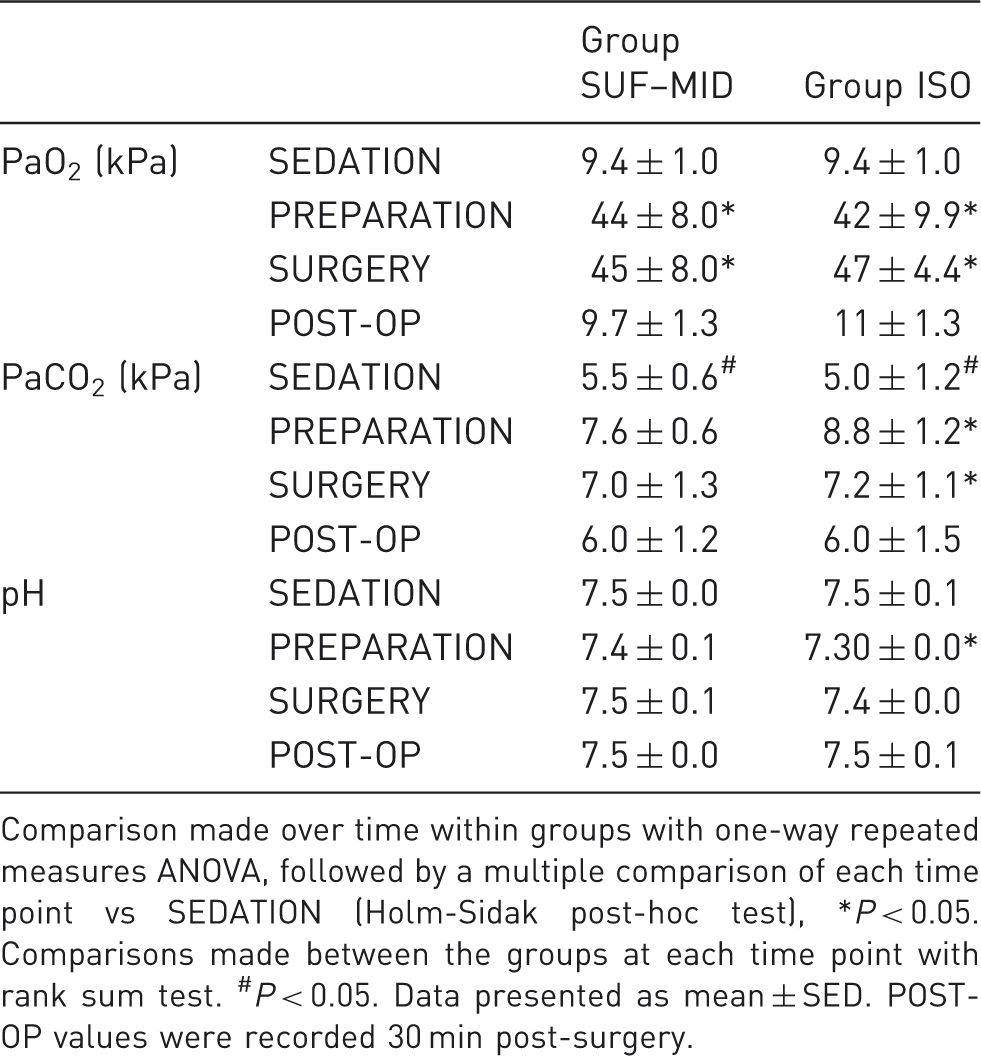
Comparison made over time within groups with one-way repeated measures ANOVA, followed by a multiple comparison of each time point vs SEDATION (Holm-Sidak post-hoc test), *P < 0.05. Comparisons made between the groups at each time point with rank sum test. #P < 0.05. Data presented as mean ± SED. POST-OP values were recorded 30 min post-surgery.
Lactate levels were <4 mmol/L at all time points and did not differ between groups or change over time. Total serum protein levels did not differ between groups at any time point (range 3.4–6.4 mmol/L). Blood glucose increased during surgery in group ISO (Table 3).
Heart rates increased during surgery in group ISO (SEDATION: 158 ± 40, SURGERY: 197 ± 26). There were no differences between the groups. In one rabbit in group SUF–MID 0.2 mg of glycopyrrolate (Robinul; Meda, Solna, Sweden) was administered intravenously when the heart rate decreased to 83 bpm during preparation for surgery. Within 5 min an increase in heart rate to 150 bpm was recorded. MAP (Figure 2) was lower in group ISO than group SUF–MID during surgery (50 ± 8 mm vs 63 ± 12 mmHg). In all but one rabbit in group ISO, MAP was severely low (<40 mmHg) at at least one time point, whereas that only occurred in one rabbit at one time point in group SUF–MID. No abnormalities were seen in the ECG measurements. Body temperature decreased in both groups during surgery (SUF–MID: 0.4 ± 0.2℃, ISO: 0.4 ± 0.3℃). Mean body weights did not differ between the groups before surgery (SUF–MID: 3.8 [3.0–4.4] kg, ISO: 4.0 [3.5–4.2] kg). Body weights were reduced by 5% during the first two postoperative days, a further 2% during postoperative days 2–7, and 2.7% during the second postoperative week. Thus the mean body weight loss was 9.7% (0.38 ± 0.15 kg). There were no differences between the groups. After two weeks the body weights stabilized. Two rabbits (one per group) had to be euthanized 15 and 18 days postoperatively due to infection which did not respond to antibiotic treatment with enrofloxacin. Post-mortem examination revealed sparse growth of Staphylococcus aureus and beta-haemolysing streptococci in the joint capsule with intermediate resistance against enrofloxacin.
Mean arterial pressure (MAP) in medetomidine-premedicated New Zealand White rabbits during surgery with sufentanil–midazolam (SUF–MID) or isoflurane (ISO) anaesthesia.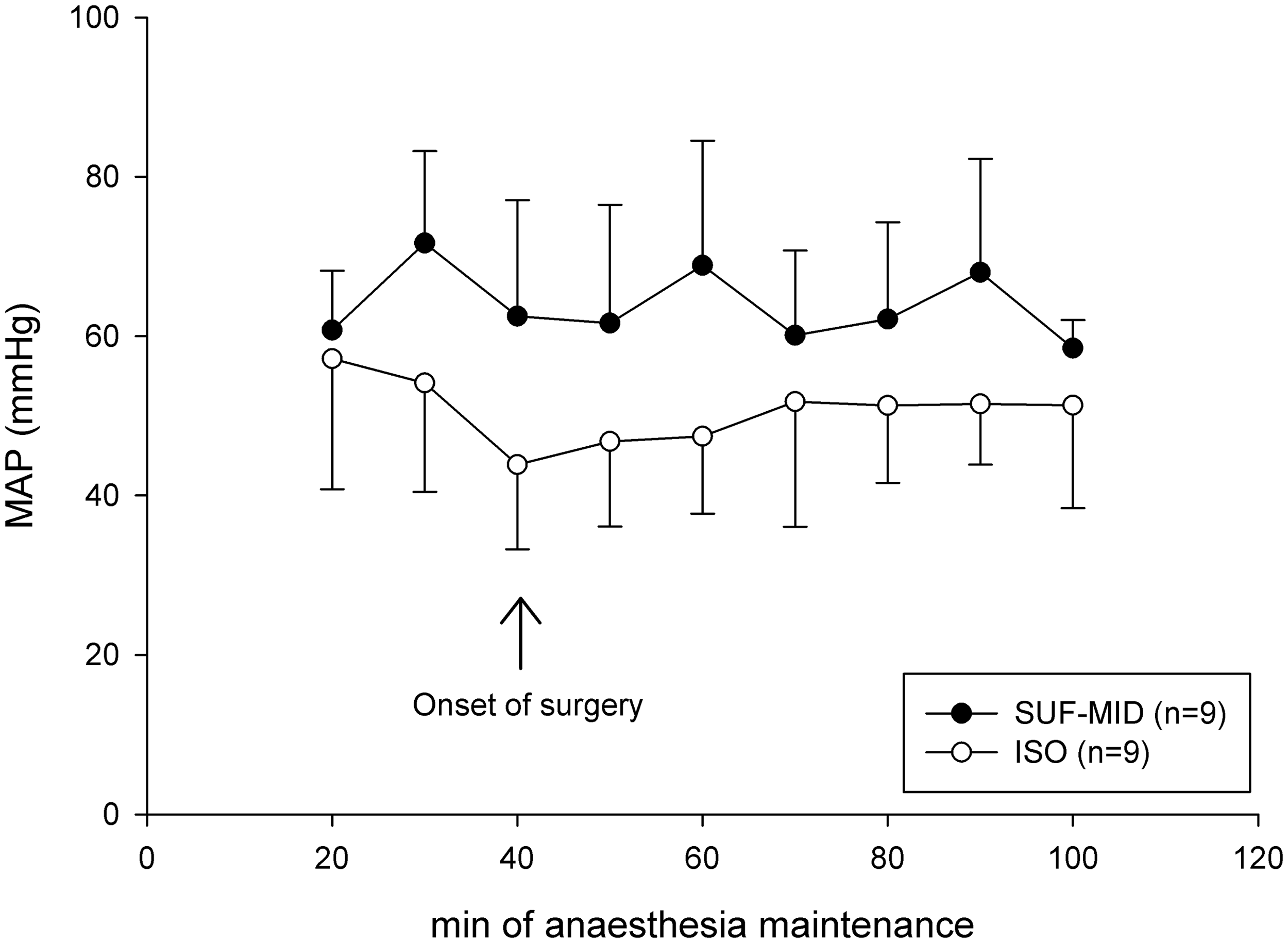
Discussion
In this orthopaedic study in NZW rabbits, continuous infusion with sufentanil–midazolam was as effective as isoflurane in producing surgical anaesthesia in medetomidine-premedicated rabbits, and produced less hypotension. It is important to develop safe intravenous infusion anaesthesia protocols for circumstances in which volatile agents cannot be used. Examples are imaging or extracorporeal studies, or studies using animals with a limited cardiovascular reserve, such as animals with cardiac disease or geriatric animals.
Induction with sufentanil–midazolam was rapid and smooth, up to an infusion rate of 0.9 mL/kg/h. At a higher infusion rate muscle rigidity occurred, which also occurs in pigs during rapid induction with sufentanil. 23 Muscle rigidity was however short lasting and no seizures were produced, in contrast to what was reported from a study on intravenous anaesthesia using a similar combination (midazolam–xylazine–alfentanil) in NZW rabbits. 24
Blood pressure was higher during surgery with sufentanil–midazolam than isoflurane anaesthesia, which rendered the rabbits clinically hypotensive. No intervention was performed since the aim was to evaluate the effects of the anaesthesia protocols on physiological parameters. In the research setting hypotension may well go unnoticed if blood pressure is not monitored during anaesthesia. MAP was considerably higher in group SUF–MID (mean 63 mmHg) than in our previous study in NZW rabbits (mean 46 mmHg), in which no surgery was performed and a much lower infusion rate was administered. 20 This is likely to be due to differences in nociceptive stimulation. Rabbits are commonly hypotensive during anaesthesia (MAP < 60 mmHg), which may contribute to high peri-anaesthetic mortality. Further, during surgery heart rate and blood glucose levels increased only in rabbits under isoflurane anaesthesia. There were no differences in glucose levels between the groups, and the biological significance of the increase in the ISO groups is questionable. It is known, however, that surgery activates the neuroendocrine system which leads to tachycardia and hyperglycaemia intra- and postoperatively. 16 Opioids are known to inhibit the stress response and decrease glucose levels during and after surgery in humans. 25
Comparison of serum glucose levels in medetomidine-premedicated New Zealand White rabbits undergoing surgery during sufentanil–midazolam (SUF–MID) or isoflurane (ISO) anaesthesia.

The number of animals varies between six and nine at different time points. Comparison made over time within groups by one-way repeated measures ANOVA, *P < 0.05, multiple comparison vs SEDATION (Holm-Sidak post-hoc test). Data are presented as mean ± SD. POST-OP values were recorded 30 min post-surgery.
Conclusion
In spite of the limited number of animals in this study, intravenous use of sufentanil–midazolam seems to provide effective anaesthesia in rabbits undergoing surgical procedures, and is especially useful in cases when inhalation anaesthesia is not an option.
Footnotes
Acknowledgements
The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (FORMAS) and the Research Fund for Companion Animals at the Swedish University of Agricultural Sciences funded this project. Part of the results has been presented at the Association of Veterinary Anaesthetists’ Spring meeting in Bari, Italy, April 2011 and in an undergraduate student project at the Swedish University of Agricultural Sciences 2011.
