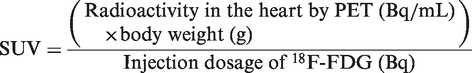Abstract
Positron emission tomography (PET) is useful for evaluating the cardiac metabolism of free fatty acid, glucose and oxygen both in human clinical practice and in experimental animal models. However, no data are available for such an evaluation in a model of stable compensated left ventricular hypertrophy in 14-month-old spontaneously hypertensive rats (SHRs). This study was designed to assess the metabolism of myocardial glucose in SHRs using 2-deoxy-2-[18F]fluoro-D-glucose (18F-FDG) using PET. The study was performed on 14-month-old male SHRs (n = 4) and age-matched Wistar Kyoto (WKY) rats (n = 4). PET scans were performed after the administration of anaesthesia with isoflurane and injection of a bolus of 39.37 ± 3.25 (mean ± SD) MBq (1.06 mCi) of 18F-FDG. The standardized uptake value (SUV) was used to evaluate 18F-FDG uptake by the heart. The analysis of SUV showed increased metabolism in the left ventricle of SHRs compared with WKY rats. Our results show that small animal PET using 18F-FDG can be performed in 14-month-old SHRs to evaluate new therapies in the regression of left ventricular hypertrophy in SHRs because pathological myocardial metabolism in the SHR differs from the normal metabolism of the WKY rat.
Small animal positron emission tomography (PET) makes it possible to investigate myocardial metabolism in experimental animal models in much the same way as in the human heart. Altered fatty acid and carbohydrate metabolism in the myocardium have been associated with cardiovascular disease (chronic ischaemic heart disease, dilated cardiomyopathy and ventricular hypertrophy). 1 Under normal conditions, the heart uses glucose (30%), fatty acids (60%) and lactate (10%) as primary energy sources; however, glucose metabolism is increased in cardiac hypertrophy. 2 Consistent with this observation, several studies have shown a higher rate of glucose uptake in animal models of cardiac hypertrophy.3–5 However, data are not available for spontaneously hypertensive rats (SHRs), which are a model of hypertension-induced left ventricular hypertrophy (LVH), 6 analysed using PET. The aim of our study was to explore myocardial glucose metabolism in 14-month-old male SHRs using 2-deoxy-2-[18F]fluoro-D-glucose (18F-FDG) using PET. We evaluated the potential of 18F-FDG–PET for measuring the response to treatment of LVH in this experimental model.
The animals studied were 14-month-old male SHRs (NHsd, n = 4) and normotensive control Wistar Kyoto (WKY/NHsd, n = 4) rats. The animal lines were developed by Laboratorio Harlan (Castellar del Valles, Barcelona, Spain), and they were bred at the animal house of Universidad Autónoma (Madrid, Spain) (EX021-U). All the rats were supplied with standard rat chow and drinking water ad libitum and were maintained on a 12 h/12 h light/dark cycle. The animals were housed at a constant temperature of 24℃ and relative humidity of 40%. Twenty-four hours before PET, the rats were moved to the Experimental Medicine and Surgery Unit of Hospital Gregorio Marañón (ES280790000087) and housed in individual cages. The study was performed in accordance with European Union guidelines for the protection of animals used for experimental and other scientific purposes (Guideline 2010/63/UE and Spanish Royal Decree 53/2013). Our institutional Animal Care and Ethics Committee approved the protocol.
All images shown in this study were obtained using a small animal PET/computed tomography (CT) scanner (Sedecal®, Madrid, Spain). The animals were deprived of food but allowed water ad libitum for 8 h before the study. Systolic blood pressure (SBP) and heart rate (HR) were monitored by the tail cuff method with a photoelectric sensor (NIPREM 645, Cibertec, Madrid, Spain)
7
using software, every 15 min during the PET study, and levels of blood glucose were measured before and after. The animals were anaesthetized with isoflurane (Forane, Inibsa, Barcelona, Spain) (3% induction and 1.5% for maintenance in 100% oxygen) using an SA1 respirator (Dräger Medical AG, Lübeck, Germany). 18F-FDG (Molipharma, Madrid, Spain) (39.37 ± 3.25 MBq [1.06 ± 0.08 mCi]) was administered via a venous catheter in the tail vein. PET scans were performed for 54 min. After the last scan, the rat was sacrificed using carbon dioxide. AMIDE 0.9.3 for windows was used in the image analysis. Following image processing, the standardized uptake value (SUV) was used to evaluate 18F-FDG uptake by the heart.
4
The SUV was calculated and normalized according to body weight and the injected dose of 18F-FDG
4
:
The SUV for each region of interest namely, heart, dorsal muscle and background, was used for analysis. The background value subtracted from that of the heart and the dorsal muscle, and the SUVheart and SUVdorsal muscle were determined, as was their quotient. Variables were expressed as mean ± SD. The statistical analysis was performed using SPSS 17.0 for Windows (SPSS Inc, Chicago, IL, USA) and S-PLUS 6.1. A t-test for independent samples was used to assess differences between the two groups. A value of P < 0.05 was considered to be statistically significant.
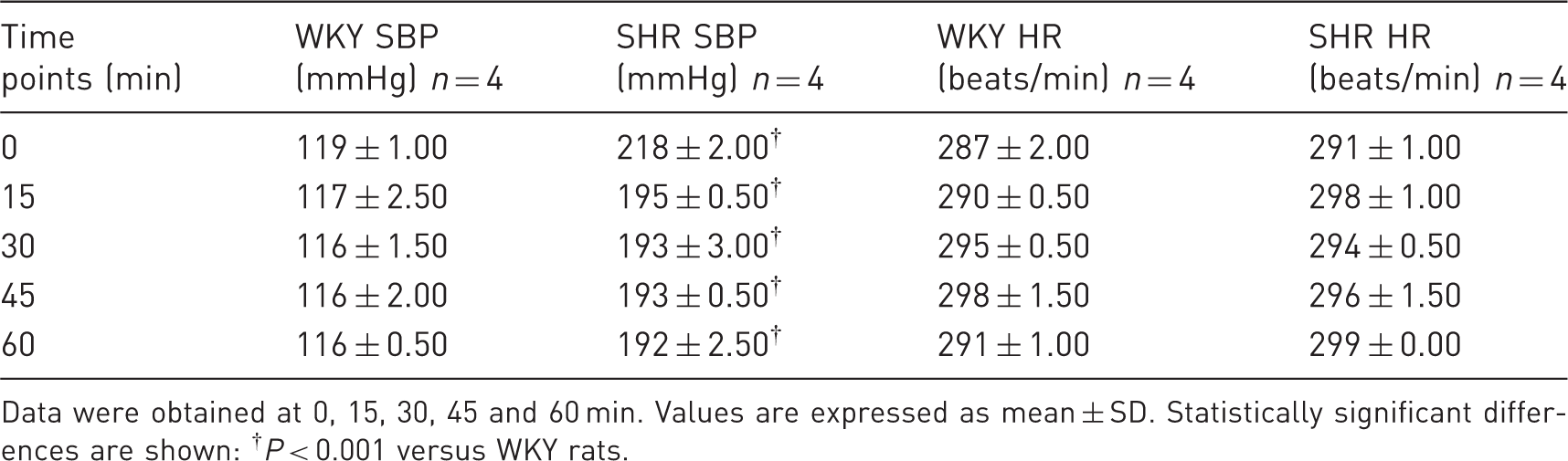
Body weight was higher in WKY rats (WKY rats 450.25 ± 6.70 g versus SHRs 392.25 ± 34.05 g; P = 0.016). The level of blood glucose remained unchanged in SHRs compared with WKY rats before the PET study (WKY rats 83.50 ± 17.40 mg/dL versus SHRs 80.25± 29.89 mg/dL; P = 0.858) and after the PET study (WKY rats 115.66 ± 18.33 mg/dL versus SHRs 89.66 ± 4.61 mg/dL; P = 0.076). Values of SBP and HR are shown in Table 1. SBP was higher in SHRs than in WKY rats, and HR remained unchanged in both SHRs and WKY rats during the PET study (0, 15, 30, 45 and 60 min). The SUVheart and SUVheart/SUVdorsal muscle were significantly higher in SHRs than in WKY rats (P < 0.05 and P < 0.01 respectively) but the SUVdorsal muscle remained unchanged in both SHRs and WKY rats. PET acquisitions in SHRs revealed higher 18F-FDG uptake than in WKY rats and increased regional metabolism in the hearts of SHRs compared with the hearts of the WKY rats (Figure 1).
Examples of positron emission tomography (PET) images of myocardial 18F-FDG uptake from a Wistar Kyoto (WKY) rat and a spontaneously hypertensive rat (SHR). Note that 18F-FDG uptake was lower in WKY rats than in SHRs (a). Standardized uptake value (SUV) in WKY rats (n = 4) and SHRs (n = 4). Values are expressed as mean ± SD. Statistically significant differences are shown: **P < 0.01 versus WKY rats (b). Systolic blood pressure (SBP) and heart rate (HR) of the Wistar Kyoto (WKY) rats and spontaneously hypertensive rats (SHRs) during data acquisition in positron emission tomography (PET). Data were obtained at 0, 15, 30, 45 and 60 min. Values are expressed as mean ± SD. Statistically significant differences are shown: †P < 0.001 versus WKY rats.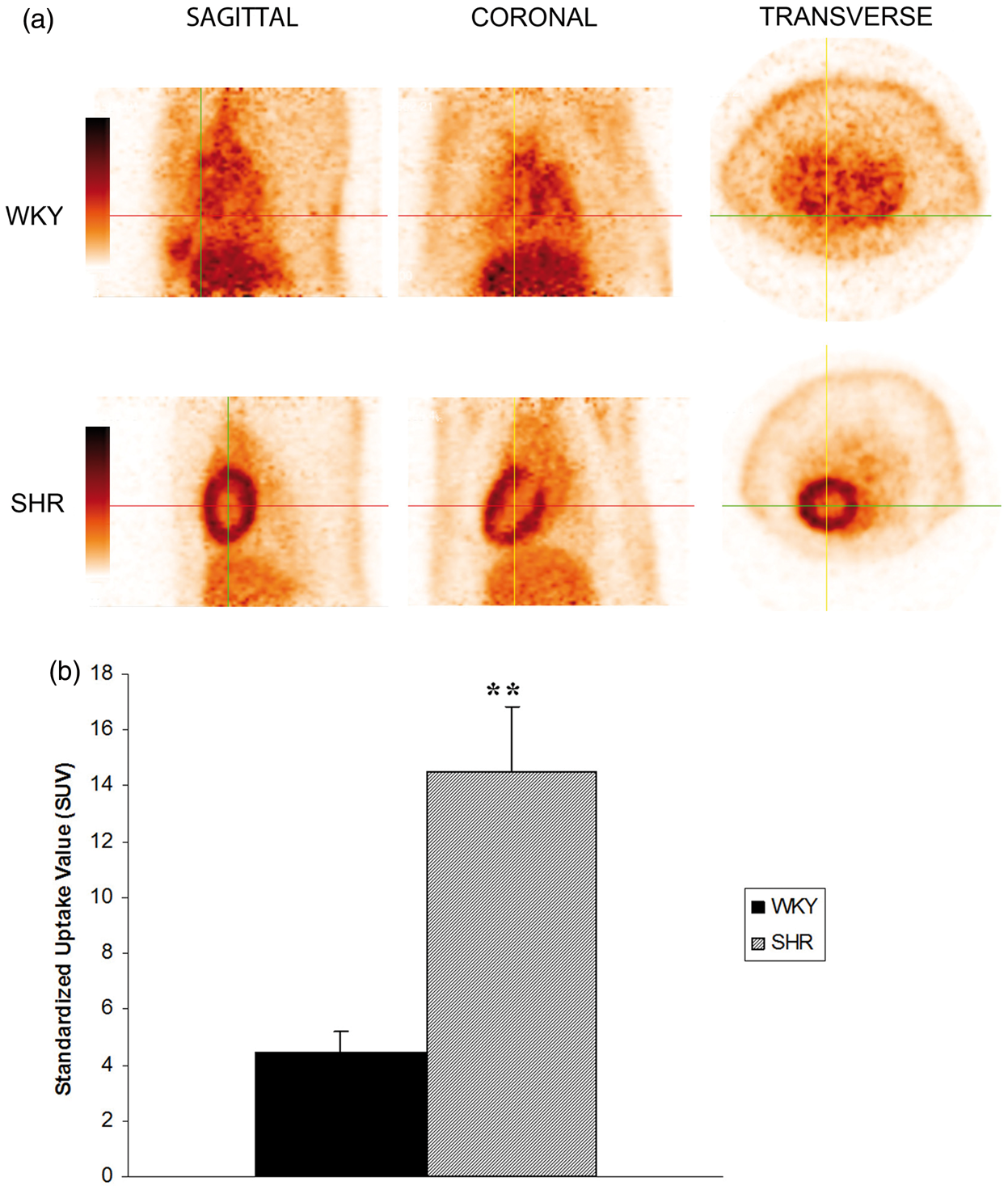
We used two different expressions to evaluate 18F-FDG uptake by the heart: SUVheart (quantitative) and SUVheart/SUVdorsal muscle (semiquantitative). SUVheart/SUVdorsal muscle requires a reference tissue (the tissue with the lowest uptake of the drug), which in our case was the dorsal muscle (i.e. in a PET scan of the brain, the reference tissue would be the cerebellum). In our study, both methods provided similar results (both quantitative and semiquantitative values showed an increase in myocardial metabolism in SHRs).
Our results show that small animal PET with 18F-FDG can be performed in 14-month-old SHRs to evaluate LVH. Previous studies showed a higher rate of glucose uptake in animal models of cardiac hypertrophy (salt-sensitive Dahl rats 4 and Wistar rats with ascending aortic constriction3,5); however, this observation has not been investigated using PET in SHRs. Despite increased glycolysis, one study found decreased glucose oxidation in cardiac hypertrophy in rabbits, 8 with severe hypertrophy before ventricular dilation and heart failure (impaired glucose uptake was detected before progression from compensated to decompensated hypertrophy). SHR progresses to heart failure during the last six months of their lifespan (about 2 years); therefore, we applied this model of human hypertrophy at 14 months of age (when SHRs display stable compensated concentric hypertrophy, diastolic dysfunction and concomitant supernormal systolic function). The results of our study show an increase in myocardial glucose uptake in SHRs at 14 months, possibly because LVH has not yet progressed to heart failure. Some authors suggest that metabolic changes occur in parallel with the degree of cardiac hypertrophy. 9
LVH is an independent clinical factor for heart failure, sudden death, and myocardial infarction. As regression of cardiac hypertrophy is associated with a lower likelihood of cardiovascular events, it is recognized as a therapeutic objective. 10 However, measurement of regression of left ventricular mass may not be sufficient for the evaluation of the effect of treatment, and an improvement in cardiac metabolism is necessary. 11 Although studies have shown regression of LVH in SHRs after treatment, 12 cardiac metabolism has not been analysed by 18F-FDG–PET. The importance and relevance of our findings is that analysis of 18F-FDG uptake by PET could prove useful for evaluating new treatments in the regression of LVH in preclinical studies.
In conclusion, our results show that 18F-FDG–PET can be performed in 14-month-old SHRs to evaluate LVH. Small animal PET could prove useful for evaluating treatment of LVH in SHRs because pathological myocardial metabolism in the SHR differs from the normal metabolism of the WKY rat.
Footnotes
Funding
This work was supported by a grant from FIS PI10/02831.