Abstract
This study was undertaken to refine the techniques of in vivo collection of sperm in the mouse. The principal objective was to offer a viable, safe and reliable method for serial collection of in vivo epididimary sperm through the direct puncture of the epididymis. Six C57Bl/6J males were subjected to the whole experiment. First we obtain a sperm sample of the right epididymis, and perform a vasectomy on the left side. This sample was used in an in vitro fertilization (IVF) experiment while the males were individually housed for 10 days to let them recover from the surgery, and then their fertility was tested with natural matings until we obtained a litter of each one. After that, the animals were subjected another time to the same process (sampling, recover and natural mating). The results of these experiments were a fertilization average value of 56.7%, and that all the males had a litter in the first month after the natural matings. This study documented the feasibility of the epididimary puncture technique to in vivo serial sampling of sperm in the mouse.
In vivo retrieval of mouse sperm has always posed numerous difficulties leading to collection normally being carried out postmortem. Regardless of this, there are cases where it is essential to obtain sperm while keeping the animal alive, as for example when it is wished to make repeated sperm sampling from a single male, or situations where the male is of great value, as occurs with founder animals or males belonging to strains in danger of extinction.
There are currently several approaches described for the above cases:
Electroejaculation, which is commonly used in other animal species has a limited use for mice, as it often results in the animal’s death (up to 22%), either due to the electrical stimulus or to uraemia deriving from urethral obstructions caused by the coagulating part of the sperm. Furthermore, the complexity of the technique requires trained staff to perform it, and it has also been proved that the sperm obtained has poor viability, a low sperm count and low motility.
1
–4
Injecting drugs (most commonly a combination of pernosterone and yohimbine) to cause ejaculation is another option, but it has undesirable side-effects (up to 21% of animals subjected to this method die) and variable success rates. Therefore, it is little used due to low performance.
5
Another possibility is to obtain sperm from the uterus of females used in directed matings. This may be the most commonly used technique nowadays, although it has several drawbacks, such as having to bind the oviducts in females previously, postcoital sacrifice of the females and the need for a very laborious protocol on hormone treatment, mating and observation of the vaginal plug.
6
–9
There is only one option when faced with these techniques, although it cannot really be considered an alternative for making serial collections since it consists in unilateral extirpation of an epididymis from the male in order to obtain sperm. Therefore, the technique is limited to two samplings, resulting in the male becoming infertile and requires surgery to extract the organ, where it is essential to control bleeding.
Following Russell and Burch’s three Rs, we decided to refine the technique to enable in vivo sperm collection through minor surgery that posed the least possible danger to the animal’s life. Thus, we aimed to obtain sperm directly from the epididymis without removing it, and causing minimal trauma so that the technique could be repeated as required, with fertility remaining intact.
Materials and methods
Animals
All procedures for animal experimentation were reviewed and approved by the Biosafety and Bioethics Committee of the Instituto de Neurociencias de Alicante CSIC-UMH.
The mice used in the experiments were C57BL/6J, and were used as sperm donors (12 weeks old), oocyte donors (3–4 weeks old) and females for mating (8–12 weeks old).
The animals were born and housed in the RMG-SPF animal facility in the Instituto de Neurociencias de Alicante CSIC-UMH, under stable, controlled environmental conditions, according to standards contained in national regulations, with a 12-hour light/dark cycle (lights on at 8:00), constant temperature and relative humidity of 22 ± 1℃ and 55 ± 5% and free access to food and water.
Mediums
The media used for the in vitro fertilization (IVF) and to preload the capillaries in preliminary experiments was Research Vitro Fert (K-RVFE-50, Cook Medical). Paraffin oil (NidOil, Nidacon) was used to cover the drops of medium and subsequent incubation.
Preliminary experiments
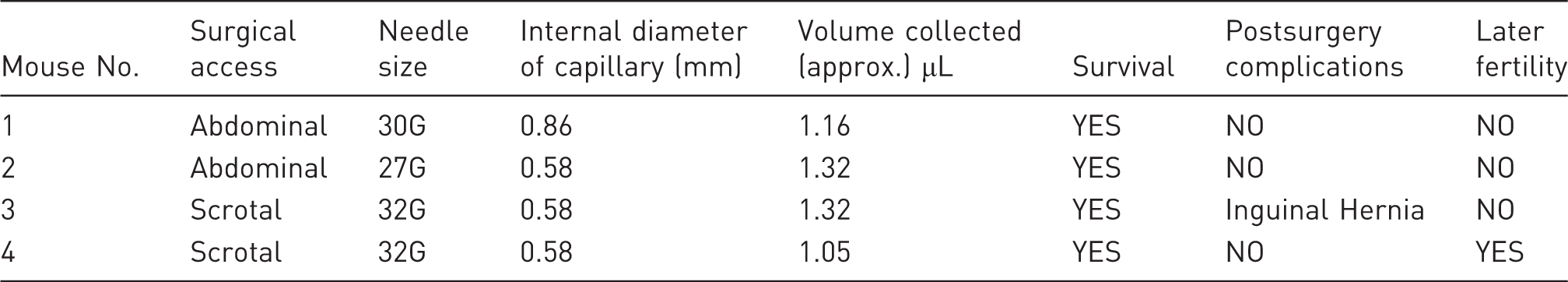
In order to be able to evaluate the viability of our proposal, we performed a pilot experiment on 4 C57BL/6J males. The objective was to test various surgical accesses, needle sizes for puncturing, diameter of the collection capillary, whether there is a need to preload the capillary with preincubated medium for the collection and to remove unnecessary items from the technique to ensure minimal trauma for the male and its survival and fertility. For these animals, routine sperm sampling was performed in the epididymal tail on the right side, and vasectomy on the left side, in order to be able to effectively evaluate the effects from puncturing.
The volume of pure extract of sperm from the epididymis was calculated using the formula V = Πr 2·h, taking into account the radius of the capillary and the height reached by the sample inside it. After surgery, the animals were housed individually for 10 days, after which they were mated with two C57BL/6J females of 8–12 weeks old in the hope of obtaining a litter.
Finally, the use of the sperm samples for IVF was evaluated, by introducing new variables, such as the size of the fertilization drop.
Experiment 1. In vivo serial sampling of epididymal sperm
We designed an experiment with 6 C57BL/6J males, in which we collected sperm from the right side epididymis and vasectomized the left side. The sample collected was used for IVF, while the operated males were allowed to rest for 10 days, after which they were mated naturally with two C57BL/6J females of 8–12 weeks old, until a litter was obtained. The experiment was repeated with the same males to assess the possibility of making serial collections.
The collection technique requires the male used for sampling to be anaesthetised first. Therefore, we weighed the animal in order to calculate the dose of anaesthetic, and prepared a Ketamine/Medetomidine mixture in a sterile vial with 0.38 mL of Ketamine (Imalgene 1000, Merial), 0.25 mL of Medetomidine (Domitor, Pfizer) and 4.37 mL of a sterile physiological saline solution. The mixture was administered to the mouse at 10 µL/gr intraperitoneally.
The animal was placed in an incubator at 37℃ to prevent loss of body temperature, and was injected subcutaneously with Meloxicam (Loxicom, Norbrook), a presurgical analgesic, at 2 µL/g when it started to fall asleep. From then on, all handling of the animal outside the incubator was performed on a heated plate. We started to prepare the animal for presurgery by covering its eyes with an ophthalmic vaseline preparation (Lubrifilm, Cusí), shaving, cleaning and disinfecting the scrotum area with 70% alcohol and then with iodized povidone.
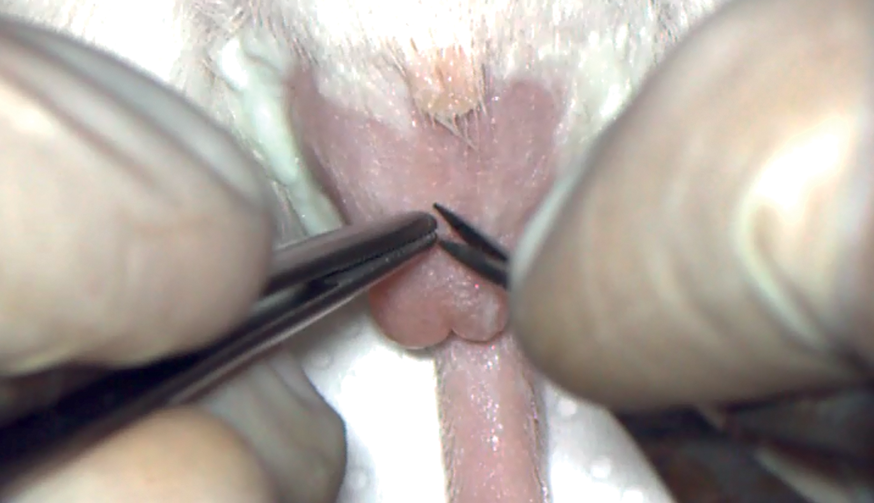
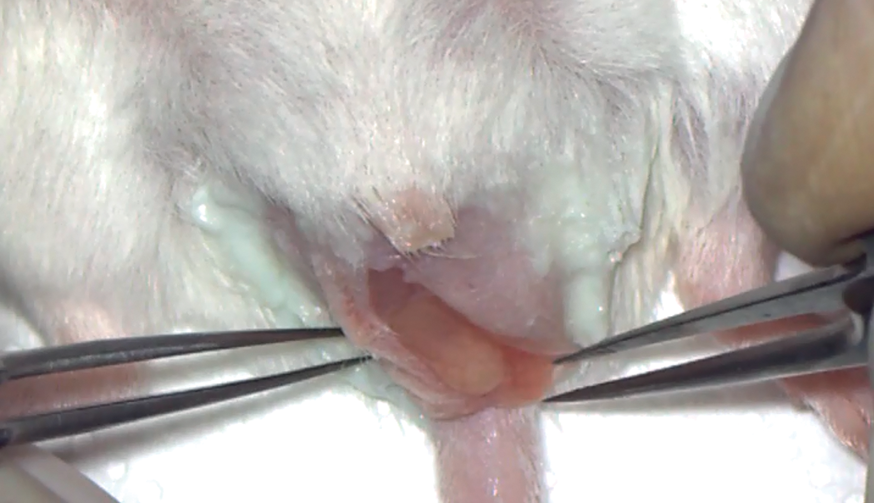
In order to take samples, we had to access and isolate the tail of the epididymis, for which we had to make scrotal access to the testis, which is used routinely in vasectomies in mice.
The reproductive organs were always handled with meticulous care in order not to damage them, and once the testis was located, it was taken outside carefully, avoiding pinching the epididymis, the ductus deferens or any other structure that could be damaged. We found very useful to locate a fatty region that we could clamp and which was located between the head of the testis and tail of the epididymis.
Once the fatty region had been located, it was pulled out to reveal the tail of the epididymis, which we tried to isolate from the adjoining tissues to prevent other bodily fluids from contaminating the sperm sample taken with the capillary, and thus facilitate subsequent puncturing.
Following this, we had to position the tail of epididymis so that, when making the puncture, the sample could rest on the organ without sliding off, which meant that it had to lie completely parallel to the flat surface on which the mouse was lying.
When the correct position was accomplished, the adjoining fatty region was clamped in place with a bulldog serrefine (Fine Science Tools).
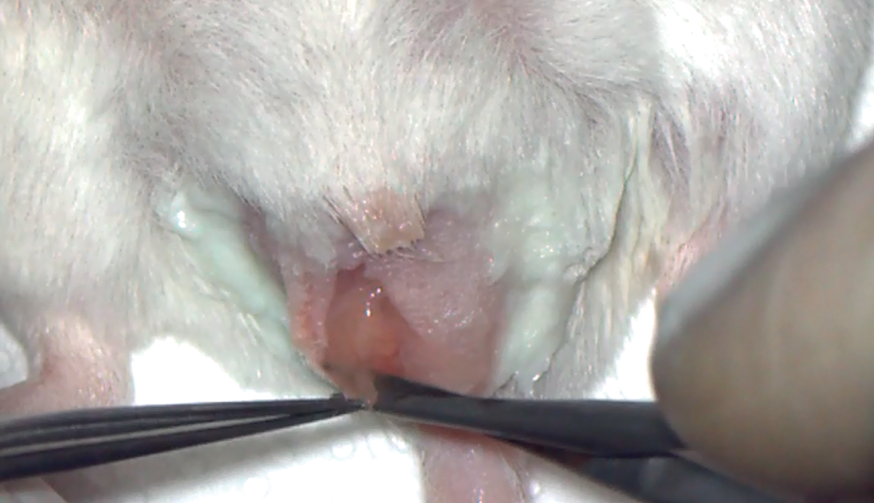
We next prepared an oral pipette with a 0.58 mm internal diameter capillary to collect the sample, and placed it in the mouth, and a 32G hypodermic needle ready to make the puncture.
The mouse was placed under a stereo microscope so we could see the tail of the epididymis in order to find the best spot to puncture, which had to comply with the following characteristics:
To be apart from the surface veins of the epididymis; To be located in the flattest part possible of the epididymis so that the sperm would not spill.
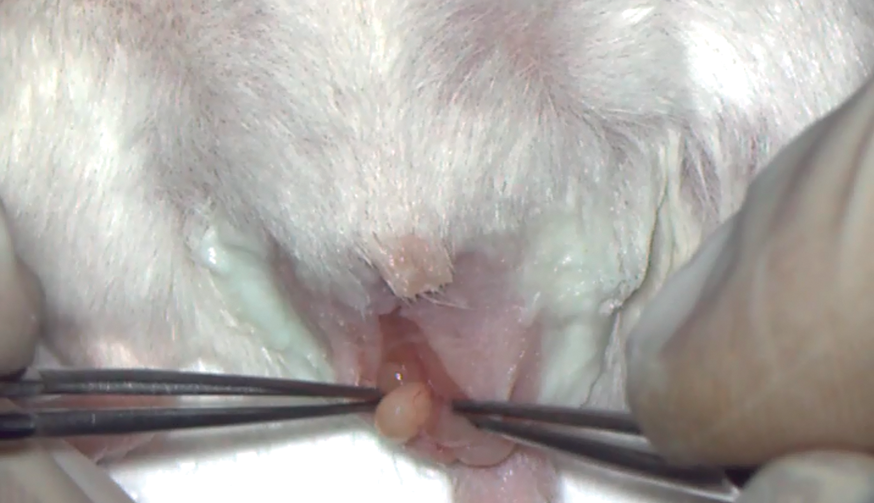
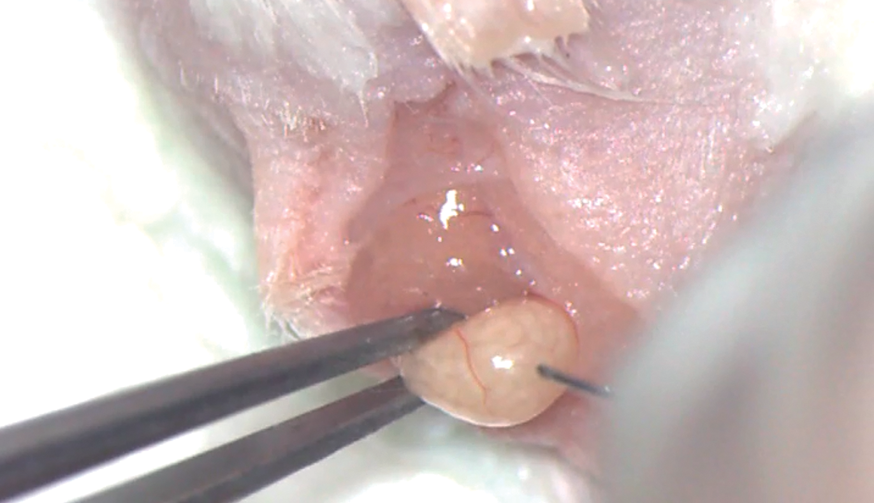
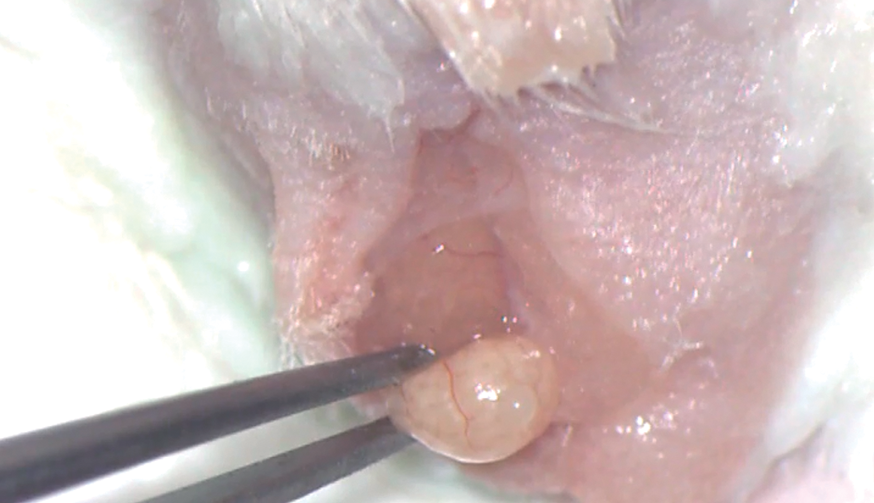
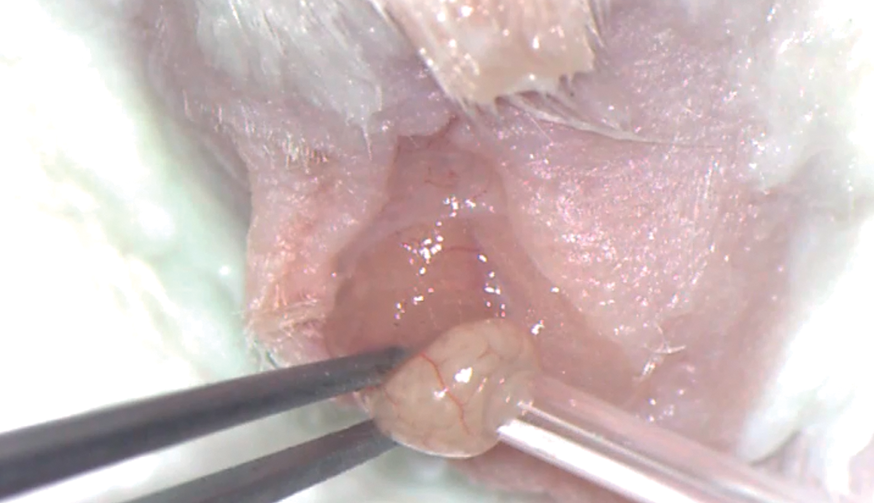
Once everything was ready, we very carefully clamped the tail of the epididymis with 2.2 mm internal diameter ring forceps (Fine Science Tools), and with the other hand used the hypodermic needle to make the puncture at the selected point. We rapidly removed the needle, and immediately afterwards a drop of epididymal sperm arose due to the pressure to which it was subjected inside. We waited for a few seconds while the drop formed, and next put the prepared capillary in contact applying slight suction and avoiding contact with any other liquid. This sample could be used directly.
Finally, we moistened the tail of the epididymis with a sterile physiological saline solution before replacing it in the body, sutured the incisions in layers, and cancelled the sedative by subcutaneous injection of 2 µL/g of Atipamezole (Antisedan). The animal was replaced in the incubator until it woke up and moved about actively. It was then put in an individual cage with food and water and provided with a buprenorphine gelatine dish.
Experiment 2. Using the sample in IVF
To perform IVF, 2 C57BL/6J females were superovulated for each sampled male with 5 IU of PSMG (Folligon, Intevet), followed by 5 IU of hCG (Coriogan, Lab, Ovejero) 48 hours later. The day before the IVF, a fertilization dish was prepared for each male to be sampled, with 4 × 150 µL drops of VitroFert covered with NidOil paraffin oil and incubated overnight.
About 10 minutes before the 13 hours post-HCG had elapsed, we had to obtain the sperm sample from the place where the epididymal puncture was made. As soon as the sample was collected in the capillary, we removed the fertilization dish from the incubator and placed it under the stereo microscope. We introduced the capillary into the fertilization drop through the oil layer, and once inside, we applied slight suction until the fertilization medium penetrated the pipette a little and displaced the sperm sample. Next, we emptied the content into the fertilization drop, taking great care that no air escaped too. We put the dish at 37℃ in the incubator for 10 minutes to disaggregate the sperm mass, and after that, we added the oocytes and continued the IVF experiment as normal. To this end, at approximately 13 hours post-HCG, the females were killed by cervical dislocation and their oviducts quickly dissected for transfer to the oily stage of the fertilization dish, where the cumulus-oocyte complexes were removed from the ampulla with watchmaker’s forceps (Fine Science Tools) and placed in the fertilization drop. The whole procedure should be completed before 13.5 hours post-HCG have elapsed. After 4–5 hours of co-incubation of the gametes, the oocytes were washed with two drops of VitroFert and then placed into the last clean drop where they were left to incubate overnight.
Statistical analysis
In order to be able to compare the values obtained from IVF in the first and second samplings, a two-sample Student’s t-test was used. The differences were marked as significant when P < 0.05. The fertilization percentage was set at the number of 2-cell embryos compared with total oocytes after gametes were co-incubated for 24 hours.
Results
Preliminary experiments
Experimental design for the preliminary experiments, and results.
Using the sperm sample in IVF
The six males that were subjected to the full procedure of experiments, with two sperm samples taken from each one, survived and were fertile following the surgery, with litters obtained from all in the first month of mating.
Results of the in vitro fertilization technique in the preliminary experiments.

Results of the use of the sperm sample collected in experimented 1 in the in vitro fertilization performed in experiment 2.
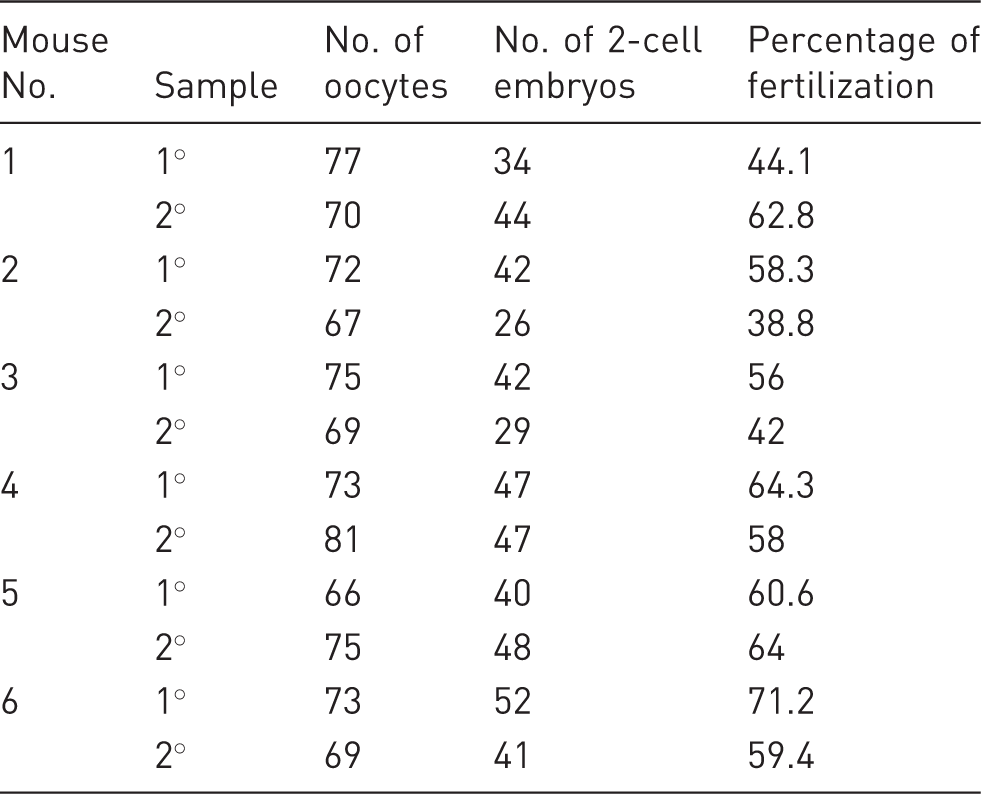
Furthermore, the experiments prove that there is no significant difference between fertilization percentages obtained after the use of the first (IVF1) and second (IVF2) epididymal sperm samples in IVF (Figure 1; P < 0.05).
Percentage of fertilization with the sperm samples from the experiments 1 and 2.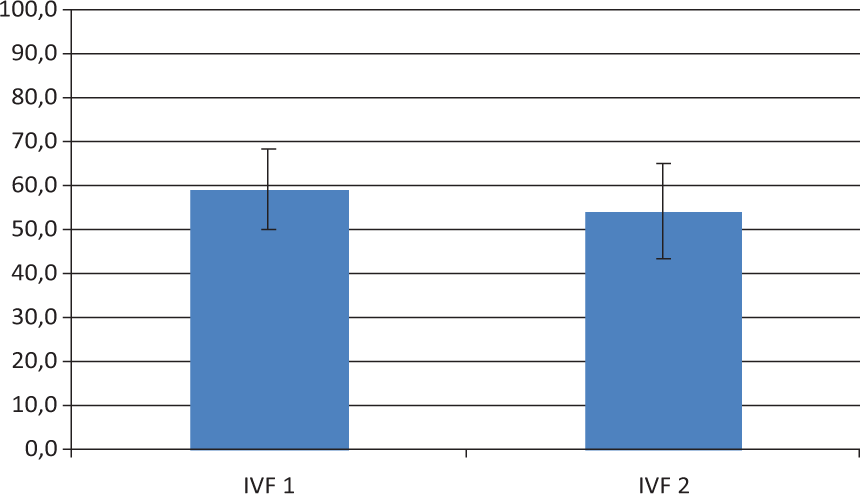
Discussion and conclusions
The results from this study show that it is possible to take in vivo serial samples of epididymal sperm from mice with a new and more advantageous technique. Current techniques for in vivo sperm collection from mice present several problems:
Group 1: Poor reliability and moderate mortality (Electroejaculation and drug-induced ejaculation); Group 2: Large number of animals required, meticulous programming and laborious works (Obtaining sperm by directed mating); Group 3: The ejaculated sperm is of lower quality than epididymal sperm, and also cannot be used directly, as the seminal plasma and urine must be removed first. (Electroejaculation, drug-induced ejaculation and sperm obtained by directed mating).
Regarding the first group, our technique solves both problems, since it is safe and reliable. Thus, in the 16 experiments performed, no animal died. There was only one case of postoperational complications (inguinal hernia), which we attributed to the fact that the surgery technique was not fully established at the time, and the same problem did not occur again in any of the 13 subsequent scrotum operations. Furthermore, the technique is reliable, as a sperm sample was obtained in all cases, which was successfully used for IVF, and the males were fertile after the surgery.
With regard to the second group, the technique also solves all the problems faced, as it reduces the number of animals used, because females are not required for sperm extraction and the animals can be their own control in serial sperm collections. Also, it is a relatively simple, fast and cheap technique that does not require previous programming or laborious work. Neither does it require complicated machinery or highly specialized staff, as occurs with electroejaculation.
As for the third group, various articles have stated a loss of sperm quality in ejaculation retrieval methods compared with epididymal sperm. Thus, lower viability, motility, percentage of fertilization and fewer live births, 3,8 –10 more susceptibility to sperm DNA damage 11 and the possibility of the sample containing urine 4 have been observed. However our technique has been proven useful in obtaining in vivo epididymal sperm which can be used directly with acceptable results, as the fertilization rate obtained in the IVF experiments does not greatly differ from that published in other studies for the same background. 12
In conclusion, we can state that in vivo serial sampling of epididymal sperm in mice using the technique described in this article is viable. It is safe, reliable and useful, as 100% of the males survived, sufficient sperm was retrieved and embryos were obtained from IVF.
There are multiple possible uses for this technique, such as repeated sperm sampling from the same male for studies on mutagenesis, reproduction, pharmacology, pharmacokinetics and toxicology, using the sample in intracytoplasmic sperm injection experiments
11,13,14
or in IVF for rederivation
15
or in the study of transmission to germ-line chimaeras.
16
Oral device for collection. Scrotal access to the testis. Testicular package exteriorization. Opening of the testicular package Figure. Exteriorization of the epididymis tail. Epididymis puncture. Sperm drop after the puncture. Retrieving of the sperm drop. Releasing the sperm sample in the fertilization drop.

Footnotes
Acknowledgements
The authors would like to thank our colleagues of the Servicio de Experimentación Animal-UMH for their work and support, especially those who works in the Instituto de Neurociencias CSIC-UMH Animal Facility, Alejandro Botella, Antonio Caler, Ma Carmen Checa, Sandra González, Verónica Jiménez, Antón Núñez, Ana Lorena Marín, Ma Jesús Molina, Rebeca Ortiz, Raúl Pardo, Sonia Segura, Abigail Segura, Ma Ángeles Soler, and Lucia Yuste. We also wanted to thank Dr Juan Galcerán and Dra Eva Rodriguez for their help with the statistical analysis. Finally we are grateful to Dra Belén Pintado and Dr Juan de Dios Hourcade for their constant orientation, and help.
