Abstract
Articular cartilage has a limited capacity for self-repair in adult humans, and methods used to stimulate regeneration often result in re-growth of fibrous cartilage, which has lower durability. No current treatment option can provide complete repair. The possibility of growth factor delivery into the joint for cartilage regeneration after injury would be an attractive treatment option. A full thickness osteochondral defect of 4 mm in diameter and 2 mm deep was created by mechanical drilling in the medial femoral condyle in 20 female adult New Zealand White rabbits. In an attempt to improve regeneration a hyaluronic hydrogel system, with or without bone morphogenetic protein-2 (BMP-2) was delivered intraarticularly. The contralateral joint defect was treated with saline as control. Throughout the study, rabbits were clinically examined and after 12 (n = 6) or 24 (n = 9) weeks, the rabbits were euthanized and the joints evaluated by histology. The defects healed with fibrocartilage like tissue, and the filling of the defects ranged from less than 25% to complete. The healing of the defects varied both inter- and intra-group wise. Treatment with hyaluronan gel with or without BMP-2 had no effect on cartilage regeneration compared with controls. Instead, severe ectopic bone formation was found in seven joints treated with BMP-2. In conclusion, the present study shows that neither treatment with hyaluronic gel alone, nor in combination with BMP-2, improves the healing of an induced cartilage defect in rabbits. It further shows that BMP-2 can induce ectopic bone formation, which severely affects the functionality of the joint.
The repair of cartilage is a clinical challenge as regeneration in human adults is limited. Treatment options include microfracture technique, 1 autologous cell transplantation 2 and osteochondral mosaicplasty, 3 but many patients still require prosthetic joint replacement. 4 Although progress has been made during recent years, present methods include surgery or complicated cell therapies and cannot provide complete regeneration. The healing often results in formation of fibrocartilage, which contains less proteoglycans and more type I collagen, having inferior properties in terms of morphology, biochemistry and biomechanics compared with hyaline cartilage. 5 The fibrocartilage formation could be due to failure of chondrogenic differentiation of the recruited stem cells, and may be improved by appropriate stimuli of the endogenous cells. For instance, application of growth factors to the damaged site may improve healing. Bone morphogenetic protein-2 (BMP-2) has been shown to promote chondrogenic differentiation in vitro and in vivo.6–9 However, the effect of BMP-2 is highly limited due to its short half-life. To increase the effect, BMP-2 may be presented in a delivery system allowing protection and a slow release into the tissue. Commonly used are formulations based on natural or synthetic polymers. Hyaluronan is a polysaccharide known to induce chondrogenic differentiation of mesenchymal stem cells 10 which makes it an attractive material to use for intraarticular delivery.
Induced full thickness defects in the medial femur condyle or trochlear groove of rabbits are commonly used for screening cartilage repair techniques. Rabbits represent a species that is practical for early evaluation of biomaterials and therapies due to its relative cost-effectiveness, ease of handling and a reasonable joint size for surgical procedures. Rodent models are of limited use for cartilage repair studies due to their small size, lifelong open growth plates and a high potential for intrinsic repair. 11 Large animal models (dogs and small ruminants) are more costly and of a higher ethical concern, and therefore mainly used in late preclinical development. 12
A two-component hydrogel system based on hyaluronan has successfully been used for delivery of BMP-2 in studies on bone regeneration.13,14 Further, we have demonstrated that chondrocytes and mesenchymal cells cultured in the hyaluronan hydrogel form cartilage-like tissue that is rich in glucosaminoglycans, collagen type II and aggrecan. 15 In a recent rabbit pilot study, the injection of the hydrogel, with or without addition of BMP-2, improved the healing of a full-thickness cartilage defect in the stifle joint. 15 In the present study, this finding was further examined in a surgically-induced, full thickness osteochondral defect, in the femorotibial joints of 20 New Zealand White rabbits. The intra-articular injection of the hyaluronan hydrogel with and without BMP-2 was evaluated by clinical examination throughout the study. After 12 weeks (n = 6) or 24 weeks (n = 9) following surgery, the animals were sacrificed and the stifle joints, including the osteochondral defects, were evaluated macroscopically and by histology.
Materials and Methods
Animals
Twenty New Zealand White rabbits (6–7-month-old females, average weight 3.9 kg; range 3.5–4.4 kg) were purchased from a specific pathogen-free colony (Lidköpings Kaninfarm, Sweden). The colony was examined for the following agents according to FELASA recommendations: 16 rabbit haemorrhagic disease virus, rabbit rota- and coronavirus, Bordetella bronchiseptica, Clostridium piliforme, dermatophytae, Pasteruella multocida, other Pasteruellaceae, Salmonella spp, ectoparasites, endoparasites and Encephalitozoon cuniculi.
Rabbits were housed two or three together in floor pens with an area of 1.4–2.0 m2 per rabbit, enabling free movement as well as social contact. The animals were fed a standard pelleted rabbit diet (Lactamin K3, Lantmännen, Stockholm, Sweden) and autoclaved hay, and had access to water ad libitum. The light–dark cycle was 12:12 h with lights on at 06:00. Room temperature was 20 ± 2°C and humidity 55 ± 10%. The experiments were approved by the ethical committee for animal experiments in Uppsala (C158/10).
The animals were acclimatized for three weeks, and weighed and handled daily for four days preoperatively. At the time of surgery, the mean age of the rabbits was seven months (range 6.5–8.3; average weight 3.9 kg).
The rabbits were clinically examined and blood samples taken from the ear artery after treatment with a local anaesthetic cream (EMLA®, AstraZeneca, Södertälje, Sweden) 24 h before surgery. Haematology and complete blood cell counts were performed with ADVIA®Automated Solutions (Siemens Healthcare Diagnostics, Tarrytown, NY, USA), analysis of serum amyloid A (SAA) with an ELISA kit (Tridelta Development Ltd, Maynooth, Ireland), serum lactate with a GM7 analyser (Analox Instruments Ltd, Middlesbrough, UK) and total serum protein with a refractometer. One animal was isolated in a pen and treated with the antibiotic ceftiofur (Excenel® vet, Orion Pharma Animal Health, Sollentuna, Sweden) 10 mg/kg BW, once a day intramuscularly, due to inappetence, small fight wounds and moderate neutrophilia (5 × 109/L). After seven days of treatment it was found dead and necropsy revealed severe liver lipidosis and excessive fat deposits.
One rabbit died during the night after surgery (necropsy showed circulatory failure), two had to be killed due to postoperative joint infection (growth of Staphylococcus aureus) and one due to a sudden onset of non-weight-bearing lameness (caused by patellar luxation). Hence, the final number of animals in the study was 15 (group hyaluronan hydrogel; n = 8, group hyaluronan hydrogel + BMP-2; n = 7).
Preparation of gel components
The hyaluronan hydrogel is a two-component system based on aldehyde-modified hyaluronan (HAA) and hydrazidemodified polyvinyl alcohol (PVAH), which is rapidly crosslinked forming a gel in situ upon mixing. The HAA and PVAH components were synthesized and prepared as described previously.17,18 Gel formation was achieved before injection by extensive premixing of the two reactive components and the gel subsequently injected into the joint space. For the BMP-2 group, BMP-2 was dissolved in the PVAH component and encapsulated in the gel during cross-linking.
Experimental design and surgical procedure
In all rabbits, a defect was created on the medial femoral condyle in both stifle joints. In a randomized fashion, one defect was treated with either (1) the hyaluronan hydrogel alone or (2) the hyaluronan hydrogel containing BMP-2. The contralateral defect was treated with saline to serve as a control. The rabbits were killed 12 weeks (n = 6; 3 from each treatment group) or 24 weeks (n = 9; 5 treated with hyaluronan hydrogel and 4 treated with hyaluronan hydrogel containing BMP-2) postoperatively.
Preoperatively, the rabbits were sedated with 0.1 mg/kg BW medetomidin subcutaneously (Domitor® vet, Orion Pharma Animal Health) and administered intramuscularly with the antibiotic ceftiofur (Excenel® vet, Orion Pharma Animal Health) 10 mg/kg BW. The rabbits’ ears were pretreated with a local anaesthetic cream (EMLA®, AstraZeneca) to enable insertion of intravenous and intra-arterial catheters without pain. General anaesthesia was induced with a mixture of sufentanil 2.3 μg/mL (Sufenta, Jansen-Cilag, Sollentuna, Sweden) and midazolam 0.42 mg/mL (Midazolam, Actavis AB, Stockholm, Sweden) through the ear vein catheter. The trachea of the rabbits was intubated and anaesthesia maintained by administration of 1.5–3% isoflurane in a mixture of 40% oxygen and 60% air at a flow rate of 1.5 L/min (n = 10), or via intravenous infusion of 4.1 μg/kg/h sufentanil and 0.8 mg/kg/h midazolam (n = 9). A comparison of the anaesthesia protocols is reported elsewhere (manuscript in preparation). Blood pressure, heart rate, breathing rate, body temperature, end tidal oxygen and carbon dioxide, and oxygen saturation were monitored during anaesthesia but are not reported here. Warm replacement fluids were administered intravenously during surgery (10 mL/kg/h, Ringer-Acetate®, Fresenius Kabi, Uppsala, Sweden). The rabbits were kept on a heating pad during anaesthesia. Blood was sampled before and at predetermined time points during and after anaesthesia for analyses of arterial blood gases, pH, blood glucose, serum lactate, total protein, SAA and drug concentrations. Arthrotomy of the femorotibial joint was performed under aseptic conditions by a medial longitudinal parapatellar incision. The patella was dislocated laterally to expose the surface of the femoral condyles. A defect of 4 mm in diameter and 2 mm deep was created centrally in the medial condyle by careful drilling down to the subchondral bone using a Midas Legend mechanical drill (Medtronics, Kista, Sweden). After assuring access to stem cells from the subchondral bone by visible bleeding, the patella was relocated and the joint capsule tightly sutured with resorbable 4-0 Vicryl (Ethicon products, San Angelo, TX, USA). A volume of 0.4 mL of the hyaluronan hydrogel alone (n = 10) or hyaluronan hydrogel containing 150 μg/mL BMP-2 (n = 9) was injected into the joint, before the skin wound was closed. The same procedure was performed in the control joint, only 0.4 mL of saline was injected instead. Postoperative pain relief was provided by administering 0.03–0.05 mg/kg BW buprenorphine (Temgesic®, 0.3 mg/mL, Schering-Plough AB, Sweden) subcutaneously twice a day for 3–5 days. As soon as surgery was completed, the first dose was administered; 0.03 mg/kg intravenously (for immediate pain relief) and 0.02 mg/kg subcutaneously.
The first three weeks postoperatively the rabbits were examined daily. The stifles were examined for signs of infection and the grade of lameness was estimated on a scale from 0 to 3; grade 0 denoting no detectable lameness, grade 1 minor lameness, grade 2 major lameness and grade 3 non-weight-bearing lameness. Once body weight had stabilized and no lameness was seen, the rabbits were examined and weighed once a week. The rabbits were killed 12 weeks (n = 6) or 24 weeks (n = 9) postoperatively with an intravenous overdose of pentobarbital (Apoteket Produktion & Laboratorier AB, Malmö, Sweden), and necropsies, including histopathology, were performed.
Postmortem macroscopic and microscopic examinations
The stifle joint was opened, photographed and macroscopically examined. The entire joint with the distal femur, patella and proximal tibia was immersed in 10% neutralbuffered formalin. The bone tissue was decalcified in 3.4% (w/v) sodium formiate and 15.1% (w/v) formic acid. A sample from the synovial membrane, dorsodistal of the patella and slabs from the medial and lateral distal condyles of the femur were embedded in paraffin, cut into 4 μm (synovial membrane) and 6 μm (femoral condyle) sections, and stained with haematoxylin and eosin (H&E), The bone sections were also stained with toluidine blue and safranin-O-fast green. The sections were evaluated blindly, and semiquantitatively analysed according to a modified histological grading scale, 19 previously used in the pilot study. 15 The scoring values ranged from 0 (normal hyaline cartilage) to 31 points (no repair tissue); for details see results. Sections were only evaluated in the centre of the defect, and the articular cartilage stains (toluidine blue and safranin-O) were compared with the contralateral control.
Statistical procedures
Results are presented as mean (SD). Mean body weights were compared between the day of surgery and day 14 after surgery with a paired t-test. A P value <0.05 was considered significant. Lameness scores were compared between the treated and control group as well as between treatments. For comparisons, sums of scores and median scores were calculated for each animal for days 1–21 after surgery and comparisons were made with a Mann-Whitney rank sum test.
Results
Clinical findings
The body weight of the animals decreased by 0.38 (0.15) kg during the first two weeks postoperatively, after which it stabilized for the remaining time of the experiment. Levels of the acute phase protein SAA before surgery were low or below the detection limit (<4.7 ng/mL) in all animals. Three days postoperatively the values showed a pronounced increase (87 [49] mg/mL), and four weeks postoperatively the levels had decreased to preoperative values (Figure 1).
Serum SAA concentrations. Serum SAA concentrations (ng/mL) before surgery, three days and four weeks postoperatively. Three days postoperatively values are depicted differently according to treatment [hyaluronan hydrogel alone (▼) or hyaluronan hydrogel with BMP-2 (⧩)]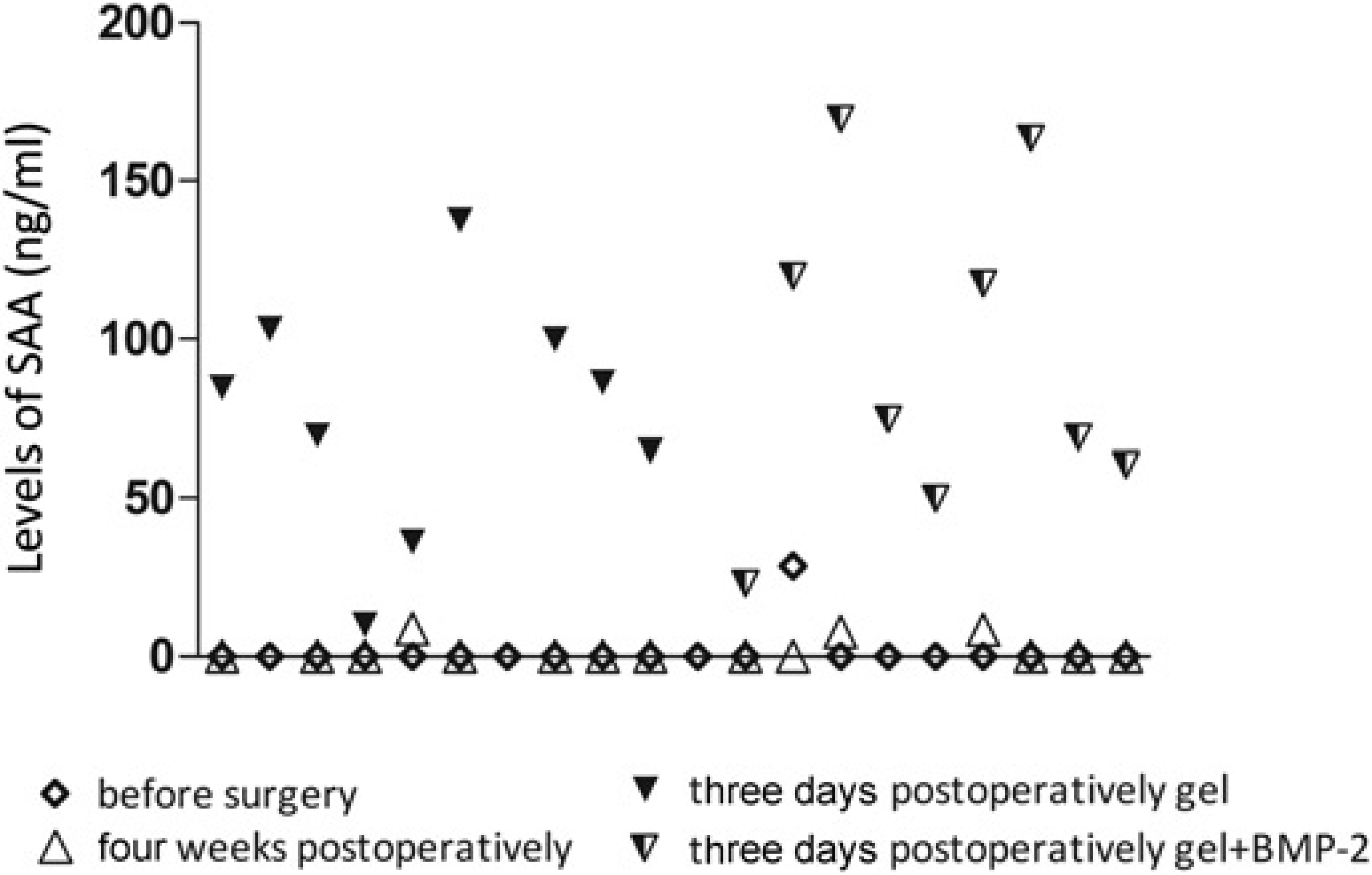
Postoperatively, seven (out of 18) animals were completely devoid of lameness, while 11 rabbits showed signs of lameness on one or several occasions (Table 1). No significant difference could be seen between groups or when comparing treated and untreated stifles in either group. Eleven rabbits received postoperative treatment with the antibiotic enrofloxacin (Baytril® vet, Bayer AS, Animal Health Division, Copehagen, Denmark), 5 mg/kg BW, subcutanously twice a day, for two to three weeks, due to signs of wound infection (lameness, swelling, body weight loss, fever, secretion of pus). Two rabbits did not respond to treatment and had to be euthanized. A third rabbit had to be euthanized due to a sudden onset of non-weightbearing lameness.
Number of animals observed with lameness, patellar luxation and ectopic bone formation
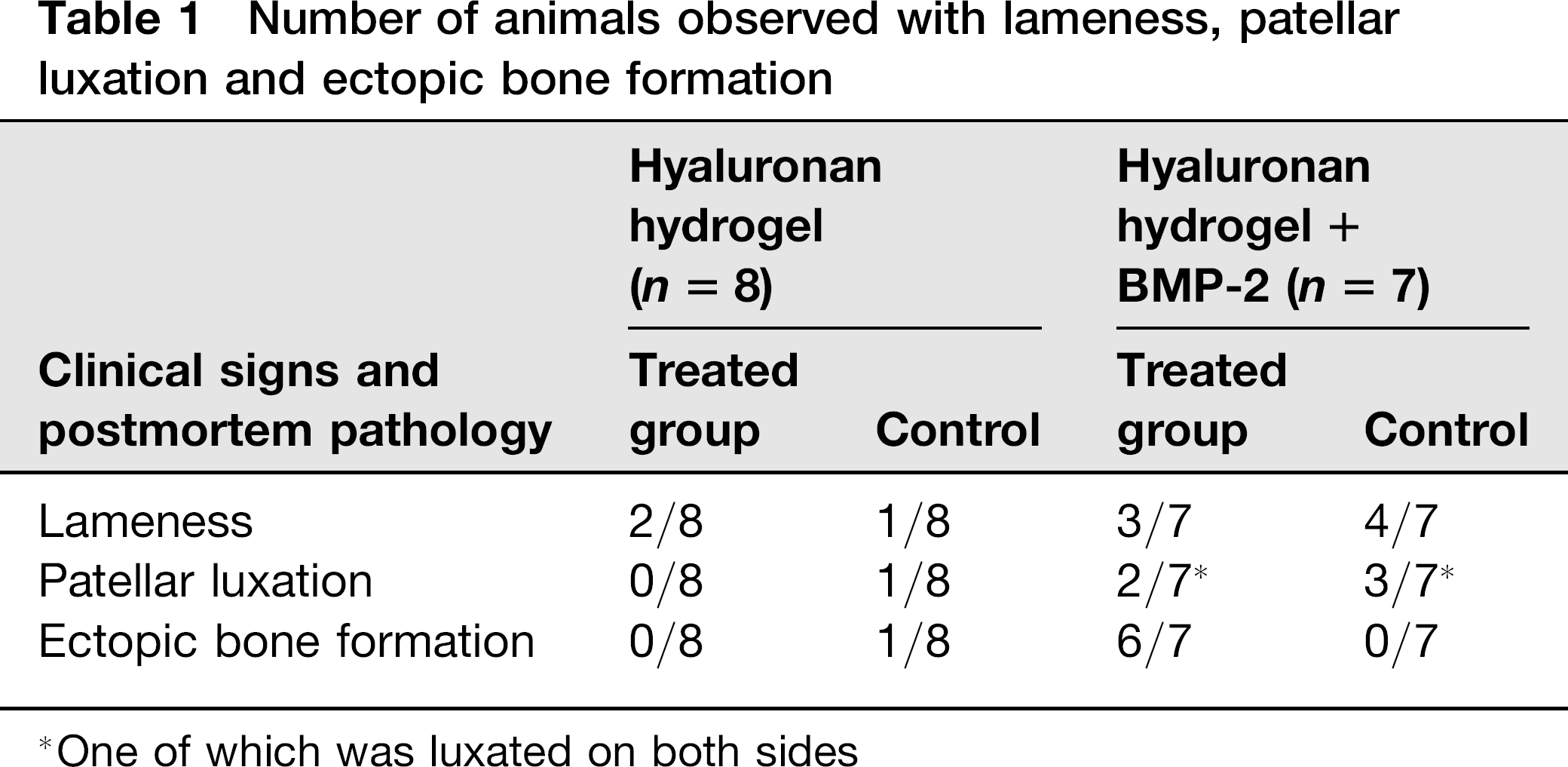
One of which was luxated on both sides
Postmortem macroscopic and microscopic findings
In all animals, the macroscopic appearance of the condyle lesions was similar, and the defects were filled with a dull cartilage-like material. All defects were easily seen as a roundish area in the medial condyles (Figures 2a, c and 3). Some of the defects were indented, in treated as well as control joints. Extensive ectopic bone formation in the synovial capsule and periarticular on the distal femur was found in six of the BMP-2-treated joints (Figure 2b). These comprised two of three BMP-treated joints examined after 12 weeks and all four BMP-treated joints examined after 24 weeks. Lateral patellar luxation with different amounts of periarticular osteophyte formation could be seen in six joints. Four of these were control joints, of which two were examined after 12 weeks and two after 24 weeks. Two of the six joints with patellar luxation were from the BMP-treated group after 12 weeks (Table 1). In these two joints, the periarticular osteophytes could be seen together with the ectopic bone formation.
Macroscopic pictures of the stifle joints in one rabbit euthanized 24 weeks after treatment. The upper panel shows the distal femoral condyles from a rabbit treated with (a) hyaluronan hydrogel + BMP-2 and (c) its contralateral control treated with saline. The lower panel shows the corresponding femoral trochlea. (b) Shows severe ectopic bone formation (arrow) in the synovial capsule. (d) Shows the normal contralateral control. The dashed lines indicate the area of the lesion. med: medial aspect, lat: lateral aspect of the joint Gross morphology and histology in two rabbits euthanized 24 weeks after treatment. The upper panel shows gross morphology of the lateral and medial femur condyles. The lower panel shows cross-section histology of the medial condyles stained with safranin-O-fast green. In both treated and control groups the defect was filled with repair tissue. The subchondral bone healed well but the tidemark was not restored and none or weak proteoglycan staining (red) was detected in the articular cartilage area. The dashed lines indicate the area of the lesion in the upper panel and the tidemark in the lower panel. Refer to online edition for colour reference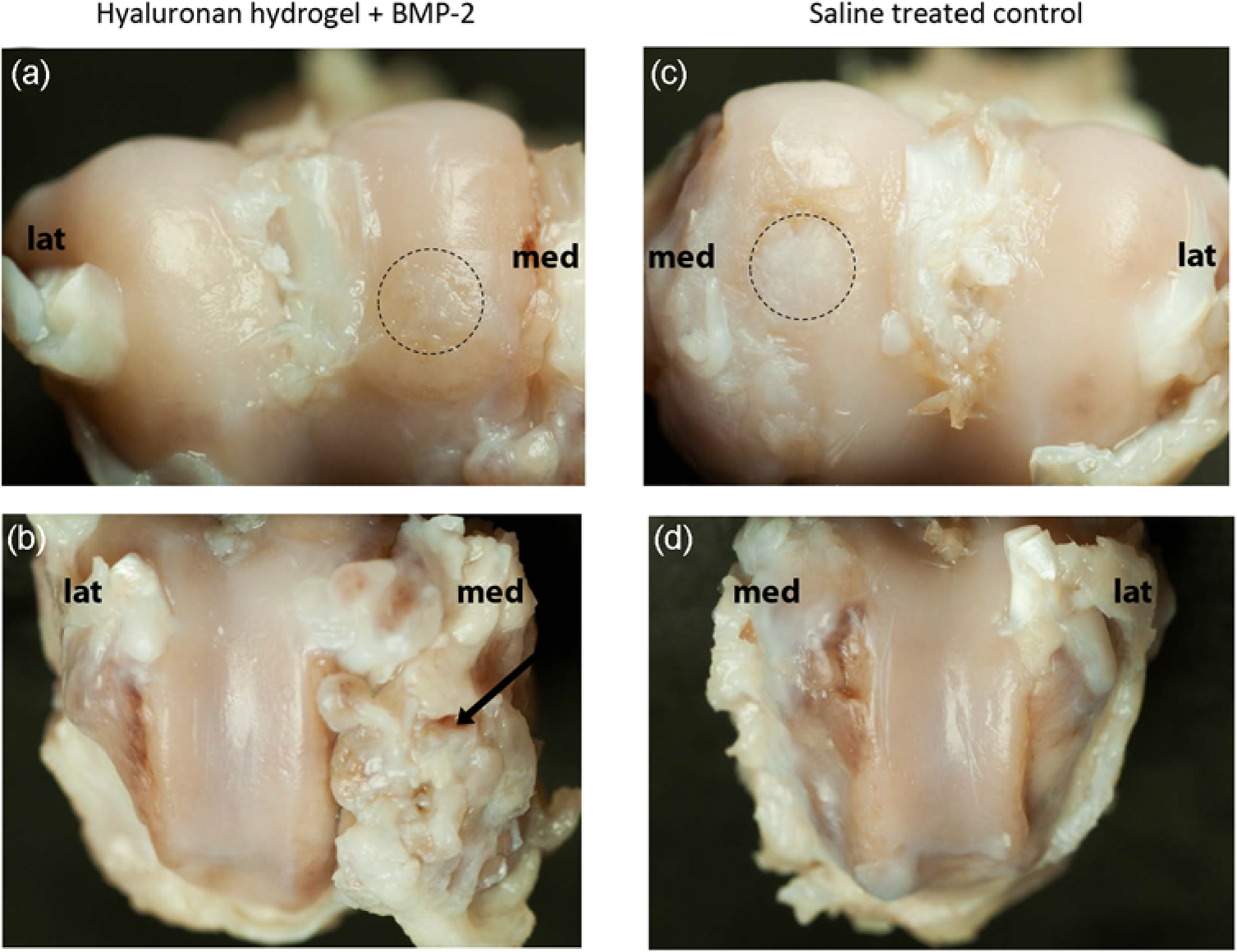
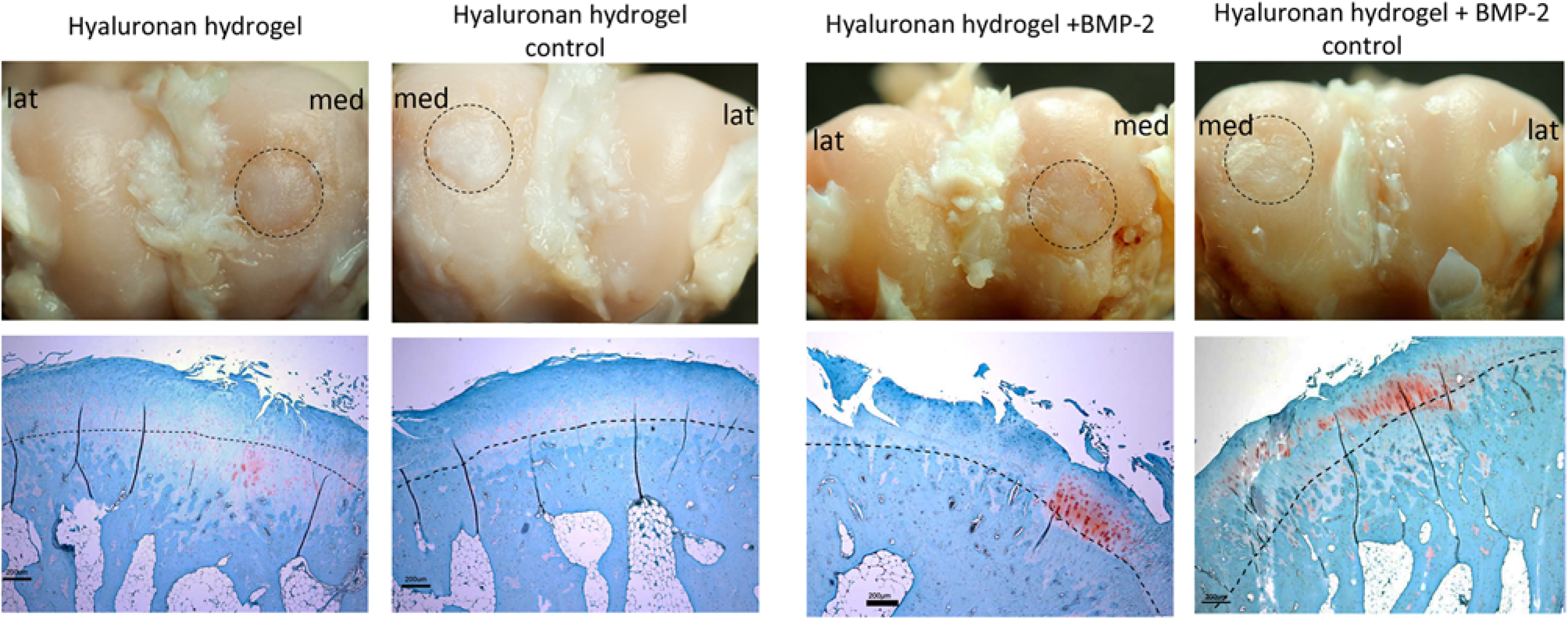
Cross-section specimens from the centre of the defects in the distal femur condyles were sectioned and evaluated histologically (Figure 3). Healing of defects varied both inter- and intragroup wise.
The defects were categorized and graded according to the filling of the articular cartilage defect, the integration with surrounding articular cartilage, the matrix staining (safranin O-fast green), cellular morphology, architecture of the entire defect and the surface as well as the restoration of the subchondral bone defect. The cell morphology was graded 0 when cells were mostly rounded with chondrocyte appearance and 5 when they were mostly spindle shaped (fibroblast like). The overall repair scoring values ranged from 0 (normal hyaline cartilage with subchondral bone including a tidemark) to 31 points (no cartilage or bone repair tissue). The filling of the subchondral bone defect was also evaluated separately according to percentage of new subchondral bone and formation of tidemark. The scoring of bone replacement ranged from 0 (complete filling with tidemark) to 4 (less than 25% of bone and tidemark). The treatment groups and the controls did not show any clear differences in the healing process. The total scores are presented in Figure 4. Statistical analysis was not performed due to the large intragroup variations.
Histological scoring of the regenerated tissue. A low value represents a high degree of regeneration. Individual scores are shown as dots and the arithmetic means of treatment groups as horizontal bars. (a) The scores (0–31) represent the overall repair in each joint. (b) The scores (0–4) represent healing of the subchondral bone defect in each joint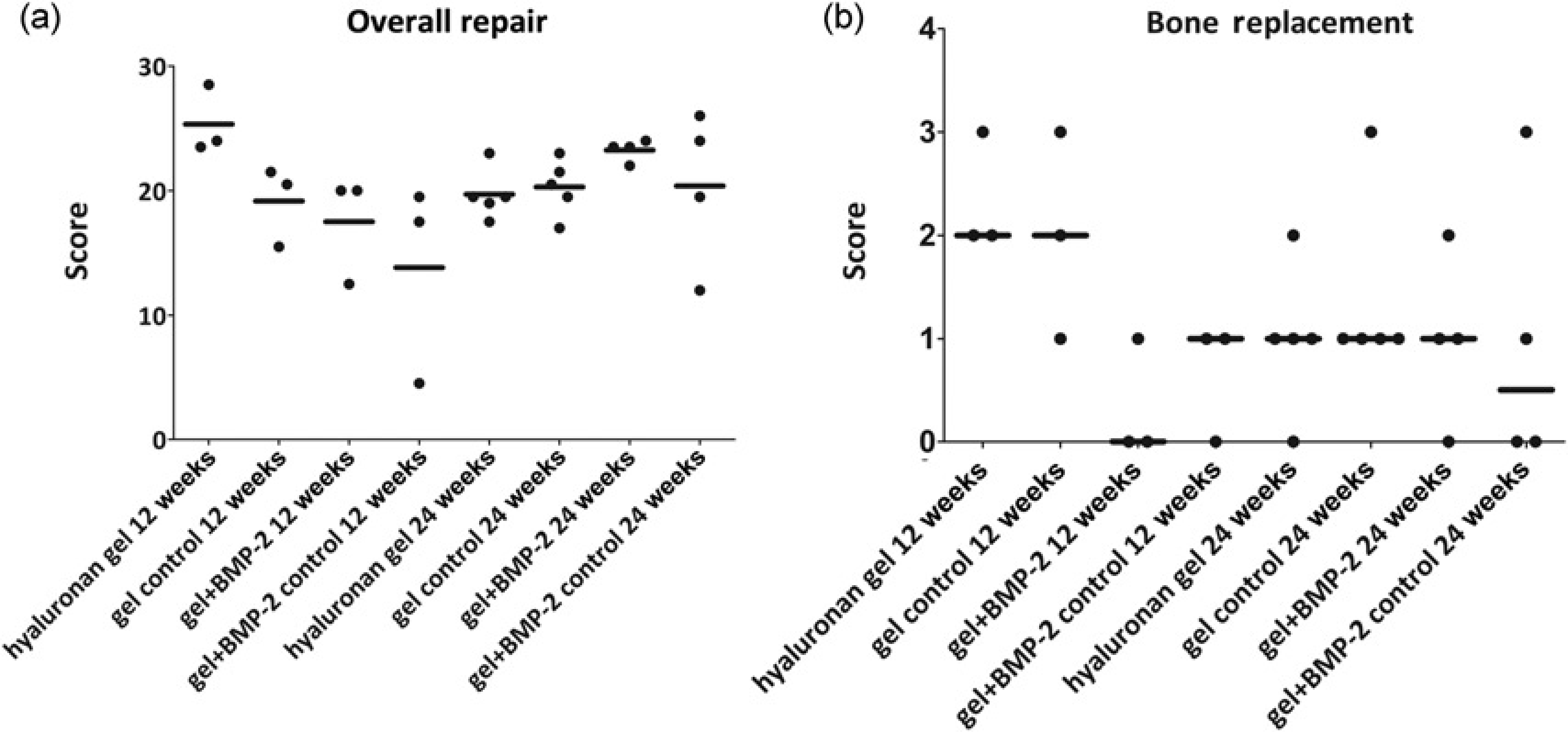
Synovial membranes showed a mild synoviopathy 20 with slight villi formation covered by two cell layers of synoviocyte proliferation, slight hyperplasia of the synoviocytes and mild to moderate diffuse to perivascular lymphoplasmocytic infiltration. Areas of mild fibroblastic and vascular proliferation could also be seen in some joints. In joints with patellar luxation, the cell infiltration was focally moderate. In six joints, multifocal to coalescent areas of ectopic bone formation with lamellar, trabecular to compact bone with areas of intertrabecular bone marrow were present in the joint capsule.
Discussion
The possibility of growth factor delivery into the joint for cartilage regeneration after injury would be an attractive treatment option to currently used techniques. The present study investigated the possibility of cartilage repair by intra-articular delivery of BMP-2 using an in situ cross-linkable hyaluronan hydrogel. The hydrogel represents a minimal invasive technique for local treatment of cartilage damage. We found that the healing of an induced cartilage lesion was not improved by treatment with hyaluronan hydrogel alone or hyaluronan hydrogel with BMP-2 compared with controls. A large inter-individual difference in the degree of healing was found in both groups, treated and control.
Not only was there no positive effect from either treatment, but joints treated with BMP-2 showed considerable ectopic bone formation, which constitutes a severe side-effect. The ectopic bone formation was seen both medially and laterally in the joint, suggesting it may have started from the joint capsule. Stem cells present in the synovial membrane and synovial fluid will after incubation with specific medium differentiate into osteogenic cells. 18 It can be speculated that the treatment with BMP-2 will influence these cells to become osteogenic and produce bone matrix.
BMP-2 has been used extensively to enhance bone formation and it is also known to stimulate chondrocyte proliferation, matrix production and aggrecan synthesis. BMP-2 plays contradictory roles, on one hand protecting from cartilage degeneration and on the other hand promoting chondrocyte hypertrophy and subsequent endochondral ossification. 21 Both dosage and presentation of BMP-2 may influence the fate of stem cells 22 and the risk when using potent growth factors such as BMP-2 is high, as seen from the ectopic bone formation in this study. The severity of the ectopic bone formation seen here may also in part be contributed to the relatively high concentrations used. When applying BMP-2 locally inside a defect with the flap-technique (often within a scaffold), the concentrations are often 10–20% of the amount used in this study.8,23 The high concentration of BMP-2 present in the joint may have shifted the chondrocytes toward a more degenerative phenotype, driving hypertrophy and ectopic bone formation rather than maintaining the chondrocyte phenotype.
In the joints treated with hyaluronan gel without BMP-2 our findings do not support earlier studies which have shown a positive effect of hyaluronan on cartilage repair, including the preceding pilot study using a hyaluronan gel.24–26A possible and important issue that may affect the diverse outcomes from the two studies is the altered preparation of the hydrogel. In the present study, gel formation was achieved before injection by extensive premixing of the two reactive components whereas in the previous study the hydrogel was mixed in situ by the use of dual barrel syringes and a mixing tip. The gel components carry aldehyde and hydrazide functional groups for the gel formation. If the mixing is inadequate, as assumed in the pilot study, unreacted components reside in the final gel and reactive sites on the polymers are still accessible. 27 The negative impact of endogenous aldehydes, such as 4-hydroxynonenal, on chondrogenesis has been demonstrated in a number of studies.28–30 An interaction and neutralization of endogenous aldehydes in the joint cavity by polymers that are functionalized with aldehyde-reactive hydrazide groups is possible and may have affected cartilage healing. Additional studies on the effects of hydrazidelinked polymers on cartilage repair are underway. On the other hand, the osteogenic effect of BMP-2 was boosted in the present study, which indicates that a premixed highviscous hydrogel gives a more favourable pharmacokinetic profile of the growth factor with enhanced effect as compared with the less mixed hydrogel.
Hyaluronan hydrogel and BMP-2 have been used alone or in combination for cartilage regeneration both in vitro and in vivo with varying results.19,31–33 The hydrogel itself is degraded within four weeks, with all BMP-2 released within that time. 13 Once released from the hydrogel, BMP-2 is rapidly cleared in vivo. From this study, the ectopic bone formation from released BMP-2 outweighed any potential regenerative effects. However, the possibility of using intra-articular injections treatment for cartilage regeneration after injury would be an attractive alternative to techniques currently used, in which a periosteal or collagenous flap is used to cover or seal the treatment material inside the defect. At the time of necropsy, no inflammation was detected in any of the animals. Initially after surgery, the acute phase protein SAA was increased as expected in all rabbits, and within one month postoperatively values had regained baseline levels, further supporting the absence of inflammation. The lack of inflammation together with the uncomplicated procedure of application showed that the hyaluronan gel system functions satisfactory as a local delivery system for the joint. This technique would be relevant also for treatment of joint disorders other than local injuries, such as osteoarthritis.
Other adverse effects, apart from ectopic bone formation, seen in five animals were patellar luxation and periarticular osteophyte formation, all but one in the BMP-2 treated group, but not restricted to the treated side. Osteophytes are known to develop in unstable joints, e.g. after tearing of the anterior cruciate ligament. 34 Growth factors such as transforming growth factor beta (TGF-β) are known to potentiate the development of osteophytes. 34
During surgery, a medial parapatellar incision was made into the joint capsule and the patella was moved laterally to gain access to the medial femur condyle, which extends the patellar tendon that keeps the patella in place. In the pilot study (n = 6), patellar luxation did not occur, which leads us to believe that luxations were a result of postsurgical housing conditions. An identical surgical technique was used in the pilot, but postoperatively, the rabbits were housed in single cages (0.42 m2) during the first two weeks, followed by pair-housing in double cages for the remaining period. 15 The floor area of the pens used in the current study is >3 times as large as that of the single pen, allowing the animals to move around freely. Some of the rabbits were easily frightened, despite frequent handling, and ran across the pen when someone entered the animal room. The excessive movement may have caused damage to the repair sutures, causing a lateral re-luxation of the patella.
Free movement is however important for regeneration of cartilage, inability to move may inhibit regeneration and result in muscle atrophy. 35 To reduce the risk of patellar luxation, it may be beneficial to house the rabbits in separate cages initially after surgery for 2–3 weeks, followed by pair-housing in double cages for 3–4 weeks, and then transfer them to larger pens for long-term recovery. In comparison, the recommendation from the American College of Veterinary Surgeons after surgical repair of patellar luxation in dogs is restricted exercise (walking) for at least six weeks. 36
The risks associated with social housing are evident from the rabbit that showed signs of fighting and died of hepatic lipidosis. The remaining rabbits, however, did not show fighting or fight wounds, indicating that social housing predominantly worked well. All rabbits were obese, evident at necropsy. This may be a problem for some studies. Results from rodent studies have shown that the use of fat and lean animals can result in opposite conclusions. 37
All but one rabbit recovered well from surgery and were eating within 24 h. The death of the first rabbit to undergo surgery was likely due to anaesthetic overdose. For induction of anaesthesia an intravenous combination was used which is new to rabbits, and for maintenance of anaesthesia this rabbit received a much higher isoflurane concentration than the subsequent rabbits.
During the first two weeks, all rabbits lost body weight (mean 10%) which may be expected after this kind of invasive surgery. The rabbits received at least three days of postoperative opioid treatment, and additional doses if needed. The study did not permit the additional use of a non-steroidal anti-inflammatory drug, which likely would have improved the rate of recovery. The formation of ectopic bone and patellar luxation most likely added to the pain caused by surgery.
Several rabbits received antibiotic treatment. Two that were euthanized due to response failure were diagnosed postmortem with joint infections caused by S. aureus and beta-haemolytic streptococci. In the successfully-treated rabbits, infections were most likely superficially located, not involving the joint. This was further confirmed at necropsy. It may be that not all treated rabbits were infected, it can however be difficult to differentiate between an infectious and non-infectious inflammatory state even with the help of a white blood cell count.
In a similar study of induced cartilage defects in rabbits, a 1/18 rate of joint infection was reported. 38 The rate of postoperative infection in stifle surgery in dogs is 4–7%. 39
Whereas most rabbits were free from lameness after surgery, some were lame for a longer period. In all cases but one, the lameness could be derived from patellar luxation or ectopic bone formation, although a few rabbits suffered from luxation or ectopic bone formation without showing any lameness. There was no difference in lameness between treated stifles and controls, indicating that the hyaluronan hydrogel did not have an analgesic effect, such as lubrication or ‘cushioning’.40,41 The possibility of detecting a difference in lameness between treatment groups may have been hampered by the fact that bilateral surgery was undertaken.
Many variables affect the outcome of induced cartilage lesion studies, including the response of the tissue to injury, age of the animals and size of the defect. 42 Spontaneous healing of subchondral bone is better in younger animals, 43 whereas intrinsic cartilage repair is not satisfactory even in young rabbits. 44 Epiphyseal growth plates are still detected in six-month-old rabbits, therefore 7–8-month-old rabbits were used in the current study.
The importance of the duration of follow-up in cartilage repair studies in rabbits was identified by Shapiro (1993), 45 who noticed that early regeneration of hyaline cartilage was followed by increasing signs of degeneration of the repair tissue, seen as early as 12 weeks after injury. This has led to the recommendation to leave at least six months to evaluate the defects. In the present study some regeneration was seen after 12 weeks, and it was neither improved nor degenerated after 24 weeks, which does not support this recommendation.
In conclusion, the present study shows that neither treatment with hyaluronic gel alone, nor in combination with BMP-2, improves the healing of an induced cartilage defect in rabbits. It further shows that BMP-2 can induce ectopic bone formation, which severely affects the functionality of the joint.
The rabbit can be a useful model for the study of cartilage repair. Rigorous aseptic procedures are important to prevent postoperative infections and housing space should be restricted for the first weeks following surgery to avoid postoperative patellar luxation.
Footnotes
Acknowledgements
The authors acknowledge Beate Hillmann and Tapio Nikkilä (both from Biomedical Sciences and Veterinary Public Health, SLU) for technical assistance in gross morphology and histology preparations. The authors also thank Dr Kristoffer Bergman and Dr Dmitri Ossipov (both from Materials Chemistry, Uppsala University) for providing the hyaluronan hydrogel components. This work has benefited from research funding from St Jude Medical AB research grant, Foundation Lars Hiertas Minne and FORMAS. The authors have no professional or financial affiliations that may have biased the presentation.
