Abstract
Specialty drugs treat complex, severe diseases and offer significant therapeutic advances. Despite their potential to transform patient care, little is known about the drivers of their diffusion. In this study, the authors develop a framework for the diffusion of specialty drugs that is motivated by the drugs’ novelty, complexity, and importance, with a focus on the role of scientific evidence production. They propose that the effects of scientific evidence on specialty drug diffusion are multifaceted and generated through the three stages of the scientific evidence production process: unpublished clinical studies, publications in medical journals not cited in clinical guidelines, and clinical guidelines. The findings from the empirical analysis of two specialty drugs validate the framework, supporting the idea of multistage scientific evidence effects. In contrast, marketing activities do not have a significant effect. Although this could be attributed to specialty drug prescribers discounting information from commercial sources, it may also be due to limited marketing support. An additional analysis on a nonspecialty drug further validates the proposed framework. Calculations of scientific evidence contributions to trial prescriptions indicate that scientific evidence production can generate returns beyond the publication stage, which should provide specialty drug manufacturers with strong incentives to commit to quality and innovation.
Keywords
A notable trend in the pharmaceutical industry is the development of specialty drugs, which are designed to treat complex, severe diseases, often with a limited number of patients (Kober 2008). Between 2014 and 2018, of the 219 new active substances that were launched in the United States, 136 (62%) belonged to specialty classes (IQVIA 2018). In 2017 alone, specialty drugs accounted for 32 of the 42 new active substances. Specialty drugs are characterized by novelty, complexity, and importance (e.g., Kesselheim et al. 2014). They are produced using advanced biotechnology and often fill unmet patient needs (Chambers et al. 2014; Lucio 2015). They treat complex, critical diseases and offer significant therapeutic advances, but they also come with high risks of adverse events. In addition, they are extremely costly (Kirchhoff 2015; Lucio 2015). For example, in 2018 specialty drugs in the United States accounted for 49.5% of total medicine net spending ($344 billion), even though they represented only 2.2% of the total 5.8 billion prescriptions (IQVIA 2019). Because of their high costs, their prescription is subject to prior insurance authorization, which requires documentation on the appropriateness of the treatment for the patient (Robinson, Howell, and Pearson 2018).
Despite the potential of specialty drugs to transform patient care, our knowledge regarding the drivers of their diffusion is limited. Most extant research has focused on nonspecialty drugs, providing valuable insights regarding the contributing factors to their growth and adoption (e.g., Azoulay 2002; Manchanda, Xie, and Youn 2008; Sood, Kappe, and Stremersch 2014; Van den Bulte and Lilien 2001). However, the study of specialty drug diffusion requires a framework that is motivated by their defining characteristics of novelty, complexity, and importance. In this study, we develop and validate such a framework, which is guided by two core principles:
The combination of the novelty, complexity, and importance of specialty drugs necessitates that prescribing physicians must quickly develop an extensive knowledge base to make appropriate and timely evidence-based decisions (e.g., Bergethon and Wasfy 2019; Peay and Peay 1994). Specialty drug prescribers are predominantly specialist physicians (SPs) who have the motivation, opportunity, and ability to directly source and process scientific information to make evidence-based decisions (Jadad and Enkin 2007). They treat critical illnesses, are in close proximity to research, focus on a narrow range of clinical problems, and have the expertise to assimilate knowledge developments (Harrold, Field, and Gurwitz 1999; Peay and Peay 1994; Stremersch, Landsman, and Venkataraman 2013).
Thus, scientific evidence production should be critical for tapping the limited market potential of specialty drugs and, consequently, their diffusion in the marketplace.
A distinctive feature of our framework is the acknowledgment that scientific evidence production is a process, which can influence diffusion by providing information to specialty drug prescribers through its various stages. The process begins with clinical studies (first stage), some of which are published in medical journals (second stage). Eventually, a subset of the published articles will form the basis of clinical practice guidelines (third stage), defined as “systematically developed statements to assist practitioner and patient decisions about appropriate healthcare for specific clinical circumstances” (Institute of Medicine 1990, p. 8). Prior research, mostly on nonspecialty drugs, has focused mainly on publications and has not examined the role of scientific evidence production as a process. Here, we argue that because of the novelty, complexity, and importance of specialty drugs, extensive sourcing of information from all three stages of the scientific production process is essential for prescribing physicians.
The proposed framework links the scientific evidence production process to the drug's diffusion process. It also accommodates marketing activity effects. However, we argue that such effects should be weaker or even negligible because (1) specialty drug prescribers would tend to discount information from commercial sources, which can be partial and lag behind scientific developments (Ching and Ishihara 2010; Ziegler, Lew, and Singer 1995), and (2) practically, marketing support for specialty drugs may be limited due to the limited accessibility of specialty drug prescribers (Khedkar and Sturgis 2016). We empirically validate the framework by analyzing the diffusion of two specialty drugs in Canada. We adopt a macro-level diffusion approach since our data are at the product level. Our findings support the idea that the influence of scientific evidence production on specialty drug diffusion is multifaceted and manifested through all three stages of its production process. We also find that both journal advertising and detailing have no significant effect on diffusion, confirming our expectations. To further validate our findings, we study the diffusion of a nonspecialty drug and find that in terms of scientific evidence production, uncited publications (i.e., publications in medical journals not cited in clinical guidelines) and clinical guidelines influence prescriptions, whereas unpublished clinical studies do not. Moreover, separate analyses by physician specialty status reveal that clinical guidelines influence only SP prescribers, and detailing has a significant effect only on general practitioner (GP) prescribers.
Overall, our study claims the following contributions: (1) It develops and validates a framework for studying the diffusion of specialty drugs, which links the scientific evidence production process to the diffusion process. (2) It provides empirical support for multifaceted scientific evidence production effects that are generated through the three stages of the scientific evidence production process. Our study is the first, to our knowledge, to register a significant effect of unpublished clinical studies on specialty drug prescriptions. (3) It offers insights, elaborated in the “Implications and Recommendations” section, regarding the absolute and relative contributions of each stage of the scientific evidence production process to drug prescriptions over time and hence their return on investment (ROI) potential.
The rest of the article is organized as follows. In the next section, we provide a background on the diffusion of new pharmaceuticals and scientific evidence production effects. We then propose a conceptual framework for the diffusion of specialty drugs, followed by our empirical analysis. Finally, we discuss the implications of our findings and recommendations for marketers and conclude with a discussion of research implications and future research.
Background
Diffusion of New Pharmaceuticals
The diffusion of new pharmaceuticals has been extensively reviewed elsewhere (e.g., Landsman, Verniers, and Stremersch 2014; Wieringa et al. 2014). Here, we focus on studies most relevant to ours. Diffusion models for new pharmaceuticals have been developed at both the macro and micro levels, depending on the availability of data (product level vs. physician/patient level). They examine the effects of social contagion through measures of prior cumulative adoption, direct-to-physician (DTP) and direct-to-consumer (DTC) marketing activities, and scientific evidence.
Lilien, Rao, and Kalish (1981), Rao and Yamada (1988), and Hahn et al. (1994) introduce foundational modeling approaches to macro-level diffusion of pharmaceuticals. They distinguish between trial and repeat prescriptions and examine the effects of marketing activities such as detailing and medical journal advertising. Trials are modeled using variants of the Bass model (Bass 1969), whereas repeats are modeled as a fraction of cumulative adoption. The findings of these studies suggest that DTP marketing activities have a positive effect on diffusion. Shankar, Carpenter, and Krishnamurthi (1998) and Kolsarici and Vakratsas (2010) build on these approaches. The former show that late entrants introducing innovative drugs can overcome pioneer advantages with higher trial growth and repeat rates, whereas the latter provide evidence for the dynamic effects of detailing and direct-to-consumer advertising (DTCA). Ruiz-Conde, Wieringa, and Leeflang (2014) test for the most appropriate specification for the effects of marketing activities (detailing, medical journal advertising, DTCA, and physician meetings) in trial-repeat models. They examine 34 drugs in various therapeutic categories and find that promotional activities affect mostly the trial rate through external influence.
In terms of micro-diffusion models, Van den Bulte and Lilien (2001) revisit the study of Coleman, Katz, and Menzel (1966) and find that the contagion effect estimated in the latter dissipates after accounting for marketing efforts in a hazard model. Manchanda, Xie, and Youn (2008) use a discrete choice model and find that both contagion and detailing affect the utility of adoption for a drug mostly prescribed by GPs. Iyengar, Van den Bulte, and Valente (2011) use a hazard model and find evidence for the role of opinion leadership in contagion effects on the adoption of a drug treating the chronic form of a specific viral infection. Liu and Gupta (2012) find evidence for the effects of marketing activities, social contagion, patient requests, and physician characteristics on physician adoption of a drug that treats a chronic condition and affects a large number of patients.
The only study that examines scientific evidence effects, using a macro-level diffusion approach, is by Conti, Bernstein, and Meltzer (2012). They investigate the impact of clinical studies initiated around the drug's approval date and the drug's efficacy according to the U.S. Food and Drug Administration (FDA)-approved drug label on the diffusion of seven new specialty (cancer) drugs. Hence, their scientific evidence measures are time-invariant. The authors estimate a generalized Gompertz hazard model on data pooled across drug classes and find that the count of clinical studies has a significantly positive effect on the rate of diffusion, but efficacy does not. They interpret the former as an initial quality signal effect, whereas they attribute the latter to substitutability with the social learning effect of cumulative past adoption. However, they do not distinguish between published and unpublished clinical studies, nor do they consider clinical guidelines and marketing activities.
Scientific Evidence Effects
Table 1 provides a summary of prior empirical studies on scientific evidence effects on pharmaceutical demand. The summarized studies have focused mostly on published evidence, including results of clinical studies published in medical journals or released by health authorities, which they frequently term “clinical trials/studies.” The only exceptions are Conti, Bernstein, and Meltzer (2012), which has already been discussed, and Shapiro (2018), who studies published evidence and clinical guidelines as demand and detailing informational shocks.
Empirical Studies on Scientific Evidence Effects on the Demand for Drugs.

Notes: AR(1) = first-order autoregressive; UnPubStud = unpublished clinical studies; PE = published evidence; UncitPub = uncited publications; ClinGuide = clinical guidelines; (Stk)Det = (stock of) detailing; (Stk)JAd = (stock of) journal advertising.
Published evidence is often measured using characteristics such as volume, or the count of publications, as well as valence and dispersion, depending on whether published results are positive, negative, or neutral (e.g., Azoulay 2002; Ching and Ishihara 2010; Sood, Kappe, and Stremersch 2014). Other measures consider the significance of the evidence using patient enrollment in studies (Ching et al. 2016). The types of effects studied are direct (on sales, market share, diffusion, or prescription volume/probability), indirect (through marketing or physician learning of drug quality), and interactive (with DTP marketing).
Most studies find that the influence of scientific evidence is positive. Exceptions are Cockburn and Anis (2001), who find insignificant effects on market share, and Chintagunta, Jiang, and Jin (2009), who find negative effects on prescription probability. Serra-Sastre and McGuire (2013) find evidence for direct effects of published evidence, as do Slejko, Basu, and Sullivan (2018), who also include specialty drugs in their analysis. Azoulay (2002) and Sood, Kappe, and Stremersch (2014) examine both direct and indirect effects. Azoulay (2002) finds that published evidence has a positive direct effect on market share, which is smaller than the effects of marketing activities such as detailing. He also finds that published evidence has an indirect effect through marketing activities. Sood, Kappe, and Stremersch (2014) decompose the characteristics of published evidence in terms of volume, valence, and dispersion and find that volume and valence have long-term positive effects on drug sales; in contrast, promotional efforts such as detailing and medical journal advertising have smaller and short-lived effects. They further find that the volume of published evidence decreases DTCA, whereas valence (more positive outcomes) increases both DTCA and DTP marketing. In addition, the dispersion (variance of reported outcomes) of evidence decreases DTCA. Their findings allow for the calculation of the commercial value of clinical studies, which has clear implications for practice. Lastly, Blankart and Stargardt (2020) and Olson and Yin (2021) focus on the effects of valence. The former find that public evidence reporting positive results accelerates initial drug adoption, whereas the latter conclude that public evidence reporting negative or mixed effects for children lowers the drug prescriptions to them.
Shapiro (2018) also finds evidence for indirect effects of new publications and clinical guidelines, in the form of informational shocks through detailing. Ching and Ishihara (2010) find that, compared with patients’ experience with a drug, published evidence provides more precise information to physicians about the drug's quality. Ching and Lim (2020) further find that physicians’ belief in a drug's quality is positively affected by not only the published evidence favorable to the drug but also the evidence favorable to other drugs in the same class. Interaction effects of scientific evidence are studied by Venkataraman and Stremersch (2007), who find that published evidence tends to boost the effect of detailing on the number of prescriptions by reducing physician uncertainty, and Ching et al. (2016), who show that the interaction between detailing and cumulative favorable published evidence increases a drug's market share.
In summary, most of the scientific evidence literature has focused on published evidence only and has not investigated the role of scientific evidence production as a multistage process. However, there are indications that clinical studies (Conti, Bernstein, and Meltzer 2012), including unpublished ones, and clinical guidelines (Shapiro 2018) may also influence prescription growth. The influence of the entire scientific evidence production process can be especially relevant in the case of specialty drugs and will constitute the focal point of our conceptual framework.
Conceptual Framework
The extant literature discussed in the “Background” section has provided valuable insights into the diffusion of new drugs and scientific evidence effects on pharmaceutical demand. However, little is known regarding the diffusion of specialty drugs, despite their potential to transform patient care. To the best of our knowledge, besides Conti, Bernstein, and Meltzer (2012), the only other study focusing on the commercial performance of specialty drugs in the management literature is by Liu, Liu, and Chintagunta (2017). They find that detailing can have both competitive and spillover effects for HIV/AIDS drugs, but they do not examine drug diffusion or account for scientific evidence effects. Furthermore, prior work has not investigated the role of scientific evidence production as a process that can influence diffusion through its various stages.
In this section, we develop a conceptual framework for the diffusion of specialty drugs, which acknowledges the role of marketing and scientific evidence production as a multistage process. First, we provide a foundation by discussing the defining characteristics of specialty drugs and the prescription behavior of physicians, which motivate our approach.
Specialty Drugs and Physician Prescription Behavior
Specialty drugs mostly treat complex, severe diseases. Although a small number of specialty drugs can be lifestyle-enhancing rather than treating a medical need (Marinova et al. 2017), we focus on specialty drugs treating a limited number of patients with serious medical conditions. Specialty drugs are more complex than nonspecialty drugs because they are often biology-based mixtures that are not easily identified (Lucio 2015). They offer significant therapeutic advances but also come with important risks (Kesselheim et al. 2014). Moreover, their cost is high, ranging from $15,000 to as much as $750,000 per year per patient (Barlas 2014). In contrast, annual costs for branded statins, which have been extensively studied by marketing researchers, range from $2,040 to $7,824 per year per patient (Consumer Reports Best Buy Drugs 2014). Due to these high costs, insurers may impose restrictions on specialty drug reimbursement. More specifically they may accept certain, but not all, conditions for which the drug was approved and demand that treating physicians document that their patients have one of the acceptable conditions (Robinson, Howell, and Pearson 2018).
Therefore, specialty drugs define a unique setting, characterized by novelty, complexity, and importance. In such a setting, prescribing physicians need to quickly develop an extensive knowledge base to make appropriate and timely evidence-based decisions that also meet insurer requirements (e.g., Peay and Peay 1990; Sackett et al. 1996; Straus et al. 2018). This accords with prior work on decision making, which associates novelty, complexity, and importance with extensive information acquisition (e.g., McQuiston 1989).
Because of the high potential benefits of specialty drugs and the severity of the diseases they treat, early awareness and systematic assessment of relevant information from scientific evidence are critical for prescribing physicians, who need to minimize the time between knowledge generation and helping the patient (Bergethon and Wasfy 2019; O’Connor et al. 2018). Given the well-documented time lag between the production of clinical study findings and their publication (Qunaj et al. 2018), unpublished and ongoing clinical studies can be an important source of early information for prescribing physicians. Because of the high risks of specialty drugs, recommendations in clinical guidelines, developed by leading authorities in the corresponding therapeutic areas, can also be an important supplementary source of information. Specifically, clinical guidelines are based on the appraisal of the totality of evidence by experts (Djulbegovic and Guyatt 2019; Shekelle et al. 1999, 2001). Thus, their recommendations should reduce prescribing physicians’ concerns about the risks of a specialty drug and increase their confidence in its quality.
Specialty drug prescribers are predominantly SPs. They have the motivation, opportunity, and ability to source and process information not only from publications but also from unpublished clinical studies and clinical guidelines. They cater to patients with severe conditions and are therefore highly motivated “by a need to prevent complications at all costs” (Beaulieu et al. 2008, p. 35) to assist them by quickly building a knowledge base to evaluate the appropriateness of each treatment. Because they are typically employed at research medical institutions, where clinical studies take place, they can access information even on unpublished clinical studies through their contacts with clinical investigators (Ayanian et al. 1994; Harrold, Field, and Gurwitz 1999; Peay and Peay 1990, 1994) or by attending specialty conferences and meetings where ongoing clinical study results are presented before publication (Conti, Bernstein, and Meltzer 2012; Toma et al. 2006). SPs also tend to consult clinical guidelines, developed by thought leaders in their network and published in medical journals (Harrold, Field, and Gurwitz 1999; Pollard, Bansback, and Bryan 2015). Finally, given their expertise and narrow focus on clinical problems, SPs have the ability to process and integrate such information. This is consistent with the consumer research perspective that experts have a greater ability to delineate relevant complex product information (Alba and Wesley Hutchinson 1987; Broniarczyk and Griffin 2014).
While selecting a specialty drug is an important decision for patients, prescribing SPs are usually the lead decision makers because of their expertise and intellectual mastery (Ding and Eliashberg 2008; Stremersch, Landsman, and Venkataraman 2013) and the potentially negative emotional state of their patients (Botti, Orfali, and Iyengar 2009). Therefore, specialty drug prescribing is the result of a physician-centric and evidence-based rational decision-making process (Jadad and Enkin 2007; Maxwell 2016).
Specialty Drug Diffusion
We propose a diffusion framework, depicted in Figure 1, guided by the two core principles emerging from the previous discussion: (1) Specialty drugs are characterized by novelty, complexity, and importance. Prescribing such drugs requires that physicians, as decision makers, quickly build an extensive knowledge base to make appropriate and timely evidence-based treatment decisions. (2) Specialty drug prescribers have the motivation, opportunity, and ability to process evidence-related information. As shown on the top row of Figure 1, a drug's total market consists of patients with the disease that the drug is designed to treat (hereinafter termed the “focal disease”). Often, however, the drug is approved to treat a subclass of the disease and thus can possibly treat only a proportion of its total market, which we call its “maximum penetration potential.” For example, the drug Harvoni, designed to treat hepatitis C (focal disease), is approved for treating chronic hepatitis C (subclass), and thereby its maximum penetration potential (chronic hepatitis C patients) corresponds to a subset of its total market (all hepatitis C patients). Patients move from the market potential stage to the trial stage subject to physician evaluation of the drug's utility, which is based on scientific evidence and marketing information. The utility evaluation reflects a physician-centric rational decision-making process, consistent with our discussion in the previous section. After initial trial, patients may repeat prescriptions through refills and renewals.

Conceptual Framework for Specialty Drug Diffusion.
A distinctive feature of our framework is the acknowledgment that scientific evidence production is a process, which can influence specialty drug diffusion by providing information to prescribers through its various stages. Clinical studies enroll patients that fit the eligibility criteria, such as the condition/disease, to be early adopters of the drug. As enrollment accumulates and more studies approach completion, more scientific evidence for the drug, based on the experiences of the participating patients, emerges and becomes shareable. Some of these studies will be published in scientific journals subject to the peer-review process. Journal articles further help disseminate evidence for the drug to the prescribing physician community, especially when they generate a sizeable number of citations. Evidence from journal articles is then reviewed by experts, who develop and update clinical guidelines on the use of the drug for treating the focal disease. In clinical guidelines, experts cite only a subset of those journal articles, which they deem to report adequate evidence for forming recommendations for the drug in comparison with other treatments.
The three stages of the scientific evidence production process are shown on the bottom row of Figure 1: (1) unpublished clinical studies, or clinical studies on the specialty drug's treatment of the focal disease whose results are not yet published; (2) uncited publications, or medical journal articles that report results from some of those studies and have not been cited in clinical guidelines; and (3) clinical guidelines. From a diffusion perspective, the scientific evidence production process provides an opportunity for social learning from prior adoption (e.g., Young 2009), through the communication of drug treatment outcomes. Such outcomes are based on the experiences of clinical study participants and can influence prescriber evaluation of the drug's quality (e.g., Ching and Ishihara 2010). Unpublished clinical studies can offer the most timely information on medical advances, yet such information is potentially less rigorous because it has not been subjected to peer or expert review. In contrast, clinical guidelines can offer the most rigorous information that has been subjected to peer and expert review, but such information is less timely because of the time required for the review process and the accumulation of published evidence.
Based on the two guiding principles of our framework, we expect prescribing physicians to extensively source information from all scientific evidence production stages, and not only from publications in medical journals, to evaluate a specialty drug's utility. Unpublished clinical studies can provide early-stage information, which is essential due to the high benefit potential of specialty drugs and the severity of the diseases they treat. Clinical guidelines, produced by “super-representatives” of their peer group (e.g., Fink et al. 1984; Institute of Medicine 1992; Simons, Berkowitz, and John Moyer 1970), can provide prescribing physicians with credible information, which is essential due to the high risk of specialty drugs.
Finally, we expect DTP marketing (detailing and journal advertising) to have weaker or even negligible effects on utility given the ability of prescribing physicians to source information directly from the scientific evidence production process. Although detailing can include scientific evidence information, SPs would rely less on it because commercial information tends to be partial and lags scientific developments (Ching and Ishihara 2010; Peay and Peay 1990, 1994; Ziegler, Lew, and Singer 1995). Furthermore, marketing support for specialty drugs can be limited due to the limited accessibility of SPs, who constitute the vast majority of specialty drug prescribers (Khedkar and Sturgis 2016). Our expectation accords with research on SP prescription behavior in nonspecialty markets, which has found that detailing affects SPs less than GPs (Ding and Eliashberg 2008; Stremersch, Landsman, and Venkataraman 2013). It also conforms with the decision-making literature suggesting that experts use select relevant information (Shanteau 1992). The expected effects are indicated in Figure 1. In brief, we expect all scientific evidence production variables to have positive effects on the utility of each focal specialty drug and thus the trial prescriptions of the drug. DTP marketing activities for the drug, such as detailing and journal advertising, may have weaker or even negligible effects on its utility.
Empirical Analysis
Data and Variables
We collect monthly data on two specialty drugs (labeled SD1 and SD2) in Canada using multiple sources. Table 2 summarizes relevant background information. The key variables are described in Table 3 and illustrated in Figure 2. Data concerning prescriptions and marketing activities are provided by the drug manufacturers, IMS Health and IQVIA, on the condition of anonymity; hence, we cannot disclose full details. The drugs (“focal drugs”) are from different therapeutic classes, developed by different firms, and introduced at different times; thus, we model their diffusion separately. Each drug treats a subclass of a rare disease (“focal disease”), which is lethal and almost incurable and affects less than 1% of the Canadian population. The five-year survival rate for the disease class SD1 treats is 14%, whereas the corresponding rate for SD2 is 70%, indicating that the focal disease for SD1 progresses faster than that for SD2. Before entering Canada, both drugs received a “priority review” designation from the FDA, suggesting that they could offer a significant improvement compared with existing treatments. Prescribers for both drugs are predominantly SPs representing a little more than 1% of all medical specialists in Canada, with 59% (SD1) and 72% (SD2) of them being employed by academic health science centers or universities (Canadian Medical Association n.d.).
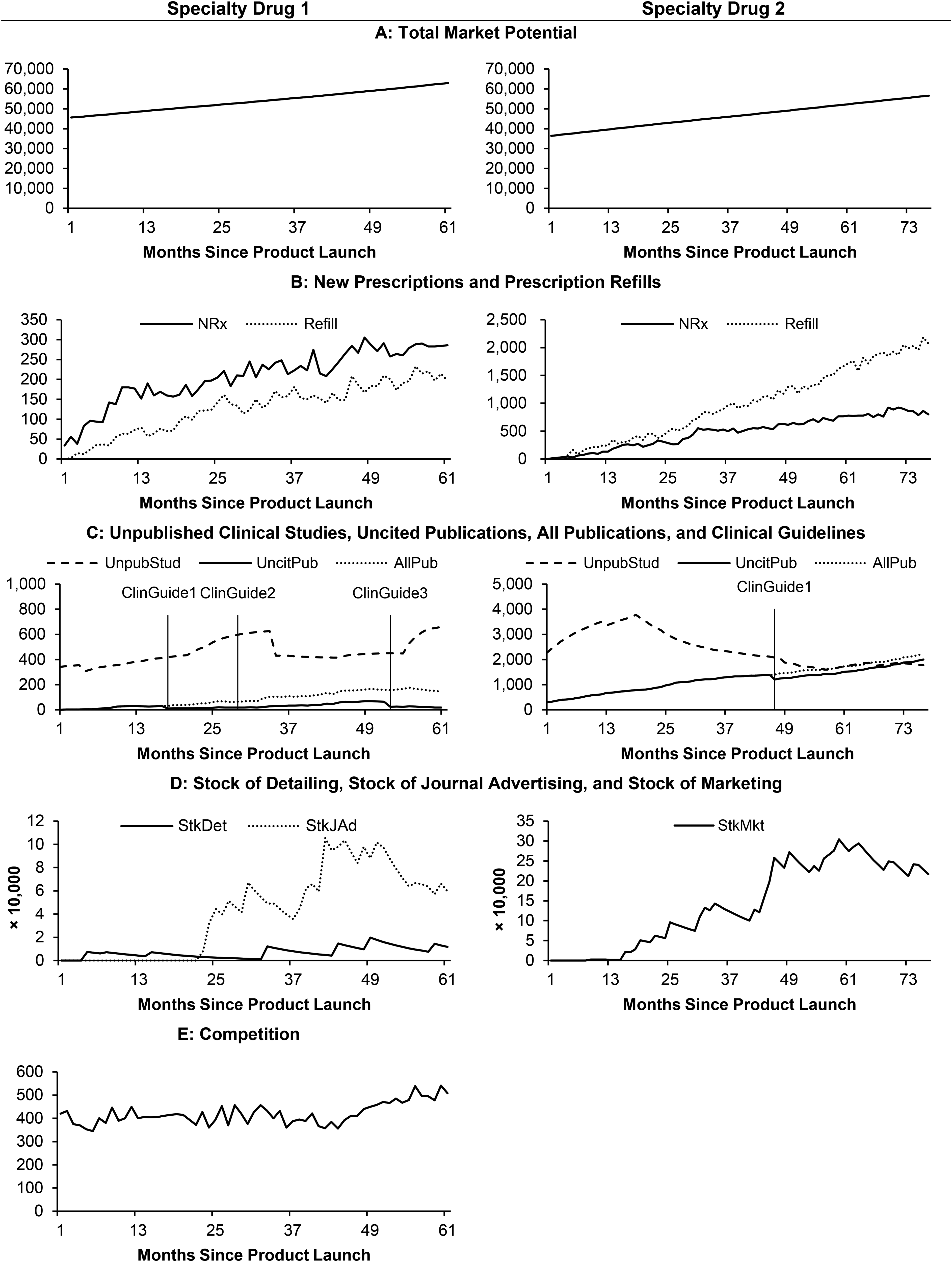
Monthly Data of Key Variables for the Specialty Drugs Since Product Launch.
Descriptive Information for the Specialty Drugs.

Clinical studies on the focal drug in treating the focal disease.
Journal articles publishing results from the focal clinical studies.
First-line treatment refers to the initial treatment given to patients for a disease. Typically, the first-line treatment is the one that worked best in clinical studies. After the first-line treatment has failed, stopped working, or has side effects that are not tolerated, a different treatment (called a second-line treatment) may be given.
Description of Variables.

The disease that the focal drug is designed to treat.
Clinical studies on the focal drug in treating the focal disease.
Journal articles publishing results from the focal clinical studies.
Prescriptions
For each month t, we observe new prescriptions (NRxt) and prescription refills (Refillt) of each drug; NRxt contains both trials (Trialt) and prescription renewals (Renewalt) that cannot be separately observed. The observation period for each drug spans at least 61 months and starts with its launch. About 23% of patients treated with SD1 and 84% of patients treated with SD2 show no sign of health condition deterioration within the first year of treatment. Given that patients tend to continue with the same treatment for as long as it works, the difference between the two drugs in those figures may explain why Refillt is lower than NRxt for SD1 but not for SD2. During the respective observation periods, the number of rival drugs for SD1 grew from one to six, while SD2 remained a monopolist. SD1 became the market leader within one year of its launch and maintained its leading position for the remainder of the observation period with monthly total prescriptions (NRxt + Refillt) frequently higher than those of the competing drugs combined (Compt).
Total market potential
To calculate the total market potential of each focal drug in month t, TotMktPtnlt, we collect data from Statistics Canada. We rely on annual statistics on new cases and deaths related to the focal disease (the most granular level of available data) and use the year that statistics were first available as the starting year. We compute TotMktPtnlt as the number of alive patients with the focal disease by accumulating net new cases, calculated as the number of new cases minus the number of deaths, assuming both figures are uniformly distributed within each year.
Unpublished clinical studies
Data on unpublished clinical studies up to month t, UnpubStudt, are mainly collected from the clinical study registry ClinicalTrials.gov. This site was created following the Food and Drug Administration Modernization Act of 1997, which requires registration for clinical studies on drugs for serious or life-threatening diseases. For each study, the registry reports (1) the condition (i.e., the disease under study), (2) the intervention (i.e., drugs that are either investigational or already available), (3) the NCT number (i.e., the unique code given to each study registered on ClinicalTrials.gov), (4) the starting date, (5) the primary completion date (i.e., the date when the last participant in a clinical study was examined to collect final data for the primary outcome measures), (6) the enrollment (i.e., the total number of participants), (7) the recruitment status (e.g., whether the study is ongoing, completed, withdrawn before enrolling, terminated, or unknown because it has not been verified within the past two years), and (8) the published journal articles associated with the clinical study as identified by the NCT number.
We collect information on the clinical studies related to the focal disease and the focal drug. We first use the focal drug and the focal disease as keywords and identify 98 studies that are initiated by the end of the observation window for SD1 and 309 studies for SD2. We then exclude studies not focused on the focal drug's efficacy, safety, or impact on patients’ quality of life; studies combining the focal drug with other treatments (because those combinations are not yet approved); and studies using the focal drug but focusing on the development of other treatments not yet approved. Thus, we keep 63 studies for SD1 and 160 studies for SD2 (i.e., the “focal clinical studies”).
Consistent with prior work (e.g., Azoulay 2002; Ching and Ishihara 2010; Ching and Lim 2020; Sood, Kappe, and Stremersch 2014), we assume that scientific evidence knowledge accumulates over time and has a long-lived effect, which could be subject to decay. However, each study's contribution to the total stock of knowledge may vary, depending on its enrollment and valence. A study is more likely to produce shareable evidence when more participants are examined. Physicians may also be more likely to gain access to a clinical study of a larger scale or consider such a study to be more important. Further, a study with no adverse events should contribute positively to the total stock of knowledge. We therefore calculate UnpubStudt as the sum of stocks of enrollment over all focal clinical studies that have not been published, weighed by their valence.
More specifically, we assume that between its start month Si and primary completion month Ci, study i can examine participants at a uniform speed, and thus the number of participants in month t is Eit = TEi/(Ci − Si), where TEi is its reported enrollment; outside the period between Si and Ci, Eit = 0. The stock of enrollment is calculated as SEit = δCS × SEi(t − 1) + Eit, where δCS is the carryover parameter, assumed to be the same across all clinical studies. Based on its recruitment status, we assign the valence of study i (VEi) to be −1 if it is terminated because scientific data show adverse events, showing early evidence against the drug (Williams et al. 2015); 0 if it is withdrawn before enrolling any participants, terminated for other reasons, or with an unknown status; and 1 otherwise. Finally, we calculate UnpubStudt in month t by summing SEit × VEi over only focal clinical studies whose results have not been published in journals up to month t.
Uncited publications
Data on uncited publications up to month t, UncitPubt, are mainly from ClinicalTrials.gov, which provides a list of journal articles associated with each clinical study by the NCT number. In addition, we use NCT to further validate the list and search for additional medical journal articles on PubMed.gov, a free resource developed and maintained by the U.S. National Center for Biotechnology Information. Table 2 reports the number of publications and the total citations associated with the focal clinical studies (i.e., the “focal publications”), which are further classified by whether the published results are positive or negative. Unsurprisingly, publications reporting negative results are rare, with fewer than three articles for each drug.
Following the logic applied to UnpubStudt, we calculate UncitPubt as the sum of citation stocks over all publications that have not been cited by clinical guidelines, weighed by their valence. The rationale is that physicians may consider a journal article that received more forward citations from publications to be more impactful or may be more likely to access such an article. Further, an article reporting positive results is expected to have a positive contribution to the total stock. More specifically, let the number of forward citations received by article j from publications other than clinical guidelines in month t be Cjt; then the stock of citations is SCjt = δP × SCj(t − 1) + Cjt, where δP is the carryover parameter assumed to be the same across all publications. The valence of article j (VCj) is assumed to be 1 if it reports positive results regarding the focal drug's efficacy, safety, or impact on patients’ quality of life; −1 if it reports negative results on those aspects; and 0 if it reports inconclusive results or results irrelevant to those aspects. Finally, we compute UncitPubt in month t by summing SCjt × VCj over focal publications not cited in clinical guidelines up to month t. For model comparison purposes, we also compute a variable termed all publications (AllPubt), which is the sum of SCjt × VCj over all articles, including those cited by clinical guidelines. Figure 2 shows the trajectories of AllPubt and UncitPubt, with the gap between the two due to the publications that are cited by clinical guidelines.
Clinical guidelines
We search for guidelines issued by the Canadian association of each focal disease from PubMed.gov. These associations gather top Canadian experts to review the available evidence and develop or update guidelines. Table 2 lists the publication months of guidelines that review the focal drugs and their main recommendations. All versions give the focal drugs positive recommendations. In line with Shapiro (2018), we use dummy variables, ClinGuide l t, to indicate whether the lth version of clinical guidelines is in effect in month t.
Marketing variables
We construct stock variables to account for the carryover effect of marketing activities. Specifically, let the marketing spending in month t be Mktt. Then the corresponding stock is defined as StkMktt = δM × StkMktt − 1 + Mktt, where δM is the carryover parameter. Using this approach, we create the stock of detailing (StkDett) and stock of journal advertising (StkJAdt) till month t for SD1. For SD2, journal advertising spending is nonzero for only two months during the observation window; we thus combine spending on the two activities to form a single stock variable for marketing activities.
Empirical Model
To validate our conceptual framework, we employ a trial-repeat model. We model Trialt using a utility-based diffusion approach (Krishnan, Seetharaman, and Vakratsas 2012). Specifically,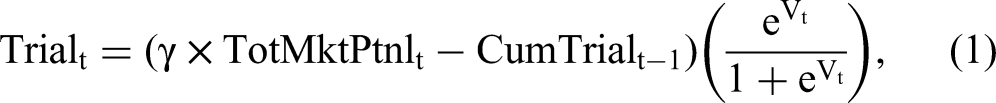


The utility-based diffusion model has a firm decision-theoretic foundation, consistent with the principles of our conceptual framework. It is also analogous to prior micro-diffusion approaches (Liu and Gupta 2012; Manchanda, Xie, and Youn 2008), which, however, do not consider scientific evidence effects. The consideration of scientific evidence in the utility is consistent with our social learning perspective, according to which adoption decisions flow from the rational evaluation of evidence based on prior adoption (Young 2009).
Empirically, we cannot observe Trialt; we can only observe NRxt, which includes both Trialt and Renewalt. Thus, we decompose NRxt as follows:
We note that endogeneity concerns are limited in our study. One potential concern is the endogeneity of marketing activities due to drug-specific demand shocks observed by the firm but not by the researchers. The most common unobserved demand shock discussed in the literature is new clinical information (Azoulay 2002; Ching and Ishihara 2010). However, this is not an issue here, since we account for scientific evidence production. Furthermore, not including prices is not a concern because in Canada they are highly regulated and remain stable during the observation window. Finally, when controlling for the competition effect, we use lagged values for total prescriptions of rival drugs to mitigate the potential endogeneity due to the simultaneity of prescriptions of focal and rival drugs.
Preliminary Analysis: Log-Log Model
To form a baseline of findings regarding the effects of the three scientific evidence production stages on prescriptions, we estimate a log-log model by regressing NRt on variables used in our analysis in the following form:
Log-Log Model Estimation Results for the Specialty Drugs.

*p < .1.
**p < .05.
***p < .01.
Proposed Model Results
Table 5 summarizes the results from the proposed model (Model 1) and an alternative model (Model 2), which accounts for the effect of AllPubt, rather than the three scientific evidence production stages, on trial prescriptions. Both root mean squared error (RMSE), which is adjusted for the number of parameters, and mean absolute deviation (MAD) indicate that Model 1 outperforms Model 2 for both drugs. This lends further validity to our conceptual framework, which advocates multistage scientific evidence production effects. We therefore discuss the results from Model 1 in detail.
Estimation Results for the Specialty Drugs.

*p < .1. **p < .05. ***p < .01.
We estimate α, the parameter corresponding to the maximum penetration level γ = 1/[1 + exp(−α)].
Renewal rate is the coefficient corresponding to lagged prescription refills of the focal drug.
Notes: Bold values indicate the best model for the drug based on each fit statistic.
All scientific evidence production variables have significantly positive effects on trial prescriptions (SD1: β1 = .054, p < .001; β2 = .166, p < .001; β3,1 = .227, p = .014; β3,2 = .309, p = .006; β3,3 = .888, p < .001; SD2: β1 = .028, p = .009; β2 = .142, p < .001; β3,1 = .477, p < .001). We therefore confirm our expectation that for specialty drugs, scientific evidence can influence diffusion through all stages of its production process due to the need for prescribing physicians to quickly build a comprehensive knowledge base to make appropriate and timely treatment decisions. To our knowledge, this is the first study to register a significant effect of unpublished clinical studies on trial prescriptions.
To gain further perspective on these findings, we calculate average elasticities of trial prescriptions with respect to unpublished clinical studies and uncited publications using their analytical expressions (.5β1(UnpubStudt).5{1 – [eVt/(1 + eVt)]} and .5β2(UncitPubt).5{1 − [eVt/(1 + eVt)]}, respectively). The resulting unpublished clinical study elasticities are .559 for SD1 and .672 for SD2, and the uncited publication elasticities are .397 for SD1 and 2.300 for SD2. For SD1, the higher unpublished clinical study elasticity, compared with the one for uncited publications, suggests that prescribing physicians emphasize timeliness more than rigor. This can be justified based on the low survival rate (fast progression) of the disease treated by SD1, which necessitates a timely intervention. For SD2, prescribing physicians appear to prioritize rigor over timeliness (suggested by a higher uncited publication elasticity), which can be afforded by the higher survival rate (slower progression) of the disease. Based on the corresponding formula, the size of the uncited publication elasticity for SD2 can be attributed to the early high volume and high growth of the uncited publication stock (Figure 2).
Our findings on scientific evidence effects suggest that for specialty drug manufacturers, sponsoring scientific evidence production can generate returns that extend beyond the publication stage. Thus, not accounting for the role of scientific evidence production as a multistage process may result in not only a misattribution of scientific evidence effects (to publications only) but also their underestimation. For example, Model 2 yields average all publication elasticities of .409 for SD1 and 1.932 for SD2, which are comparable to the average uncited publication elasticities from Model 1. However, the additional effects of UnpubStudt and ClinGuide l t, estimated with Model 1, suggest that scientific evidence production can generate additional returns at no additional cost and therefore boost ROI. We further explore this issue in the “Implications and Recommendations” section.
In terms of marketing activities, we do not find a significant effect on trial prescriptions. This may be attributed to specialty drug prescribers discounting information from commercial sources due to its potential subjectivity and time lag. However, it could also be due to the limited marketing support for these drugs. Overall, our findings indicate that scientific evidence production is the dominant force in driving the diffusion of new specialty drugs, lending support for the evidence-based, rational prescribing focus of our framework.
Validation: Diffusion of a Nonspecialty Drug
To provide further validation for the proposed framework, we analyze the diffusion of a nonspecialty drug. Introduced in Canada in March 2012, this drug treats a disease that is recurrent and chronic. Like the two specialty drugs, it received a priority review designation from the FDA before entering the Canadian market. We collected data in the same way we did for the case of specialty drugs. The only exception is the data collection for the total market potential. More specifically, we use PubMed.gov to search for studies that report the prevalence (%) of the focal disease in Canada, which we multiply by the demographic statistics from Statistics Canada to come up with the market potential. The data are described in Table 6 and illustrated in Figure 3. We note that data on prescriptions and detailing are separated by physician type (GP vs. SP). Medical literature suggests that GPs are more likely to receive clinical information from commercial information sources and respond to it by prescribing nonspecialty drugs (Beaulieu et al. 2008; Greving et al. 2006). Further, GPs, compared with SPs, may exercise greater caution in accepting emerging information regarding a treatment (Harrold, Field, and Gurwitz 1999; Hirth, Fendrick, and Chernew 1996). Finally, because GPs do not focus on specific clinical fields, they are more likely to perceive that there are too many clinical guidelines, which are hard to access, and that consulting guidelines is bothersome or time consuming (Carlsen and Bringedal 2011). Hence, we expect that GPs may rely more on detailing and less on unpublished clinical studies or clinical guidelines.
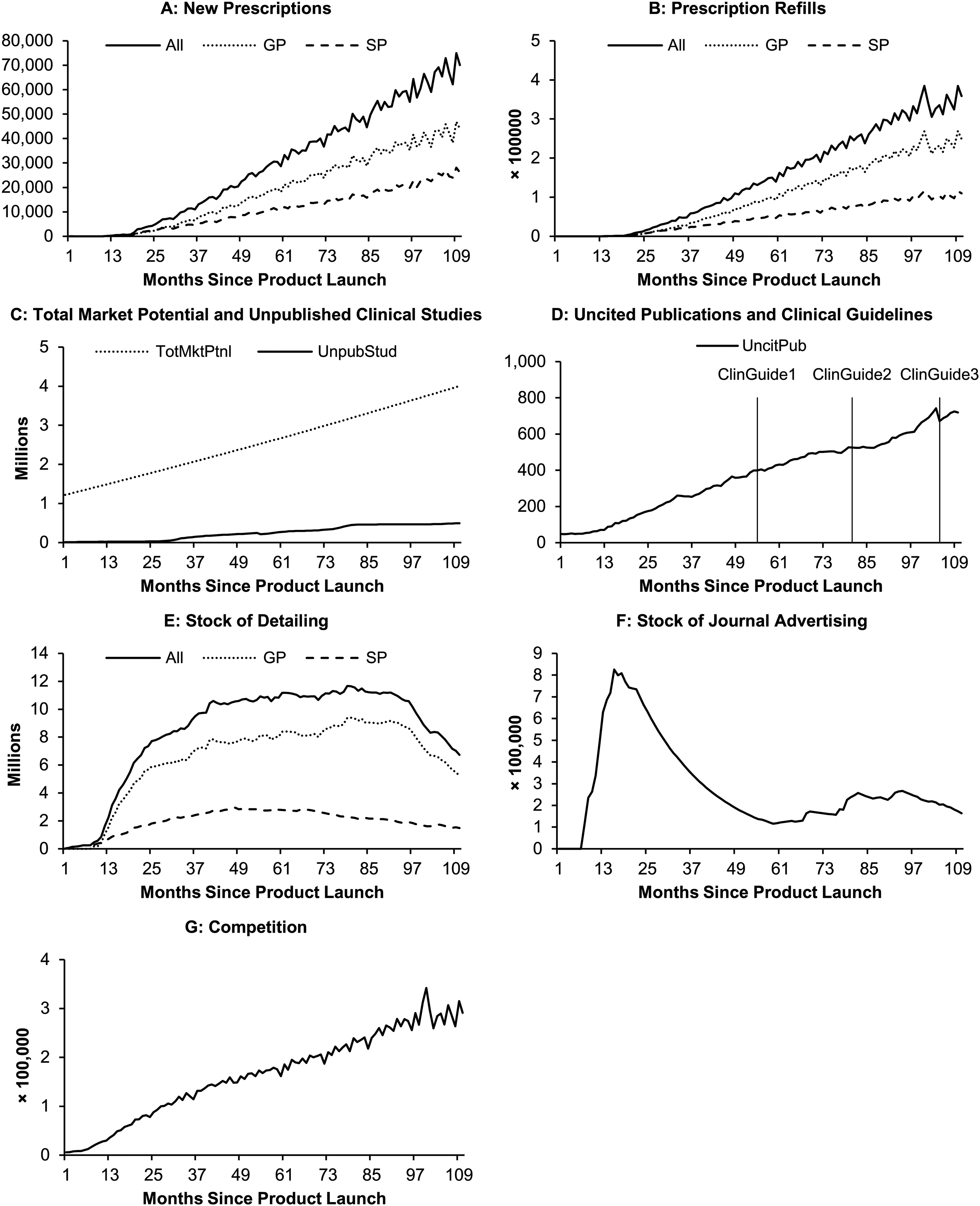
Monthly Data of Key Variables for the Nonspecialty Drug Since Product Launch.
Descriptive Information for the Nonspecialty Drug.

Clinical studies on the focal drug in treating the focal disease.
Journal articles publishing results from the focal clinical studies.
Table 7 presents the results. An initial empirical analysis showed that the maximum penetration level is close to 1; we therefore set γ = 1 without loss of fit or a notable change in the estimates. The findings suggest that uncited publications and the third version of the clinical guidelines have significantly positive effects on trial prescriptions (β2 = .331, p < .001; β3,3 = .482, p = .062). Moreover, the stock of detailing has a significantly positive effect on trial prescriptions (β4 = .001, p = .034), consistent with the positive detailing effects reported by many prior studies on nonspecialty drugs.
Estimation Results for the Nonspecialty Drug.

*p < .1. **p < .05. ***p < .01.
The carryover parameters are 1 for clinical studies, .97 for publications, and .95 for marketing activities.
Renewal rate is the coefficient corresponding to lagged prescription refills (by all physicians when using all data or by the type of physician when using data only from GPs or SPs) of the focal drug.
We then estimate separate models for GPs and SPs. Table 7 shows that although uncited publications still have a significantly positive effect for both GPs (β2 = .294, p = .001) and SPs (β2 = .271, p < .001), the third version of clinical guidelines has a significantly positive effect only for SPs (β3,3 = .473, p = .064), whereas detailing has a significantly positive effect only for GPs (β4 = .001, p = .016). The findings for GPs regarding the effects of published evidence and detailing are in line with those of prior work on nonspecialty drugs or GP prescriptions. Moreover, the findings on clinical guideline effects on SPs accord with the medical literature discussed previously.
Our findings on SPs suggest that although they source evidence from uncited publications and clinical guidelines, they do not respond to unpublished clinical studies. Hence, their response lies between the response of GPs for nonspecialty drugs (balance of published evidence and detailing) and the response of SPs for specialty drugs (focus on all three scientific evidence production stages). This suggests that differences in the drivers of the diffusion of specialty and nonspecialty drugs are due not only to the specialty status of the physician but also to the specialty status of the drug. More specifically, the high benefit potential of specialty drugs, coupled with the severity of the diseases they treat, prompts prescribing SPs to respond to early-stage scientific evidence (unpublished clinical studies). Such urgency can be further explained by the fact that many specialty drugs fill unmet needs (Kesselheim et al. 2014), whereas new nonspecialty drugs usually have viable alternatives (Peay and Peay 1994). Thus, when prescribing nonspecialty drugs, physicians may not rely on information from unpublished clinical studies but rather wait for less timely but more rigorous information from uncited publications. Overall, these findings provide further validation for our framework. We also explored an alternative specification of a trial-repeat model and obtained results consistent with the ones we reported. For further information, please refer to the Web Appendix.
Implications and Recommendations
Scientific Evidence Contribution to Trial Prescriptions
To further explore and quantify the implications of our empirical findings, we use the estimation results from Model 1 to calculate absolute and relative contributions to trial prescriptions from each stage of the scientific evidence production process for specialty drugs. Specifically, we first predict cumulative trials for the first five years, by using the parameter estimates of all variables from Model 1 in Table 5 (“full prediction”). Given that clinical guidelines constitute the last stage of the scientific evidence production process, we derive their contribution first, by muting their effects (setting ClinGuide l t = 0 for all l) and then predicting cumulative trials. The difference between the full prediction and the prediction without the effect of ClinGuide l t thus represents the contribution of clinical guidelines to cumulative trials. Next, we mute both ClinGuide l t and UncitPubt and predict the corresponding cumulative trials; the difference between the prediction without ClinGuide l t and that without both ClinGuide l t and UncitPubt represents the contribution of uncited publications. Finally, we mute ClinGuide l t, UncitPubt, and UnpubStudt and make another prediction of cumulative trials, which represents the base prediction. The difference between the prediction without ClinGuide l t and UncitPubt and the base prediction represents the contribution of unpublished clinical studies.
The contributions of each scientific evidence production stage for SD1 and SD2 are illustrated in Panels A and D of Figure 4. The overall contribution of scientific evidence production to cumulative trials is substantial, reflecting our findings on the dominance of scientific evidence effects. Specifically, five years after the drug's launch, UnpubStudt, UncitPubt, and ClinGuide l t collectively contribute 8,010 trials for SD1 and 16,758 trials for SD2 (corresponding to the difference between the full and base predictions in Month 60). Notable is the relative contribution of unpublished clinical studies for SD1 (see Figure 4, Panel B), which is higher at the beginning. This is in line with our arguments that unpublished clinical studies provide critical early-stage information for specialty drug prescribers. As uncited publications pick up for SD1, the relative contribution of unpublished clinical studies drops but remains at a high level. On average, the relative contribution of UnpubStudt to cumulative trials is lower than that of UncitPubt (43.19% vs. 46.35%).

Contribution of Evidence to Predicted Cumulative Trials of the Specialty Drugs for the First Five Years.
The contributions for SD2 exhibit a different pattern, with uncited publications having the highest relative contribution throughout the observation window (see Figure 4, Panel E). This should be largely attributed to the early high volume and steady growth of publications for SD2. The relative contribution of unpublished clinical studies to SD2 trial prescriptions is limited but, as in the case of SD1, higher early on. For both drugs, the relative contribution of clinical guidelines at the end of the five-year window is also considerable (23.58% for SD1 and 12.20% for SD2). Finally, we note that base contributions are low, especially for SD2, because these are due to purely innovative behavior (corresponding to the utility intercept that represents a constant baseline hazard), which is unlikely in drug adoption.
We also compare the contributions implied by Model 1 with those implied by Model 2, which considers only all publications (panels (c) and (f) of Figure 4). Model 2 predicts that all publications contribute 4,089 trials for SD1 and 14,117 trials for SD2 over the five-year window. In both cases, the contributions implied by Model 2 are much lower than the collective contributions of all scientific evidence production stages suggested by Model 1 (8,010 for SD1 and 16,758 for SD2). Consistent with our elasticity discussion, our calculations suggest that assuming that the ROI in clinical studies is realized only through publications, as most previous studies on nonspecialty drugs do, may underestimate scientific evidence ROI for specialty drugs. The high ROI potential of scientific evidence production should provide incentives for specialty drug manufacturers to commit to innovation and quality.
Recommendations for Marketers
Our findings suggest that scientific evidence is the dominant driving force for the diffusion of specialty drugs. Given the prominence of scientific evidence as the primary source of information for prescribing physicians and its focus on drug quality, an opportunity exists for prelaunch marketing to contribute via its integration with research and development (e.g., Becker and Lillemark 2006). More specifically, marketing can contribute by engaging physicians and patients in clinical study protocol design to produce customer-led innovation (Gudiksen et al. 2008; O’Hagan and Farkas 2009). In fact, firms increasingly engage physicians in the development of new drugs (DiMasi, Grabowski, and Hansen 2016), whereas patient engagement may not only increase enrollment and reduce protocol amendment costs but also increase drug relevance to the target patient population (Gudiksen et al. 2008; Levitan et al. 2018). Because engaging physicians and patients in clinical studies can create conflicts of interest, firms should proactively disclose such conflicts and adhere to related research ethics guidelines.
In addition, marketing can contribute by installing processes to systematically monitor patient and physician input and assess its impact on scientific evidence production (Becker and Lillemark 2006). Such a shift, with a focus on engaging with patients and physicians, has the potential to optimize marketing efforts given marketing's capabilities to glean customer insights. Finally, a closer involvement in the production of innovation may lead to increased marketing influence within the firm (Verhoef and Leeflang 2009).
Conclusion and Future Research
Our study proposes and validates a framework for the diffusion of specialty drugs that is motivated by their novelty, complexity, and importance, with a focus on the role of scientific evidence production process. The core finding from the empirical analysis of two specialty drugs is that the effects of scientific evidence are multifaceted and manifested through the three stages of its production process. Our study is the first, to our knowledge, to register a significant effect of unpublished clinical studies on prescriptions, which we claim to be specific to specialty drugs due to their high benefit potential and the severity of the disease they treat. Our findings on uncited publications and clinical guidelines effects further suggest that, although both are based on published evidence, they correspond to distinct stages of the scientific evidence production process to which physicians respond differently. Therefore, researchers studying the commercial performance of specialty drugs should distinguish between uncited publications and clinical guideline effects.
Our empirical analysis of a nonspecialty drug further validates our framework, albeit on a limited basis, suggesting that differences in the effects of diffusion drivers between specialty and nonspecialty drugs cannot be solely attributed to the specialty status of the prescriber but also the specialty status of the drug. More specifically, a spectrum of physician prescription behavior has emerged from the collective findings of our empirical analysis. On one end of the spectrum, GPs prescribing nonspecialty drugs respond to a balance of detailing and published evidence. On the other end, SPs prescribing specialty drugs respond to information from all stages of the scientific evidence production process, with negligible marketing influence. In between, SPs prescribing nonspecialty drugs respond only to scientific evidence except for unpublished clinical studies. The emergence of a prescription behavior spectrum warrants further investigation via replications with more therapeutic classes since it can carry important implications for returns on scientific evidence and effort allocation by firms managing a portfolio of specialty and nonspecialty drugs.
From a broader perspective, our study offers a characterization of the diffusion process for specialty drugs that can be used by both academics and pharmaceutical firms to evaluate relevant sources of influence and returns on scientific evidence production. On the latter, we underscore the need for a change in the evaluation model from a focus on journal publications only, as suggested by prior research on nonspecialty drugs, to the consideration of all three stages of the scientific evidence production process. Our implications illustrate that focusing only on publications will lead to an underestimation of the returns on scientific evidence. Thus, from a research perspective, not accounting for the effects of all scientific evidence production stages will cause potential omitted variable bias. From a practical perspective, the prospect of a high scientific evidence ROI for specialty drugs due to multistage effects should provide pharmaceutical firms with strong incentives to continue committing to innovation and quality. This, in turn, will improve the quality of life for patients, enhance productivity, and minimize the burden of the disease (Sullivan 2008).
Our findings on the prominence of scientific evidence effects also point toward new opportunities for marketing. Specifically, given the growing trends in physician and patient engagement, marketing can contribute to customer-led innovation via its integration with research and development. A systematic approach to customer-led innovation with marketing's input could increase the relevance of new drugs to patients, which may be more important for commercial success than the novelty of the drug itself (Gudiksen et al. 2008). It could also increase marketing's influence within the firm via direct involvement in the production of innovation.
A limitation of our study is that it focuses on two specialty drugs. Future research should extend applications to different specialty therapeutic categories in various countries. Although the proposed framework is generalizable because it is based on broad defining characteristics of specialty drugs, the intensity of scientific evidence effects may be different across different settings. Therefore, we identify five potential areas of future research:
Global perspective: Do scientific evidence effects vary across a wide range of countries? Could uncertainty avoidance (Stremersch and Lemmens 2009) moderate the impact of unpublished clinical studies on the diffusion of specialty drugs? For example, relying on unpublished clinical studies may be perceived as risky by prescribing physicians in countries with high uncertainty avoidance because such studies have not been subjected to the peer review process. In addition, in countries without national health insurance (unlike Canada, which provided the context of the current study), pricing may have a significant effect on diffusion. Life-saving versus life-enhancing: As mentioned in our conceptual framework section, a small number of specialty drugs, such as growth hormones, are used to treat healthy patients who wish to improve the natural capacity of their body or mind (Marinova et al. 2017). In such cases, the treatment decision-making approach may involve patients and their families with SPs having more of a consulting role. This in turn may affect the relative contribution of scientific evidence production and increase the potential influence of not only DTP marketing but also DTCA. Volume and timing of published evidence: Our implications show a strong contribution of uncited publications for SD2, which we attributed to a relatively high volume of publications early on (prelaunch). Future research should investigate more extensively the role of these potential moderators to provide generalizations regarding the (relative) contribution of scientific evidence. Macro- versus micro-diffusion: Given the availability of product-level data only, we were not able to investigate how the diffusion of specialty drugs may be affected by the dyadic interactions between individual physicians and their patients or the heterogeneity among physicians in terms of their experience and patient population. Future research should analyze physician/patient-level data to further explore these topics. Marketing support and effectiveness: Marketing support for the focal specialty drugs we examined is limited, which could be due to the limited population of prescribing physicians and their low accessibility. We suggested that the lack of marketing effectiveness in our study could be related to low marketing support. Future research should investigate more specialty drugs, with varying levels of marketing support, to shed more light on how marketing support relates to the effectiveness of marketing activities.
Supplemental Material
sj-pdf-1-jmx-10.1177_00222429231177627 - Supplemental material for Scientific Evidence Production and Specialty Drug Diffusion
Supplemental material, sj-pdf-1-jmx-10.1177_00222429231177627 for Scientific Evidence Production and Specialty Drug Diffusion by Demetrios Vakratsas and Wei-Lin Wang in Journal of Marketing
Footnotes
Special Issue Editor
Harald van Heerde
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bensadoun School of Retail Management Scholar Fund.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
