Abstract
Keywords
T
The immunohistochemical transmitter identification of traced neurons has made many important contributions to neurosciences. Carbocyanine postmortem tracing was successfully combined with different immunocytochemical protocols (Elberger and Honig 1990; Holmquist et al. 1992; Papadopoulos and Dori 1993). Interestingly, data on the validity of the combination of DiI and immunohistochemistry are absent.
This study was performed to optimize tracing distances by variation of technical parameters such as incubation time and incubation temperature. The differences in tracing distances among the carbocyanines DiI, DiA, and DiO and their use in both the central and peripheral nervous systems of humans and guinea pigs of different ages were studied. Examples of anterograde and retrograde postmortem tracing are presented, and the validity of the combination of DiI tracing and immunohistochemistry is evaluated.
Materials and Methods
Guinea pigs
Thirty male guinea pigs, including newborn, about 4-week juveniles, and adult animals were sacrificed with an overdose of sodium pentobarbital injected
Human Tissue
At 24–72 hr postmortem, four cadavers (62–87 years of age) of body donors were perfusion-fixed with 4% PF in 0.1 M PB (pH 7.4). Two donated cadavers (68 and 75 years of age) were obtained from dissection courses and had been perfusion-fixed with 4% carbol and 0.5% PF. To study the extent of carbocyanine travel, the spinal cord, both sciatic nerves, and both brachial plexuses were removed, partly as isolated structures and partly as tissue blocks. These tissues were cut transversely into pieces 50 mm long. This study complied with the tenets of the Declaration of Helsinki and included proper consent and approval.
Tracing to Analyze the Extent of Carbocyanine Travel
Tracer Application. Before tracer application, human and guinea pig tissues were stored in the fixative used for perfusion at 4C overnight. Under a surgical microscope, in humans and animals either carbocyanine DiI (1,1'-dioctadecyl-3,3,3'3'-tetramethylindocarbocyanine perchlorate), DiO (3,3'-dioctadecyloxacarbocyanine perchlorate), or DiA (4-(4-dihexadecylaminostyryl)-N-methylpyridinium iodide; all from Molecular Probes, Eugene, OR) was applied for tracing in a proximodistal direction to both sciatic nerves exactly below the pyriformis muscle and to one of the three brachial plexus cords of both sides. To trace ascending and descending spinal tracts, either of the tracers was applied to the lateral or posterior funiculus or dorsal columns of cervical spinal cords (cervical segments C6-C7). A fine microglass pipette was used to place carbocyanine crystals (0.5-3 mm, size depending on the fiber tract traced) precisely on transverse sectional planes of the above described neural tissues. The pipette was also used to insert the crystals slightly into the tissues. To reduce undesirable leakage of tracers, 20% agaragar (Merck; Darmstadt, Germany) in distilled water (40C) was applied to the tracing site with a little spatula. Agaragar was allowed to harden for a few minutes before specimens were incubated in 4% PF in 0.1 M PB (pH 7.4) at 37C or at 40C in the dark for different periods (3, 5, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 20, 25, 30, 35 or more weeks up to more than 1 year). To a few tissues the tracer was reapplied several times during the incubation period. To assess the influence of fixative concentration, right sciatic nerves of several animals were stored in 2% PF and those of the left side in 4% PF in the same buffer (0.1 M PB, pH 7.4).
Measurement of Tracing Distances. In case tissue blocks had been traced, nerve(s) or spinal cords, respectively, were prepared or removed from their bony canal. In guinea pigs, the corresponding branches of sciatic nerves and brachial plexuses were removed as well. Thereafter, all traced human and guinea pig tissues were prepared in the following manner. Tissues were measured macroscopically in the longitudinal direction of nerve fiber orientation, transferred to a buffered solution of 30% sucrose for 12 hr, and cut into small pieces, each 5 mm long (i.e., nerve fiber length). All tissue blocks were frozen in cold 2-methylbutane (Merck) on dry ice. The investigation started with the piece next to the application site. Several cryostat sections (10 m μ) were made of that part of each piece which was more distant from the application site. Sections were mounted on gelatin-coated slides and immediately viewed using an epifluorescence microscope (see below). When carbocyanine tracing was observed, this procedure to investigate 5-mm pieces went on until that piece was identified in which no carbocyanine was detected (in the sections more distant from application site). Complete serial sections of this particular piece were cut and the most distant point of tracing site was determined under the microscope. The exact tracing distance was calculated, knowing the number and thickness of sections. Parts of each tissue block were cut longitudinally to control specific tracing over the full length of all kinds of neural tissues used in this study.
Retrograde Tracing of Primary Afferent Neurons from Guina Pig EOM
Afferent nerve fibers from EOMs are known to travel with the oculomotor nerves. Within the cavernous sinus, EOM afferents join the ophthalmic nerve (N V/1) to the anteromedial part of the TG, where their first-order perikarya are located. To trace these afferents retrogradely, the skulls of six animals were opened, the brain removed, and DiI was applied to the anteromedial part of the TG. After different times of incubation (5, 10, 11, 12, 13, 15, 20 or more weeks), transverse and longitudinal cryostat sections of EOMs and the ipsilateral orbital parts of N V/1, the oculomotor (N III), trochlear (N IV) and abducens (N VI) nerves were cut at 10 μm.
Anterograde Tracing of Primary Afferent Neurons from Guinea Pig EOMs
The orbits of the guinea pigs were prepared using a surgical microscope. DiI was applied to the oculomotor nerves close to their entrance to EOMs to reveal the neuronal pathways of their afferent innervation. After different times of incubation (5, 10, 13, 15, 20 or more weeks) the TGs as well as the N III, N IV, N VI, and N V/1 were removed. After equilibration in a 20% followed by a 30% sucrose solution, the TGs were frozen in cold 2-methylbutane (Merck) on dry ice. Horizontal cryostat sections of the TG and the orbital and intracavernous parts of N III, N IV, N VI, and N V/1 were cut (10 μm). As far as possible, care was taken that the carbocyanine would not be allowed to diffuse away from the original tracing site. The brainstems were either cryo- or vibratome-sectioned longitudinally and vertically to reveal EOM second-order afferents. Their exact localization is still unknown.
Microscopic Analysis
All sections were immediately mounted on gelatin-coated slides, allowed to dry, and examined using an epifluorescence microscope (Zeiss Axioskop) equipped with a 50-W mercury lamp. For best viewing the red-orange DiI-fluorescence, we used a rhodamine filter set (Zeiss 15: BP 546, FT 580, LP 590). The green fluorescence of DiA and DiO was detected using an FITC filter set (Zeiss 9: BP 450-490, FT 510, LP 520). All sections were viewed with a × 40 Zeiss Plan-Neofluar (1.3 oil iris) and a × 100 Zeiss Plan-Neofluar (1.3 oil iris) oil immersion objective, partly without cover-slips. Several mounting media, i.e., PBS, glycerine-PBS (1:10), DePeX (Serva; Heidelberg, Germany), Entellan, Aquatex (both from Merck), Dako Fluorescence Mounting Medium (Dako; Glostrup, Denmark), Gel/Mount (Biomeda, Foster City, CA) were compared with each other on sequential sections. Microscopic analysis of covered and uncovered sequential sections revealed that all mounting media tried resulted in a more rapid fading of the carbocyanines or enhanced autofluorescence. Comparing water and oil immersion on uncovered sections, oil immersion was superior for detecting the faintest labeling. This is mainly explained by the higher aperture of oil immersion objectives.
Immunohistochemistry
To identify traced neurons in the TG, carbocyanine tracing was combined with CGRP-IR. After the procedure described above, sections were thoroughly examined for the presence of traced neurons and photodocumented. As a control group, animals were perfusion-fixed a few seconds postmortem and the TG was removed, cryosectioned (10 μm), and immediately prepared for CGRP-IR. All cryostat sections were processed in the same manner. Several primary antisera against CGRP were tested and the one providing the best results was finally used.
Sections were rinsed in PBS containing 0.3% Tween 20 (polyoxyethylensorbitanmonolaurate, ICI, Merck A 822184; Merck-Schuchardt, Munich, Germany) or, alternatively, 0.3% Triton X-100, three times (5 min each). A small part of the sections of immediately processed TGs were also prepared for immunohistochemistry without using detergents, without further fixation, or after fixation in Zamboni's fixative (2% PF, 15% saturated picric acid in 0.1 M PB, pH 7.0) on the slide. Tween 20 proved to be superior as a detergent and therefore was mainly used in this approach. Commercially available CGRP antiserum (Amersham International, code RPN. 1842; Poole, UK) was reconstituted with 100 μl of distilled water. The reconstituted antiserum was diluted to 1 ml with antiserum diluent solution (PBS, pH 7.2; 1:10). Working dilutions of 1:250 for the primary antiserum and 1:100 for the second antibody proved to be most useful. One hundred μl of the primary antiserum was applied with a cover plate system and sections were incubated for 3 days (21C), followed by rinsing three times in PBS for 5 min each. The second antibody (donkey anti-rabbit Ig, fluorescein-linked whole antibody; Amersham International, code 1034) was applied and sections were incubated for 2 h (21C). Sections were rinsed three times in PBS (5 min each). In controls, the primary antiserum was omitted and FITC-conjugated antiserum was used exclusively. Slides were mounted in glycerin-PBS (1:10).
Sections were immediately viewed using the abovedescribed microscope. True double-fluorescent perikarya exhibited a red fluorescence with the rhodamine filter and an orange-green fluorescence with the FITC filter. In few cases of a very high content of DiI, a breakthrough of the red fluorescence was observed with the FITC filter such that no clear differentiation from true double fluorescence was possible. In these cases, an interference red barrier filter which extinguished all red light, together with the FITC filter, enabled an unambiguous identification of true double-fluorescent neurons. Breakthrough of the green fluorescence with the rhodamine filter was never observed. With the help of interactive software (Kontron), DiI-positive cells and double-fluorescent (DiI-positive and CGRP-IR) cells were counted in the anteromedial part of the TG by serial reconstructions. A perikaryon was measured where its nucleus was most evident.
To assess the validity of CGRP-IR after postmortem tracing, systematic random samples of cells in previously DiItraced TGs were compared with samples in immunostained, not previously DiI-traced TG, thereby applying the rules of a physical dissector. The easily visible nucleus was used as a profile. Starting randomly, corresponding sample areas (not in the anteromedial part of TG) on every fifth serial TG section were studied. Profiles present in this area in the first and not in the following fifth section were counted (Q- in the stereological literature; for overview see Coggeshall and Lekan 1996). This strategy was also used to compare random samples in Tween 20-treated sections and Triton X-100-treated sections.
Statistical Methods
Distributions of variables of interest in the complete experiment and for various subgroups were described by means, standard deviations, minimal and maximal values and percentages. A split-plot factorial design was used. It has two between-block factors, kind of material and time of measurement, and one within-block factor, kind of nervous system. The outcome variable was the mean of tracing distance (in mm) between the left and the right side of neural tissues. The difference in CGRP-IR of the TG with and without previous carbocyanine tracing was assessed by a t-test. All p values are the results of two-tailed tests. A p value of less than 0.05 was considered to indicate statistical significance. The SAS statistical software system (SAS Institute; Cary, NC) was used for calculations.
Results
Tracing to Analyze the Extent of Carbocyanine Travel
Human and guinea pig neural tissues were carefully analyzed to determine the extent of carbocyanine travel. After different periods of incubation, traced axons and/or perikarya were observed in both human and guinea pig neural tissue. Under the conditions investigated, DiI proved to be the most effective tracer in fixed tissues (Figure 1A). Therefore, the results presented here were mainly achieved after the application of DiI.
Influence of Different Parameters on Tracing Distance. The only highly significant influence on the extent of carbocyanine travel was the incubation time, as shown in Figures 1A and 1B. Tracing distances continuously increased during the first 15 weeks. Incubation of 12–15 weeks proved to be optimal. After this time, DiI had traveled up to 29.2 ± 2.2 mm in guinea pigs (Figures 1B, 2, and 3) and 28.5 ± 2.3 mm in optimally fixed human tissues. No noticeable difference was observed between maximal distance for labeling cells and for labeling axons after tracing of cervical spinal cords of humans and animals. Incubation times longer than 15-20 weeks did not lead to a further increase in tracing distances. Thus, tracing did not show a linear time course but resembled a lateral diffusion curve of phospholipids in biological structures (Figures 1A and 1B).
The quality of fixation was also important. Formol-carbol fixation, only used in parts of the human tissues, resulted in shorter tracing distances, ranging from 15 to 20 mm. Storage of tissues at 40C during incubation did not lead to an increase of tracing distances as compared to 37C. No significant difference was observed between central and peripheral nervous system pathways [p>0.9; 95% confidence interval (CI) − 0.5 mm; 0.5 mm]. Only slight differences were observed between adult guinea pigs and human tissues (Figure 1B).
Retrograde Tracing: Primary Afferent Neurons from Guinea Pig EOM
After application of the tracer to the TG, DiI was observed in horizontal sections of the orbital N III, N IV and N VI nerves and the N V/1. DiI was visible in EOMs at the entrance site of the oculomotor nerves but was not observed within intramuscular axons.
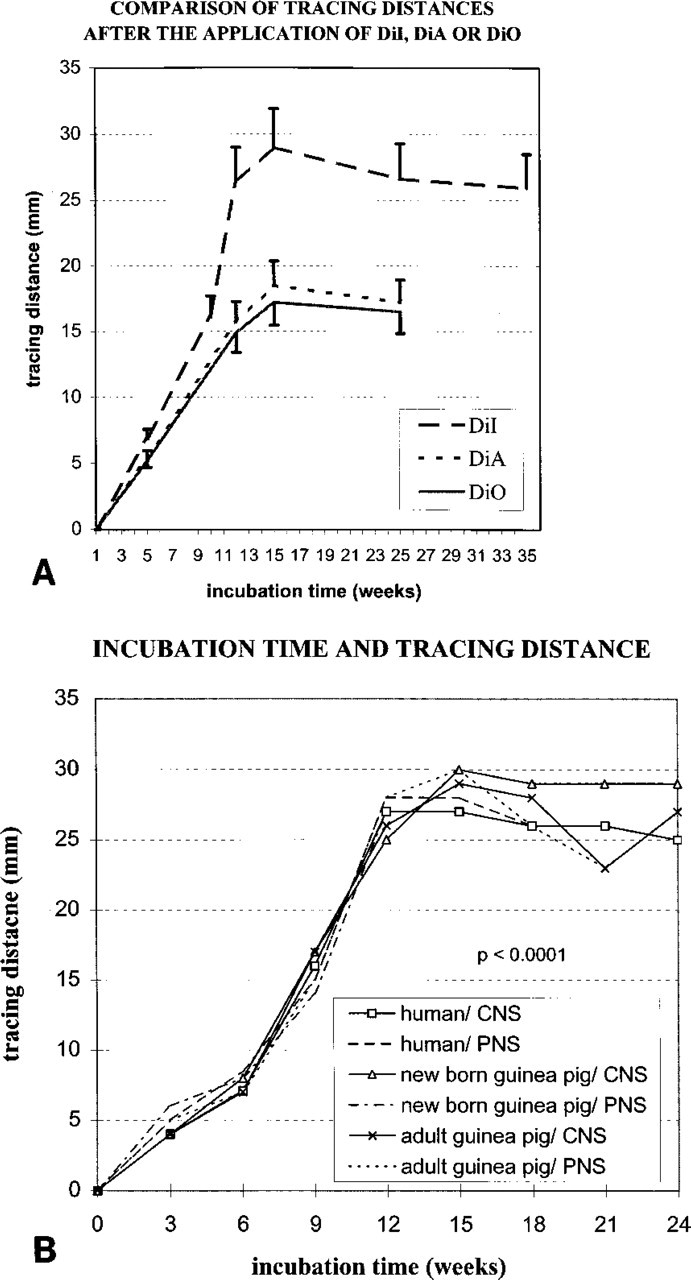
Mean ± SD of tracing distances (mm) in humans and guinea pigs after postmortem application of carbocyanines DiI, DiA, and DiO to the cervical spinal cords of guinea pigs (see Materials and Methods). Longest distances were observed after incubation of tissues for 12-15 weeks. Under the conditions investigated, DiI proved to be the most effective tracer in fixed tissues. Therefore, the investigation focused mainly on DiI.
Anterograde Tracing: Primary Afferent Neurons from Guinea Pig EOM
After application of tracers to orbital nerves innervating the EOMs, perikarya and axons were observed in the anteromedial part of the TG (Lukas et al. 1993). A DiI-positive perikaryon exhibited an intense diffuse or granular cytoplasmatic red fluorescence as observed using the rhodamine filter (Figures 4 and Figures 5). The morphology of traced somata, including their sizes and nuclei could be studied rather exactly. A total of 12-15 weeks of incubation proved to be optimal also to study this question, exhibiting the highest number of traced somata. At this time, tracing of either the N III or the N VI revealed about 270 perikarya of different sizes and tracing of the N IV about 100 somata. An original article on this item, demonstrating a somatotopic organization (Aigner et al. 1996) in the TG for each of the six EOMs, is in preparation. The tracing distances in these cases were 17-22 mm. Even after longer incubation times, some as long as 12 months, no DiI was detected in the brainstem in either cryostat or vibratome sections.
Immunohistochemistry
Anterogradely labeled neurons in the TG were identified by CGRP-IR. CGRP-IR cells were observed among DiI-traced and untraced neurons of the TG (Figures 6A-6D). Immunocytochemistry after 15 weeks of incubation revealed 26.2 % double-fluorescent perikarya containing the tracer carbocyanine DiI (red fluorescence, rhodamine filter) and CGRP (green fluorescence, FITC filter) of all DiI-positive neurons in the anteromedial part of the TG. CGRP-IR perikarya were confined to the groups of medium (30-50 μm) and small-sized cells (<30 μm) (Figure 7). More detailed results have been published elsewhere (Aigner et al. 1997). In a large series of experiments, no greater interindividual or right to left side differences were evident.
Validity of CGRP-IR After Postmortem Tracing
Random samples, consisting of about 150 cells, of the TGs of animals previously exposed to postmortem tracing were compared to analogous samples of animals exclusively processed for CGRP-IR. Under both conditions, about 45 cells (30%) of the samples of all TG cells exhibited CGRP-IR. It is important to note that only an insignificant difference of 1.2 percentage points in the percentage of CGRP-IR cells between animals traced and those not previously traced was evident [p = 0.73; 95% CI (-5.7 percentage points; 8.1 percentage points)]. Furthermore, those sections of the TG immediately processed for CGRP-IR that had not been exposed to Tween 20 or Triton X-100 in the buffer contained comparable proportions of CGRP-IR cells.
Problems of Specificity of Tracing
After 15–20 weeks of incubation, unspecific tracing occurred occasionally and increased with time, particularly in those cases in which the spinal cord had been removed from its bony canal before tracing. In these cases, a few peripherally located axons were traced for a certain distance discontinuous from the original carbocyanine application site. Thus, a few carbocyanine crystals dispersed in the incubation solution had reached fibers of the anterior or posterior horn of the spinal cord, and from that site these crystals had initiated another tracer diffusion process. This undesirable spread of tracer resulted in even shorter continuous tracing distances. During longer incubation times, fading of DiI might occur as well. Apart from this, no evidence for transaxonal travel of carbocyanines was observed. In general, after 6, 8, 12, or more months of incubation no increase in DiI tracing distances was observed. Moreover, storage of sciatic nerves at a lower concentration of buffered formaldehyde (2%) compared to 4% during incubation had no noticeable influence on the extent of carbocyanine travel.
In some cases of anterograde EOM labeling, undesired tracing due to leakage occurred. This was evidenced by many more clearly traced perikarya in the TG and, without exception, evidence of carbocyanines in sections of at least one of those orbital nerves not originally traced. This was more frequently observed after a more extensive preparation in the orbit. Obviously, several sheets of orbital structures had been injured. Best results were achieved after tracing of oculomotor nerves close to their entrance to EOMs without greater manipulations. Even in cases of more extensive preparation, specific tracing was achieved in 80-85%.
In general, the use of Tween 20 in the buffer proved to be most useful for the combination of postmortem tracing and immnunohistochemistry. No noticeable fading of the DiI was observed. Other detergents, such as Triton X-100, or postfixation with Zamboni's fixative on the slide proved to be less helpful. In particular, the proportion of double labeled cells was about 10% with Triton X-100 and 26% with Tween 20.
Discussion
Postmortem neuronal tracing with carbocyanines has proved to be a very useful tool for exact mapping studies. However, in any tracing experiment not all axons will transport the tracer. Maximal cell counts were nearer the total of afferent fibers in oculomotor nerves. Under best conditions, tracing distances were improved by up to 30 mm in the present study, allowing many questions about neuronal pathways to be answered. Carbocyanines proved to produce rather consistent results. Exact correlations between incubation time and tracing distance provided in the present study might be useful for the design of postmortem tracing studies in mammals. A protocol for the optimal method is given at the end of this section.

Longitudinal section of a guinea pig spinal cord: distant point of DiI travel that had reached 29 mm after 12 weeks of incubation at 37C in the dark. Bar = 650 μm.
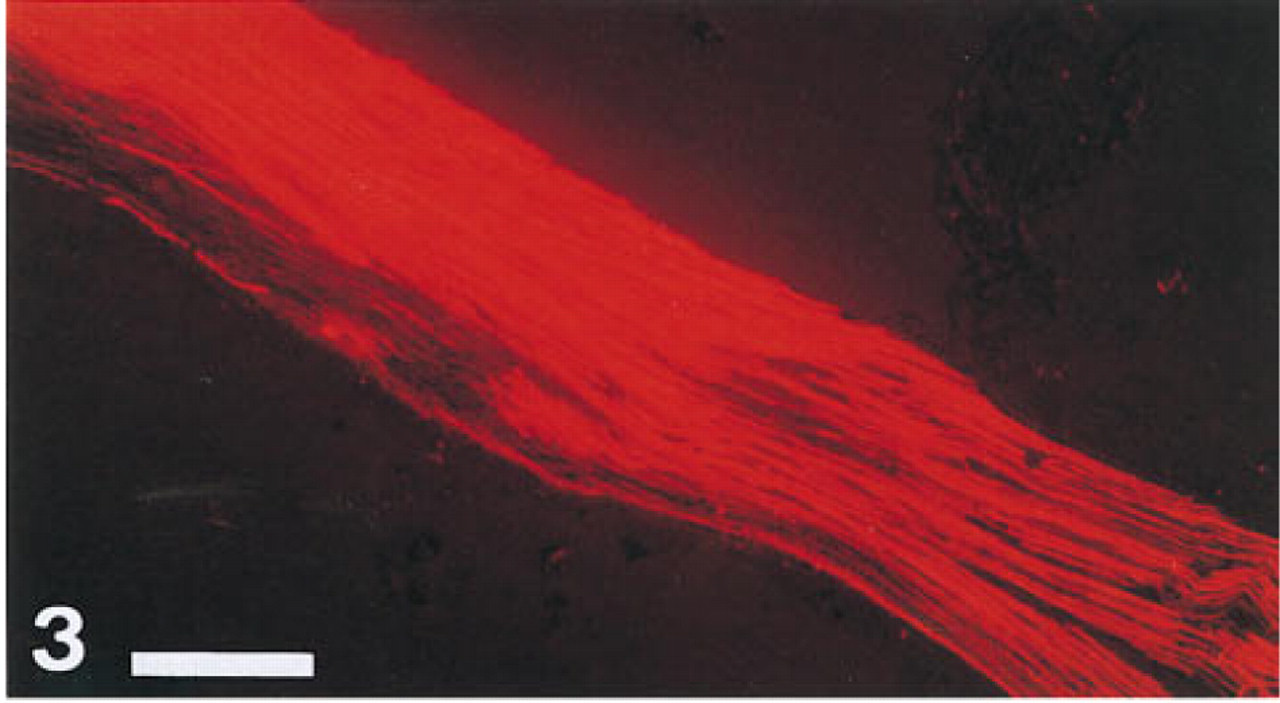
Longitudinal section of an adult guinea pig sciatic nerve, proximal part traced with DiI. Total extent of DiI travel was 22 mm after 10 weeks of incubation. Bar = 650 μm.
Several observations of the present study are in accordance with those described by several authors (Godement et al. 1987; Honig and Hume 1986,1989). Incubation of traced tissues at 37C enhanced the speed of carbocyanine travel, which also was previously reported by Friedmann et al. (1991) and Heredia et al. (1991). Earlier postmortem neuronal tracing studies did not focus on tracing distances and no detailed data on this question have thus far been published. One might estimate that postmortem tracing distances reported previously have been shorter than 10-15 mm (Friedmann et al. 1991; Wassef et al. 1992; Magoul et al. 1994; Yanez and Anadon 1994; Byers and Sugaya 1995). Therefore, within certain limits, tracing distances were improved in the present study. Precise comparison is difficult because relatively few postmortem DiI studies have been carried out in mammalian tissue. Moreover, incubation times were adopted for each particular question in each study as reported by several authors (e.g., incubation times of 5 months and more have been indicated which in this study proved to be unnecessarily long). Because of restricted knowledge about carbocyanines as specific tracers, they are still under debate. In particular, DiI was reported to travel transaxonally or transcellularly (Godement et al. 1987; Honig and Hume 1989; Fritzsch and Wilm 1990). Although several recent studies did not focus on the questions of specificity of carbocyanines, they revealed the specificity of DiI tracing by the simultaneous use of DiI and other tracers (Godement et al. 1987; Easter and Taylor 1989; Honig and Hume 1989; Wizenmann and Thanos 1990; Yanez and Anadon 1994). On the basis of their long-chain lipophilic structure, carbocyanines travel along the plane of plasma membranes by diffusion, enabling tracing in anterograde and retrograde directions. Godement et al. (1987) emphasized that carbocyanine “staining would progress slower and slower as it goes farther from the point of tracing.” This typical phenomenon of a lateral diffusion process in membranes was presented for DiI in Figure 1. In the present study, the diffusion coefficient of DiI was found to be 2.5 × 10-7cm2sec-1 which is in accordance with previously reported data obtained by extrapolation (Godement et al. 1987; Honig and Hume 1989). One might expect that the concentration at the application site and the time of incubation will be the most relevant factors for the extent of carbocyanine travel. In the present study, only the incubation time had significant influence. Repeated reapplication at the original application site did not result in longer distances. Adequate concentrations of tracers or, presumably, even saturation with the tracer had been achieved by the first application.
The maximal distances presented reflect easily reproducible results. The potential diffusion limit of carbocyanines observed in the present study raises several questions. The detection of carbocyanines is a function of physical factors such as the choice of illumination, filters, and objectives. Because the FITC filter set and the mercury arc lamp of the microscope used are not optimally suited for the detection of DiO, we cannot exclude the posibility that DiO traveled farther than was actually detected in this study. This might, to a lesser extent, be true also for DiA. It is important to note that the filter set used for DiI is well suited. The only caveat for DiI remains that cryostat sections may be less stable than vibratome sections for detecting the weakest label. Experiences with vibratome sections after DiI tracing in this laboratory (Schickinger 1993), however, do not indicate that farther distances could be attained, whereas tissue quality is of course by far better for documentation. In addition, the tracing limits observed explain why no carbocyanine was detected in the brainstem after DiI application to oculomotor nerves.
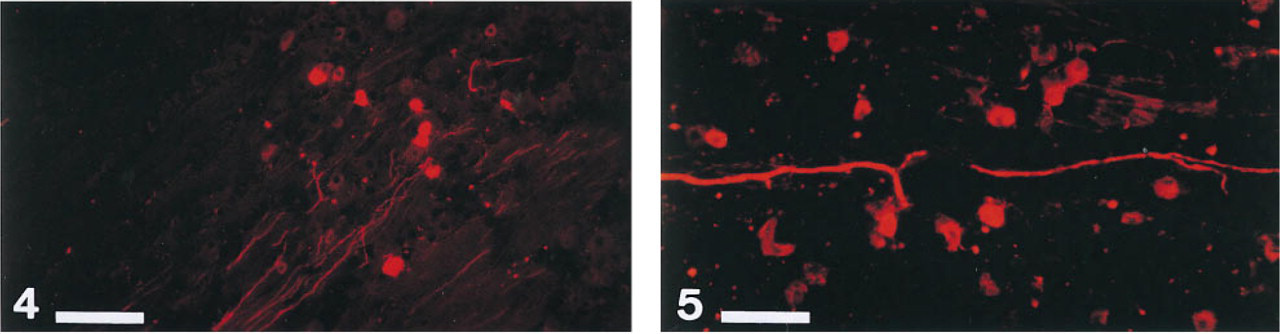
Primary afferent fibers and perikarya from extraocular muscles in horizontal sections of the guinea pig trigeminal ganglion revealed by DiI application to the ipsilateral
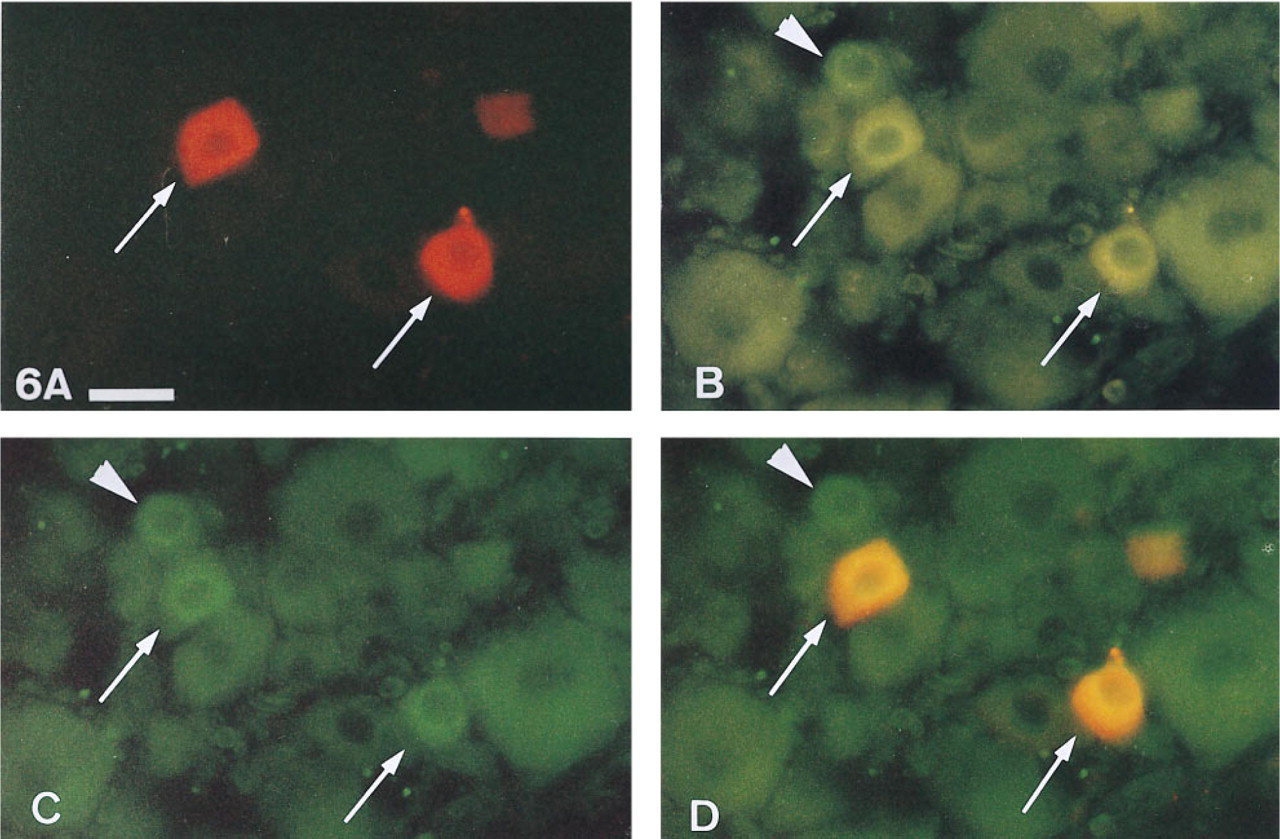
Calcitonin gene-related peptide-immunoreactivity (CGRP-IR) of primary afferent neurons from guinea pig extraocular muscle identified by DiI tracing. Same horizontal section of the trigeminal ganglion viewed with different filter positions enables an unambiguous identification of true double-fluorescent cells (DiI and CGRP-IR).
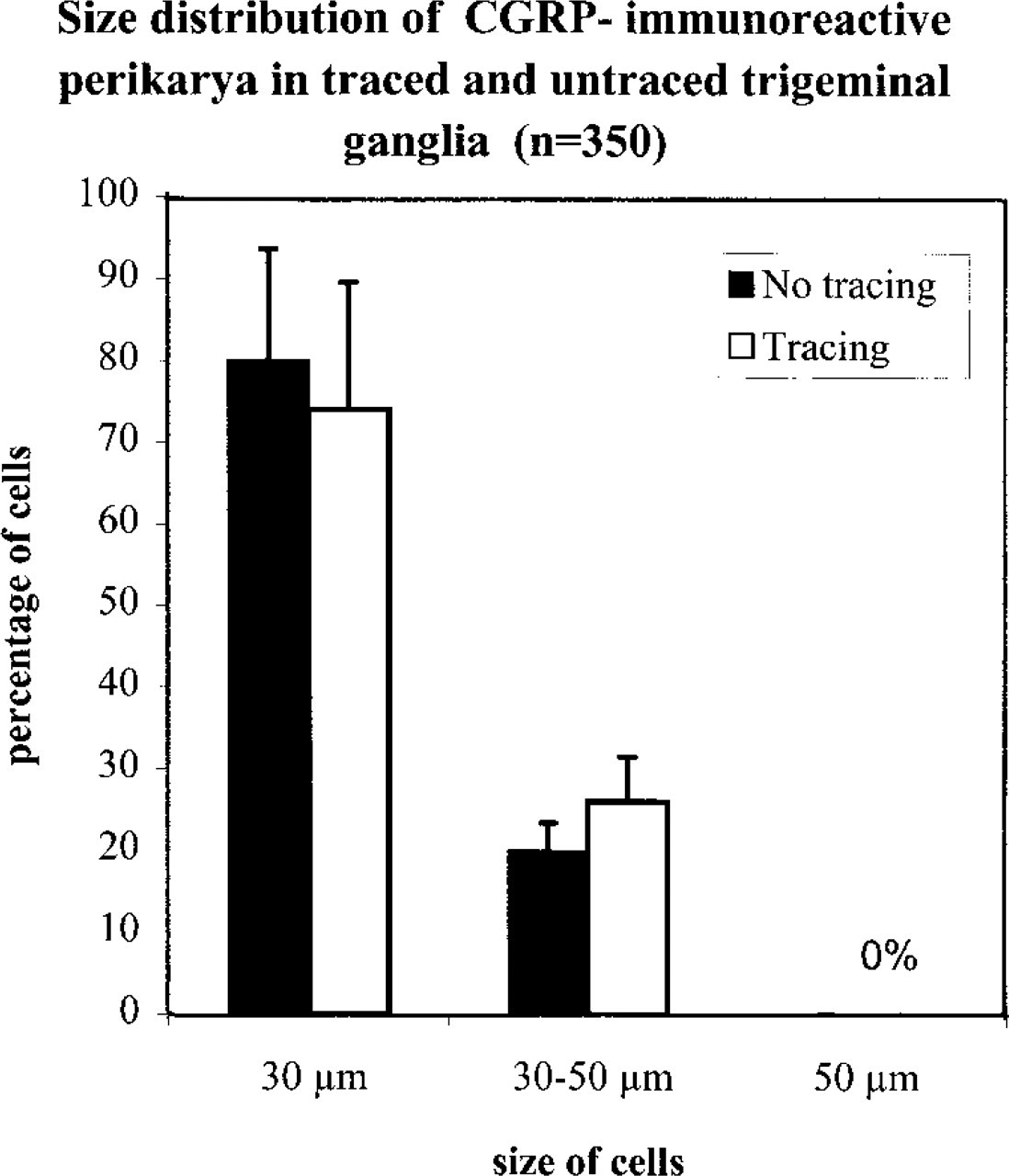
Size distribution of CGRP-immunoreactive perikarya in random samples of trigeminal ganglion cells of guinea pigs. Columns indicate means and bars SD of the percentages of each group of cells. No CGRP-immunoreactive perikarya larger than 50 m were found. No main differences in size distribution were observed between trigeminal ganglia obtained from animals traced and those obtained from animals not previously exposed to postmortem tracing.
Overfixation by prolonged storage of tissues in 4% buffered paraformaldehyde might also contribute to ultimately limiting diffusion (Honig 1993). In the present study we did not observe any difference between tissues stored in 2% or 4% of buffered formaldehyde. Furthermore, an additional immersion-fixation for several days after perfusion-fixation before tracer application compared favorably to tracing shortly after perfusion-fixation. In preliminary investigations we found that perfusion fixation of animals with 2% buffered formaldehyde was less useful for postmortem tracing. Parts of the human tissues were obtained from dissection course cadavers and had been fixed with carbol and paraformaldehyde. Analogously, markedly smaller tracing distances were observed in this material. In conclusion, it appears unlikely that overfixation might have greatly influenced tracing distances. Short postmortem times before fixation proved to be important. Even in optimally fixed human tissues, tracing distances were slightly shorter than in guinea pigs. This might be explained by the fact that immediate postmortem fixation was achieved only in animals. Glutaraldehyde in the fixative may reduce the speed of carbocyanine travel (Schlessinger et al. 1977; Bartheld et al. 1990). Moreover, even as little as 0.5% of glutaraldehyde may enhance autofluo-rescence (Elberger and Honig 1990) and may affect the immunocytochemical results (Holmquist et al. 1992). Glutaraldehyde has proved useful for photo-conversion of DiI to DAB for subsequent ultrastructural investigation (Bartheld et al. 1990).
In our laboratory, postmortem carbocyanine tracing was successfully applied for the confirmation of a vestibulocochlear anastomosis (Schickinger 1993), originally described by Burian et al. (1989) using HRP, and for questions on the proprioceptive innervation of EOMs (Aigner et al. 1997). Quantitative results after DiI application to oculomotor nerves were well in accordance with previous tracer studies in other species (for an overview see Aigner et al. 1997), indicating that DiI had not traveled transcellularly. Furthermore these results demonstrate that cryosectioning had not led to a greater diffusion of DiI from the original site of tracing. Convergence of afferents from two different EOMs to one TG cell was determined using DiA and DiI in a postmortem doubletracing experiment in guinea pigs (Denk et al. 1995).
Several procedures for the combination of postmortem neuronal tracing with immunohistochemistry have been published (Elberger and Honig 1990; Holmquist et al. 1992; Papadopoulos and Dori 1993). Immunofluorescent labeling combined with carbocyanine tracing provides the advantage of rapid and precise localization of these two labels only by switching between filter positions. In the present study, the results of the combination of carbocyanine postmortem neuronal tracing with immunohistochemistry were compared to the results of immunocytochemistry without previous postmortem tracing. The most important question was whether preservation of tissues in fixative during several weeks might influence the results of immunocyto-chemistry. Long incubation times in a fixative might reduce the antigenicity of several neuronal protein constituents. Moreover, the penetration of antisera in long-term fixative-exposed tissues is reduced, necessitating the use of detergents to enhance antibody penetration. Detergents such as Triton X-100 saponin, or dimethylsulfoxide have been reported to cause fading of the DiI labeling (Elberger and Honig 1990; Holmquist et al. 1992; Papadopoulos and Dori 1993). Therefore, only a sequential staining and visualization of DiI in one section and of immunocytochemistry in the following section were possible. In the present study, the penetration of the primary antiserum was analogously improved with Tween 20, but almost no fading of DiI was observed, allowing simultaneous viewing of DiI and CGRP-IR. It remains unclear whether Tween 20 would be equally useful for immunocytochemical detection of other proteins or peptides whose epitopes might be affected by prolonged fixation. Freezing of the tissue should also enhance antibody penetration (Holmquist et al. 1992). Vibratome sections are therefore not necessarily the better choice. These modifications of widely used protocols of fluorescence immunocytochemistry contributed to more valid results in these double labeling experiments, and even the quantitative results were in accordance with the literature (for an overview see Aigner et al. 1997).
The major advantage of postmortem tracing is that it enables experiments in human tissues, especially in embryonic tissues. Moreover, several questions can be solved in the same animal using different parts of the nervous system, as demonstrated in the present study. Thus, this technique contributes significantly to reduction of the number of animals required for scientific research and to reduction of harm to animals. The most important disadvantages are limited tracing distances and long incubation times. Research should focus on possible improvements of tracing distances when carbocyanines are used as postmortem tracers. Moreover, there might be other or similar substances that could enable longer tracing distances. Already, however, carbocyanine postmortem tracers are an indispensable tool for study of neuronal pathways.
This protocol has proved to be the optimal procedure for carbocyanine postmortem neuronal tracing:
Perfusion-fixation with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) as soon as possible postmortem. Postfix tissues in the same fixative overnight (or up to 2 days) at 4-8C.
Prepare the application site carefully with a minimum of tissue deterioration to prevent undesired tracer uptake by surrounding membranes or nerves.
Apply carbocyanine crystals to the exposed neural tissue or to transverse sectional planes of nerves after cutting, and cover application site with agaragar to reduce leakage of tracer.
Incubate tissues in the fixative used for perfusionfixation at 37C in the dark, incubation time depending on the tracing distance desired between 2 and 15 weeks (Figures 1A and 1B).
Make vibratome sections or, after cryoprotection and freezing tissues in cold 2-methylbutane, make cryostat sections and thaw-mount these on gelatincoated slides.
Immediate photodocumentation (caveat: fading) using a fluorescence microscope equipped with the filter sets aproppriate for the carbocyanine used. [DiI, rhodamine filter (e.g. Zeiss 15 or others)]. Particularly for DiO and DiA, the filter sets offered by Molecular Probes (Eugene, OR) are preferable. For documentation, 400 ASA films and exposure times ranging from 1 to 80 sec proved to be useful. A high-quality digital analyzing and printing system provides most rapid photoprints, which helps to overcome the problems of fading. Evaluation of sections with high-power oil immersion (oil 518C, aperture: 1.518; Zeiss, Oberkochen, Germany) objectives of high numeric aperture (1.3 or more) without prior coverslipping gave best results for detecting the faintest labeling.
For simultaneous visualization of carbocyanine tracing and fluorescence immunocytochemistry, the following protocol is recommeded (a basic knowledge of immunohistochemistry is required. Readers are referred to standard handbooks of immunohistochemistry):
After the procedure described above and careful photodocumentation, rinse sections in PBS containing 0.3% Tween 20 (polyoxyethylensorbitanmonolaurate, ICI, Merck A 822184, Schuchardt) three times (5 min each).
Incubate with primary antiserum for 48-72 hr on the slide (in this approach antiserum against CGRP applied with a cover-plate system).
Rinse three times in PBS, 5 min each.
Incubate sections with secondary antibody conjugated with a fluorophore that can be differentiated from the carbocyanine used by adequate filter sets (e.g., DiI tracing and FITC- or AMCA-conjugated second antibody).
Rinse with PBS, 5 min.
Mount sections in glycerin-PBS.
Immediate photodocumentation (400 ASA) with different filter positions: one with the filter position suitable for the carbocyanine, one with the filter position corresponding to the fluorophore of the antibody, and one double exposure switching between these two filter sets. Interference red barrier filters might be needed for unambiguous identification of true double-fluorescent cells when DiI is used as tracer (see also point 9 above).
Footnotes
Acknowledgements
We wish to thank Mag. I. Theyer for helpful comments and C. Krivanek and R. Meyer for valuable technical help.
