Abstract
I
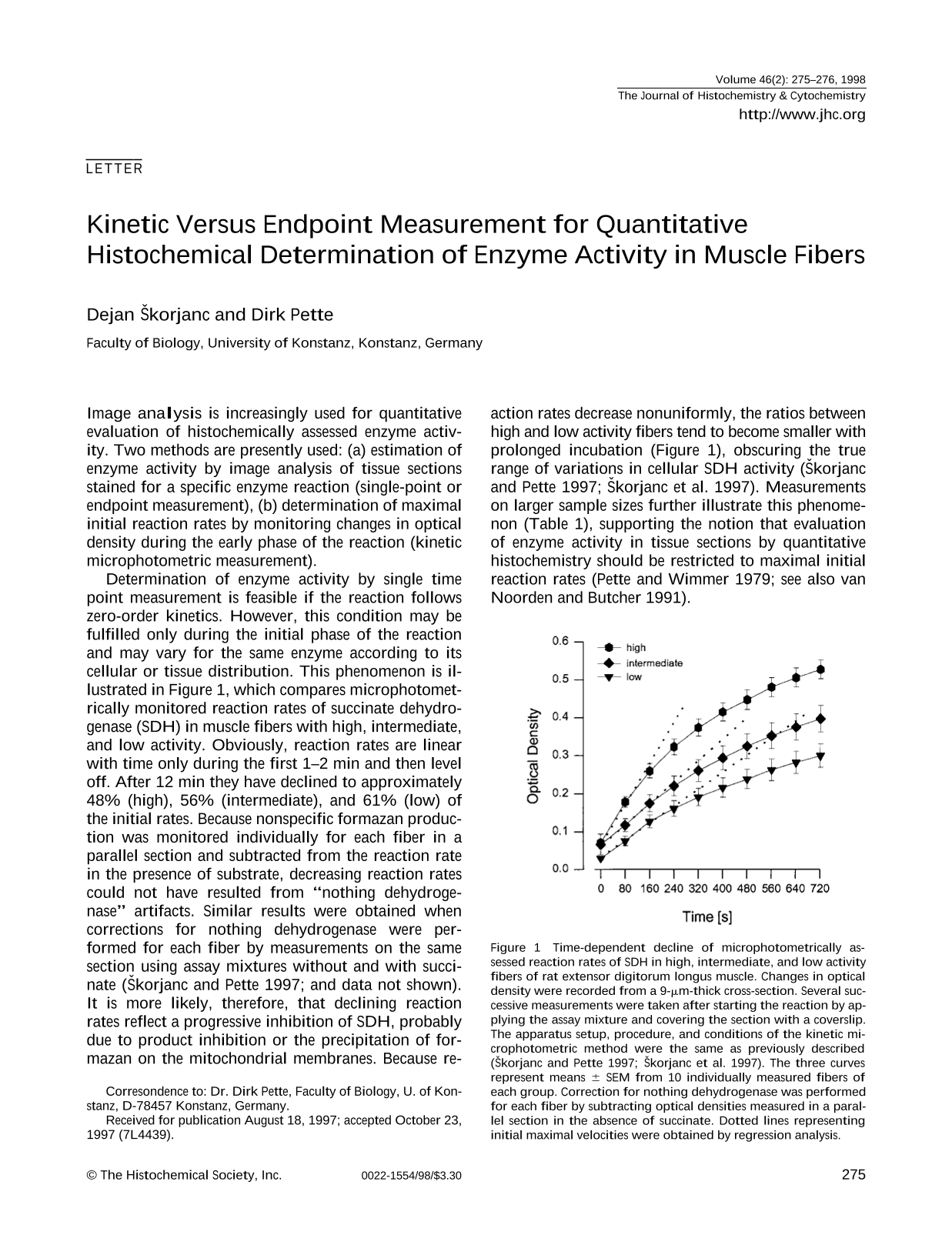
Determination of enzyme activity by single time point measurement is feasible if the reaction follows zero-order kinetics. However, this condition may be fulfilled only during the initial phase of the reaction and may vary for the same enzyme according to its cellular or tissue distribution. This phenomenon is illustrated in Figure 1, which compares microphotometrically monitored reaction rates of succinate dehydrogenase (SDH) in muscle fibers with high, intermediate, and low activity. Obviously, reaction rates are linear with time only during the first 1–2 min and then level off. After 12 min they have declined to approximately 48% (high), 56% (intermediate), and 61% (low) of the initial rates. Because nonspecific formazan production was monitored individually for each fiber in a parallel section and subtracted from the reaction rate in the presence of substrate, decreasing reaction rates could not have resulted from “nothing dehydroge-nase” artifacts. Similar results were obtained when corrections for nothing dehydrogenase were performed for each fiber by measurements on the same section using assay mixtures without and with succi-nate (Škorjanc and Pette 1997; and data not shown). It is more likely, therefore, that declining reaction rates reflect a progressive inhibition of SDH, probably due to product inhibition or the precipitation of formazan on the mitochondrial membranes. Because reaction rates decrease nonuniformly, the ratios between high and low activity fibers tend to become smaller with prolonged incubation (Figure 1), obscuring the true range of variations in cellular SDH activity (Škorjanc and Pette 1997; Škorjanc et al. 1997). Measurements on larger sample sizes further illustrate this phenomenon (Table 1), supporting the notion that evaluation of enzyme activity in tissue sections by quantitative histochemistry should be restricted to maximal initial reaction rates (Pette and Wimmer 1979; see also van Noorden and Butcher 1991).
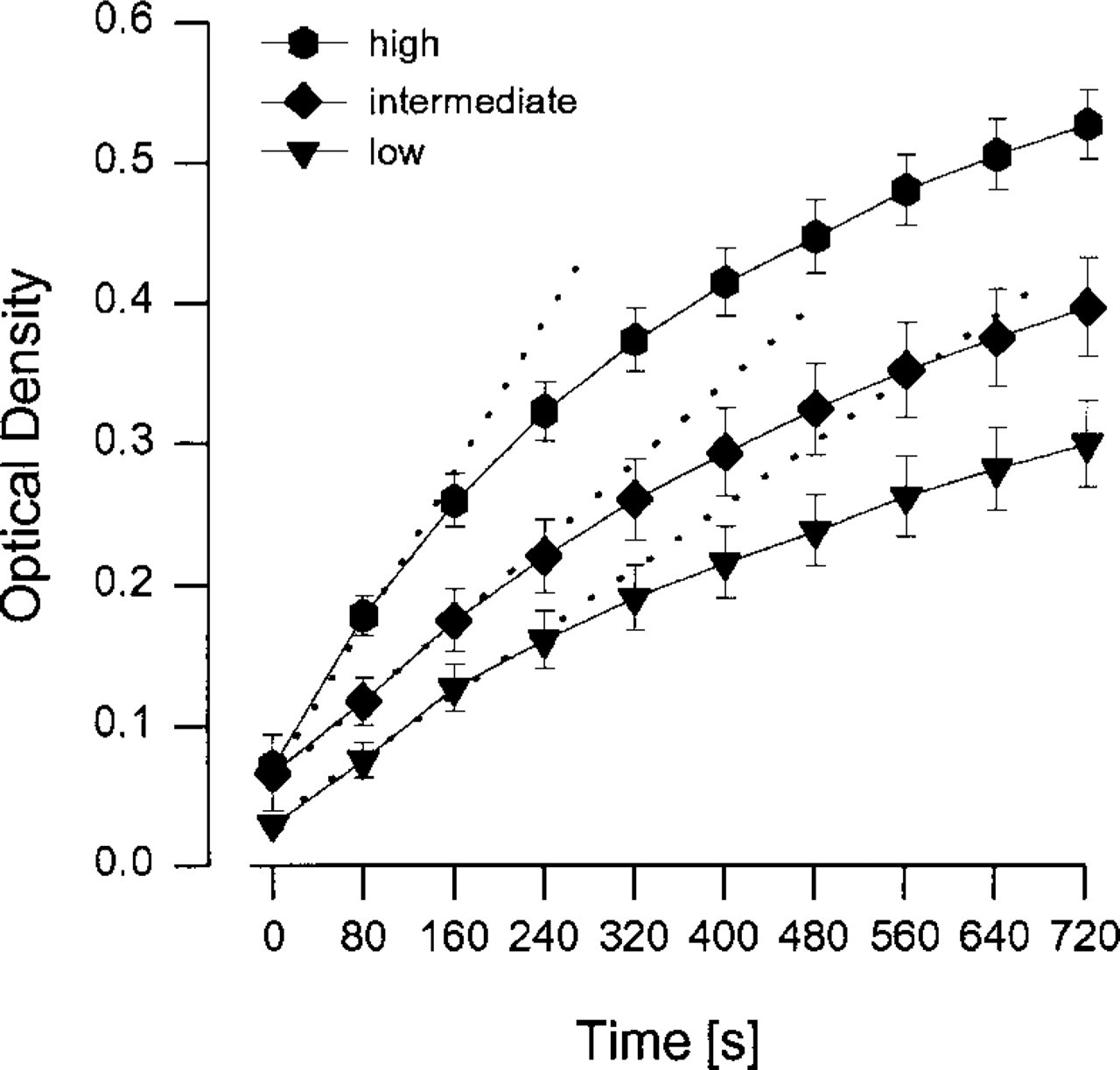
Time-dependent decline of microphotometrically assessed reaction rates of SDH in high, intermediate, and low activity fibers of rat extensor digitorum longus muscle. Changes in optical density were recorded from a 9-μm-thick cross-section. Several successive measurements were taken after starting the reaction by applying the assay mixture and covering the section with a coverslip. The apparatus setup, procedure, and conditions of the kinetic microphotometric method were the same as previously described (Škorjanc and Pette 1997; Škorjanc et al. 1997). The three curves represent means ± SEM from 10 individually measured fibers of each group. Correction for nothing dehydrogenase was performed for each fiber by subtracting optical densities measured in a parallel section in the absence of succinate. Dotted lines representing initial maximal velocities were obtained by regression analysis.
Effect of incubation time on SDH activities in high, intermediate, and low activity fibers from rat extensor digitorum longus muscle a
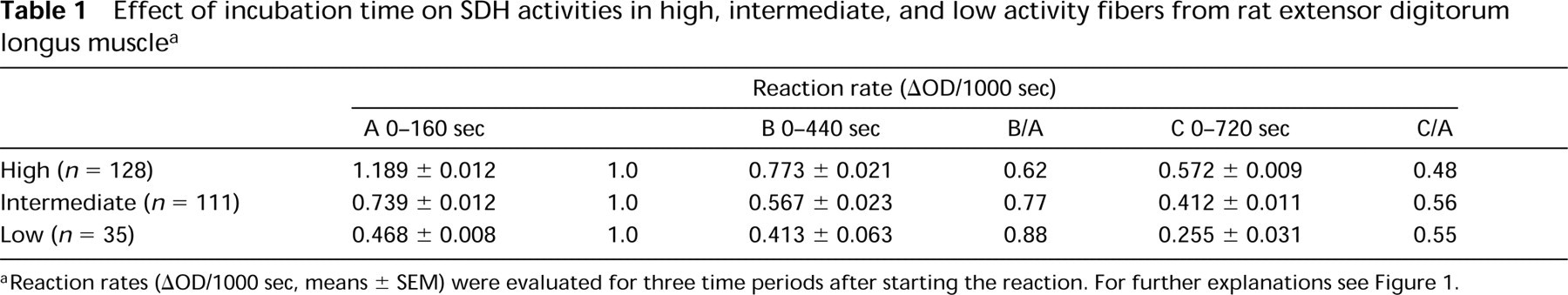
Reaction rates (AOD/1000 sec, means ± SEM) were evaluated for three time periods after starting the reaction. For further explanations see Figure 1.
The present results and previous findings from our laboratory (Pette and Wimmer 1979; Pette 1981; Škorjanc and Pette 1997; Škorjanc et al. 1997) and others (Old and Johnson 1989; Nakae and Stoward 1995) disagree with reports that the histochemically assessed SDH reaction exhibits linear rates of up to 10 min and that determination of SDH activity in muscle fibers can therefore be performed by endpoint measurements using 6–10-min incubations (Martin et al. 1986; Sieck et al. 1986; Blanco et al. 1988; Edgerton et al. 1995; Michel et al. 1996). The reason for this discrepancy is not clear, but our measurements on large sample sizes in this and previous studies (Škorjanc and Pette 1997; Škorjanc et al. 1997) clearly demonstrate that SDH reaction rates are linear with time only during a short initial phase of the reaction. Moreover, we show that the decline in reaction rates is nonuniform in fibers of high and low activity, which emphasizes the necessity of measuring initial maximal velocities.
In summary, evaluations of endpoint measurements may yield reliable results only when incubation periods are restricted to that phase of the reaction during which enzyme activity and product formation display a linear relationship. This condition must be individually validated for each measurement. This is easily ascertained by kinetic microphotometry. Because image analysis greatly facilitates such measurements, we see no reason not to apply this method to studies in quantitative enzyme histochemistry.
Footnotes
Acknowledgements
Supported by Deutsche Forschungsgemeinschaft Pe 62/ 18–2 and Pe 62/25-1.
