Abstract
We characterized the distribution of CD40 and CD40 ligand (CD40-L) in the adult and developing murine thymus. Before birth, CD40 was almost exclusively localized to scattered foci of medullary cells. By birth there was a dramatic upregulation of CD40 expression by cortical epithelial cells, which was accompanied by a consolidation of medullary epithelial foci. CD40-L+ thymocytes displayed a medullary location. Analysis of mice deficient in CD40-L expression indicated that CD40-L/CD40 interactions were not required for development of the medullary compartment. Overexpression of CD40-L targeted to thymocytes altered thymic architecture, as reflected by a dramatic loss of cortical epithelial cells, expansion of the medullary compartment, and extensive infiltration of the capsule with a mixture of CD3+ cells, B-cells, and macrophages/dendritic cells. Reconstitution of lethally irradiated normal mice with
Keywords
The role of CD40 interactions in T-cell development is less clear. The absence of significant alterations of T-cell development in CD40null or CD40-Lnull mice suggests that CD40/CD40-L interactions are not absolutely required for T-cell development to occur (Kawabe et al., 1994; Xu et al., 1994). However, disruption of thymic negative selection in mice chronically treated with an anti-CD40-L MAb or in mice deficient in CD40-L expression suggests that CD40/CD40-L interactions may play a role in shaping the TCR repertoire (Foy et al., 1995). Overexpression of CD40-L perturbs normal thymocyte development and results in a lethal wasting disease with mononuclear infiltrates accumulating in multiple organs (Clegg CH et al., manuscript in preparation). Ligation of CD40 expressed by cultured human thymic epithelial cells affects cytokine production by these cells (Galy and Spits, 1992) and, based on the impact of CD40 ligation on B-cell differentiation, might be expected to affect the differentiation of thymic epithelium as well.
In this study we characterized the expression of CD40 and CD40-L within the developing and adult normal murine thymus and assessed the effects of either the lack of CD40-L expression or elevated levels of CD40-L expression on the development and organization of the thymic environment. Although thymic tissue from mice lacking CD40-L displayed reduced expression of Class II MHC antigens and accessory molecules within the medullary compartment, the over all thymic architecture and phenotype was essentially normal. In contrast, thymic tissue from mice overexpressing CD40-L was profoundly affected, with B-cell infiltration/expansion, disorganization and loss of cortical epithelium, and dramatic increases of the medullary compartment.
Materials and Methods
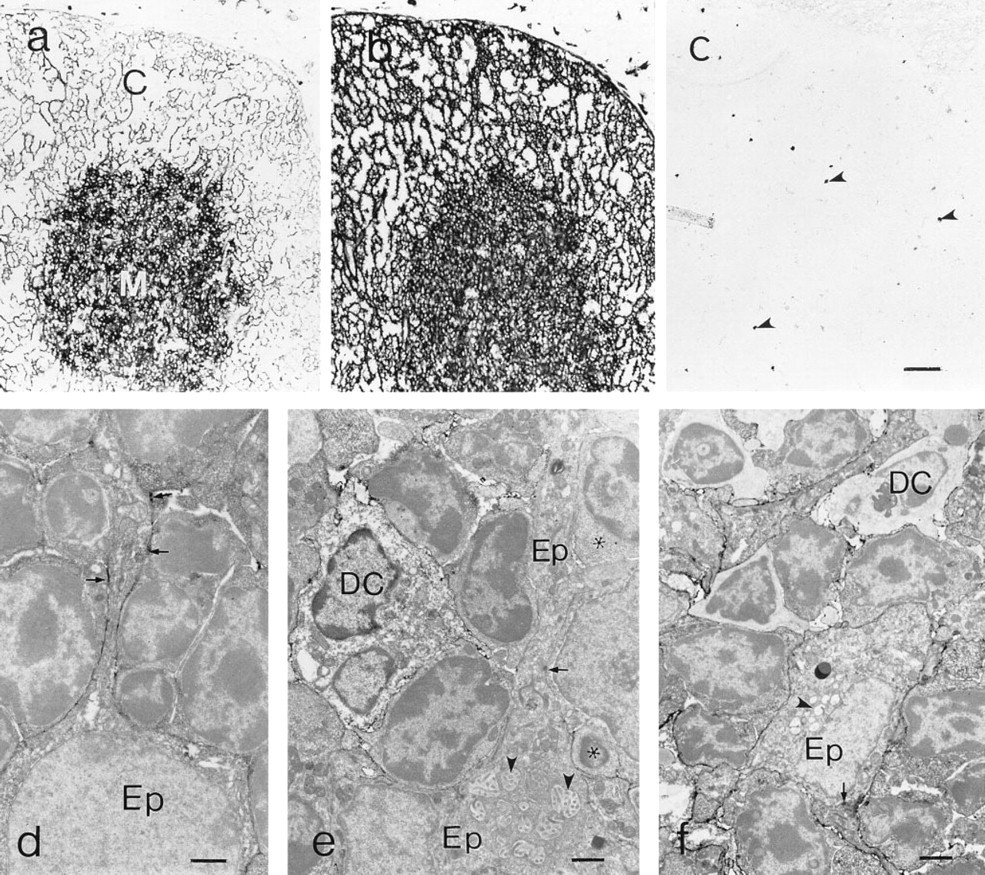
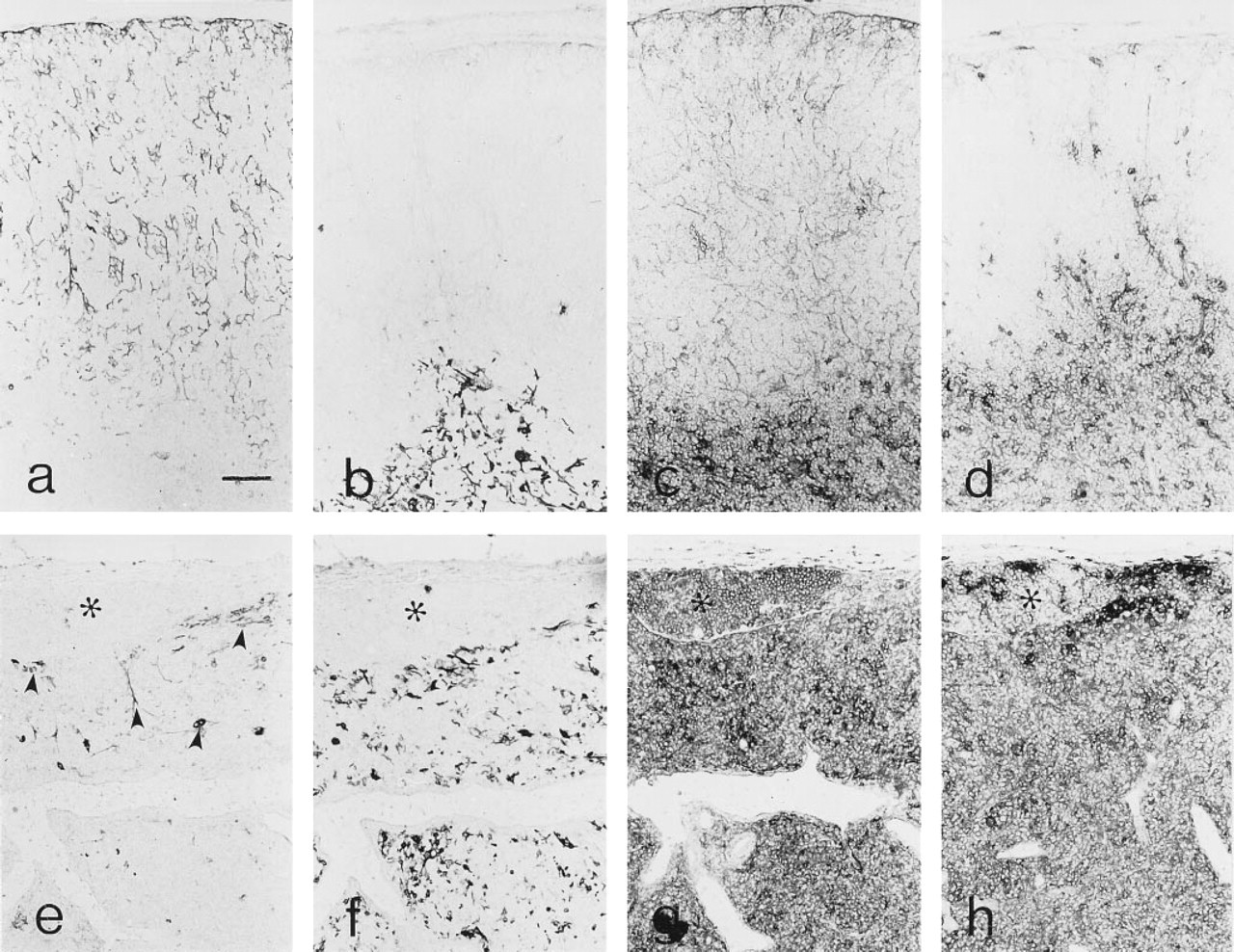
Thymic expression of CD40. (a) CD40 displays a delicate reticular pattern in the cortex (C) and a more confluent pattern in the medulla (M). (b) Class II MHC expression is qualitatively similar to that of CD40. (c) Labeling with an irrelevant rat MAb. Endogenous peroxidase is indicated by arrowheads; no blocking of endogenous peroxidase activity. (d) Immunoelectron microscopy of CD40 expression by cortical epithelial cells. Note the patchy labeling along the epithelial cell process (arrowheads). (e) Immunoelectron microscopy of a dendritic cell (DC) and epithelial cell (EP) in the medullary area. Epithelial characterization is based on the presence of distinctive cytoplasmic vacuoles with microvilli (arrowhead) and desmosomes (arrow). (f) Immunoelectron microscopy of a dendritic cell (DC) and epithelial cell (EP) in the medullary area. Epithelial characterization is based on the presence of distinctive cytoplasmic vacuoles with microvilli (arrowhead) and desmosomes (arrow). Bars:
Results
Thymic CD40 Expression
Ultrastructural immunohistochemistry confirmed that the CD40 expression in the thymic cortex was associated with epithelial cells (Figure 1d). For the most part, there was a delicate linear distribution of the CD40 reaction product along the epithelial cell processes, which was consistent with the relatively light labeling of cortical CD40 at the light microscopic level. Interestingly, there were small segments of the epithelial cell processes that bore larger amounts of the reaction product, resulting in a patchy labeling pattern. In the medullary compartment, dendritic cells and epithelial cells, identified by ultrastructural features, were labeled with anti-CD40 MAb (Figures 1e and 1f). In contrast to dendritic cells, which appeared to uniformly label well with anti-CD40 MAb, some medullary epithelial cells displayed very little reaction product (Figure 1e), whereas the labeling of others approached that displayed by dendritic cells (Figure 1f).
Thymocyte Expression of CD40-L
Previous flow cytometric analyses of normal young adult Balb/c thymocytes demonstrated low levels of CD40-L expressed by a small population of thymocytes, which increased following exposure to ConA or anti-CD3 MAbs (Nelson et al., 1996). Three-color flow cytometry was employed to examine thymocyte expression of CD40-L more closely. As shown in Figure 2a, labeling of normal thymocytes with MR-1 MAb resulted in a shoulder of dully stained cells. Using a channel number marker established with an irrelevant digoxigenin-modified primary MAb, about 9% of the thymocytes were MR1+, although the frequency of thymocytes expressing lower levels of MR-1 would be higher. In three-color analyses of CD4, CD8, and CD40-L expression, most of the CD40-L expression was associated with CD4+8- (∼60%) and CD4+8+ (∼25%) thymocyte populations (Figure 2b). Within the CD4+8+ thymocyte subset, CD40-L was preferentially expressed by cells expressing higher levels of CD4 and CD8 (Figure 2b).
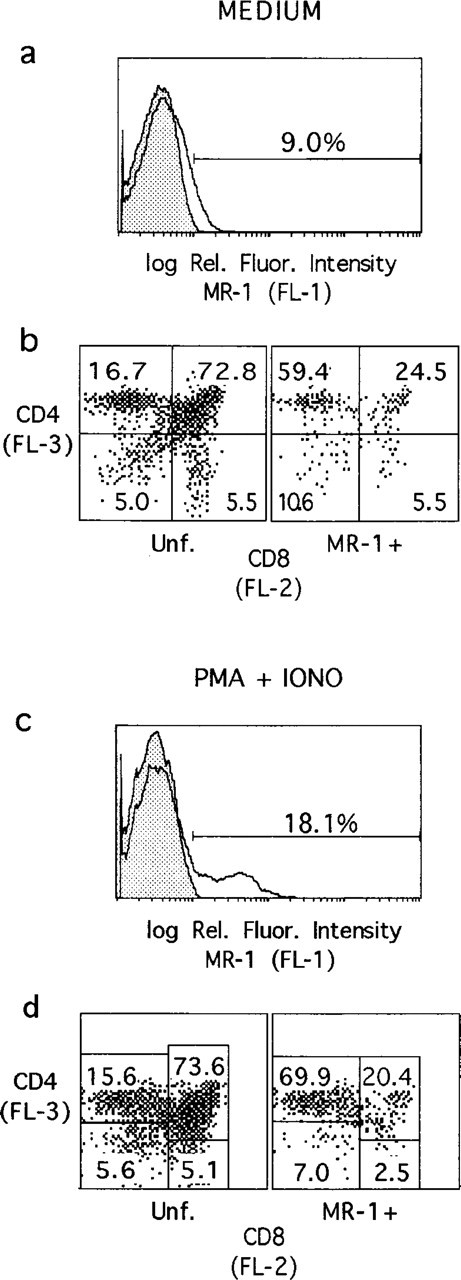
Flow cytometric analyses of CD40-L expression by thymocytes. (
Exposure of thymocytes to PMA and ionophore for 1 hr resulted in a rapid upregulation of CD40-L (Figure 2c). The distribution of CD40-L+ cells among thymocyte populations defined by CD4/CD8 expression was similar to that of untreated thymocytes, although there was further skewing of MR1+ cells to the CD4+8- population (Figure 2d). Although this treatment had little effect on the representation of thymocyte subsets defined by CD4 and CD8 expression, marked downregulation of CD4 labeling was observed after PMA/ionophore treatment.
Immunohistochemical analysis of gp 39 expression in frozen sections of adult thymus tissue detected rare, scattered CD40-L+ cells in the cortex and more labeling in areas corresponding to the medulla (Figure 3a). A punctate anti-CD40-L labeling pattern predominated among medullary thymocytes, although profiles of partial circumferential labeling were also observed (Figure 3b). Demonstration of CD3 expression on adjacent thymic sections (Figure 3c) confirmed that the location of the majority of the CD40-L+ thymocytes coincided with the distribution of strongly labeled CD3+ cells in the medulla.
Ontogeny of Thymic CD40 and CD40-L Expression
At Day 14 of gestation, the earliest time point examined, expression of CD40 was preferentially associated with aggregates of cells in the central portion of the thymus (Figure 4a). With advancing gestational age (Days 16 and 18), increased levels of CD40 labeling were associated with cords of medullary cells (Figures 4b and 4c, respectively). Levels of cortical CD40 expression observed in adult thymic tissue were not observed in fetal thymic tissue through Day 18 of gestation, but were dramatically increased by birth (Figure 4d). As shown in Figures 4e and 4g, CD40-L+ thymocytes were not detectable in Day 18 fetal thymic tissue. By birth (Figures 4f and 4h), CD40-L+ thymocytes were readily detectable and were preferentially associated with the medullary compartment, although isolated CD40-L+ thymocytes were scattered throughout the cortex. The upregulation of cortical CD40 expression and the appearance of CD40-L+ thymocytes coincided temporally with consolidation of the medullary thymic compartment. Scattered foci of ER-TR5+ and CD40+ epithelial cells present at Day 18 of gestation (Figure 4i) had coalesced into fewer but larger foci by birth (Figure 4j). The distribution of ER-TR5+ and medullary CD40+ cells was largely overlapping, suggesting that many of the ER-TR5+ stromal cells also expressed CD40 at this stage of development.
Targeted Overexpression of CD40-L to the Thymus Results in Altered Cortical, Medullary, and Subcapsular Compartments
The temporal coincidence of the appearance of CD40-L+ thymocytes and the organization of the medulla raised the possibility that CD40-L/CD40 interactions might contribute to the differentiation or expansion of this tissue compartment. Therefore, we examined the organization and phenotype of thymic tissue in which the thymocyte expression of CD40-L was abrogated (Xu et al., 1994) or elevated through targeted transgene expression.
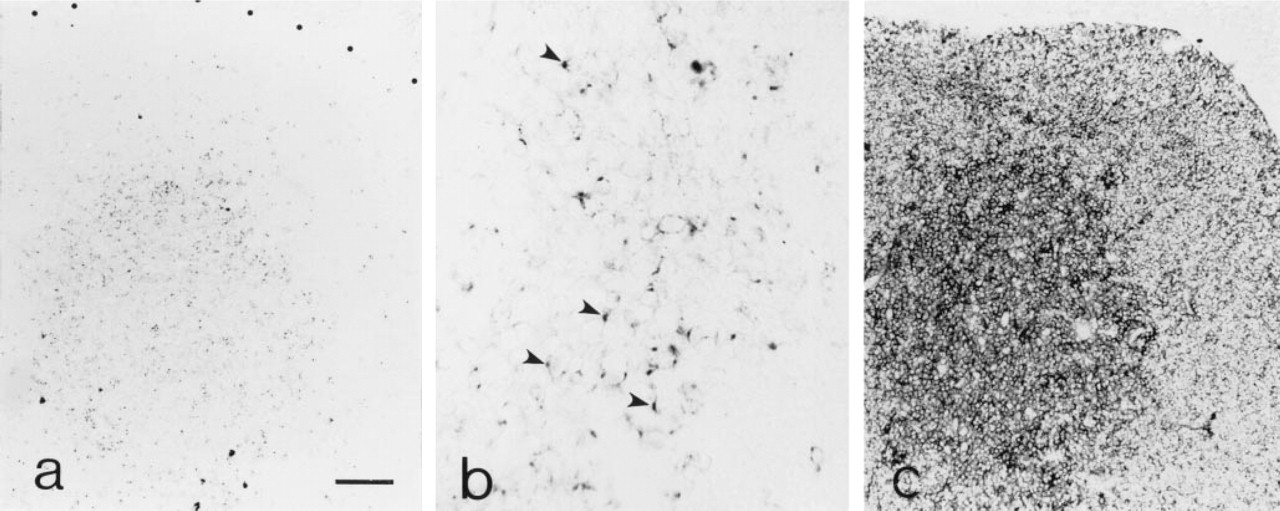
Thymic expression of CD40-L. (
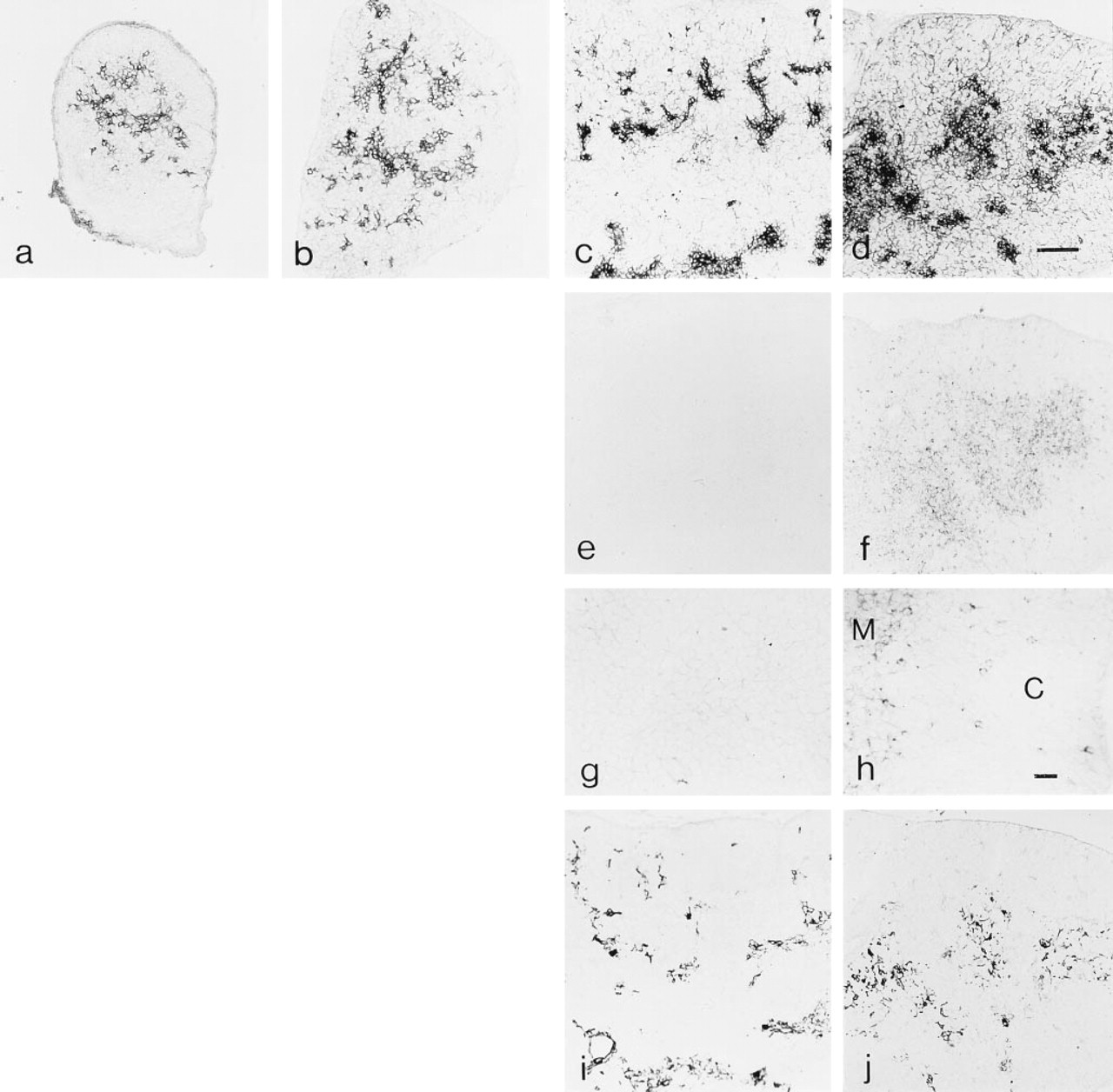
Ontogeny of thymic CD40 and CD40-L expression. CD40 expression at fetal Days 14 (
Thymic tissue from mice bearing a disruption of the CD40-L gene displayed fairly normal histology, with well-defined cortical and medullary compartments and no obvious defect in thymocyte development (Xu et al., 1994), although some aspects of the selection process appear to be altered (Foy et al., 1995). Immunohistochemical analyses of the thymic environment of CD40-L-deficient mice detected subtle decreases in medullary expression of B7–2 (Foy et al., 1995) CD40, B7–1, and Class II MHC molecules (data not shown), but clearly indicated that CD40-L/CD40 interactions did not play an obligate role in establishment of cortical or medullary thymic compartments.
Thymic tissue from transgenic mice expressing different levels of CD40-L transgene under control of the proximal
In addition to the disrupted segregation/representation of cortical and medullary epithelial cells, the expression patterns of several putative cell interaction molecules were elevated in the
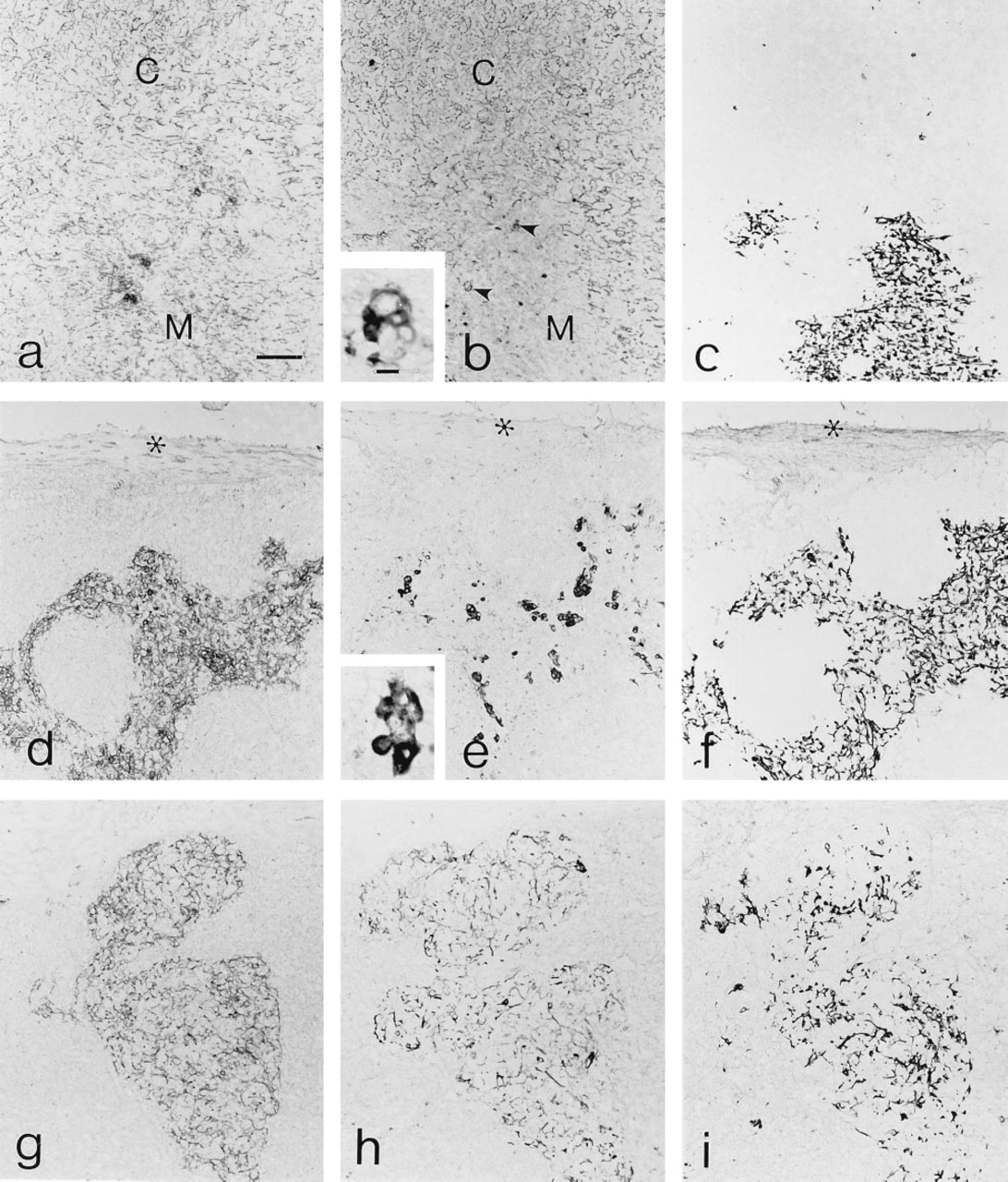
The expanded extraepithelial subcapsular space defined by the absence of E-cadherin+ epithelium (Figure 6a) contained accumulations of B220+ (Figure 6b) and CD40+ (Figure 6c) cells. Widespread CD40 expression was also evident within the epithelial compartment of the thymus (Figure 6c). The extraepithelial compartment also contained substantial numbers of CD3+ (Figure 6d) cells expressing CD40-L (Figure 6e). Although the level of CD40-L expression was dramatically elevated in the transgenic thymic tissue, the punctate labeling pattern was similar to that observed in normal mice (inset to Figure 6e). In addition to B-cells and CD3+ cells, these areas also contained significant numbers of CD11c+ cells (Figure 6f) and thus resembled diffuse peripheral lymphoid tissue. Endogenous peroxidase activity was predominantly associated with the epithelial component of the transgenic thymic tissue (Figure 6g). As in normal thymic tissue, this activity was due largely to eosinophils, as determined by ultrastructural cytochemistry (data not shown).
Elevated intrathymic CD40-L expression could be affecting the initial expansion of different stromal cell populations or the differentiation program of TE subpopulations. If promiscuous CD40-L/CD40 interactions were primarily altering the expansion potential of different TE populations, thymocytes overexpressing CD40-L would not be expected to significantly alter the representation of relatively radio-resistant cortical and medullary elements in an established thymic environment that had developed normally. On the other hand, if the loss of cortical TE and expansion of the medullary compartment within the
Discussion
The pattern of CD40 expression in the murine thymus was similar to that described for the human thymus (Galy and Spits, 1992) and resembled the distribution of Class II MHC antigens within the adult thymus (Farr and Nakane, 1983; Rouse et al., 1979). Whereas cortical CD40 was predominantly associated with epithelial cells, cells expressing CD40 in the medulla were heterogeneous and included dendritic cells and thymic epithelial cells. The basis for the variable expression of CD40 by medullary epithelial cells is presently not clear and may represent additional epithelial heterogeneity or local microenvironmental influences on the levels of CD40 expression.
Flow cytometric and immunohistochemical analyses indicated that CD40-L is expressed at low levels by thymocytes, but preferentially by medullary CD4+8- and, to a lesser extent, CD4+8+ subsets. The distinctive punctate nature of much of the MR1 labeling may represent CD40-L sequestered in a cytoplasmic compartment within thymocytes or redistribution of CD40-L on the thymocyte cell surface as a consequence of crosslinking induced by interactions with CD40+ cells in the thymus. Dynamic regulation of CD40-L expression by CD4+ thymocytes is suggested by reports that CD40-L/CD40 interactions lead to downregulation of CD40-L expression by CD4+ T-cells (Yellin et al., 1992), and that human CD4+ memory T-cells contain preformed CD40-L that is rapidly brought to the cell surface after TCR-mediated activation (Casamayor-Palleja et al., 1995). The increased levels of CD40-L expression observed 1 hr after PMA/ionophore stimulation would be consistent with the possibility that CD40-L can be mobilized from intracellular sites.
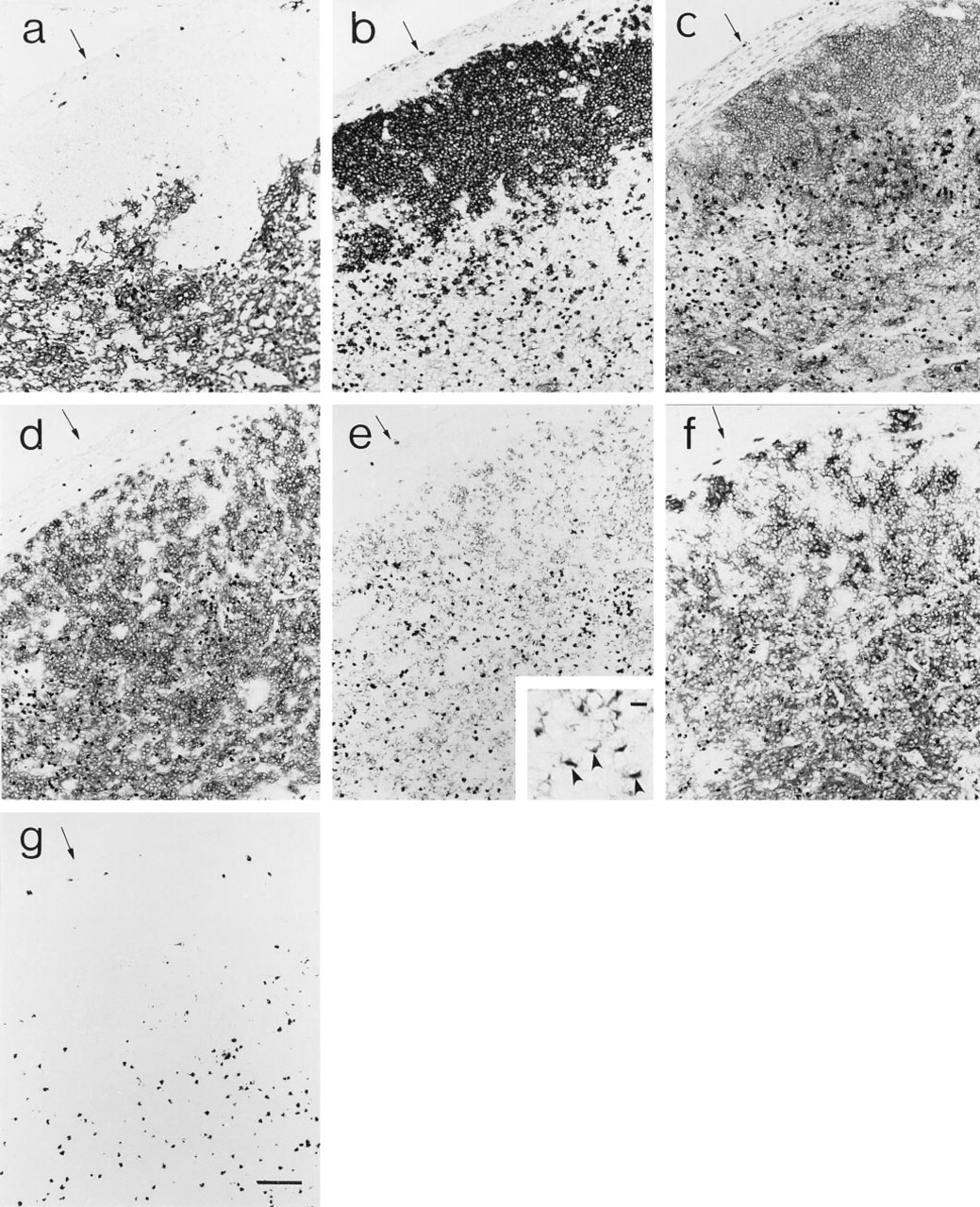
Thymic alterations due to targeted overexpression of CD40-L in thymocytes. Normal thymic tissue (
Thymic capsular connective tissue acquires characteristics of secondary lymphoid tissue in CD40-L transgenic mice. Adjacent sections of transgenic thymus tissue were processed to demonstrate E-cadherin (
Reconstitution of normal irradiated mice with bone marrow from transgenic mice results in an altered thymic phenotype. C57B1/6 mice were lethally irradiated and then reconstituted with T-cell-depleted bone marrow from syngeneic (
Previous studies of SCID or RAG-deficient mice demonstrated that normal development and organization of medullary, but not cortical, thymic epithelium was dependent on the participation of mature CD3+ thymocytes (Shores et al., 1991,1994; Surh et al., 1992). In normal mice this is reflected by conversion of scattered medullary foci present in the fetal thymus to a more consolidated adult pattern at about birth, when mature single positive thymocytes expressing αβ TCR begin to accumulate (Havran and Allison, 1988). The augmented expression of cortical CD40 and the appearance of predominantly medullary CD40-L+ thymocytes during late fetal development occurred simultaneously with reorganization of the medullary epithelial compartment, and may reflect an interdependence of these processes. Expression of CD40 by murine thymic stromal cells can be upregulated in vitro by exposure to TNFα or combinations of IFNγ and other proinflammatory cytokines (Farr, unpublished observations). If the cytokine requirements for CD40 expression by TE cells in vitro reflect physiological requirements for this process in vivo, the neonatal upregulation of CD40 expression may be related to the accumulation of CD4 and CD8 single positive thymocytes, which can produce high titers of IFNγ and TNFα in vitro after activation with calcium ionophore and PMA (Fischer et al., 1991) and which may be releasing these cytokines in response to physiological stimuli in situ.
Foy et al. (1995) have reported that negative selection in some model systems, but not in others, is disrupted in CD40-L-deficient mice and in mice chronically treated with anti-CD40-L MAb. They also documented a reduction of B7–2 expression within the medullary compartment of CD40-L-deficient mice and suggested that CD40-L/CD40 interactions may exert their effects in part by modulating expression of other cell interaction molecules within the thymic environment. Our demonstration that medullary, but not cortical, levels of other potential cell interaction molecules, including Class II MHC antigens, CD40, and B7–1, are reduced in gp 39-deficient mice lends additional support to this hypothesis. Lower levels of these cell interaction molecules within the thymic medulla, particularly MHC molecules, might decrease the efficiency of negative selection of some self-reactive thymocytes without having a significant impact on the overall production of thymocytes.
Targeted overexpression of CD40-L in thymocytes resulted in reduced thymic cellularity, disrupted thymocyte development, and profound alterations in the phenotype and organization of the thymic environment, the severity of which increased with increased transgene expression. At low levels of transgene expression (<12 copies) thymic cellularity was reduced two- to 10-fold, with fairly normal representation of thymocyte subsets defined by CD4 and CD8 expression. With high copy number there was a preferential loss of CD4+8+ thymocytes (up to 4000-fold reduction) and skewing of the mature thymocyte representation to the CD8 single positive subset. Thymocyte development in radiation bone marrow chimeras repopulated with transgenic bone marrow yielded similar results (Clegg CH et al., submitted for publication). Based on the work of Foy et al. (1995) demonstrating that CD40/CD40-L interactions participate in negative selection, it is tempting to speculate that this process is perturbed in the
A potential role for CD40-L/CD40 interactions in regulating epithelial cell growth/differentiation is suggested by the progressively increased representation of medullary TE cells and the decline in the frequency of cortical TE cells as levels of transgene-derived CD40-L expression increased. It is presently not known if cortical and medullary epithelia derive from a common 3rd pharyngeal pouch endodermal epithelial progenitor population (Le Douarin et al., 1984) or reflect expansion of separate epithelial cell populations derived from 3rd pharyngeal pouch endoderm and ectoderm from the 3rd pharyngeal cleft (Cordier and Haumont, 1980). Studies of thymic epithelial cell differentiation in vitro have described terminal differentiation of medullary epithelial cells (Lobach et al., 1987), but acquisition of a medullary phenotype by cortical epithelial cells has not been demonstrated in vitro. The congruent localization of cortical and medullary epithelial cells in both low and high copy-number mice, and the replacement of cortical with medullary epithelium in some areas of thymic tissue from high copy-number mice, may result from changes in the developmental program of thymic epithelium, resulting in the differentiation of cortical epithelium to express a medullary phenotype or from alterations within the thymic environment that favor the growth/maintenance of a discrete medullary epithelial population. The ability of T-cell-depleted
The observed alterations in the thymic environment may not represent a direct sequelae of CD40/CD40-L interactions. Alterations of thymocyte development as a consequence of dysregulated expression of CD40L could modify other stimuli that in turn regulate the differentiation of thymic epithelial cells. A loss of cortical epithelium has been reported in transgenic mice that are homozygous for a human CD3∊ transgene and display a block in thymocyte differentiation at the CD44-CD25- stage (Hollander et al., 1995). However, in contrast to the CD40-L transgenic thymus, the lack of cortical epithelial development in the CD3∊ transgenic mice does not appear to be accompanied by expansion of the medullary compartment or capsular involvement, suggesting that the requirements for expansion and maintenance of cortical TE may be different. Interestingly, thymic tissue from transgenic mice with thymic overexpression of oncostatin M (Clegg and Farr, manuscript in preparation) or LIF (Shen et al., 1994) exhibits alterations that resemble some aspects of the
Footnotes
Acknowledgements
This work was supported by grants from the National Institutes of Health (AI-24137 and AG-04360) and by the Bristol-Myers Squibb Pharmaceutical Research Institute.
We thank Dr Roger Perlmutter for critical review of the manuscript.
