Abstract
We developed a rapid and reliable technique for specifically staining intact chloroplasts using the fluorescent dye carboxyfluorescein diacetate. Intact, import-competent chloroplasts were isolated simply and rapidly from soil-grown Arabidopsis thaliana plants, with yields of 20 ± 5 μg chlorophyll per g FW, greater than previously reported yields from soil-grown Arabidopsis. Traditional chloroplast isolation buffers sometimes contain low concentrations (<10 mM) sodium ascorbate as a general-purpose anti-oxidant, but we found that only Arabidopsis chloroplasts isolated in the presence of high concentrations (50–100 mM) of sodium ascorbate in the initial grinding buffer were import-competent.
U
Arabidopsis thaliana ecotype Columbia was grown on compost under low light (100 μmol photon m–2 sec–1) with an 8-hr photoperiod at 20 ± 2C for 4–5 weeks. Chloroplasts were isolated using a linear Percoll gradient essentially according to the barley method described in Brock et al. (1993) with the following modifications: 10 g healthy leaves were homogenized in 200 ± 50 ml grinding buffer containing 50 mM L (+) sodium ascorbate, in four 2-sec pulses in a kitchen blender equipped with disposable razor blades (Kannangara et al. 1977), and all centrifugation except the Percoll gradient step was reduced to 1000 × g. Larger quantities of leaf material (20–40 g tissue) required 100 mM sodium ascorbate in the initial grinding buffer. Typical yields were 20 ± 5 μg chlorophyll per g FW, which are greater than published yields from similar tissue [5 μg/g FW (Fitzpatrick and Keegstra 2001); 4–6 μg/g FW (Rensink et al. 1998)]. Traditional grinding buffers have sometimes included 1–10 mM sodium ascorbate or isoascorbate as a general-purpose anti-oxidant (e.g., Walker et al. 1987). We propose an additional role for sodium ascorbate in protecting Arabidopsis chloroplasts from damage by the breakdown products of glucosinolates. Glucosino-lates are abundant in Arabidopsis; tissue damage leads to their hydrolysis by myrosinases (Wittstock and Halkier 2002). It is well documented that glucosino-late breakdown products are biologically active and can cause enzyme and organelle damage (e.g., Lykkesfeldt and Møller 1993).
Chloroplast intactness, necessary for protein import assays, is often estimated by phase-contrast microscopy (Walker et al. 1987). Intact chloroplasts are surrounded by a more pronounced halo (Figures 1A and 1C) compared with broken chloroplasts (Figure 1B). One drawback with this method is that chloroplast envelopes can break, releasing stromal contents, and then reseal, retaining the appearance of intactness (Walker et al. 1987). Arabidopsis chloroplasts were equilibrated for 5 min with an equal volume of carboxyfluorescein diacetate (CFDA; Molecular Probes, Leiden, The Netherlands), final concentration 0.0025% w/v, and were examined with a Zeiss Photomikroskop II equipped with FITC filters (excitation filter BP 455–490, beam splitter FT 510, and emission filter, either “fluorescein-specific” BP 520–560 or “nonspecific” LP 520). CFDA fluoresces strongly when de-esterified to carboxyfluorescein (CF). “Ester loading” of nonpolar esterified derivatives of fluorescein is a standard test for cell viability, depending on an intact membrane that prevents leakage of the polar dye into the medium and on the presence of esterases (Oparka and Read 1994). Chloroplasts that had taken up CFDA fluoresced intensely green (Figure 1E) and corresponded exactly to the ones delineated by a bright halo under phase-contrast (Figure 1D). Up to three Arabidopsis carboxylesterases are predicted to be targeted to the chloroplast stroma (Swissprot Q9M899, Q8L8W1, Q9LT10; Emanuelsson et al. 2000), implying that the green-fluorescing organelles retain stromal contents as well as an intact envelope. Interestingly, the non-carboxylated dye fluorescein diacetate did not cause fluorescence of chloroplasts (data not shown). Chloroplasts that did not take up CFDA (Figures 1D–1F, marked with arrows) were red when all emission wavelengths (F) or only chlorophyll autofluorescence (not shown) were detected using appropriate filters. Red chloroplasts corresponded exactly to the ones without a marked halo under phase-contrast. With the nonspecific filter settings, intact chloroplasts appeared yellowish owing to the presence of both green fluorescein and red autofluorescence. Chloroplasts isolated in the absence of ascorbate (Figures 1G–1I) were not visibly damaged and showed a similar degree of intactness as those isolated in the presence of ascorbate (Figures 1D–1F).
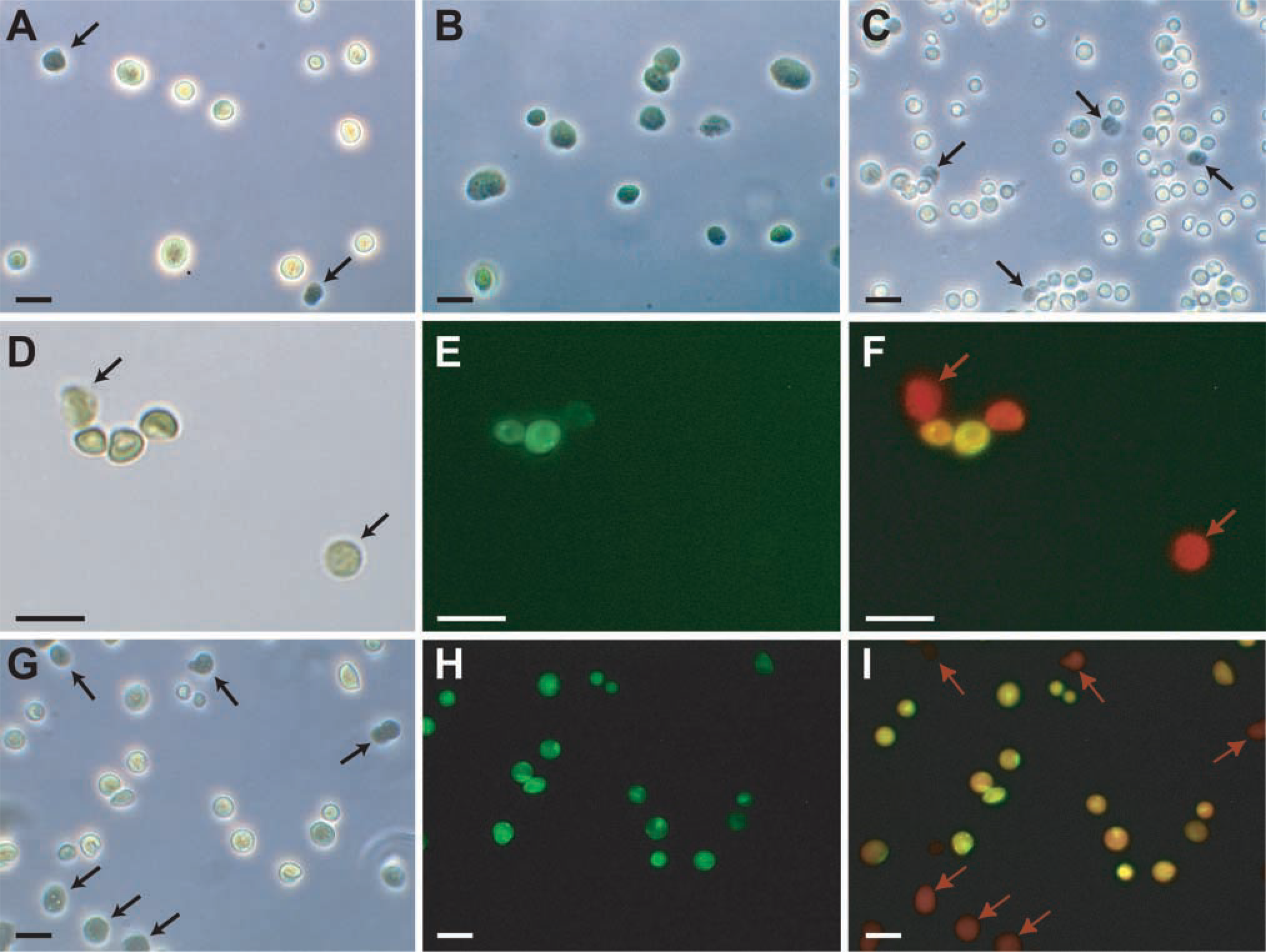
Images of isolated Arabidopsis and pea chloroplasts analyzed by brightfield, phase-contrast, and fluorescence microscopy. (
There was, however, a marked difference in the chloroplasts' ability to import precursor proteins (Figure 2). Import assays were carried out according to Robinson and Mant (2002) using the pea precursor of Rubisco small subunit (pSSU, a stromal protein), and the wheat precursor of the 33-kD oxygen-evolving complex subunit (p33K, a thylakoid lumen protein). Chloroplasts were fractionated after import to determine the location of imported protein. For chloroplasts isolated without ascorbate, the majority of chloroplast-associated radiolabeled protein was precursor, bound to the outside of the organelle (Figure 2A, no ascorbate). Little SSU was found in the stromal fraction, and almost no 33K in the thylakoid lumen. By contrast, for chloroplasts isolated in the presence of 50 mM ascorbate (Figure 2A, with ascorbate), the majority of radiolabeled protein was protected from protease digestion and correctly targeted to the stromal and thylakoid lumen compartments, respectively. A parallel experiment using pea chloroplasts isolated by standard procedures without ascorbate (Figure 1C) showed that Arabidopsis import efficiencies were on a par with those of pea (Figure 2B).
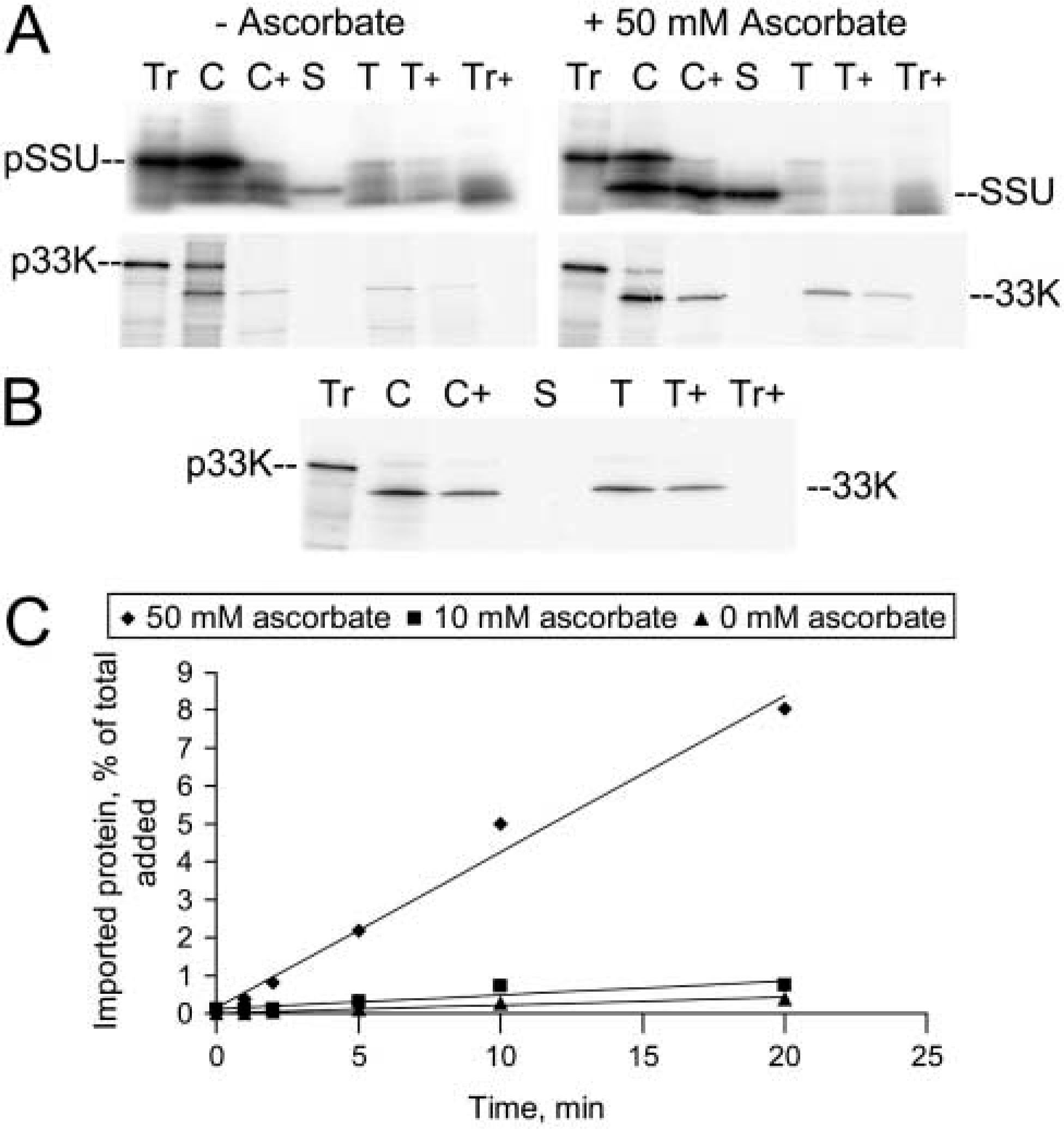
Comparison of the protein import capabilities of chloroplasts isolated in the presence and absence of sodium ascorbate. (
We compared the rate of pSSU import by Arabidopsis chloroplasts isolated in the absence of ascorbate and in the presence of 10 mM and 50 mM ascorbate (Figure 2C). Chloroplasts isolated in the presence of 50 mM ascorbate imported pSSU at a rate 19-fold and 11-fold higher than in the absence of ascorbate or presence of 10 mM ascorbate, respectively.
We conclude that inclusion of 50 mM ascorbate in the grinding buffer enables the isolation of import-competent chloroplasts. The use of CFDA to identify intact chloroplasts may also be extended to other isolated organelles containing carboxylesterases.
Footnotes
Acknowledgements
Supported by an EMBO Long Term Fellowship to AM.
We thank Mitsuru Akita and Kenneth Keegstra for giving the clone encoding pea pSSU, Birger L. Møller for invaluable advice and Lynda Fitzpatrick for helpful discussions.
