Abstract
The aim of this work was to evaluate by immunohistochemistry (IHC) the expression of both LRP-1 and urokinase-type plasminogen activator receptor (uPAR) at different developmental stages of rat prostate disease by using a prostate cancer model previously developed in our laboratory. We found that LRP-1 was weakly expressed in normal prostates and in rats with hyperplastic glands. The expression of this receptor increased and correlated with the degree of premalignant lesions (PIN I, II, and III). The IHC for uPAR in normal prostates and in premalignant lesions showed a score of immunostaining that correlated with the expression of LRP-1. On the other hand, in prostates with adenocarcinomas and undifferentiated carcinomas, LRP-1 was undetectable or weakly detected, whereas uPAR showed a significantly higher level of expression. Based on the IHC results in rat prostates with premalignant and malignant lesions and considering that LRP-1, by mediating the internalization of uPAR, is involved in the regulation of extracellular matrix remodeling and cell migration, we conclude that a decreased expression of LRP-1 could be involved with the increasing activation of plasminogen activators shown in cancers.
Keywords
I
LRP-1 is a member of the low-density lipoprotein receptor gene family, which is synthesized as a single polypeptide chain of about 600 kD and in the trans-Golgi network is cleaved by furin into subunits of a 515-kD α-chain and an 85-kD transmembrane β-chain (Strickland et al. 2002). The 515-kD α-chain of LRP-1 contains binding sites for proteinases such as tPA, uPA, and MMPs, for proteinase inhibitors including activated α2-macroglobulin (α2-M), pregnancy zone protein (PZP), and plasminogen activator inhibitor type 1 (PAI-1), or for ligands that are functionally diverse, such as lipoprotein lipase (Herz and Strickland 2001; Howell and Herz 2001; Sánchez et al. 2001; Chiabrando et al. 2002a, b; Strickland et al. 2002). Ligands bound to LRP-1 are internalized via clathrin-coated pits and degraded in lysosomes, and LRP-1 recycles back to the cell surface (Krieger and Herz 1994). It has been shown that uPA is predominantly cell-associated, interacting with a cellular receptor, uPAR, and that it initiates a proteolytic cascade that activates plasminogen to plasmin and other proteinases, including MMPs. This proteolytic pathway is regulated by PAI-1, which forms a stable complex with uPA. Once formed, this complex is rapidly internalized along with uPAR and is mediated by LRP-1 (Herz and Strickland 2001). Recently, it was demonstrated that uPAR also regulates cell growth and motility of tumor cells, suggesting that the uPA-uPAR system is an important pathway through which LRP-1 may modulate cell physiology (Hussaini et al. 1999). There is also accumulated evidence to suggest a prominent role for LRP-1 in signal transduction pathways that lead to cell migration and proliferation (Goretzki and Mueller 1998; Herz and Strickland 2001; Strickland et al. 2002), although the mechanisms involved in these pathways are not completely understood.
In normal tissues, LRP-1 is expressed by hepatocytes, macrophages, fibroblasts, neurons, and vascular smooth muscle cells (Moestrup et al. 1992). Curiously, the LRP-1 expression in tumor cells may be dependent on the cancer type. It has been shown that LRP-1 levels are substantially decreased in invasive subclones compared with noninvasive subclones derived from human prostate PC-3 and DV145 and melanoma A2058 cells (Kancha et al. 1994; de Vries et al. 1996). In addition, hepatocellular carcinomas are primarily LRP-1 negative (Gonias et al. 1994). On the other hand, astrocytomas express LRP-1, whereas nonneoplastic and nonreactive human astrocytes are LRP-1-negative (Lopes et al. 1994). Taken together, these and other findings indicate that the evidence from in vivo experiments is not enough to explain the biological role of LRP-1 in cancer.
Gilardoni et al. (1999) developed an experimental model of prostate gland cancer induced by long-term hormone treatment and a chemical carcinogenic agent in male Wistar rats with autoimmune prostatitis. This animal model takes into account rats with premalignant and malignant prostate lesions characterized by atypical hyperplasia (≈ 10% of animals), prostate intraepithelial neoplasm (PIN; ≈40% of animals) classified as (a) PIN I, the earliest stage of the lesion with marked variation in nuclear size or anisonucleosis, (b) PIN II, in which most nuclei are enlarged, with greater hyperchromasia, and (c) PIN III, in which cells display large, prominent nucleoli similar to those seen in invasive carcinoma as well as remarkable hyperchromasia and anisokaryosis. Finally, prostate adenocarcinomas and undifferentiated carcinomas are found in ≈50% of the remaining animals. Premalignant and malignant lesions are found exclusively in the dorsal or lateral lobe but not in the ventral lobe of the prostate or in the other accessory sex glands. This animal model has clinical correlations in terms of premalignant and malignant disease of the human prostate (Gilardoni et al. 1999).
In this study, derived from this animal model, expressions of LRP-1 and uPAR were investigated by IHC to obtain further information about the interrelation of both receptors at different developmental stages of prostate disease and prostate cancer, using specific antibodies to these proteins.
Materials and Methods
Animals
Three-month-old male random-bred Wistar rats were housed and cared for at the Department of Clinical Biochemistry, Faculty of Chemical Science, National University of Cordoba, in accordance with the guidelines for Institutional Animal Care. Animal models suffering different types of prostate lesions were prepared after the DMBA and testosterone treatment previously reported (Gilardoni et al. 1999). Formalin-fixed, paraffin-embedded sections of the entire prostate complex were prepared and examined histologically in 4-μm sections by hematoxylin-eosin staining. Tissue samples from dorsolateral prostates of animals with normal prostates (n = 4), benign hyperplasia (n = 4), PIN I (n = 4), PIN II (n = 5), PIN III (n = 9), and malignant (n = 16) lesions were appropriately selected and processed for IHC and Western blotting or were stored at −70C. Animals with malignant lesions consisted of adenocarcinomas (n = 11), undifferentiated carcinomas (n = 3), and different origin types (n = 2).
Antibodies
A mouse monoclonal antibody (MAb) against the 85-kD β-chain of LRP-1 (#3501) was obtained from American Diagnostica (Greenwich, CT). This antibody has been extensively characterized for use in IHC (Moestrup et al. 1992). A rabbit polyclonal antibody against the 515-kD α-chain of LRP (2629) was generously provided by Dr. Dudley Strickland (American Red Cross; Rockville, MD). This antibody has been previously tested by Western blotting (Zheng et al. 1994; Chiabrando et al. 2002a). A rabbit polyclonal antibody against rat uPAR (#3920) was purchased from American Diagnostica and is recommended for use in IHC in rat prostates by the manufacturer. Horseradish peroxidase (HRP)-conjugated rabbit anti-mouse IgG and goat anti-rabbit IgG polyclonal antibodies were obtained from Sigma (St Louis, MO).
Immunohistochemistry
Immunostaining was performed using biotin-streptavidin anti-mouse and anti-rabbit IgG-peroxidase (Calbiochem-Novabiochem; La Jolla, CA). Briefly, 4-μm-thick paraffin-embedded sections were cut and mounted on poly-
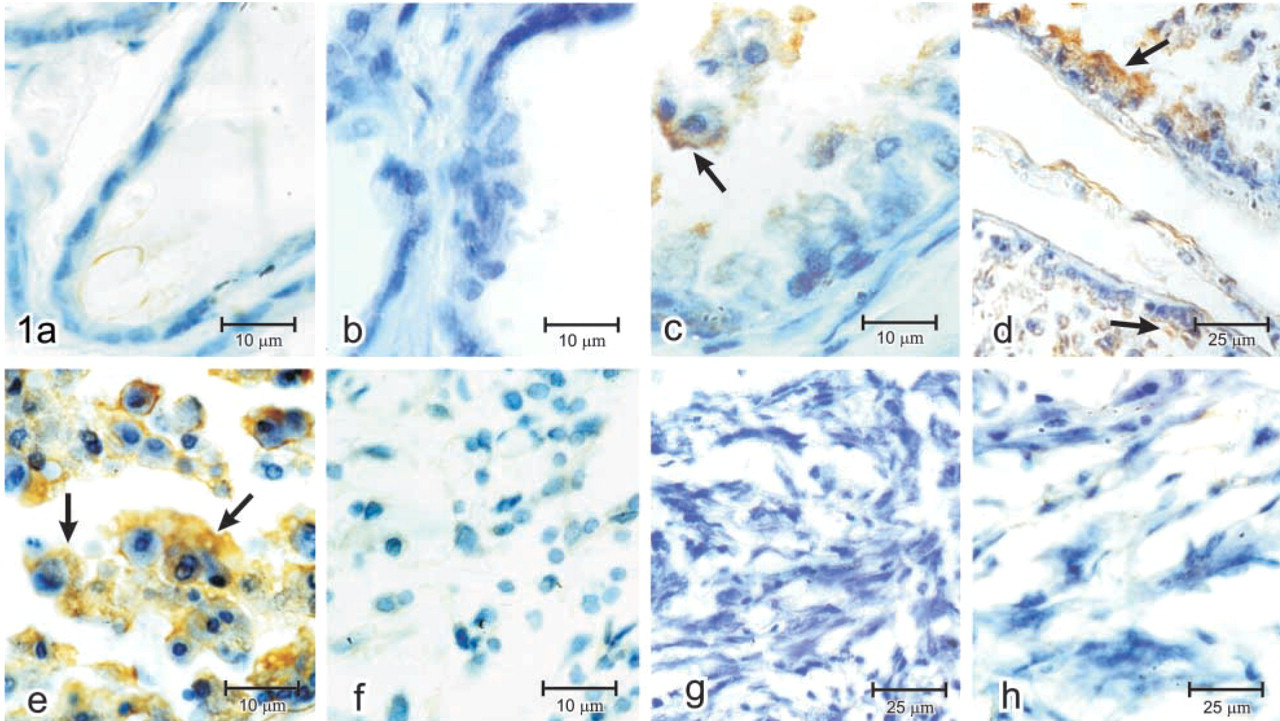
IHC peroxidase staining with MAb against the 85-kD β-chain of LRP-1 in normal, premalignant, and malignant rat prostates. (
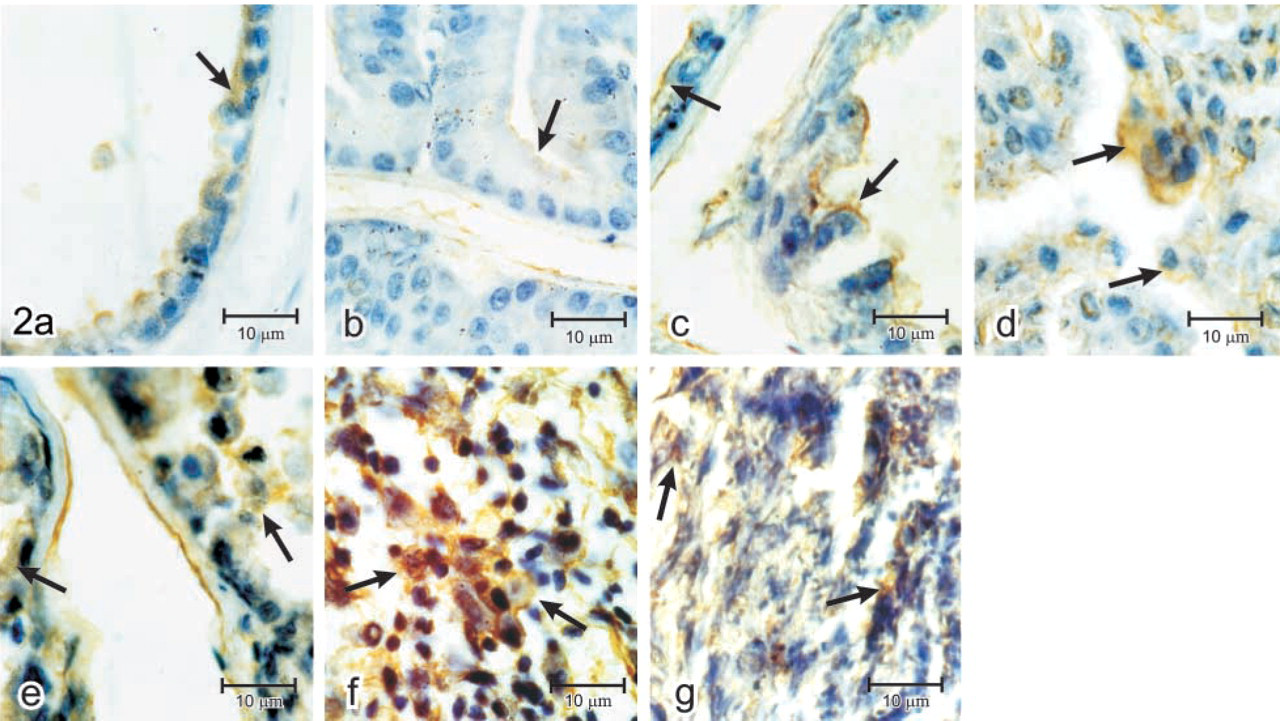
IHC peroxidase staining with polyclonal antibody anti-uPAR in normal, premalignant, and malignant rat prostates. (
Immunostaining levels of LRP-1 and uPAR in normal, premalignant and malignant rat prostates
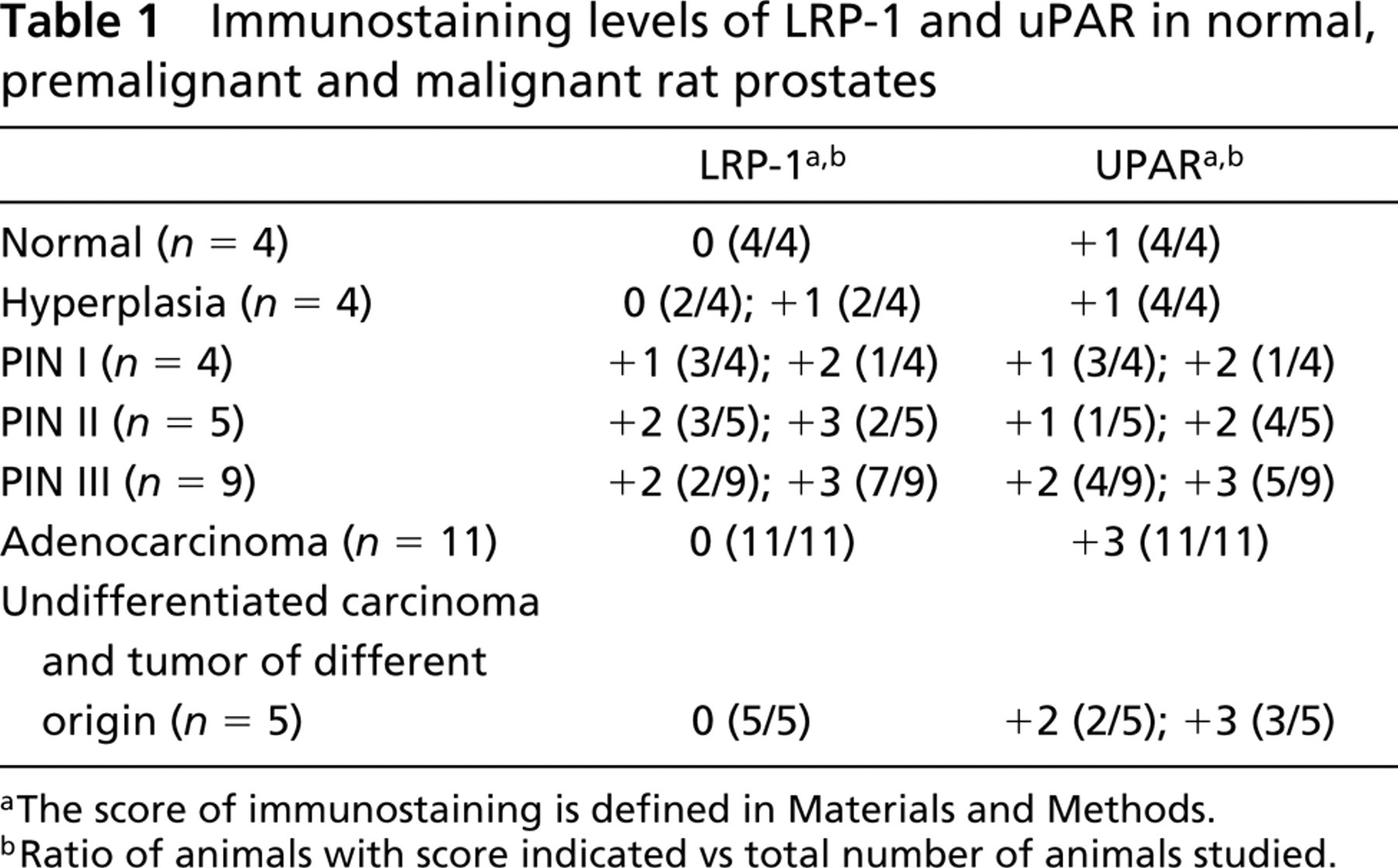
aThe score of immunostaining is defined in Materials and Methods.
bRatio of animals with score indicated vs total number of animals studied.
Negative controls for all antisera included (a) omission of primary antibody, (b) substitution of primary antibody with an MAb of irrelevant specificity, and (c) substitution of primary polyclonal antibody with normal non-immune IgG at the equivalent protein concentration.
Immunostaining was assessed semiquantitatively as the percentage of positively stained cells according to the following scale: score 0, absent (no staining present) or <10% of cells with partial staining; score +1, light intensity or >10% of cells with partial staining; score +2, moderate intensity and >10% of cells with complete membrane staining; score +3, strong reaction and >10% of cells with complete membrane and cytoplasm staining. The sections were examined and scored by two independent observers, then reviewed together, and the average data represent a consensus value of all observations.
Western Blotting Analysis
To detect the expression of LRP-1 from rat tissues at different stage of prostate diseases, prostate membranes were prepared as essentially described Jensen et al. (1988). Fifty μg of tissue protein was diluted in SDS-PAGE loading buffer (100 mM Tris-HCl), pH 6.8, 5% SDS, 1% bromophenol blue, 20% glycerol) and boiled for 5 min. Samples were size-fractionated on a 5% SDS-PAGE gel under non-reducing conditions (Laemmli 1970). Proteins were electrotransferred to nitrocellulose membranes (Towbin et al. 1979), blocked with 5% non-fat dry milk in TBS-T buffer (20 mM Tris-HCl, pH 7.4, and 0.05% Tween-20) and then incubated overnight at 4C with rabbit polyclonal antibody against the 515-kD α-chain of LRP-1. The membranes were washed and incubated with HRP-conjugated goat anti-rabbit IgG antibody for 2 hr at room temperature. After washing, antibody bound to antigen was detected by an ECL chemiluminescence system (NEN Life Science Products; Boston, MA) by exposure to a Kodak X-OMAT film.
Results
LRP-1 Immunohistochemistry
To evaluate the expression of LRP-1 in prostates of rats that experimentally develop different disease stages, IHC on paraffin-embedded tissues was done using an MAb against the 85-kD β-chain of LRP-1. In each case, the positive staining for LRP-1 was localized predominantly in epithelial cells. Figures 1a and 1b show that both normal and hyperplastic prostates contain a pattern of staining for LRP-1 equivalent to score 0, whereas premalignant lesions with PIN I (Figure 1c), PIN II (Figure 1d) and PIN III (Figure 1e) showed scores of +1, +2, and +3, respectively. On the other hand, prostates with different types of tumor, such as adenocarcinoma (Figure 1f) and undifferentiated carcinoma (Figures 1g and 1h) showed a pattern of staining equivalent to score 0.
uPAR Immunohistochemistry
The expression of uPAR at different stages of prostate disease was also studied by IHC and each imunostaining score was compared with that of LRP-1. The uPAR expression for normal, hyperplastic, and PIN I prostates showed a score of +1 (Figures 2a–2c). PIN II and III presented a score of +2 (Figures 2d and 2e), and adenocarcinoma (Figure 2f) as well as undifferentiated carcinoma (Figure 2g) showed scores of +3 and +2, respectively. Table 1 shows and compares the IHC results for LRP-1 and uPAR obtained from the total number of animals studied. These data indicate that both receptors correlate positively in normal, hyperplastic, and premalignant lesions, whereas in tumor uPAR was highly expressed, in contrast to the very low level observed for LRP-1.
Western Blotting Analysis for LRP-1
Considering that the expression of LRP-1 was studied by IHC using a MAb against the 85-kD β-chain, we evaluated the expression of this receptor on protein extracts of rat prostates by Western blotting, employing a polyclonal antibody against the 515-kD α-chain of LRP-1. Figure 3 shows a single band equivalent to 515 kD. It was significantly detected in premalignant lesions, being more evident in PIN II and PIN III lesions (Figure 3, Lanes e-g). On the other hand, the 515-kD α-chain of LRP-1 was undetected in prostates with tumor (Figure 3, Lane a), being independent of the origin of the malignant lesion studied. These results showed a strong correlation with the IHC for LRP-1.
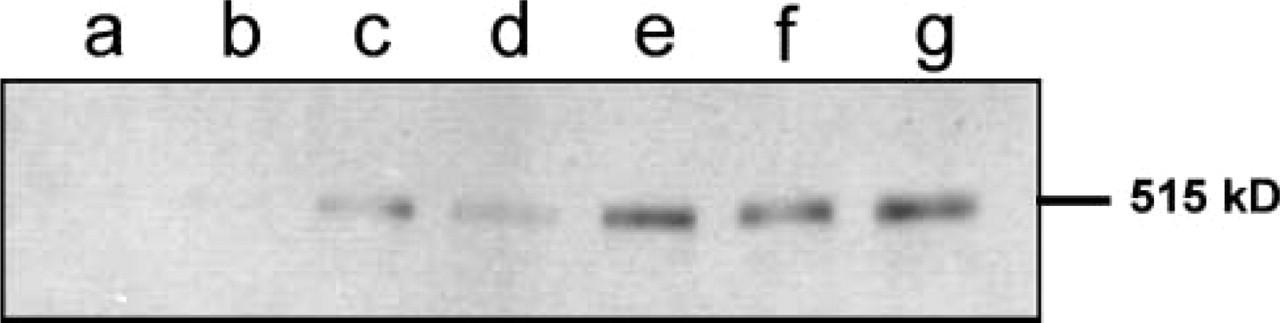
Western blotting analysis of LRP-1 level in normal, premalignant, and malignant rat prostates. Protein extract (40 μg) was subjected to SDS-PAGE under non-reducing conditions in 5% acrylamide gels and electrotransferred to nitrocellulose membranes. LRP-1 was detected with a rabbit polyclonal antibody against the 515-kD α-chain. Lane a, adenocarcinoma; Lane b, normal prostate; Lane c, hyperplastic prostate; Lane d, PIN I; Lane e, PIN II; and Lanes f,g, PIN III.
Discussion
In this study we clearly show, using IHC procedures, the expression profile of LRP-1 and uPAR in an animal model that can generate different stages of prostate disease from benign or hyperplastic to premalignant and malignant lesions (Gilardoni et al. 1999). It appears that LRP-1 is weakly expressed in normal prostates as well as in rats with hyperplastic glands, and its expression increases following the tendency of the grade of premalignant lesions (PIN I and II), being strongly expressed in animals with PIN III lesions.
Immunohistochemical assays done for uPAR in the same tissues showed, in normal prostates as well as premalignant lesions, a score of staining that correlated with the expression of LRP-1 (Table 1). However, in prostates with adenocarcinomas and undifferentiated carcinomas, LRP-1 was undetectable or weakly detected, whereas uPAR showed a significantly higher level of expression. In addition, the expression level of LRP-1, by IHC using an MAb against the 85-kD transmembrane β-chain, was also confirmed by Western blotting employing a polyclonal antibody against the 515-kD α-chain of LRP-1. The strong correlation demonstrated by both assays shows the specificity of the antibodies against LRP-1 used in this study.
The plasminogen activator system is composed of serine proteinases, such as uPA and tPA, and plays important roles in wound repair, tumor invasion, and cell migration under both normal and pathological conditions (Plas et al. 1998). At leading edge of cell migration, uPAR-bound uPA localizes the proteolytic activity and thereby facilitates cell penetration of tissue boundaries (Gyetko et al. 1994). At the cell surface, this complex binds its inhibitor PAI-1, which is localized in the matrix, and then is internalized by LRP-1 (Nykjaer et al. 1992). The co-expression of LRP-1 and uPAR on the cell surface might regulate the proteolytic activity of these serine proteinases by maintaining the homeostasis of extracellular matrix remodeling and cell motility (Zhang et al. 1998; Chazaud et al. 2000). Hence, in reference to our results, a decreased expression of LRP-1 contrasting with an increased uPAR level in malignant prostate lesions could be mainly involved with the elevated expression and activation of plasminogen activators observed in almost all human cancers (Bissell and Radisky 2001; Egeblad and Werb 2002).
Recently, a putative tumor suppressor gene was identified, termed LRP-1B or LRP-DIT (deleted in tumor) from the chromosome 2q21.2 region, which was found to be homozygously deleted in several human cancer cell lines (Liu et al. 2000,2001; Langbein et al. 2002). This receptor is a new member of the LDL receptor gene family and is closely related to LRP-1, suggesting that these giant receptors may bind similar sets of ligands and display overlapping functions (Liu et al. 2001). However, LRP-1B differs from LRP-1 in both endocytic kinetics and in its tissue expression pattern. Recently, Li et al. (2002) found that LRP-1B and uPAR form immunoprecipitable complexes on the cell surface in the presence of complexes of uPA and PAI-1, but displayed a substantially slower rate of uPA:PAI-1 complex internalization than LRP-1. Nevertheless, two aspects should be mentioned in reference to our results. First, LRP-1B was demonstrated to be expressed only on human cells and tissues. Second, there is not enough evidence to show whether the anti-LRP-1 antibodies, both anti-α-chain and anti-β-chain, used in this work can crossreact with certain structures of a putative LRP-1B protein. We are performing further studies to elucidate both aspects.
In conclusion, by using an animal model of prostate cancer we demonstrated by IHC that LRP-1 is weakly expressed in rat normal prostates and that its expression is increased in premalignant lesions, showing in each case a correlation with the level of immunostaining of uPAR. On the other hand, in prostate cancer the expression of LRP-1 is undetectable or weakly detected, whereas uPAR shows a significantly higher level of expression. Nevertheless, further studies are needed to elucidate the putative biochemical regulation of the differential expression of LRP-1 and uPAR during different stages of prostate disease and to evaluate the expression of both receptors in human glands to establish a potential use as early tumor markers for prostate cancer.
Footnotes
Acknowledgements
Supported in part by grants from SECyT (Secretaría de Ciencia y Tecnología de la Universidad Nacional de Córdoba), Agencia Córdoba Ciencia, PICTO CONICOR (Consejo de Investigaciones Científicas y Tecnológicas de la Provincia de Córdoba)–Agencia Nacional de Ciencia de la República Argentina, and CONICET (Consejo de Investigaciones Científicas y Tecnológicas de la República Argentina).
This work is dedicated to the memory of Professor Dr Miguel A. Vides, PhD; he was our light and showed our way in life. We are grateful to Dr Carlos A. Landa for critical reading and suggestions about the manuscript.
