Abstract
The liver is important for the kallikrein-kinin system modulation. This system plays a role in the inflammatory cascade with anticoagulant, profibrinolytic, and anti-adhesive attributes. The metalloendopeptidase EP24.15 is a major hepatic kininase. We studied the tissue distribution and subcellular localization of this enzyme in rat liver by cell fractionation and immunohistochemistry. Our results showed that EP24.15 is predominant in the soluble fraction of the liver homogenate and is present in the cytoplasm of hepatocytes, particularly in the perivenous zone (Z3). This localization is relevant because most hepatotoxin-induced necrosis, as well as ischemic hepatocellular injury, is predominant in Z3.
T
Adult male Wistar rats, raised and provided by the animal house of the Universidade Federal de São Paulo, were manipulated in accordance with the “International Guiding Principles for Biomedical Research Involving Animals” (Council For International Organization of Medical Sciences 1985, Geneva).
Liver exsanguination was performed at 37C as previously described (Molina et al. 2000). The rat was given an anesthetic (IP injection of urethane aqueous solution, 1.3 g/kg weight) and maintained under artificial respiration. The abdominal and thoracic cavities were opened and the portal vein and thoracic inferior vena cava were cannulated. Exsanguination was obtained with perfusion of 100 ml saline solution (NaCl 0.15 M) in an open circuit, with a 28-ml/min constant flow. Perfusion pressure was continuously monitored with a water manometer placed in the circuit before the portal cannula, ranging from 12 to 16 cm of H2O.
Cell fractionation was performed according to DeDuve et al. (1955) under the following conditions. The exsanguinated liver was homogenized (20 g/100 ml Tris buffer-HCl 0.01 M, pH 7.4, containing EGTA 1 mM, manitol 0.26 M, and BSA 0.5 mg/ml) in a Potter Elvehjeim homogenizer (four strokes). After homogenization, unbroken cells and nuclei were separated by a 3000 X g centrifugation. The supernatant was further centrifuged at various g values at 4C and separated into fractions: lysosomal (pellet 15,000 g), mitochondrial (pellet 27,000 g), microsomal (pellet 100,000 g), and cytoplasmic (supernatant 100,000 g). The fractions were sampled and maintained at −20C for activity measurement of the EP24.15, glutamate dehydrogenase, acid phosphatase, glucose-6-phosphatase, and lactate dehydrogenase enzymes, markers of the mitochondrial (Mt), lysosomal (L), microsomal (M), and cytoplasmic (C) fractions, respectively. The EP24.15 activity was measured by hydrolysis of the fluorogenic substrate Abz-RPPGFSPFRQ-EDDnp (bradykinin analogue), synthesized and provided by Dr. L Juliano (Biophysics Department, Unifesp, Brazil) as described previously (Molina et al. 2000). Briefly, aliquots (10 μl) were incubated at 37C with 10 μl substrate (1.5 μM) in a final volume of 1 ml Tris-HCl 50 mM, pH 8.0, containing 2.5 mM β-mercaptoethanol. The enzymatic activity was continuously followed for 10 min in a Hitachi F-2000 fluorescence spectrophotometer by measuring the fluorescence of the hydrolyzed substrate (ΛEM = 420 nm and ΛEX = 320 nm). The amount of protein varied among the fractions but after 10 min of incubation less than 10% of the substrate was consumed, even for the cytoplasmic fraction, which contained the highest activity, indicating first-order kinetics. The assay was performed in both the absence (total kininase activity) or presence (5 μM) of the EP24.15 specific inhibitor (EP24.15 activity), the N-[1(RS)-carboxy-3-phenyl-propyl]-Ala-Ala-Phe-p-aminobenzoate (cFP-AAF-pAB), a generous gift from Dr. M.J. Glucksman (Mount Sinai School of Medicine, New York, NY). The enzymatic activity was expressed in μmoles of substrate hydrolyzed/min and the total fraction activity obtained by multiplying the activity by the fraction volume. The results were analyzed by the GraphPad Prism software (version 1.03).
For immunohistochemistry, the exsanguinated livers were removed and fragments of 2 cm were fixed by immersion for 8 hr with 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4. Liver samples were then paraffin-embedded and 5-μm-thick sections were made and placed on poly-
The mitochondrial, lysosomal, microsomal and cytoplasmic fractions were characterized by the presence of their specific markers. Considering as 100% the EP24.15 activity measured in all fractions (N, Mt, L, M, and C), 90% of its total activity was found within the cytoplasmic fraction (Figure 1). Its specific activity (U/g protein) in this fraction was 4, 7, and 16 times greater compared to the Mt, L, and M fractions, respectively. These results are in agreement with the typical cytosolic activity of the EP24.15 found in mammalian tissues (Chu and Orlowski 1985). The internally quenched substrate used to assay EP24.15 was derived from BK; the introduction of Gln at the C-terminal site of BK displaced the cleavage to P-F bond by EP24.15 as well as by EC 3.4.24.16 (neurolysin). The relative rate of hydrolysis of this substrate by EP24.15 is significantly higher than by EC 3.4.24.16 (Oliveira et al. 2001). We additionally used β-mercaptoethanol in the assay, a substance that activates EP24.15 and inhibits neurolysin (Rioli et al. 1998). Both conditions indicate that the possibility of a major contribution of EC3.424.16 was excluded in this study.
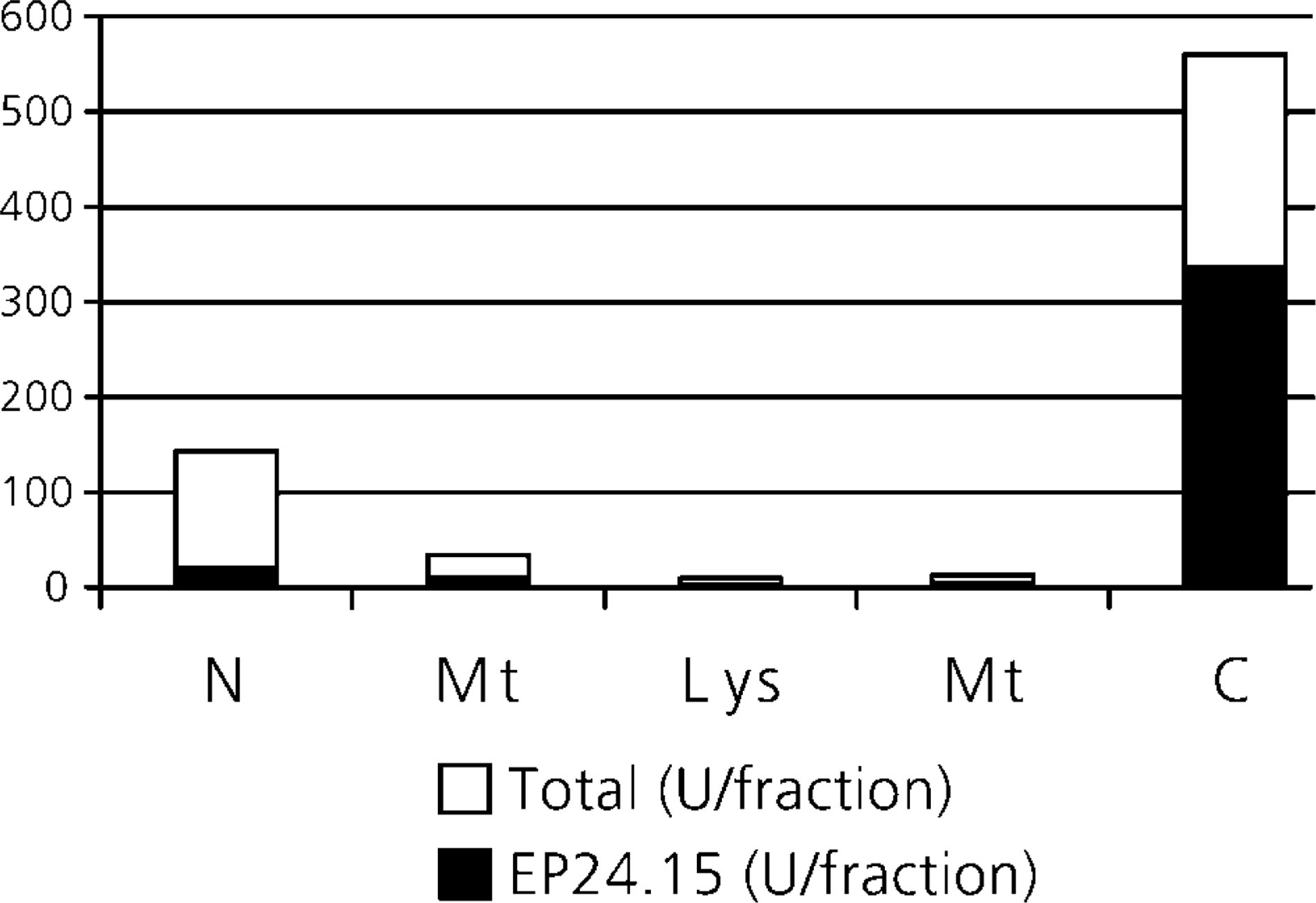
Total kininase and EP24.15 activities in the different cell fractions of rat liver. The exsanguinated liver was homogenized and the suspension was separated by centrifuging at various g values at 4C into fractions: Mt (mitochondrial), L (lysosomal), M (microsomal), and C (cytoplasmic). The fluorogenic activity was determined in the presence of β-mercaptoethanol and both in the absence (total kininase activity, sum of black and white bars) and presence of a specific inhibitor (cFP-AAF-pAB) (EP24.15 activity, black bars).
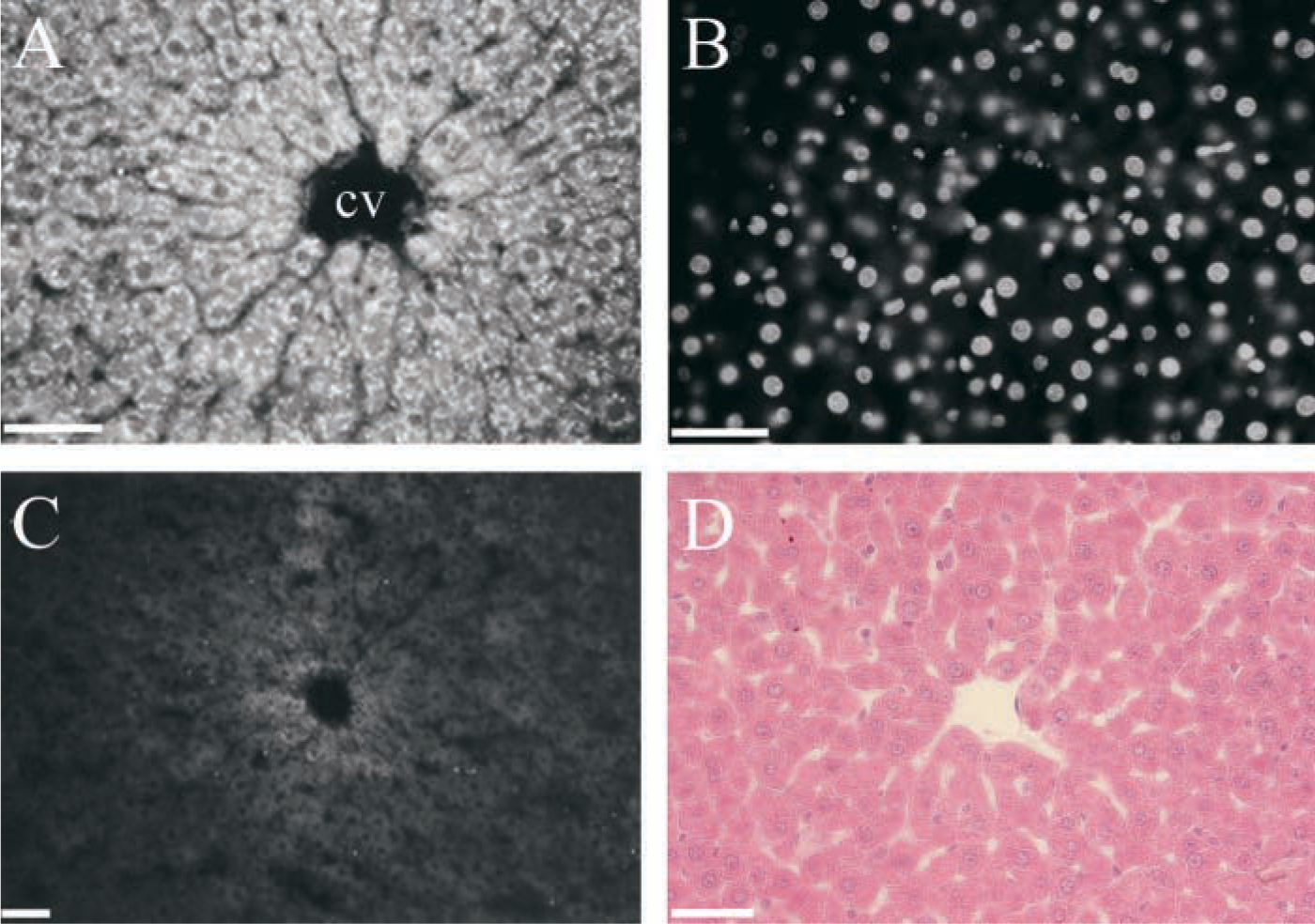
Indirect immunof uorescence of EP24.15 from rat liver. (
Immunohistochemistry demonstrated EP24.15 in the cytoplasm of hepatocytes (Figure 2A), mainly in those surrounding the centrolobular vein (zone 3). Other cell types, such as Kupffer cells and stellate cells, were not labeled. Although cells of this region receive fewer nutrients and less oxygenated blood supply compared to hepatocytes of zone 1, they are enriched with endoplasmatic reticulum and therefore have great metabolic activity. Observation with a UV filter (Figure 2B) to visualize DAPI nuclear staining ruled out the existence of EP24.15 immunoreactivity in the nucleus of the cells. We conclude that EC 3.4.24.15 is a cytoplasmic enzyme present mainly in hepatocytes of perivenous regions. This localization is relevant because most hepatotoxins induce necrosis which, in addition to ischemic hepatocellular injury, is predominant in zone 3.
Footnotes
Acknowledgements
Supported by grants from FAPESP 99/12435–2 and PRONEX (41.96.0873.00) and by FADA/UNIFESP.
We thank Dr E.S. Ferro (Biomedical Sciences Institute, USP, Brazil) who provided the rabbit antiserum against recombinant EP24.15; Dr L. Juliano (Biophysics Department, Unifesp, Brazil) for the fluorogenic substrate; and Dr M.J. Glucksman (Mount Sinai School of Medicine, NY) for the inhibitor of EP24.15; and Erika Suzuki for image editing.
