Abstract
Osteoprotegerin (OPG), a soluble member of the tumor necrosis factor (TNF) receptor family, is an osteoclastogenesis inhibitory factor. We investigated the localization of OPG in rat tibia using a specific peptide antibody to clarify the role of OPG in bone remodeling. OPG reactivity was mainly seen on bone surfaces. In bone matrices, OPG was also localized on cartilage/bone interfaces and cement lines. However, labeling was scarcely detected in the region of contact between osteoclasts and stromal cells. Some osteoblasts and osteocytes showed weak labeling. Immunoreactivity was not seen in chondrocytes or osteoclasts. Immunoelectron microscopic observation revealed that OPG is localized on the bone surfaces under osteoclasts. These findings suggest that OPG derived from osteoblast lineage cells and/or serum may be concentrated on resorbed bone surfaces and subsequently on cement lines. OPG may play an important role in the prevention of excess bone resorption by inhibiting differentiation and activity of osteoclasts in bone remodeling.
Bone volume is maintained through the balance of bone formation by osteoblasts and bone resorption by osteoclasts. Regulation of the balance between osteoblastic and osteoclastic activity is a critical component of normal bone cell biology. Osteoblasts differentiate from mesenchymal stem cells and produce bone matrix. Osteoclasts are multinuclear giant cells that arise from the monocyte-macrophage lineage of hematopoietic precursors (Nijweide et al. 1986; Suda et al. 1992). It has been suggested that cell-cell interaction between osteoblast lineage cells and osteoclast progenitors is essential for osteoclastogenesis (Ejiri 1983; Takahashi et al. 1988; Udagawa et al. 1989; Irie and Ozawa 1990; Amizuka et al. 1997). Recent research has revealed that receptor activator of nuclear factor (NF)-κB (RANK), expressed in osteoclast lineage cells and RANK ligand (RANKL) produced by osteoblast lineage and stromal cells participates in this cell-cell interaction (Udagawa et al. 1989; Lacey et al. 1998; Yasuda et al. 1998b; Hsu et al. 1999; Takahashi et al. 1999; Hofbauer et al. 2000).
Osteoprotegerin (OPG), a soluble member of the tumor necrosis factor (TNF) receptor family, works as a decoy by binding to RANKL and thus inhibits osteoclastogenesis by interrupting RANKL-mediated signaling (Simonet et al. 1997; Tsuda et al. 1997). Transgenic mice expressing high levels of OPG exhibit osteopetrosis resulting from a marked reduction in the number of osteoclasts (Simonet et al. 1997). In contrast, OPG knockout mice show severe osteoporosis associated with an increased number of osteoclasts (Bucay et al. 1998; Mizuno et al. 1998). Moreover, systemic administration of OPG produces an increase in bone density in normal rats (Yasuda et al. 1998a) and in OPG-deficient mice (Min et al. 2000), and also prevents bone loss in ovariectomized rats (Simonet et al. 1997). OPG mRNA is downregulated by stimulators of osteoclastogenesis such as 1,25-(OH)2 D3, prostaglandin E2 (PGE2), interleukin (IL)-1, and parathyroid hormone (PTH) (Murakami et al. 1998). Conversely, estrogen (Hofbauer et al. 1999), transforming growth factor (TGF)-β1 (Murakami et al. 1998), and bone morphogenetic protein (BMP)-2 (Hofbauer et al. 1998) increase OPG mRNA levels in osteoblastic cells. These findings strongly suggest that OPG mediates the effects of hormones and cytokines. Hence, OPG may serve as a physiological regulator of osteoclast-mediated bone resorption in the process of bone remodeling. Several in vivo studies have revealed the expression and the localization of OPG in the osteoblast lineage cells (Kobayashi et al. 2000; Onyia et al. 2000; Ikeda et al. 2001). However, the localization of OPG in bone matrices is still controversial.
We report here the immunolocalization by light and electron microscopy of OPG in rat tibia using a specific peptide antibody.
Materials and Methods
All animal procedures were in accordance with the Guidelines for Animal Experiments, Okayama University Graduate School of Medicine and Dentistry.
Antibody Against OPG
A cysteine-conjugated peptide corresponding to residues 233–247 (NAESVERIKRQHSSQ), which is a hydrophilic region in the death homologous domain of rat OPG (Yamaguchi et al. 1998), was synthesized and coupled via a terminal cysteine residue to keyhole limpet hemocyanin. This antigen was injected SC into rabbits and antiserum was collected. Specific antibody to OPG was purified with Affi-Gel 10 (Bio-Rad; Hercules, CA) coupled with the peptide.
Protein Extraction, SDS-PAGE, and Western Blotting
Tibias of 4-week-old rats were resected and bone segments of the proximal region were obtained. Enamel organs were also dissected out from lower incisors. Calvaria extracts were obtained from 2-day-old rats. Samples were dissolved in 100 μl of sample buffer containing 4% SDS, 20% glycerol, and 12% mercaptoethanol in 100 mM Tris-HCl (pH 6.8) and heated at 100C. We also used glutathione S-transferase (GST)-OPG as a positive control.
SDS-PAGE was carried out with a 12% polyacrylamide gel. Samples were electrophoresed at 150 V for 60 min and were then transferred to a nitrocellulose membrane using 192 mM glycine and 20% methanol in 25 mM Tris-HCl (pH 8.3) at a constant amperage of 50 mA for 60 min. The membrane was immersed in 10% skim milk in 10 mM Tris-buffered saline (TBS) for 30 min to block nonspecific binding and washed with TBS containing 0.05% Tween-20. The membrane was incubated with anti-OPG antibody (1 μg/ml) for 12 hr at 4C, followed by incubation with horseradish peroxidase (HRP)-conjugated anti-rabbit Ig G (Sigma; St Louis, MO) for 1 hr at room temperature (RT). Immunore-activity was visualized using ECL Western blotting detection reagents (Amersham Pharmacia Biotech UK; Poole, UK) according to the manufacturer's instructions.
Preparation of Tissue for Immunohistochemistry
Five 4-week-old Wistar rats were used for IHC. The rats were anesthetized with sodium pentobarbital and perfused through the left ventricle with 4% paraformaldehyde and 0.1% glutaraldehyde in 0.05 M phosphate buffer (pH 7.3). Tibias were resected, immersed in the perfusion fixative for 2 hr at 4C, and then decalcified in 5% EDTA (pH 7.3) for 1 week at 4C. Specimens from each rat were used for light and electron microscopic IHC.
Light Microscopic IHC
Specimens were dehydrated in graded ethanol and embedded in paraffin. One or two blocks obtained from each animal were cut with microtome. Seven-μm-thick sections were dewaxed with xylene and ethanol, immersed in PBS containing 10% bovine serum albumin (BSA) for 15 min, incubated in anti-OPG polyclonal antibody at a concentration of 5 μg/ml for 12 hr at 4C, and finally incubated in HRP-conjugated goat anti-rabbit Ig G diluted 1:100 for 1 hr at RT. After washing with PBS, immunoreactivity was visualized by immersion in a DAB-H2O2 solution (0.05% diaminobenzidine and 0.01% H2O2 in 0.05 M Tris-HCl buffer, pH 7.6) for 5 min at RT. For tartrate-resistant acid phosphatase (TRAP) detection, specimens were incubated in a mixture of 0.01% naphthol AS-BI phosphate (Sigma), 0.06% fast red violet LB salt (Sigma) and 50 mM
Electron Microscopic IHC
Specimens were dehydrated in graded N,N-dimethylformamide and then embedded in glycol methacrylate (GMA). Polymerization was performed under UV irradiation for 24 hr at 4C. At least one block from each rat was used for electron microscopic immunohistochemistry. Ultrathin GMA sections were cut with an Ultracut E ultramicrotome (Reichert-Jung; Vienna, Austria) and mounted on nickel grids. Specimens were immersed in PBS containing 10% BSA for 15 min, incubated in anti-OPG antibody at a concentration of 10 μg/ml for 12 hr at 4C, and finally incubated with 10-nm gold-conjugated goat anti-rabbit Ig G (British Biocell International; Cardiff, UK) diluted 1:40 for 1 hr at RT. They were then washed with PBS and distilled water and air-dried. Sections were stained with uranyl acetate and lead citrate or with 1% tannic acid and uranyl acetate. The samples were observed under a Hitachi H-800 transmission electron microscope (TEM) (Hitachi; Tokyo, Japan) at an accelerating voltage of 100 kV.
Controls
Control sections were incubated with preimmune serum or without any primary antibody. IHC procedures were performed in the same manner as for the non-control sections.
Results
Western Blotting Analysis
Western blotting analysis revealed that the OPG peptide antibody reacts with a 90-kD band in tibia and calvaria extracts. On the other hand, two bands, at 55 kD and 90 kD, were seen in enamel organ extract from lower incisors. GST-OPG, used as a positive control, reacted with this antibody and formed a band at 70 kD. This band is consistent with the size expected from GST-OPG (Figure 1).
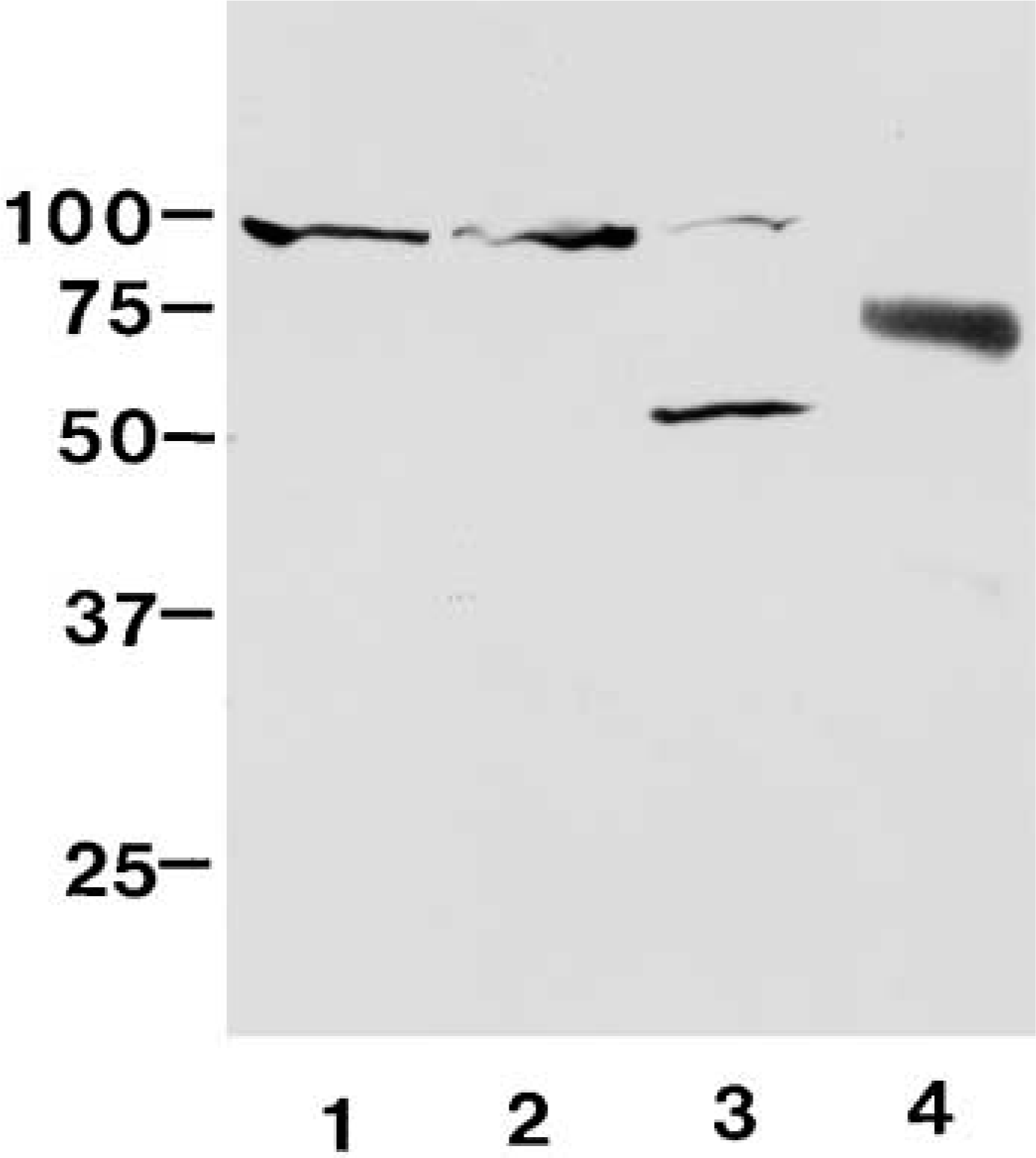
Western blotting with anti-OPG antibody under reduced conditions. Lane 1, tibia extract; Lane 2, calvaria extract; Lane 3, enamel organ extract; Lane 4, recombinant GST-OPG. OPG-positive band at 90 kD is detected in tibia and calvaria extracts. The 55-kD and 90-kD bands are seen in enamel organ extract. The antibody reacts with GST-OPG at 70 kD.
Light Microscopic IHC
We performed double labeling of OPG IHC and TRAP enzyme histochemistry to examine the relationship between OPG localization and osteoclasts.
In this report, laminar structures between new bone matrices and old ones are referred to as cement lines. Bone surfaces that have a concave shape and sometimes lead to the bone surface under osteoclasts are referred to as resorbed bone surfaces. In longitudinal sections of the proximal region of tibias from 4-week-old rats, many chondroclasts and osteoclasts were attached to cartilage matrices in the erosion zone, where blood capillaries invade and cartilage is replaced by bone. Very little OPG-positive labeling was detected in this region. OPG immunoreactivity was mainly detected on surfaces of trabecular bone slightly distant from the growth plate in metaphysis (Figure 2A). Many OPG-positive lines represented resorbed bone surfaces (Figure 2B). Weak labeling was seen in some osteoblasts and osteocytes (Figure 2C). However, osteoblast lineage cells did not always show positive labeling. Osteoclasts did not show any positive immunoreactivity for OPG without TRAP staining (Figure 2C).
In the trabecular bone of epiphyses, many OPG-positive lines were seen on the bone surface and in bone matrices. These lines in bone matrices corresponded to cement lines and cartilage/bone interfaces (Figure 2D).
Chondrocytes in epiphysial growth plate did not show positive immunoreactivity at any stage of differentiation. No reactivity was seen in cartilage matrix in the growth plate.
No specific immunoreactivity was detected in the control sections that were incubated without any primary antibody or with preimmune rabbit serum.
Electron Microscopic IHC
Under electron microscopy, many gold particles were detected under clear zones of osteoclasts (Figure 3) and resorbed bone surfaces. Gold particles were mainly distributed on the material that was slightly electron-dense. Sometimes a few gold particles were seen on the bone surface under ruffled borders. However, particles were scarcely seen in the regions of contact between osteoclasts and stromal cells.
In bone trabeculae of epiphyses, cartilage/bone interfaces were located between cartilage matrices, which contain thin collagen fibrils and amorphous ground substances, and bone matrices, which were filled with thick collagen fibrils. Cement lines were seen as lines among collagen-rich bone matrices and were only slightly stained with uranyl acetate in GMA sections. Gold particles were concentrated at cartilage/bone interfaces and cement lines (Figure 4).
Discussion
Our peptide antibody for OPG reacted with 90- and 55-kD bands in enamel organ extract by Western blotting. The 55-kD band corresponds to a monomer of OPG purified from human fibroblasts (Tsuda et al. 1997) and recombinant murine OPG produced in CHO cells (Simonet et al. 1997). However, we could not detect any other band except for the 90-kD band in tibia and calvaria extracts. These findings suggest that OPG in bone tissue may undergo different modifications, such as glycosylation, from OPG in other tissues. Although the possibility of crossreactivity of the antibody cannot be ruled out, no bone matrix protein that shows homology with our synthetic peptide sequence can be found in the database. Another possibility is that an OPG family member crossreactive with the anti-OPG antibody may be present in bone tissue.
The immunoreactivity we observed in osteoblasts and osteocytes is consistent with previous findings concerning OPG mRNA expression and OPG localization in osteoblast lineage cells (Yasuda et al. 1998a; Onyia et al. 2000; Ikeda et al. 2001). However, immunoreactivity in osteoblasts is not comparable with previous reports by Onyia et al. (2000). This difference may be due to differences in immunological characteristics between our OPG-peptide antibody and their antibody against recombinant human OPG. OPG mRNA has also been detected in various tissues other than bone, such as liver and kidney (Simonet et al. 1997; Yasuda et al. 1998a), and OPG proteins have been found in serum (Yano et al. 1999). We found that OPG is localized on resorbed bone surfaces and cement lines in bone matrices. We also detected OPG under clear zones of osteoclasts by electron microscopy. Although clear zones are believed to represent cell-matrix attachment sites and sealing structures during bone resorption, alendronate, one of the bis-phosphonates, localizes on the bone surface under osteoclasts (Sato et al. 1991). A tracer experiment using microperoxidase also demonstrated that exogenous microperoxidase infiltrates under ruffled borders and clear zones within 1 hr (Tanaka and Tanaka 1988). In addition, a network of bone canaliculi in bone matrices enables extracellular fluid to penetrate into lacunae of osteocytes (Nijweide et al. 1986). Howship's lacunae are not completely sealed against extracellular fluid. Moreover, it has been reported that serum proteins, such as α2HS-glycoprotein, osteopontin, and albumin, are absorbed in bone matrices (McKee et al. 1993; VandenBos et al. 1999). Hence, OPG synthesized by osteoblast lineage cells and/or derived from serum may be adsorbed on resorbed bone surfaces during and/or after bone resorption. Another possibility for the presence of OPG on the bone surface under the clear zone is that the osteoclast may have migrated over a previously resorbed bone matrix. In this case, OPG might prevent further bone resorption by osteoclasts.
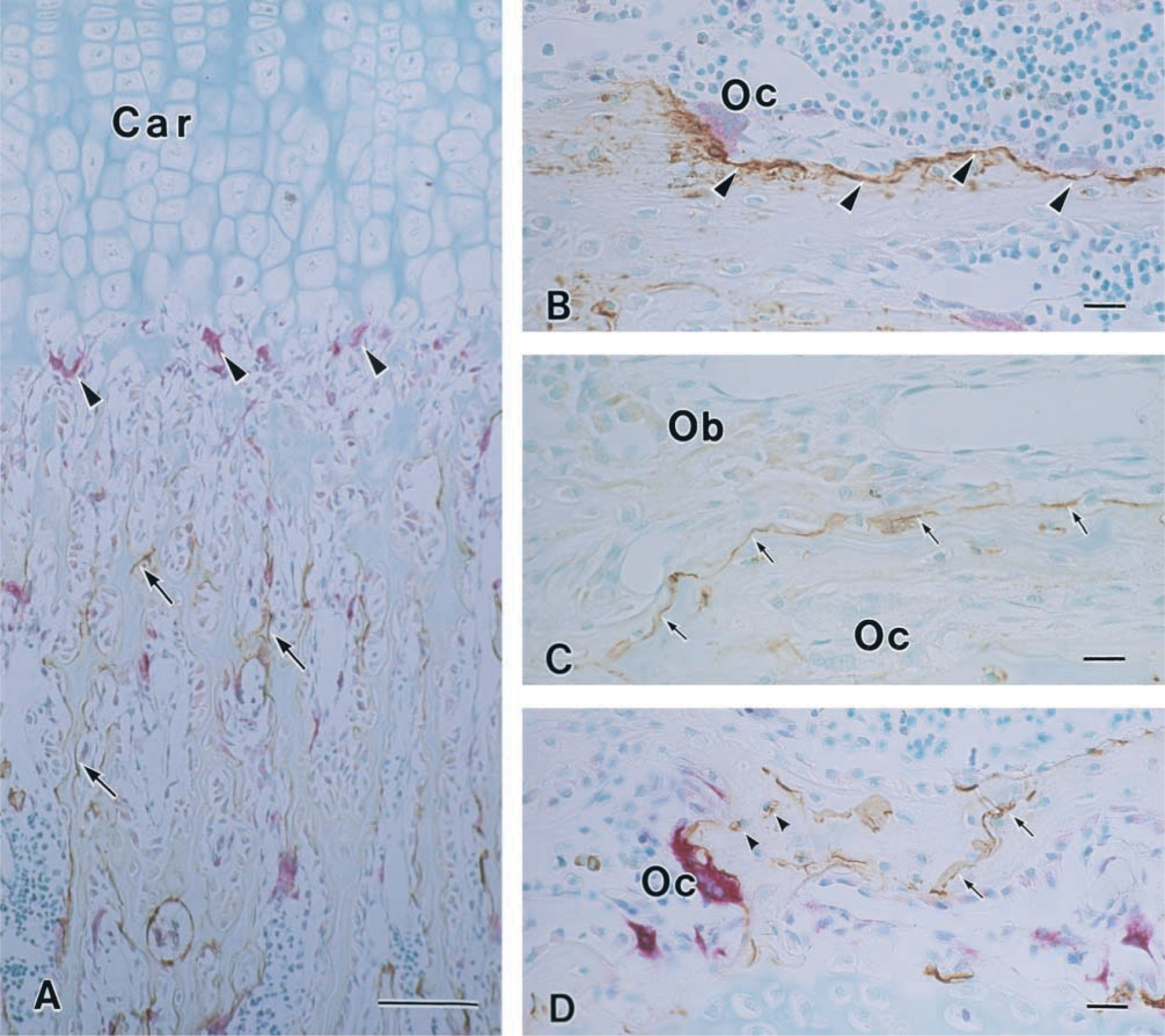
Light micrographs of the longitudinal sections of rat tibia.
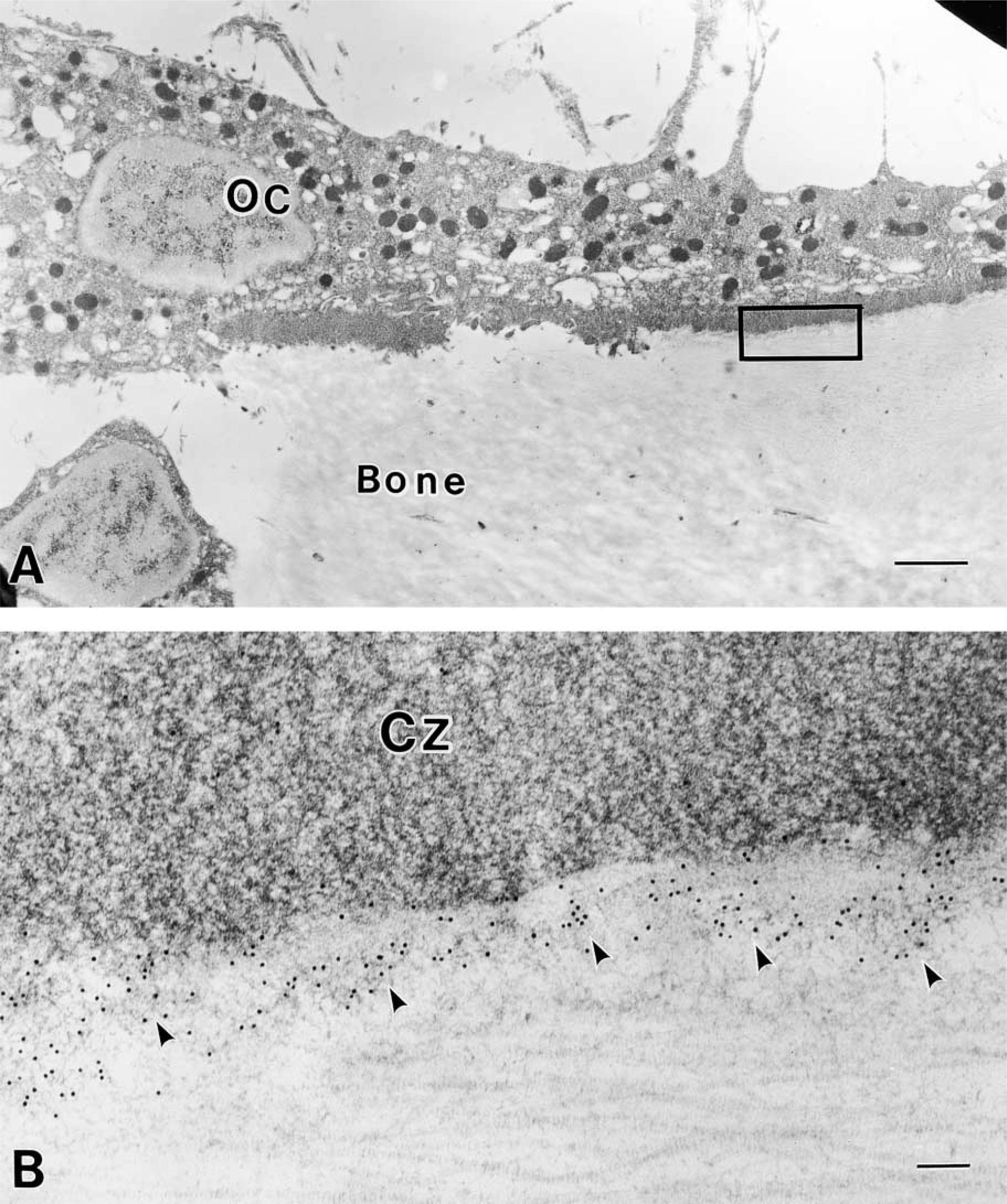
Electron micrographs indicating OPG localization.
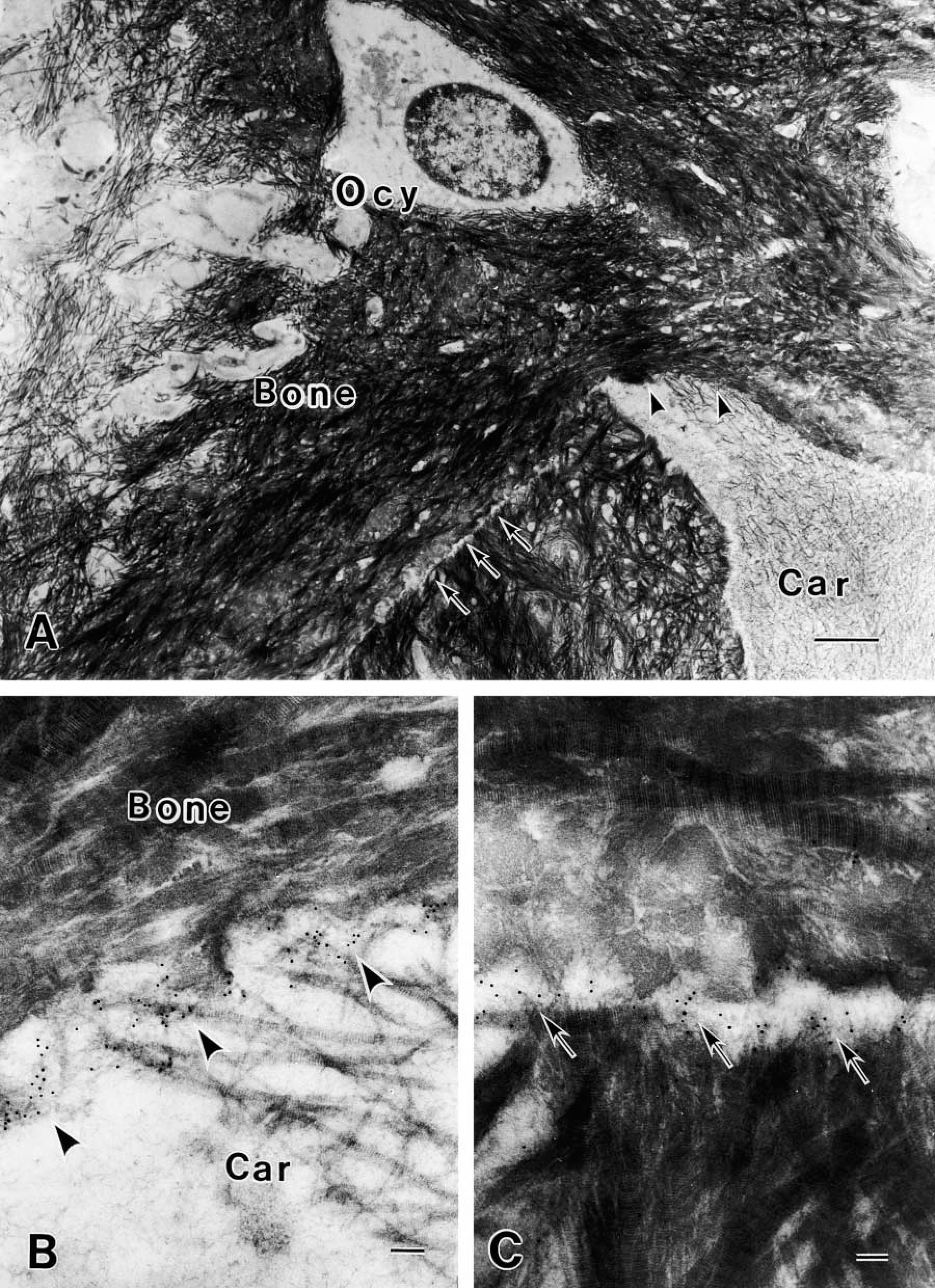
Immunolocalization of OPG in bone matrices.
There is a question of why OPG is bound to resorbed bone surfaces. Because OPG is a heparin-binding protein (Tsuda et al. 1997), one of the candidates is heparan sulfate proteoglycan (HSPG) in bone matrices. However, OPG distribution is different from that of heparan sulfate (HS) chains, as reported previously (Nakamura and Ozawa 1994). HS chains are mainly seen on the extracellular matrices between stromal cells and osteoclasts but not on resorbed bone surfaces, cartilage/bone interfaces, or cement lines, suggesting that OPG may not be bound to HSPG in bone matrices. Osteopontin and bone sialoprotein have been reported to localize on cement lines (McKee et al. 1993; McKee and Nanci 1995; Nanci 1999; Arana-Chavez and Nanci 2001). These highly charged proteins may be involved in the co-localization with OPG. Further investigation is necessary to determine whether OPG localizes on cement lines via specific binding to bone matrix proteins.
OPG is found as an osteoclastogenesis inhibitory factor that acts as a soluble decoy receptor to neutralize the RANK-RANKL interaction. OPG is believed to suppress bone resorption associated with osteoclast development. However, OPG has also been found to lower serum calcium levels in normal animals within 2 hr of administration (Yamamoto et al. 1998). Udagawa et al. (2000) reported the inhibitory effect of OPG on bone-resorbing activity in organ culture system. In addition, OPG appears to cause detachment of osteoclasts from bone surfaces (O'Brien et al. 2001), to directly inhibit resorption by osteoclasts (Hakeda et al. 1998), and to suppress osteoclast survival by promoting apoptosis (Akatsu et al. 1998; Kobayashi et al. 2000). These findings suggest that OPG may directly participate in the prevention of bone resorption by mature osteoclasts. On the other hand, OPG mRNA levels in MC3T3-E1 and ST2 cells appear to be stimulated by increasing concentrations of calcium, a condition found at sites of bone resorption (Yasuda et al. 1998a). Furthermore, OPG mRNA is downregulated by 1,25-(OH)2D3, PGE2, IL-1, and parathyroid hormone (Murakami et al. 1998), and is upregulated by estrogen (Hofbauer et al. 1999), TGF-β1 (Murakami et al. 1998), and BMP-2 (Hofbauer et al. 1998). This evidence indicates that the effects of multiple upstream hormones and cytokines converge on OPG. The balance between OPG and RANKL in a local environment might be an important determinant of osteoclastic bone resorption. Therefore, OPG on resorbed bone surfaces may provide a specific microenvironment that prevents excess bone resorption by inhibiting osteoclastogenesis and inactivating mature osteoclasts. Therefore, association of OPG with these structures may contribute to a local regulatory mechanism of bone remodeling.
Cement lines are believed to connect newly formed bone to older bone to maintain bone integrity and biomechanical strength (Schaffler et al. 1987). Resorbed bone surfaces subsequently become cement lines after bone formation. The activity of OPG in cement lines for next remodeling is uncertain because our antibody does not recognize the difference between the active and inactive forms. In bone remodeling, osteoclastic bone resorption is always followed by osteoblastic bone formation, suggesting that resorbed bone surfaces contain coupling factors that play a role in bone remodeling (Baron et al. 1984; Nakamura and Ozawa 1992). Although OPG has no effect on proliferation or differentiation of osteoblastic cells, such as MC3T3-E1, or on mesenchymal cells, at doses ranging from 0.1 to 100 ng/ml (Tsuda et al. 1997), a higher dose of OPG increases proliferation of human foreskin fibroblasts (Kwon et al. 1998). The possibility that OPG is a coupling factor needs to be addressed in future research.
Footnotes
Acknowledgements
Supported in part by a grant (no. 13671903) for scientific research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
We wish to thank Dr Noriyuki Nagaoka for technical support.
