Abstract
It has been previously reported that activin A, a homodimer of the βA inhibin subunit, is secreted by stromal cells from mouse bone marrow and causes apoptotic death of mouse plasmacytoma tumor cells. Recent in vitro studies have also implicated this cytokine in the suppression of normal B-cell lymphopoiesis. In this study we examined the occurrence of activin A in nasal polyp tissues that present a combination of epithelium, mesenchyme, and vascular endothelium, with frequent massive hemopoietic infiltration. Anti-βA-chain antibodies strongly stained epithelial mucous glands and some endothelial cells, and diffusely stained the polyp stroma. Normal adult conchae were similarly stained, whereas activin A was not detected prenatally by immunostaining of nasal tissues. Staining specificity was substantiated by ligand competition assays. Detailed examination of the inflammatory polyp infiltrate showed that activin A staining was reduced in sites of focal infiltration of B-lymphoid cells. It is therefore implied that local accumulation of a large number of B-cells is associated with relatively low activin A expression.
A
Activin A mRNA has been detected within the bone marrow stromal compartment (Yu et al. 1994) and is expressed by cultured stromal cell lines derived from mouse bone marrow. Stromal clones exhibiting an endothelial phenotype expressed a high activin A titer, whereas mesenchymal stromal cell lines produce activin A at a low spontaneous level (Zipori et al. 1986; Sternberg et al. 1995). This differential expression of activin A in various stromal cell lines and clones suggests that this cytokine may also be differentially expressed in situ. We found that the low expression of activin A in mesenchymal stromal cells could be up-regulated by external stimuli. Treatment of cultured stromal cells with basic fibroblast growth factor (bFGF) caused an increase in βA-chain mRNA (Sternberg et al. 1996). A recent study conducted in our laboratory (Shoham and Zipori 1998) showed that forced inhibition of activin A functions caused early onset of B-lymphopoiesis in long-term bone marrow lymphopoietic cultures (Whitlock and Witte 1982). It is therefore implied that activin A may be a physiological regulator of B-lineage cell generation.
To examine the possible role of activin A in regulating B-cell infiltration into inflamed tissues, we chose a model organ that presents all major types of tissue cells, i.e., mesenchyme, epithelium, and vascular endothelium, and concomitantly provides the basis for studying lymphohemopoietic infiltration. The human nasal polyp is an excellent model system for these investigations because it conforms histologically with the above criteria and often shows massive infiltrates of various hemopoietic cell types (Hellquist 1990). Nasal polyps are swellings of the lamina propria mucosa, a frequent pathology that is often treated by surgical removal of polyps that block airways, making this tissue abundant for investigation. The cause of polyposis is not entirely clear. Apart from chronic infections and allergy, asthma and cystic fibrosis have also been shown to be associated with increased incidence (Settipane 1996). The first stages of polyp formation are edema followed by proliferation of mesenchyme. Abnormal mucous glands form in about half of the polyps. The surface of the polyp facing the nasal cavity is covered with ciliated epithelium, which is often underlain by a thick basal lamina. This epithelium sometimes undergoes squamous metaplasia and hyperplasia of goblet cells (Hellquist 1990). The infiltrating cells in nasal polyps are mainly eosinophils, which are believed to play a major role in the development of this state, as well as mast cells, neutrophils, T-and B-lymphocytes, and plasma cells. A few cytokines have been detected in nasal polyps (Ohno et al. 1991, 1992; Elovic et al. 1994; Mullol et al. 1995; Eisma et al. 1997; Wang et al. 1997; Coste et al. 1998; Powers et al. 1998; Utgaard et al. 1998; Lam et al. 1999; Saji et al. 2000). However, it is not entirely clear which of them has a critical role in promoting or restricting the infiltration process.
In this study we mapped the expression pattern of activin A in nasal polyps and correlated this expression to the localization of B-lymphocytes. Abundance of B-cell agglomerates is demonstrated in sites exhibiting a relatively lower local expression of activin A.
Materials and Methods
Tissues
Nasal polyp tissues were obtained from 24 patients undergoing functional endoscopic sinus surgery for nasal polyposis at the Rabin Medical Center. Of these specimens, 19 were used for histological examination. No clear sex or age differences were observed in this group (10 female and nine male donors, 37–81 years old). Additional polyps obtained from five patients who presented last in this series were used for mRNA preparation. Normal nasal conchae specimens were obtained from eight patients undergoing septoplasty with no history of nasal polyposis. Prenatal nasal tissues were obtained from five natural miscarriages. Samples from all patients were immediately fixed in formalin and then paraffin-embedded. The paraffin blocks were stored at room temperature (RT). At least 10 5-μm serial sections were obtained from each sample and evaluated by a pathologist. Samples from five patients (30–100 mg) were homogenized using a Polytron PT1000 for mRNA preparation.
Immunohistochemistry
Paraffin-embedded sections were deparaffinized in xylene and rehydrated in graded alcohol (100%, 95%, 80%, 60%). The immunohistochemical procedure was performed with the ABC kit (Novocastra; Newcastle upon Tyne, UK), allowing detection of the antigen–antibody reaction with a biotinylated avidin amplification and a peroxidase detection system using the substrate diaminobenzidine tetrahydrochloride (DAB, code no. D-5905; Sigma, Rehovot, Israel) for brown color or the chromogen aminoethyl carbazole (AEC, code no. 00-1111; Zymed, San Francisco, CA) for red color. Different primary antibodies were used: a monoclonal antibody (MAb) (mouse IgG2b) anti-human βA-subunit of inhibin and activin (diluted 1:50, code no. MCA950S (Serotec; Oxford, UK); MAb (mouse IgG2a) anti-human α-subunit of inhibin, diluted 1:10, code no. MCA951S (Serotec); mouse IgG2b negative control, diluted 1:50, code no. MCA691 (Serotec; MAb mouse anti-α-smooth muscle actin (αSMA), ready to use, code no. 08-0106 (Zymed Laboratories); rabbit anti-human fibronectin, diluted 1:200, product no. F3648 (Sigma); mouse anti-human B-cells, clone L-26 (Zymed); MAb mouse anti-human T-cells (UCHL1) (Diagnostic Products; Los Angeles, CA).
For mouse IgG we used the secondary antibody supplied in the kit. For rabbit IgG we used goat anti-rabbit IgG biotinylated, diluted 1:500, code no. 111-065-144 (Jackson ImmunoResearch Laboratories; West Grove, PA). All dilutions were performed in PBS without Mg+2 and Ca+2. Samples were counterstained with Mayer's hematoxylin, washed extensively in tapwater, dehydrated in graded alcohol (95%, 100%), air-dried, and mounted in Entellan (code no. 107961.0100; Merck, Darmstadt, Germany) for DAB or in GVA mounting solution, code no. 00-8000 (Zymed) for AEC.
Competition Assay
During the blocking stage, the first antibodies were preincubated in test tubes with the indicated peptide (26 ng/section) for 30 min at RT and then the mixture was added to the sections for binding.
RT-PCR
Total RNA from homogenized nasal polyp tissues, 14F1.1. cells, and Balb/c mouse testis tissue was collected in TRI reagent (Molecular Research Center; Cincinnati, OH). RNA was reverse-transcribed and PCR-amplified. The following primers were used: inhibin αA-subunit, sense 5′-CAGCCCAGCTGTGGTTCC-3′; antisense 5′-TTGGAAGGAGATATTGAGAGC-3′ (Albano et al. 1994); activin βA-subunit, sense 5′-GGCTTGGAGTGTGATGGC-3′; antisense 5′-GCAGCCACACTCCTCCACAAT-3′ (Tone et al. 1990); β-actin, sense 5′-GAGCACCCTGTGCTGCTCACCGAGG-3′; antisense 5′-GTGGTGGTGAAGCTGTAGCCACGCT-3′ (Tokunaga et al. 1986). Each PCR reaction was performed using about 1 μg mRNA and 1.5 U Taq Polymerase (Promega; Madison, WI). The PCR mixture was heated at 94C for 5 min, followed by 35 cycles of 94C for 30 sec, 60C for 30 sec, and 72C for 1 min, and a final extension at 72C for 8 min.
Results
Detection of Inhibin βA-chain in Nasal Polyps
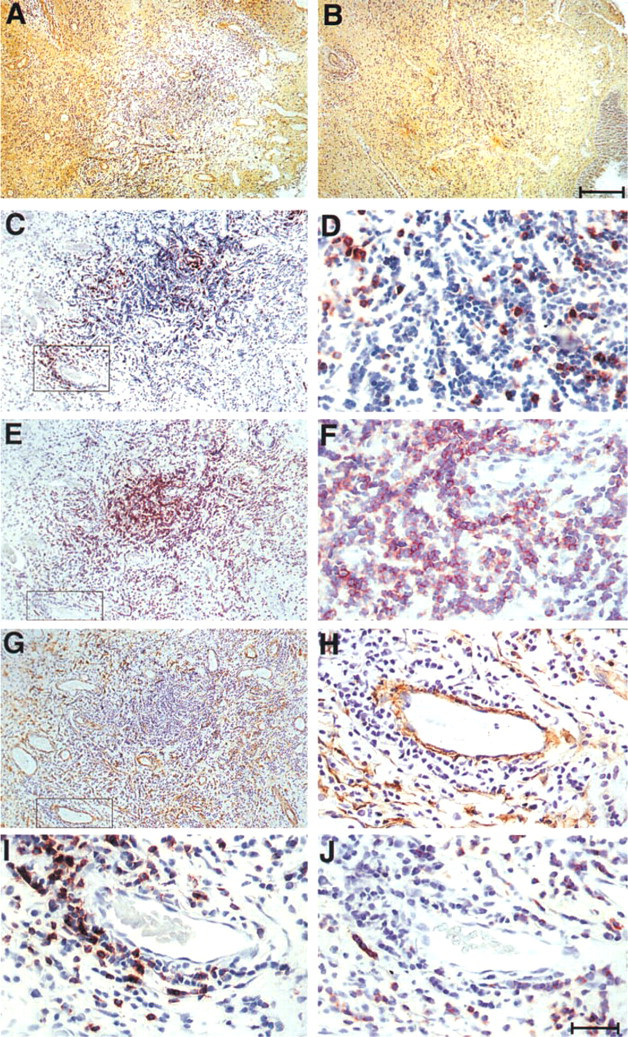
Antibodies to the βA-chain of inhibin have been used previously to detect the homodimer of this chain, activin A, in gonadal tissues where this protein is abundant (Groome and Lawrence 1991), as well as in body fluids, using ELISA determinations (Groome and Lawrence 1991; Groome and O'Brien 1993). On the other hand, only sparse information is available on the tissue expression of this molecule. We investigated the occurrence of immunologically detectable βA-chain in nasal polyps from individuals after surgical removal. The anti-βA antibody detected clear staining in the respiratory-type epithelium of the polyp (Figure 1A and 1B) and strongly stained cells within epithelial glands (Figure 1D) relative to the mesenchymal stroma, which was stained less intensely. However, the staining of the epithelial glands was variable, such that some of the glands gave only faint staining with antibodies to the βA-chain. Figure 1 (A vs C) shows that anti-α-smooth muscle actin (αSMA) highlights blood vessels, whereas activin A delineates the epithelium and is not found in the vascular tissue. The specificity of the staining was ascertained by comparison to an isotype control antibody that did not give any background staining, in contrast to the compartmentalized staining of anti-βA. To further substantiate the specificity of βA chain detection, we used the immunizing peptide originally used to produce the anti-βA antiserum (Groome and Lawrence 1991) in competition assays performed on polyp tissue sections. The tissues were either exposed to the antibody on its own (Figure 2A and 2D) or to the antibody in combination with the immunizing peptide (Figure 2B and 2E) as detailed in Materials and Methods. The immunizing peptide completely abolished the staining with the antibody. On the other hand, a peptide that is not recognized by the antibody in ELISA (data not shown) did not interfere with staining (Figure 2C and 2F). Specific antibodies to the inhibin α-chain did not detect the corresponding antigen in nasal tissues (data not shown). The above immunohistochemical detection of the βA-chain vs the absence of inhibin α-chain in nasal polyps was further substantiated by semiquantitative RT-PCR analysis. As shown in Figure 3, analysis of mRNA in randomly selected nasal polyps from five individuals showed clear βA expression in all samples, and the strength of the signal was equivalent to that observed in a positive control testis mRNA. On the other hand, no indication for α-chain expression was obtained.
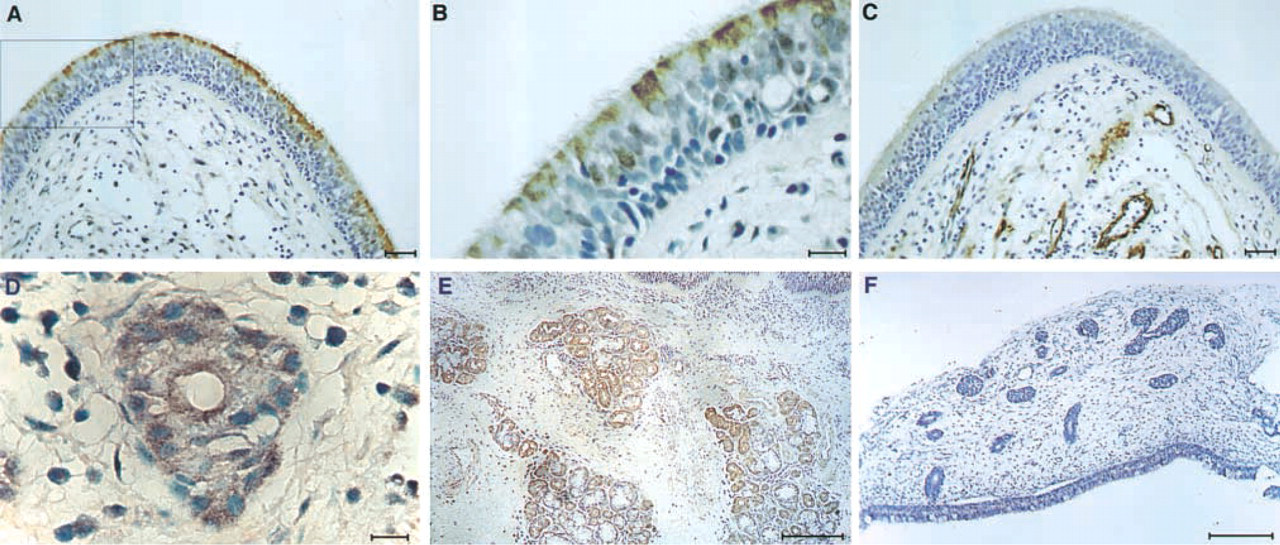
βA-chain expression in nasal tissues. Tissue sections were stained with antibodies to βA-chain (
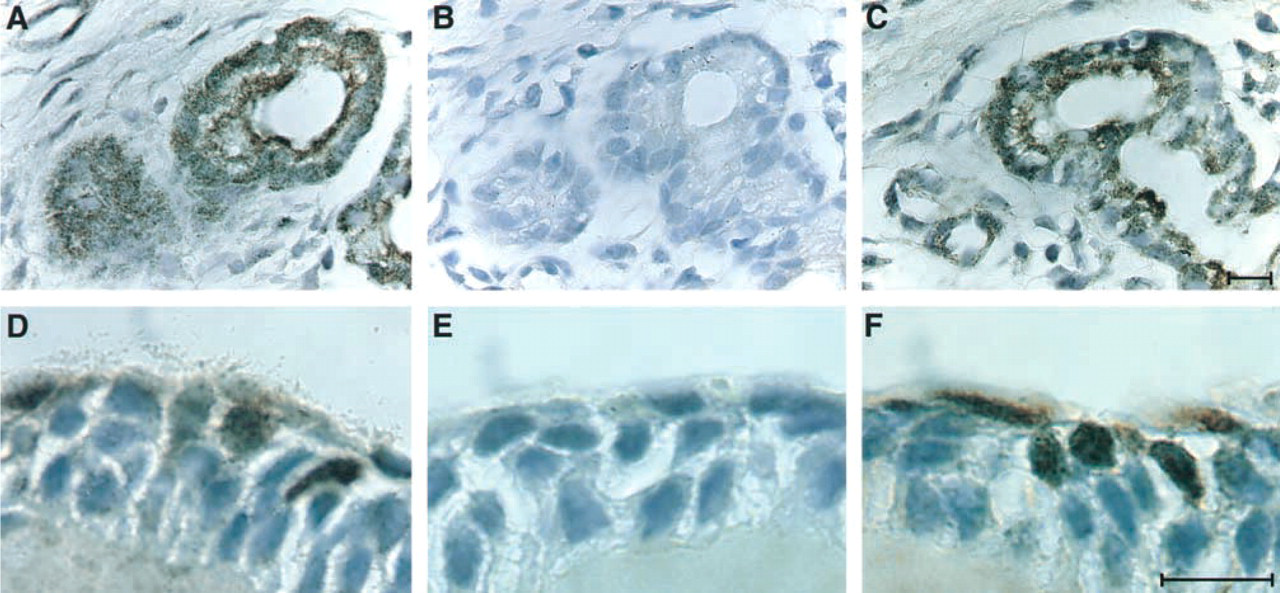
Specificity of anti-βA antibody staining demonstrated by competition with the immunizing antibody. Serial sections were stained with anti-βA antibodies in the absence of competing peptides (
Once the specificity and sensitivity limits of the detection system were established, we could turn to the study of normal tissues. Prenatal nasal tissues were obtained from five naturally aborted embryos. Tissue sections were stained with anti-βA antibodies as detailed above and consistently showed no measurable signals (Figure 1F). This finding is in sharp contrast to adult normal nasal concha, which shows clear βA staining in epithelial mucous glands (Figure 1E).
βA-chain Staining Is Excluded from B-lymphoid Agglomerates Infiltrating Nasal Polyp Tissues
We noted that the distribution of activin A within the nasal polyp, as detected by the anti-βA antibodies, was not homogeneous. Although wide areas of diffuse staining were commonly observed, these were often interrupted by unstained domains, which were occupied by infiltrating B-lymphocytes. Table 1 summarizes 19 individual nasal polyp samples examined. Figure 4 shows detailed analysis of one representative case, a relatively diffuse B-cell cluster demonstrated by staining with antibodies to human B-lymphocytes (Figure 4E and 4F). This cluster contains only a few T-cells (Figure 4C and 4D). Staining with anti-βA antibodies indicated that this B-cell cluster is positioned within an area that has reduced antibody staining (Figure 4A). A higher-magnification panoramic view of an area within the lymphocyte agglomerate showed that although this area had lower staining with the antibody, nevertheless positive staining for activin A was present (Figure 5). Because antibodies to fibronectin also produced a diffuse staining similar to that obtained with anti-βA antibodies, we used this antibody as a control. In contrast to anti-βA, antibodies to fibronectin produced a relatively uniform staining throughout the nasal polyp, including the B-lymphocyte cluster areas (Figure 4B; Table 1). Figure 4 further shows an area outside of the B-cell cluster adjacent to a blood vessel. This area, in which a relatively high level of βA-chain is found, contains mainly T-cells in the perivascular space (Figure 4C and 4I) and a few B-cells (Figure 4E and 4J). This differential accumulation of B- vs T-cells was also a common occurrence in more compact agglomerates containing predominantly B-cells positioned close to epithelial glands. βA-chain staining is pronounced in the glands, whereas within the agglomerate the staining is diffuse and barely detectable (not shown).
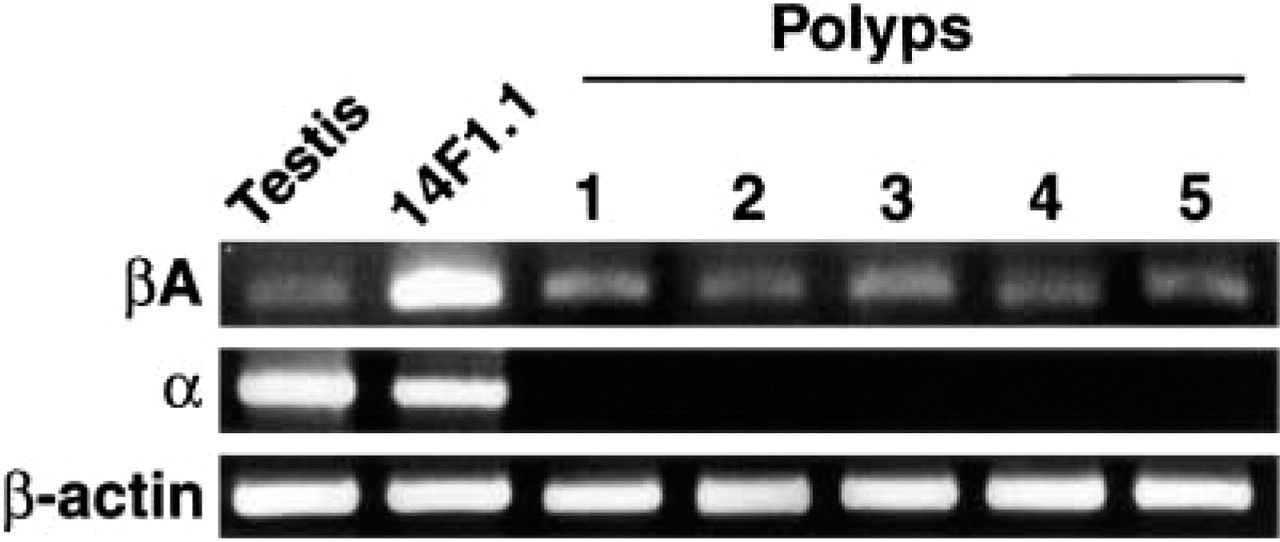
Semiquantitative RT-PCR analysis of mRNA from nasal polyps. mRNA from testis and from the 14F1.1 stromal cell line of mouse bone marrow served as positive controls for expression of the βA- and α-chain transcripts. The sizes for PCR products are as follows: activin βA-subunit, 433 bp; inhibin α-subunit 344 bp; and β-actin 311 bp.
Analysis of activin A and fibronectin expression in correlation with morphological features of lymphocytic infiltrationa
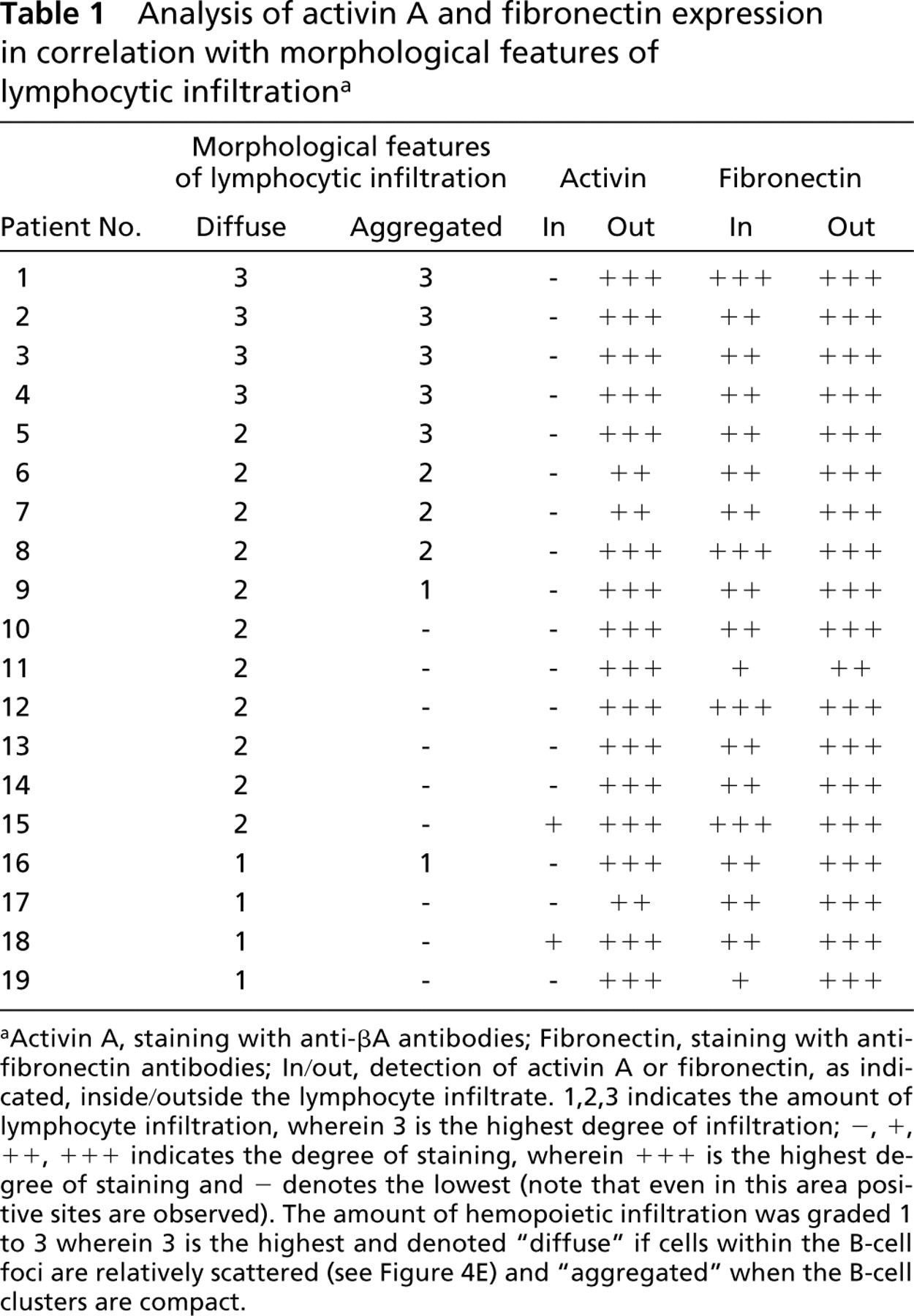
Activin A, staining with anti-βA antibodies; Fibronectin, staining with anti-fibronectin antibodies; In/out, detection of activin A or fibronectin, as indicated, inside/outside the lymphocyte infiltrate. 1,2,3 indicates the amount of lymphocyte infiltration, wherein 3 is the highest degree of infiltration; –, +, + +, + + + indicates the degree of staining, wherein + + + is the highest degree of staining and – denotes the lowest (note that even in this area positive sites are observed). The amount of hemopoietic infiltration was graded 1 to 3 wherein 3 is the highest and denoted “diffuse” if cells within the B-cell foci are relatively scattered (see Figure 4E) and “aggregated” when the B-cell clusters are compact.
Discussion
A common feature of nasal polyps is infiltration of lymphohemopoietic cells, including eosinophils, neutrophils, mast cells, T- and B-lymphocytes, and plasma cells, which may contribute to the pathogenesis of this hyperplastic and inflammatory process. The migration of blood-borne cells into the developing polyp is likely to be controlled by chemokines. Among this family of regulatory molecules, IL-8 was shown to be rapidly released from polyp endothelial cells after histamine treatment (Utgaard et al. 1998), while being inhibited by dexamethasone treatment (Mullol et al. 1995). Tumor necrosis factor (TNF)α and IL-1 were found to induce the expression of the chemokine RANTES in cultured polyp fibroblasts (Saji et al. 2000). On adhesion and invasion into the polyp mesenchymal tissue, the infiltrating cells probably survive and proliferate under the influence of locally produced IL-3 (Rudack et al. 1999), granulocyte macrophage colony-stimulating factor (GM-CSF) (Ohno et al. 1991), and vascular endothelial growth factor (VEGF) (Coste et al. 2000). IL-5 is expressed by eosinophils and induces their growth, probably in an autocrine manner (Rudack et al. 1999). Among the cytokines of the TGFβ superfamily, TGFβ1, TGFβ2, and TGFβ3 have thus far been identified in nasal polyps. Because polyposis entails fibroblast proliferation, fibrosis, and thickening of the epithelial basal lamina, it has been proposed that these processes may be supported by the action of TGFβ1, which is known to stimulate deposition of extracellular matrix (ECM) proteins. TGFβ1 mRNA was indeed detected by in situ hybridization in polyp eosinophils, and the protein was shown by immunocytochemical staining to be detectable in eosinophils, ECM, and stroma (Ohno et al. 1992; Eisma et al. 1997; Wang et al. 1997; Lam et al. 1999).
To the best of our knowledge, the expression of activin A in polyps has not yet been reported. We found that activin A is detectable by immunocytochemical staining primarily in the epithelial component of the polyp but also is abundant in the mesenchymal stroma. One of the cytokines reported to be prevalent in polyps is basic fibroblast growth factor (bFGF) (Powers et al. 1998). We have previously shown that bFGF up-regulates the expression of activin A mRNA in stromal cells (Sternberg et al. 1996) and, more recently, that this cytokine promotes the secretion of functional activin A by mesenchymal stroma (Shoham et al. in press). It is therefore likely that the high titer of activin A in the polyps is maintained by bFGF signaling. Indeed, bFGF has been found by immunostaining to be expressed in polyp sites similar to those described by us as areas highly expressing activin A (Powers et al. 1998).
We carefully verified the validity of the immunocytochemical staining for the βA-chain. It is important to note that the titer of activin A in the tissue was too low to be detected by direct immunostaining and that a highly sensitive method had to be employed. To ascertain the specificity of the staining, we first used isotype control antibodies that did not significantly stain the polyp sections. The anti-βA-chain antibody used has been raised against a synthetic peptide corresponding to 32 amino acids in the C-terminus of the βA-chain sequence (Groome and Lawrence 1991). We used this immunizing peptide, as well as a peptide which was not identified by the antibody, in competition assays that reinforced the specificity of the βA-chain detection. One major problem in identification of activin A protein is that it is a dimer of the βA-chain which is shared by the inhibitor of activin A functions, inhibin A (α/βA). Therefore, positive immunostaining may indicate the existence of either activin A or inhibin A. We therefore used anti-α-chain antibodies to specifically examine the expression of α-chain in polyps. No significant staining was obtained, in contrast to a strong positive control of mouse testis tissue (results not shown). Therefore, human polyps do not appear to harbor inhibin A detectable by immunostaining, indicating that the staining observed with anti-βA-chain antibodies is related to tissue activin A. Further to detection of activin A within the polyp tissue, we show that the expression of this molecule is clearly compartmentalized. Although activin A was widely expressed in most polyps examined, it was found in a reduced amount, as judged by immunostaining, in particular sites that were rich in B-cells. Normal conchae were positively stained with antibodies to βA-chain and had a low number of infiltrating hemopoietic cells. In addition, more than half of the polyps listed in Table 1 had no lymphocyte agglomerates but were intensely stained with antibodies to βA. Therefore, we found no correlation between activin A expression and occurrence of B-cells. It is impossible, at this stage of the research, to determine whether the reduced expression of activin A is a cause of B-cell accumulation or, alternatively, whether it is a result of this process. B-cells may elaborate signals that suppress activin A expression or reduce its binding to the ECM. The finding that activin A expression is upregulated in the nasal mucosa in adults vs embryos may imply that the expression of activin A has a protective function against B-cell infiltration. Therefore, before birth there may be no need for high expression of activin A in the nasal tissue, whereas after birth the mucosae facing the nasal cavity encounter constant irritation and are therefore prone to inflammation, infection, and over-infiltration of hemopoietic cells. In this respect, it is interesting to note that activin A is expressed by the airway epithelium and may be secreted by it. Attempts to demonstrate biologically functional activin A in nasal secretions failed owing to the toxicity of these secretions to the indicator cells used in the bioassay (not shown).
A B-lymphocyte focus in a nasal polyp area exhibiting reduced βA chain expression. Serial sections were stained with antibodies to βA-chain (
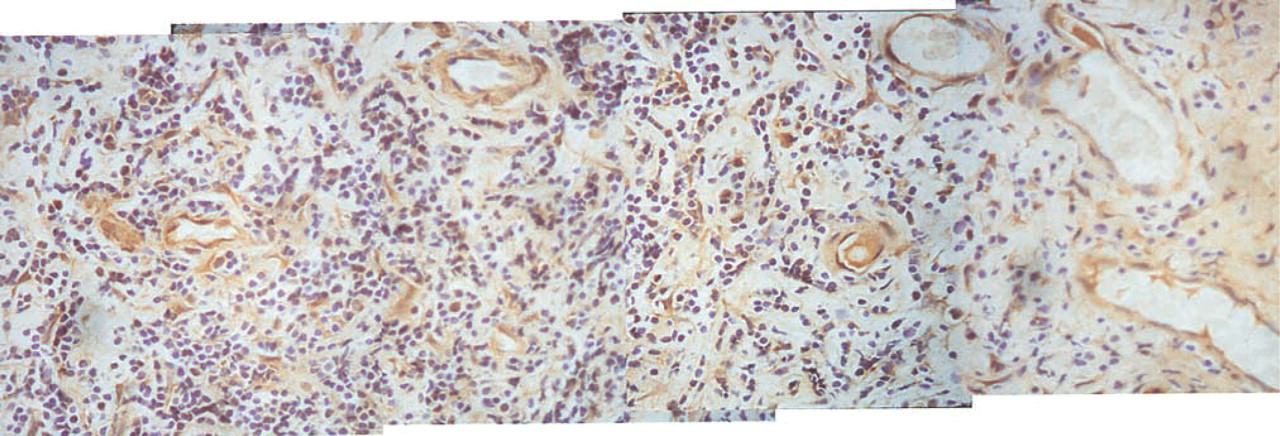
Composite panoramic view of a section within a B-lymphocyte focus (taken from Figure 4A) showing details of activin A expression in the lymphocyte agglomerate.
The unequal distribution of activin in the tissue may point to a morphogenic function of this molecule. In invertebrates, the TGFβ superfamily molecules often serve as morphogens (Pages and Kerridge 2000). Thus, decapentaplegic (DPP) is associated in Drosophila with wing development and patterning (Lecuit et al. 1996; Nellen et al. 1996). Similarly, the activin A gradient has a major role in Xenopus development (Gurdon et al. 1994; McDowell et al. 1997). In vertebrates, bone morphogenic proteins (BMPs), a subfamily within the TGFβ superfamily, determine bone development, and mutation in these genes or experimental knockouts lead to distorted bone formation (Hofmann et al. 1996; Kawakami et al. 1996). Similarly, knockout of the βA-chain causes defects in craniofacial development (Matzuk et al. 1995a,b). Several recent studies suggest a role for activin A in mammalian tissue patterning (Ferguson et al. 1998; Song et al. 1999; Kim et al. 2000). We propose that differential expression of members of the TGFβ superfamily in hemopoietic tissues is contributing to pattern formation due to their capacity to inhibit cell growth and other cell processes.
Footnotes
Acknowledgements
Supported by the Israel Science Foundation, founded by the Israel Academy of Science and Humanities, and by a research grant from the Kekst Family Center for Medical Genetics.
We thank Ms Hana Gertzman for technical assistance and Ms Rosalyn Silver for editing the manuscript. Dov Zipori is an incumbent of the Joe and Celia Weinstein professorial chair at the Weizmann Institute of Science.
