Abstract
Keywords
M
In addition to autophagic vacuoles, several other intracellular vacuoles containing high amounts of lipids occur in different cell types. These organelles are called lamellar bodies because the lipids are mostly organized in lamellar whirl-like sheets, as are observed in autophagic vacuoles (Schmitz and Müller 1991). Lamellar bodies are cell type-specific and have functions in lipid secretion and lipid metabolism. Well-known examples are keratinosomes, which are secreted from keratinocytes in the stratum granulosum of the skin stratified epithelium (Odland 1959; Wolff and Holubar 1967), or secretory vesicles in Type II pneumocytes containing surfactant (Goerke 1974; Weibel and Gil 1968). However, there is a variety of other lamellar bodies that occur under physiological or pathophysiological conditions in specific cell types (reviewed in Schmitz and Müller 1991).
Because MDC associates with acidic and lipid-rich compartments, it is unlikely that this substance can clearly discriminate among lysosomes, autophagic vacuoles, and lamellar bodies. The aim of this study was to modify the MDC molecule so as to skip the lysosomotropic property but to preserve the function of a solvent polarity probe. To achieve this, we replaced the primary amino group of monodansylcadaverine with a neutral (dansylamylamine; MDH), a polar (dansylaminopentanol; MDOH), or an acidic group (dansylaminovaleric acid; MDA). We show that only MDH stains vacuolar structures that are autophagic vacuoles, that this staining occurs in vivo and in vitro, and that it is independent of pH gradients. Therefore, we propose that MDH is a specific marker for autophagic vacuoles and probably for lamellar bodies in general.
Materials and Methods
Materials
Pentamines were purchased from Aldrich (Steinheim, Germany). All other chemicals used were obtained from Sigma (Diesenhofen, Germany).
Staining, Synthesis, and Characterization of Dansylated Pentamines
Dansylation of different pentamines was performed according to Creveling and Daly (1971), with minor modifications. In brief, 400 μl of a 110 mM sodium bicarbonate solution, pH 8.3, containing 200 μM of either amine (amylamine, 5-amino-1-pentanol, 5-aminovaleric acid, 1-aminoethan, 1-aminopropane; 1-aminobutane, 1-aminohexane, 1-aminoheptane, 1-aminooctane) was mixed with 1.6 ml of ice-cold acetone containing 50 μM dansyl chloride. The mixture was mechanically shaken for 8–10 hr at 4C until the characteristic orange color of dansyl chloride disappeared. The solute was removed in vacuum and the pellet resuspended in 1 ml DMSO. Purity was determined by thin-layer chromatography (TLC) using the upper phase of water:butanol:acetic acid (5:4:1 v:v:v) as mobile phase and high performance thin-layer chromatography (HPTLC) plates with silica gel 60 (Merck; Darmstadt, Germany) as stationary phase. Plates were analyzed with a LCD video camera (DIANA chemiluminescence module with UV transilluminator; Raytest, Straubenhardt, Germany). Concentrations of each fluorochrome stock solution were determined by fluorescence photometry with the fluorescence photometer SFM 25 (Kontron Instruments; Zurich, Switzerland). Monodansylcadaverine (MDC) was used as a standard. Excitation wavelength was set to 335 nm and emission wavelength to 525 nm unless otherwise indicated. Amplification by a high voltage setting was chosen individually in each experiment to record data within the linear scale of the photometer between relative fluorescence values (RF) of 10 and 120. Therefore, RF can differ in similar experiments.
All experiments were performed using the cell line PaTu 8902, which was established from a human primary pancreatic adenocarcinoma (Elsässer et al. 1993). Cells were cultivated to approximately 70% confluency as described elsewhere (Niemann et al. 2000). To determine uptake, the dansylated substances were added to the medium in a concentration of 0.1 mM. Cells were further incubated at 37C for the time points indicated. Subsequently, cells were washed three times with ice-cold PBS, scraped, and resuspended in 1 ml 10 mM Tris-HCl, pH 8.0. To destroy intracellular pH gradients, cells were incubated for 10 min before and during further incubation with 50 mM ammonium chloride. Before quantification of MDH, fluorescent cells were broken by passing them seven times through a Microlance 3 needle (Becton Dickinson; Heidelberg, Germany). To equalize cell numbers between different samples within one experiment, the amount of DNA was determined using Hoechst Dye 33258 (Polysciences; Warrington, PA) as described elsewhere (Niemann et al. 2000).
Fluorescence Microscopy
Staining and fixation of PaTu 8902 cells using MDC in a final concentration of 0.1 mM were described elsewhere (Biederbick et al. 1995). The other dansylpentamine derivatives were used in the same concentration as described for MDC. Cells were analyzed using a Zeiss inverted fluorescence microscope IM35 equipped with a long-distance ×40 lens and filter sets 01 and 15. To stain the lysosomal compartment with LysoTracker Red (Molecular Probes; Eugene, OR), cells were incubated for at least 30 min at a final concentration of 20 nM of the dye. Acridine Orange staining experiments were performed as described elsewhere (Niemann et al. 2000). To compare the staining pattern of MDC and MDH in vivo and in vitro, PaTu 8902 cells were cultivated on coverslips (Bachofer; Reutlingen, Germany) for 1 day and incubated with 2 μM MDH or MDC for 1 hr. Cells were washed with PBS and the coverslips were transferred into the closed perfusion open closed chamber (POC; Bachofer). Cells were analyzed using a Zeiss inverted fluorescence microscope IM35 equipped with a ×63 lens, followed by fixation with 2.5% paraformaldehyde in PBS at a flow rate of 0.1 ml/min for 20 min. After fixation, cells were washed with PBS (flow rate 0.5 ml/min for 20 min) and bleached under UV light for 5 min. The bleached cells were re-stained using 100 μM MDH or MDC (flow rate 0.1 ml/min; 5 min), washed as described above, and again analyzed by fluorescence microscopy.
Subcellular Fractionation of MDC/MDH-labeled Compartments
Fractionation was performed as described elsewhere (Biederbick et al. 1995), using a continuous sucrose gradient (15–40%).
Purification of Autophagic Vacuoles
Purification of autophagic vacuoles was performed as described previously (Niemann et al. 2000). Entire vacuoles were used for further analysis. In vitro staining of autophagic vacuoles was performed as described in Niemann et al. (2000), using 5 μM of the fluorochromes indicated.
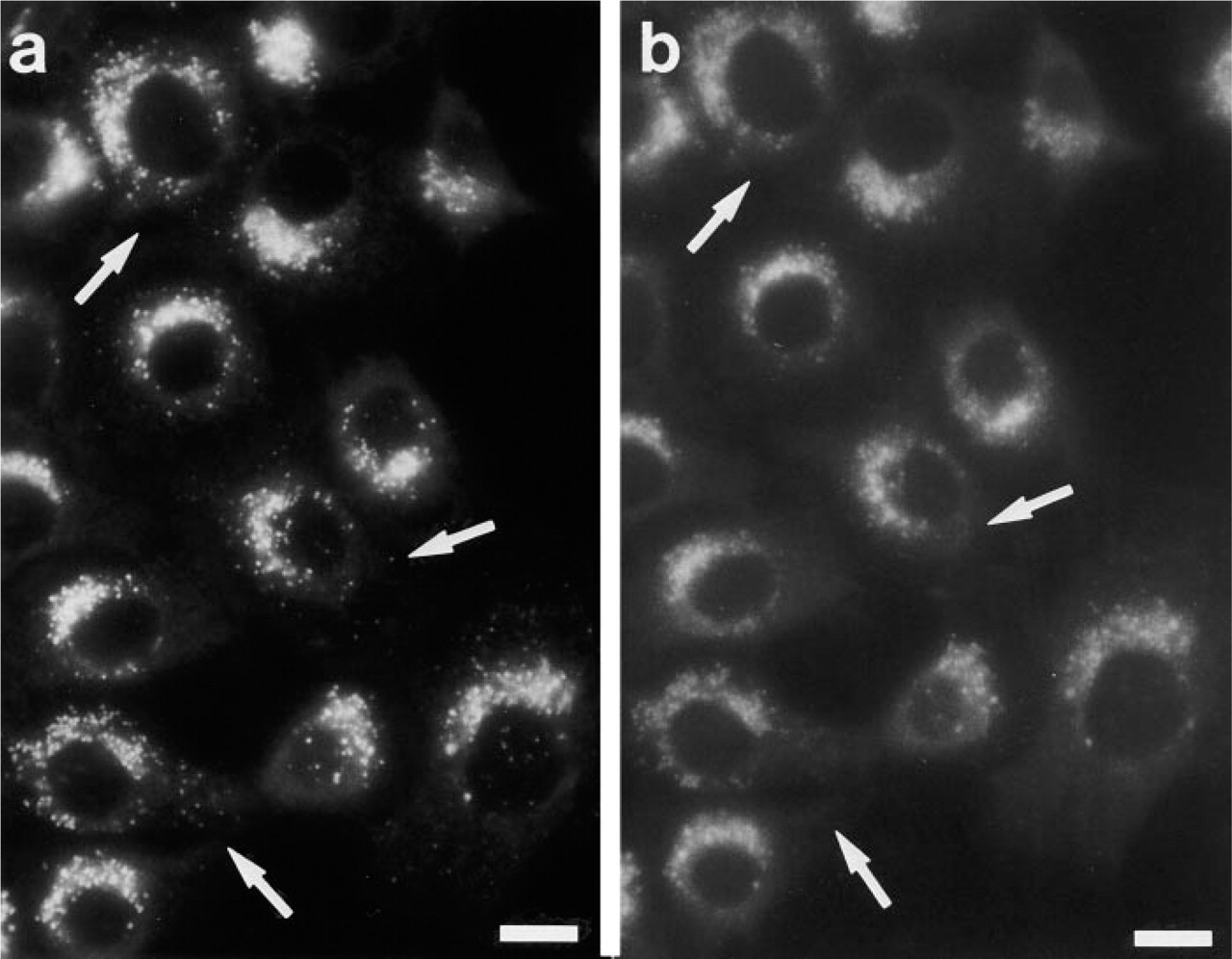
PaTu 8902 cells were stained with 2 μM MDC under in vivo conditions (
Results
The autofluorescent substance MDC specifically labels autophagic vacuoles (AVs) (Biederbick et al. 1995), based on two mechanisms (Niemann et al. 2000): ion trapping in acidic compartments mediated by the primary amino group, and changing of the fluorescent properties of MDC induced by membrane lipids that are highly concentrated within AVs. Therefore, staining properties should be different when cells stained in vivo are compared with cells stained after fixation, in which pH gradients are no longer present. Figure 1 shows that after fixation part of the MDC-positive structures observed under in vivo conditions disappeared, whereas others remained positive, indicating that MDC stains lysosomal/autophagic vacuoles of different qualities.
To analyze the contribution of the primary amino group to these staining properties of MDC, we substituted this amino group with a hydrogen atom (dansylamylamine; MDH), a hydroxyl group (dansylaminopentanol; MDOH), or a carboxylic acid group (dansylaminovaleric acid; MDA). Synthesis was performed as described in Materials and Methods. Purity was determined by TLC. Plates were analyzed using a LCD video camera under UV light (Figure 2). The purity of the dansylated pentamines was determined by comparing the RF on the plate of dansyl hydroxide (unreacted dansyl chloride) with the RF of the dansylated derivative. Whereas the MDH and MDOH reaction led to a 98% pure product, MDA showed 86% purity (see Figure 2).
Morphologically, MDH revealed in vivo fluorescence staining of AVs similar to that with MDC (Figures 3a and 3b). In contrast, MDOH exhibited only weak and blurred vacuolar staining, which disappeared quickly when cells were washed (Figure 3c). MDA did not show any staining of vacuolar structures (Figure 3d). Finally, as a control, we stained the cells for 1 hr with 0.1 mM dansyl hydroxide. Even at this high concentration, dansyl chloride showed only weak, blurred staining of the cytoplasm (data not shown). Therefore, an effect of the dansyl hydroxide contamination on the synthesized dansyl derivatives can be excluded. Because only MDH revealed a staining pattern comparable to MDC, we further concentrated on the comparison of MDC and MDH.
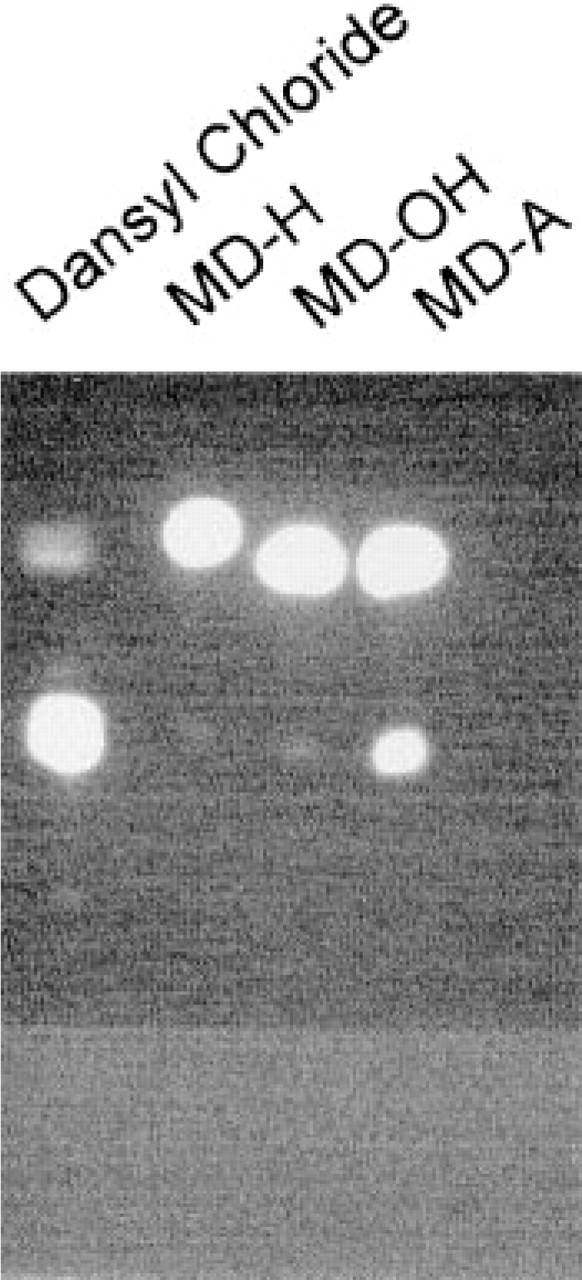
Thin-layer chromatography of synthesized and purified dansylamine (MDH), dansylaminopentanol (MDOH), and dansylaminovaleric acid (MDA). Only MDA contained a significant amount of free dansyl chloride (14%), whereas MDH and MDOH each revealed a purity of 98%, as quantitated with an LCD video camera under UV light.
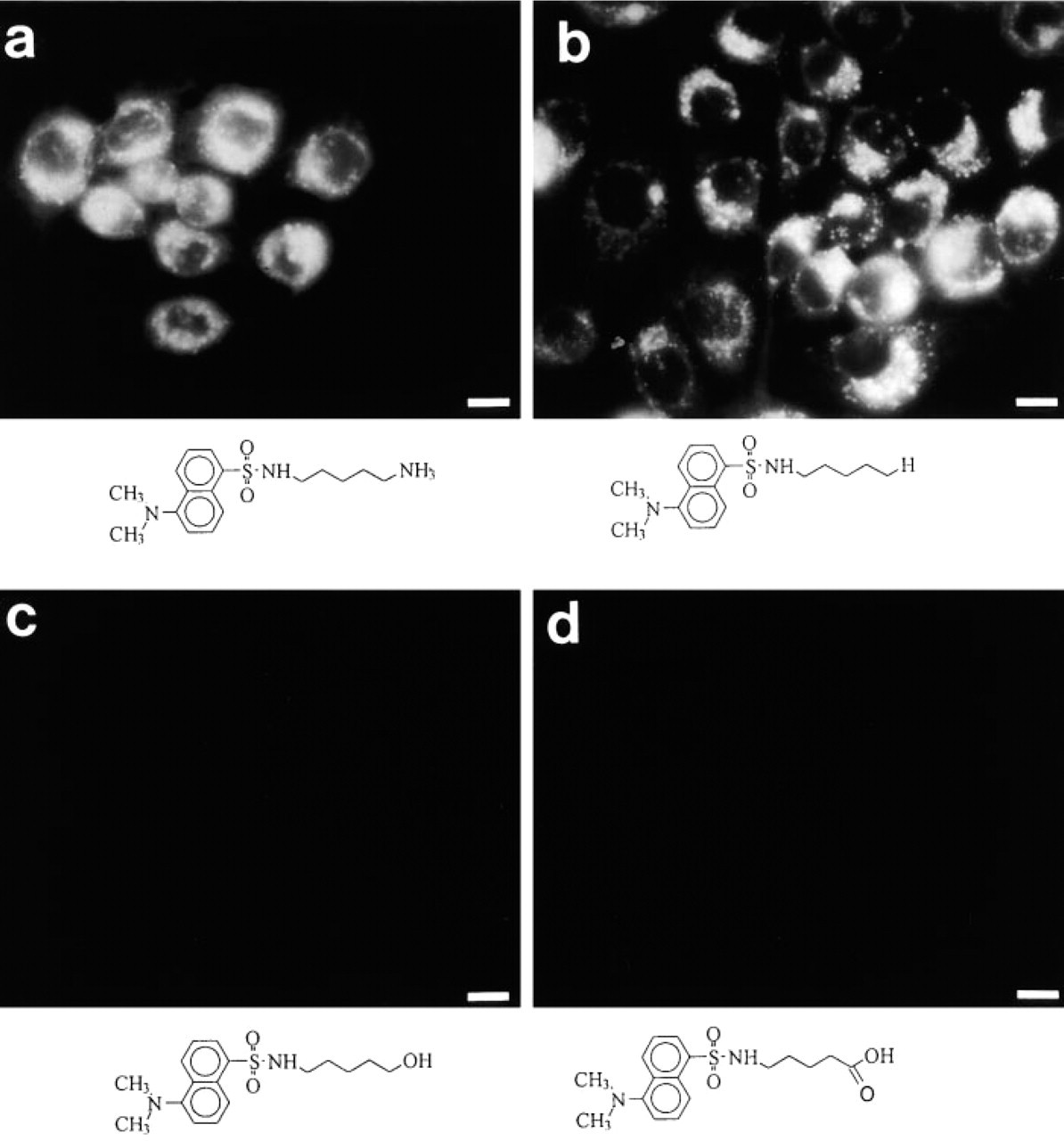
In vivo staining of PaTu 8902 cells with MDC (
To further confirm the interaction of MDH with AVs, we used isolated AVs from cells that had not been incubated with either MDC or MDH. As previously shown for MDC (Niemann et al. 2000), incubation of MDH with isolated AVs led to an increased quantum yield, expelling MDH as a solvent polarity probe. When MDOH or MDA was incubated with isolated vacuoles, no increase in RF could be observed (Figure 4). We conclude that the interaction of MDC and its structural homologues MDH, MDOH, and MDA with AVs, and hence with lipid bilayer structures, is dependent on the charged end group, with a positively or uncharged group favored over negatively charged groups.
In the next experiment, we examined the influence of the length of the MDH acyl side chain. We synthesized derivatives of dansylpentamine, changing the length of the polycarbon chain from C2 to C8. Although morphologically all derivatives showed a staining pattern of the same quality as MDC or MDH (equal to the C5 derivative), quantitation of the fluorochrome uptake revealed that the acyl chain length is crucial for uptake of the dansyl derivatives, with an acyl chain of n = 5 (MDH) being superior to all other derivatives (Figure 5).
To test the pH dependence of the accumulation of MDH within the cells, we measured the uptake of MDH in cells treated or untreated with ammonium chloride. As shown in Figure 6a, the disturbance of the lysosomal acidic pH had no effect on MDH uptake. This is in contrast to MDC uptake, which is inhibited by at least 30% with ammonium chloride. As a lysosomotropic agent, MDC itself is believed to affect the lysosomal pH in vivo in a dose-dependent manner. We used Acridine Orange as an alternative lysosomotropic agent and tested the effect of MDC and MDH on the Acridine Orange staining pattern. Cells incubated with 200 μM MDH for 1 hr showed the same Acridine Orange staining pattern as untreated cells (Figures 6b and 6d), whereas cells treated with MDC did not show any vacuolar Acridine Orange staining (Figure 6c). This indicates that the uptake of MDH into subcellular compartments does not affect the pH of these compartments and that it is independent of the lysosomal pH. Therefore, MDH did not exhibit the properties of a classical lysosomotropic agent.
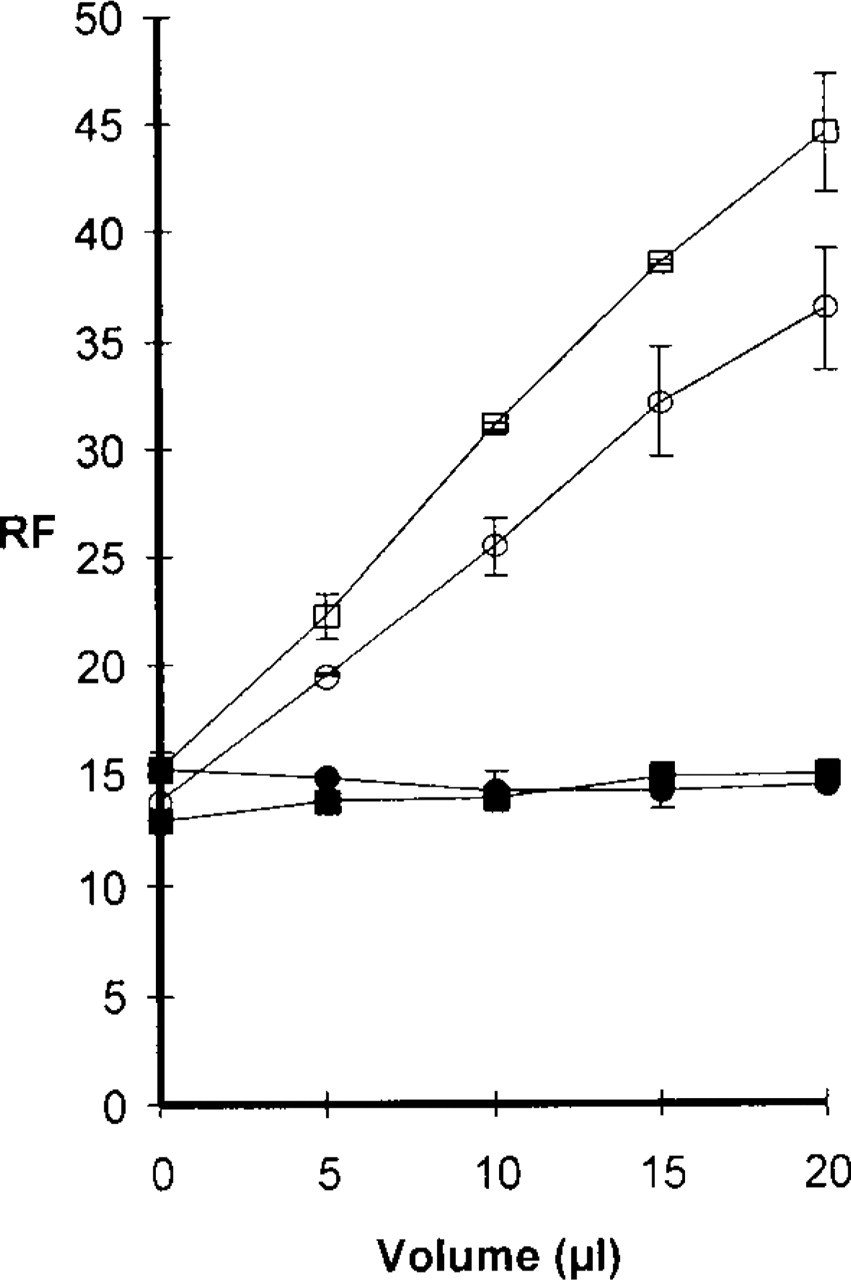
Increasing amounts of isolated autophagic vacuoles were incubated with a fixed concentration (5 μM) of either MDC (□), MDH (○), MDOH (▪), or MDA (•). Whereas MDC and MDH relative fluorescence (RF) increased with increasing amounts of autophagic vacuoles, MDOH and MDA fluorescence was not altered, indicating that the latter substances cannot interact efficiently with the lipids of the autophagic vacuoles.
Although MDH showed a comparable in vivo staining pattern compared with MDC (Figures 2a and 2b), the question remained whether MDH really stains the same compartments. To test this, cell fractionation was applied to PaTu 8902 cells that had been incubated for 1 hr with either 0.1 mM MDC or MDH. The pellets of the postnuclear supernatants were separated by centrifugation on continuous sucrose gradients (15–40% sucrose) as described elsewhere (Niemann et al. 2000). Figure 7 shows the RF of MDC and MDH dependent on the sucrose concentration. The RF distributions of MDC and MDH were the same, with a maximum between 19.5% and 24% sucrose, indicating that both substances accumulate in AVs. Furthermore, when cells were double stained with LysoTracker Red, exhibiting the identical staining properties as MDC, and with MDH, LysoTracker Red and MDH fluorescence co-localized in a certain subset of structures (Figure 8), indicating that MDH is associated with organelles that are labeled by MDC according to its property as a solvent polarity probe rather than as a lysosomotropic agent. This is further confirmed by the observation that, in contrast to MDC (Figure 1), there is no difference in the staining pattern of MDH when cells are compared before and after fixation (Figure 9).
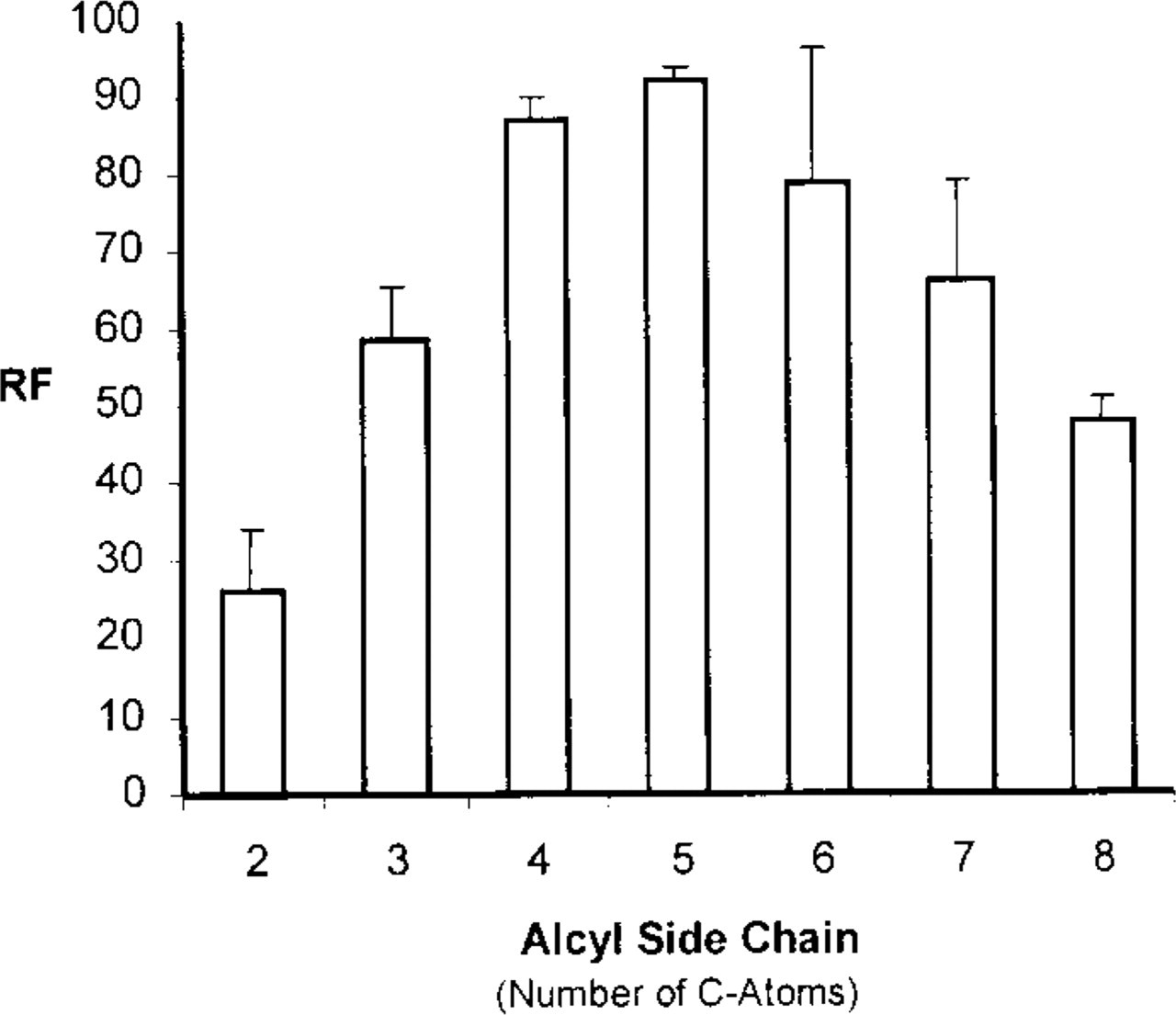
In vivo uptake of MDH derivatives differing in their alkyl side chain length. PaTu 8902 cells were incubated for 30 min with equimolar concentrations (100 μM) of the MDH derivatives indicated. Under this condition, the relative fluorescence per cell (RF) measured revealed a maximum with a side chain length of 5 C-atoms, representing MDH.
Discussion
Monodansylcadaverine (MDC) has been shown to be a useful autofluorescent marker for lysosomal/autophagic vacuoles in in vivo and in vitro applications (Biederbick et al. 1995, 1999). We recently showed in vivo that this property is based on two independent effects (Niemann et al. 2000), i.e., on ion trapping in acidic compartments via the amino group and on fluorescence light emission enhancement of the dansyl moiety in a hydrophobic environment, typically provided by AVs as lamellar membrane whirls (Papadopoulos and Pfeiffer 1987). The latter property makes MDC a solvent polarity probe (Niemann et al. 2000). Whereas under in vitro conditions only labeling of AVs or other lamellar bodies is achieved, in vivo application of MDC can lead to an enrichment of this lysosomotropic agent in other acidic compartments. This is shown in Figure 1, in which the same MDC-stained cells are documented before and after fixation. Only a subset of labeled vacuoles remained after fixation. Therefore, we synthesized the MDC analogue monodansylpentane (MDH) by dansylating 1-aminopentane, so that the amino group responsible for the ion trapping of MDC was substituted by a hydrogen atom, generating an uncharged C-5 alkyl side chain. In contrast to MDC, the same structures were labeled by MDH in vivo and in vitro (Figure 9), suggesting that only AVs were labeled. Evidence that MDC- and MDH-positive vacuoles are identical comes from a variety of experiments. The staining pattern of both fluorochromes was the same, exhibiting an array of perinuclear vacuoles (Figures 3a and 3b). Moreover, in double-staining experiments MDC was completely co-localized with the lysosomotropic agent LysoTracker, and all MDH-positive vacuoles were also positive for LysoTracker Red (Figures 8c and 8d). Furthermore, when cells labeled either with MDC or with MDH were used for cell fraction analysis, the fluorescence appeared in the same fractions (Figure 7). Finally, isolated AVs generated a similar enhancement of fluorescence light emission with MDC and MDH (Figure 4). Therefore, under in vivo conditions MDH is a more stringent marker for AVs than is MDC.
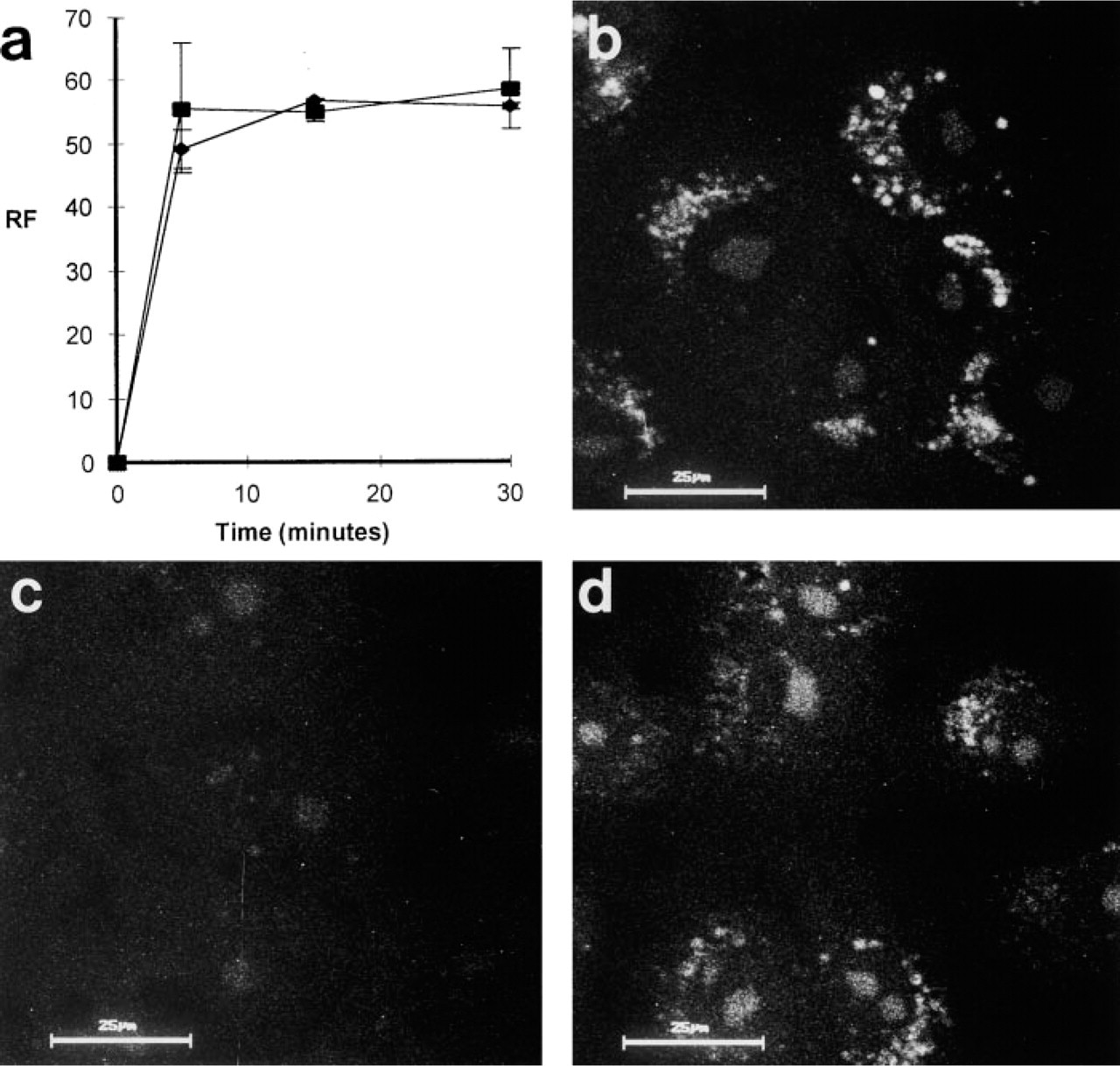
The accumulation of MDH within intracellular vacuoles is not pH dependent. (
As a classical lysosomotropic agent, MDC is a weak base. Like other weak bases, such as ammonium chloride or chloroquine, MDC is believed to influence the lysosomal pH when it enters this compartment. This is shown in the experiment of Figure 6, in which cells first stained with MDC were subsequently treated with the lysosomotropic substance Acridine Orange, which did not result in an accumulation of Acridine Orange in lysosomes in MDC-pretreated cells. The same effect is observed when cells are treated with inhibitors of the +H-V-ATPase (Muori et al. 1994) or with ammonium chloride (Niemann et al. 2000). In contrast, MDH does not interfere with the Acridine Orange staining, indicating that this substance does not alter the pH in the lysosomal compartment. Because a change of the pH in acidic compartments such as autophagolysosomes can interfere with the physiological function of these compartments (Seglen and Gordon 1980; Verhoef and Sharma 1983; Vandenbroucke–Grauls et al. 1984), MDH appears to be more suitable for the functional analysis of autophagy in living cells.
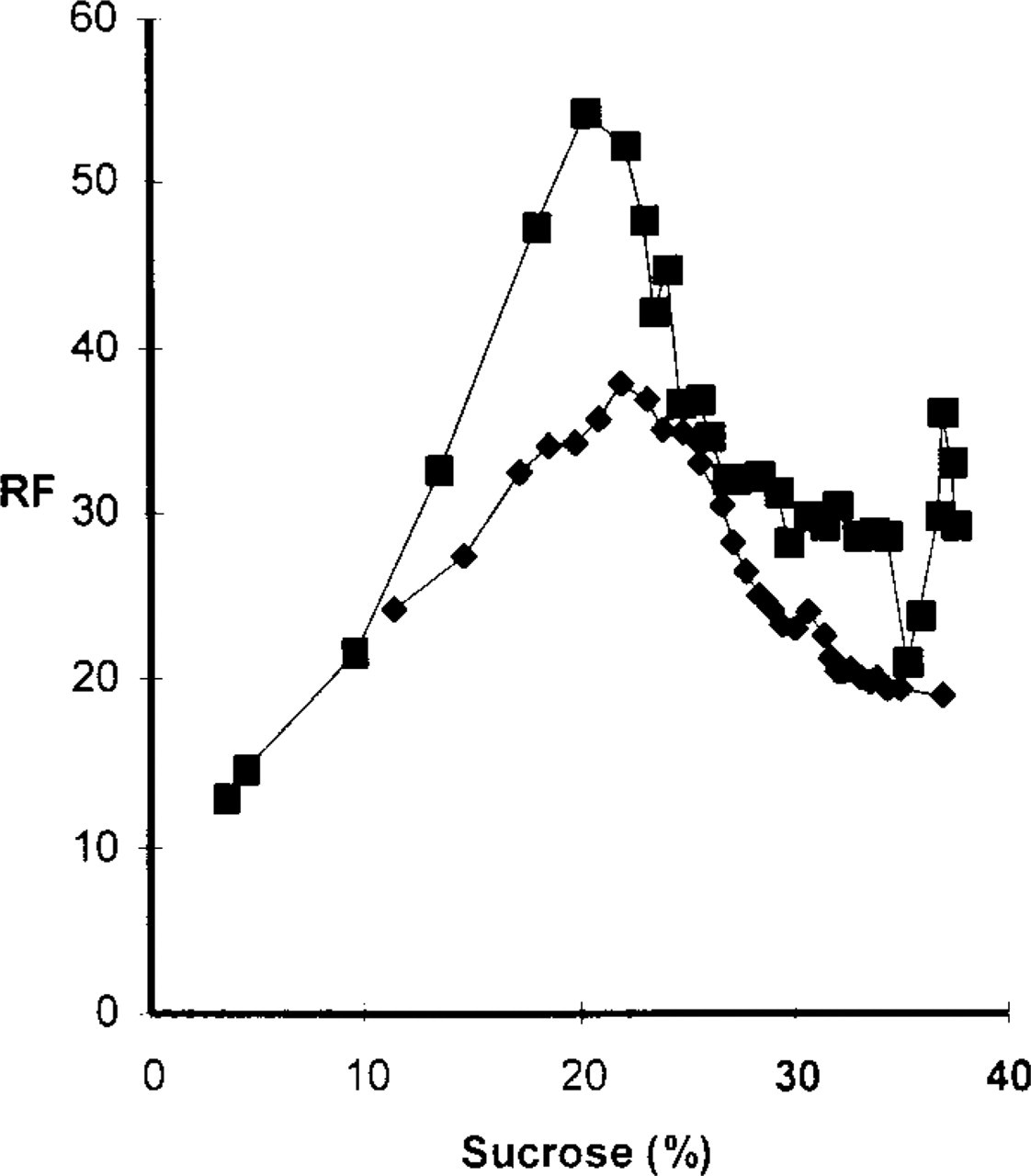
PaTu 8902 cells were stained in vivo with either MDC (▪) or MDH (♦) and subsequently used to isolate autophagic vacuoles. The MDC and MDH distribution in the sucrose gradient revealed a maximum between 19.5% and 24%, indicating that MDC and MDH are trapped in the same organelles.
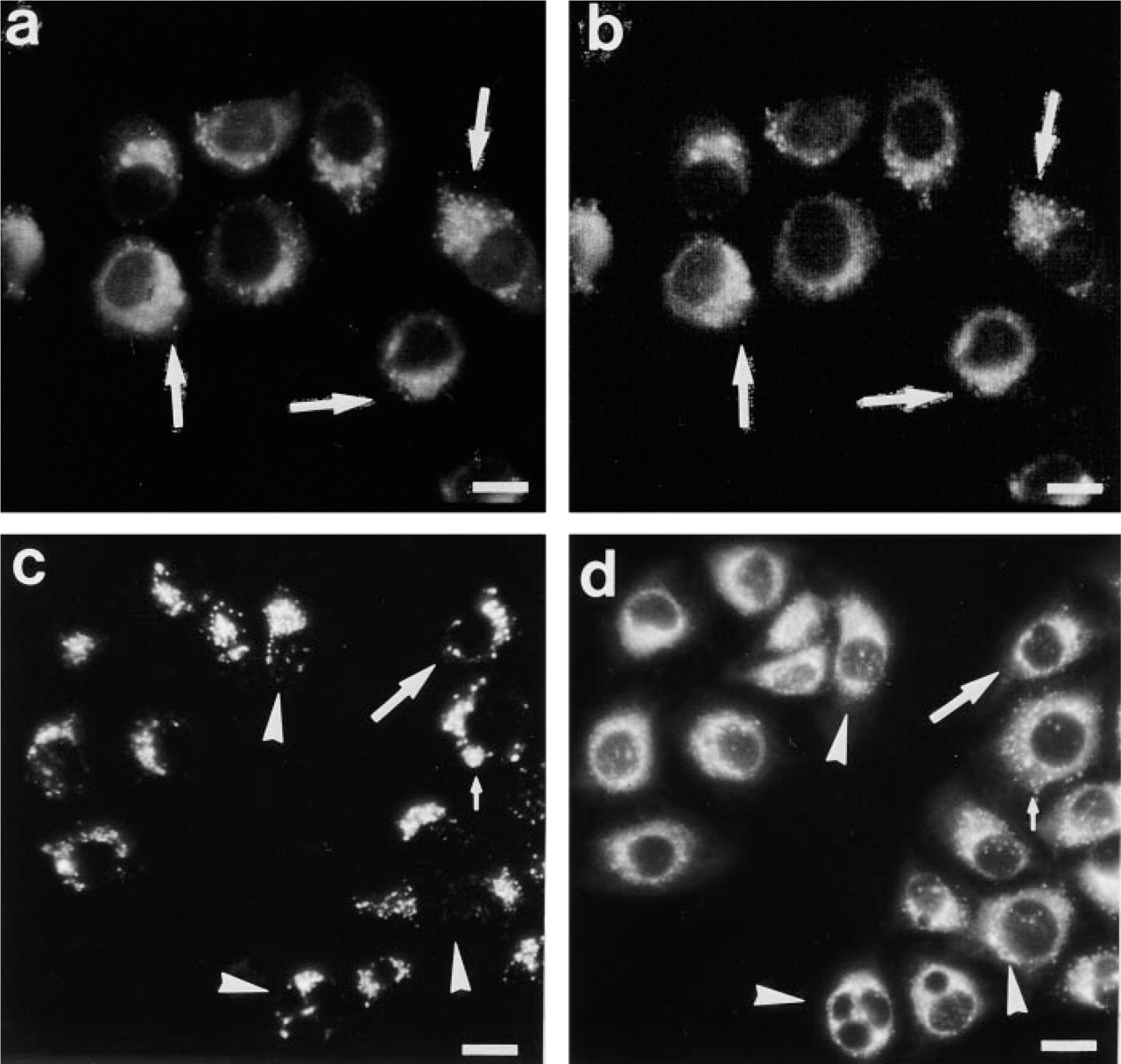
Staining of PaTu 8902 cells with LysoTracker and MDC (
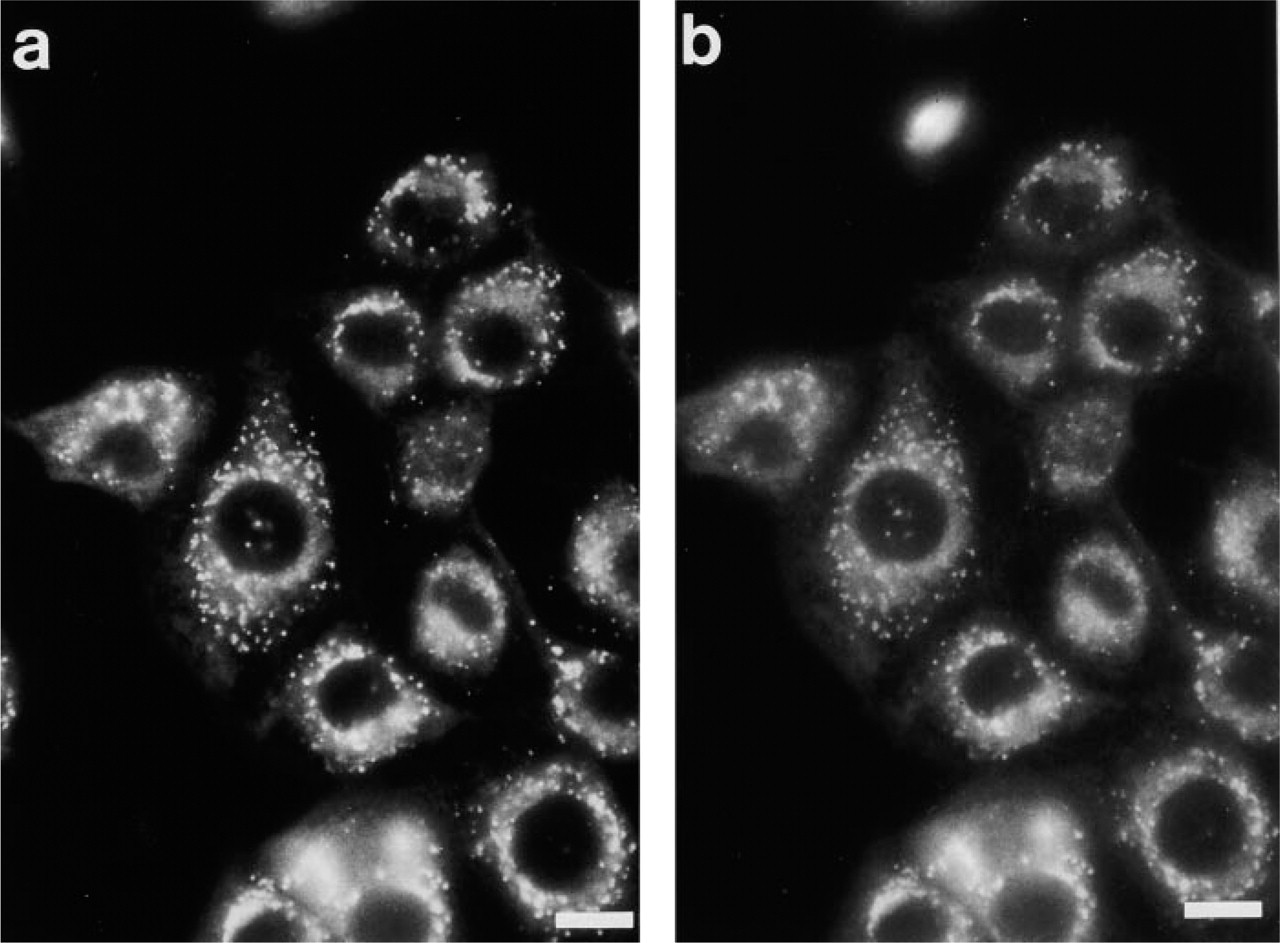
PaTu 8902 cells were stained with 2 μM MDH under in vivo conditions (
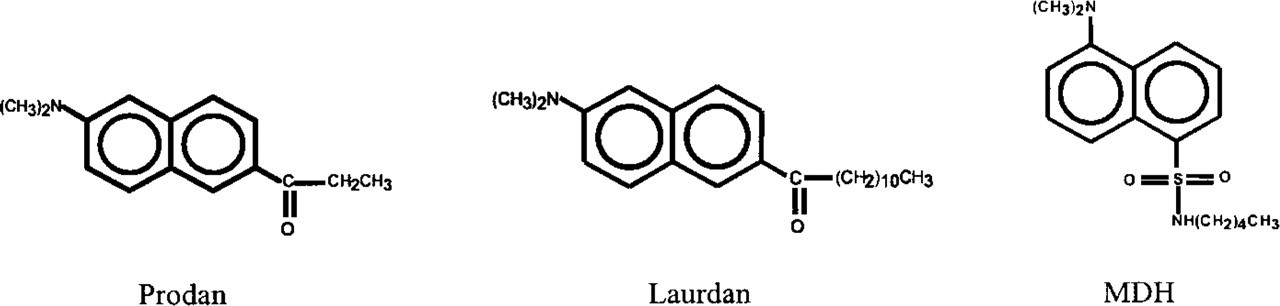
Chemical structures of Prodan, Laurdan, and monodansylpentane (MDH).
Although MDC and MDH stain the same subcellular structures when applied to fixed cells, there are some technical differences in their properties. When cells are again fixed with paraformaldehyde after MDC application, MDC is retained in the vacuolar structures even when longer staining protocols for immunohistochemistry follow. Under the same conditions, MDH fades, so that the background increases over time and the vacuolar structures become hazy. However, in freshly stained fixed cells MDH staining is more brilliant because MDC cause background by faint cytoplasmic staining, which does not occur with MDH. Nevertheless, MDH must be used with PBS as mounting medium, because with other mounting media, such as Mowiol, MDH is partly extracted from the stained structures, which also leads to increased background and decreased contrast. This has not been observed with MDC, most likely because MDC is immobilized by aldehyde fixation via the amino group. Under in vivo conditions, the staining with MDH is much more rapid than that observed with MDC. Whereas after a 10-min exposure to MDH cells are saturated with the fluorochrome (Figure 6a), MDC takes more than 45 min to reach the same level of saturation (Biederbick et al. 1995; Niemann et al. 2000).
Our extended experiments with a variety of MDC derivatives revealed that both the charged end group and the alkyl side chain length are important for the interaction of these molecules with biological membranes (Figures 3–5). Interestingly, none of the compounds used led to fluorescent staining of single lipid bilayers, such as the plasma membrane or other endomembranes. In multilamellelar bodies such as AVs, many membrane bilayers are stacked with only a small, if any, amount of aqueous phase between them. Therefore, many fluorochrome molecules are tightly packed, emitting light that combines to yield a strong signal. The amount of fluorochrome molecules in a single membrane bilayer might be in a range at which they are not detectable by the fluorescence microscopy methods applied in this study.
Fluorescent probes that are structurally similar to MDH have been described previously. Two of them, Prodan and Laurdan (Figure 10), have been extensively used to study biophysical aspects of biological membranes. Whereas the alkyl side chain of MDH is linked to the dansyl moiety via a sulfonamide group, Prodan and Laurdan possess a carbonyl group to bridge the gap between the alkyl side chain and the fluorescent group. Furthermore, Prodan has a C2 side chain length, while Laurdan has a C11 side chain length (Figure 10). On the basis of the experiment shown in Figure 5, we assume that the association of Prodan and Laurdan with intracellular membranes of living cells is not very efficient. However, both probes have been used in vitro, either as single compounds or in combination with each other, predominantly to investigate different lipid phases in biological membranes, applying the method of generalized polarization (Narayanan and Balaram 1976; Parasassi et al. 1991). In addition, aging of cell membranes in cultured cells has been investigated using Laurdan (Levi et al. 1997). To our knowledge, staining of lysosomal or related compartments with Prodan or Laurdan has not been described.
Footnotes
Acknowledgments
Supported by Studienstiftung des Deutschen Volkes and by P. E. Kempkes Stiftung Marburg.
We thank Ursula Lehr for expert technical assistance, Volkwin Kramer for preparation of the microscopic reprints, and Annette Biederbick for critical comments and helpful discussions.
