Abstract
The periodontal ligament is the connective tissue that fills the space between the tooth and its bony socket. It is abundantly innervated by the sensory and sympathetic nerves. We first investigated the immunoreactivity of TrkA, which is a high-affinity receptor of nerve growth factor (NGF), in the periodontal ligament of rats. Immunoreactivity was observed at the epithelial cells in the cervical and furcation regions of the molars. These epithelial cells, which gather together to form clusters or networks, are known as the epithelial rests of Malassez. Immunoreactivity was not observed in other non-neuronal cells, such as osteoblasts, fibroblasts, odontoblasts, cementoblasts, endothelial cells, and/or osteoclasts. On the basis of these findings, we investigated the possible involvement of sensory nerve innervation in the immunoreactivity of the epithelial cells. Denervation of the inferior alveolar nerve resulted in a marked decrease in the distribution area and size of the clusters of immunoreactive cells compared with those of sham-operated rats. These findings suggest that sensory nerve innervation may have a regulatory role in maintenance of the epithelial rests of Malassez expressing TrkA in the periodontal ligament.
T
Nerve growth factor (NGF) is essential in promoting the development, maintenance, and survival of sensory and sympathetic neurons (Levi–Montalcini 1987). NGF acts on responsive cells through specific membrane-bound proteins that serve as low- and high- affinity receptors (Barbacid 1994; Chao and Hempstead 1995). The high-affinity binding requires the presence of the trk family of proto-oncogenes, and the trkA product is a functioning receptor for NGF (Kaplan et al. 1991). TrkA is expressed in various non-neuronal tissues (Shibayama and Koizumi 1996), such as vascular smooth muscle cells (Nemoto et al. 1998), epithelial subcapsular and medullary cells in the human thymus (Parrens et al. 1998), dendritic cells and blood vessel walls in human lymph nodes (Garcia et al. 1997), and the basal layer of the epidermis in human skin (Lopez et al. 1998). However, TrkA immunoreactivity has not been investigated in the periodontal ligament. In this study we first investigated the immunoreactivity of TrkA in the periodontal ligament of the rat. Immunoreactivity was found at the epithelial rests of Malassez in the cervical and furcation regions of the molars.
Previous ultrastructural observation revealed an intimate relationship between the sensory nerve endings and the basal lamina of the epithelial rests of Malassez (Heyeraas et al. 1993; Lambrichts et al. 1993). The periodontal ligament is abundantly innervated by sensory nerves (Kato et al. 1990; Fristad, 1997). However, the possible interaction between the sensory nerve innervation and the epithelial rests of Malassez has not been elucidated. Therefore, we also investigated the possible influence of sensory nerve innervation on the distribution pattern of the immunoreactive epithelial cells by denervation of the inferior alveolar nerve in rats.
Materials and Methods
Tissues
Twenty 7-week-old male Wistar rats were used. The animals were maintained on a normal solid food diet and water given ad libitum. The rats were deeply anesthetized with sodium pentobarbital and transcardiacally perfused with 0.9% saline, followed by 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4. The mandibular alveolar bones were immediately excised, postfixed for 1 day in the same fixative as was used for perfusion, and decalcified in 10% ethylendiaminetetra-acetate (EDTA) solution for 14 days. Sagittal and horizontal sections 50 μm thick were cut on a cryostat (Sakura; Tokyo, Japan). Horizontal sections of 7 μm were also cut on a microtome (Leica; Oberkochen, Germany) after embedment in paraffin.
Immunohistochemistry for TrkA
Mandibles from 10 rats were used in this study. Serial sagittal and horizontal sections 50 μm thick were treated in the free-floating state through the labeling step. Serial horizontal sections of 7 μm were cut on a microtome after the decalcified bone had been embedded in paraffin. The sections were incubated with rabbit anti-TrkA antibody (sc-118; Santa Cruz Biotechnology, Santa Cruz, CA; 1:8000) at 4C for 48 hr. Antigen–antibody complexes were detected by the avidin–biotin–peroxidase (ABC) method (Vectastain ABC kit; Vector, Burlingame, CA). The specificity of the TrkA antibody (sc-118) had been previously proved (Burette et al. 1997). Visualization of the reaction product was done by immersion of the sections in diaminobenzidine (DAB; Dojindo, Tokyo, Japan) plus 0.015% H2O2. The 7-μm sections were counterstained with methyl green.
Nerve Denervation
Ten rats were used for the denervation study. Denervated rats were prepared by inferior alveolar nerve transection (Wakisaka et al. 1985). The rats were anesthetized with sodium pentobarbital (5 mg/kg bw, IP). The masseter muscle was dissected after a vertical skin incision to expose the bone surface at the mandible. Then the cortical bone overlying the inferior alveolar nerve was drilled and carefully removed, and the right inferior alveolar nerve was elevated with a blunt needle, ligated with a suture, and transected. The left inferior alveolar nerve was also exposed, but not transected, as a sham operation (Sham). After inferior alveolar nerve transection, animals were maintained on a normal solid food diet and water given at libitum. An interval of 1 or 3 weeks was allowed for nerve degeneration. Thereafter, the rats were deeply anesthetized with sodium pentobarbital and the mandibular alveolar bones were immediately excised. Transected nerve endings were confirmed in all the denervated rats when they were sacrificed. The number of experimental rats in each group was five, and sham-treated specimens were obtained from the contralateral side of denervated rats. The experiment was approved by the Animal Committee of the dental school.
Results
Immunohistochemistry for TrkA
In the 50-μm sections, immunoreactivity indicating TrkA was observed only in the epithelial rests of Malassez in the cervical and furcation regions of the molars. No immunoreactivity was observed in the nerve fibers (Figure 1).
In the 7-μm sections, strongly positive TrkA immunoreactivity was observed at the cell surface of epithelial rests of Malassez in the periodontal ligament (Figure 2). The rests were localized close to the root surface. Immunoreactivity was not observed in other non-neuronal cells, such as osteoblasts, fibroblasts, odontblasts, cementoblasts, endothelial cells, and osteoclasts, or in neural tissues. These numerous epithelial cells showed oval or round cluster forms, and some cells showed a strand form (Figure 2B and 2C). They were localized near the root surface rather than near the alveolar bone.
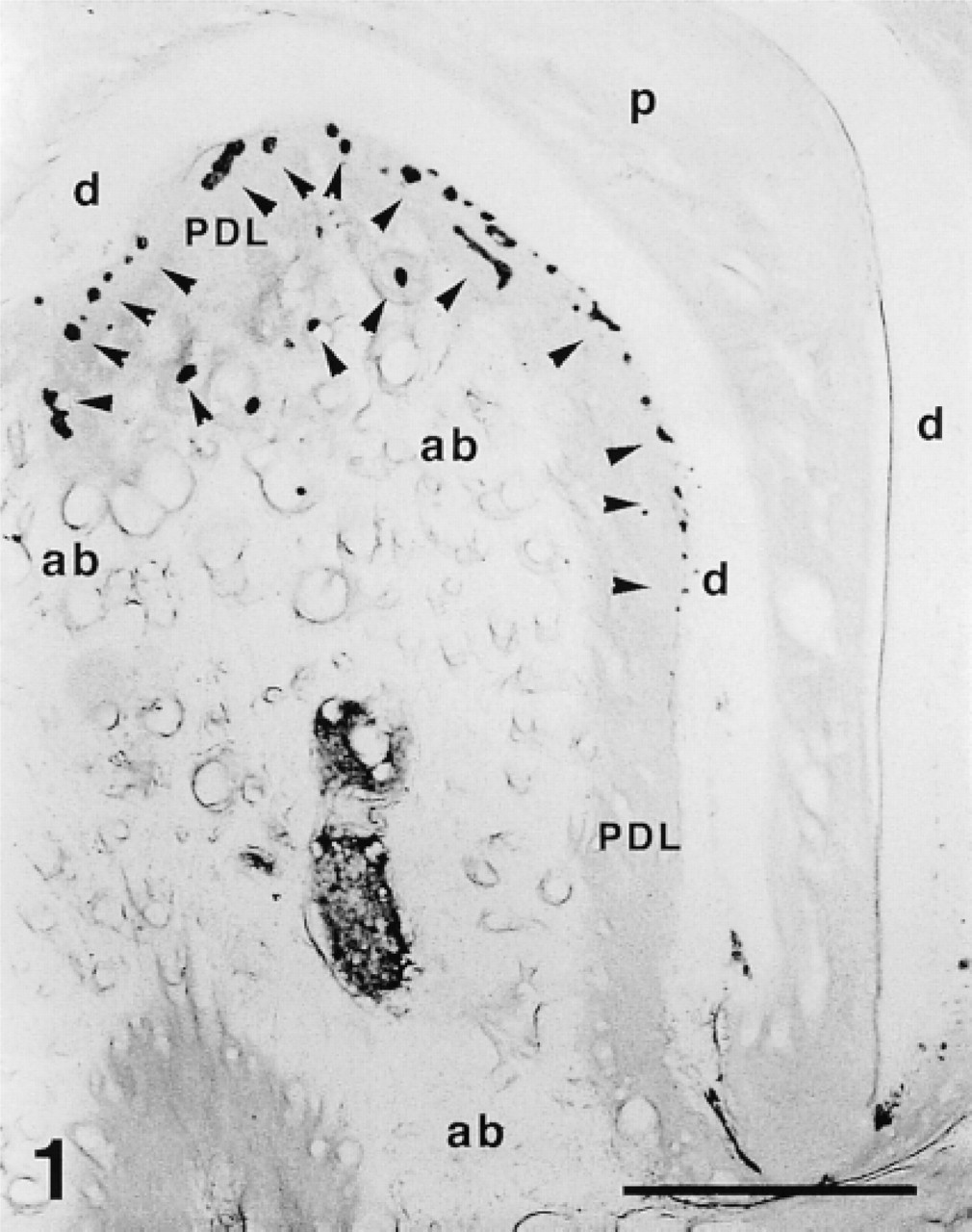
Immunoreactive expressions of TrkA in the periodontal ligament in 50-μm sagittal sections. Immunoreactivity was observed in the epithelial rests of Malassez located in the furcation region. Arrowheads indicate immunoreactive cells. PDL, periodontal ligament; p, pulp; d, dentine; ab, alveolar bone. Bar = 500 μm.
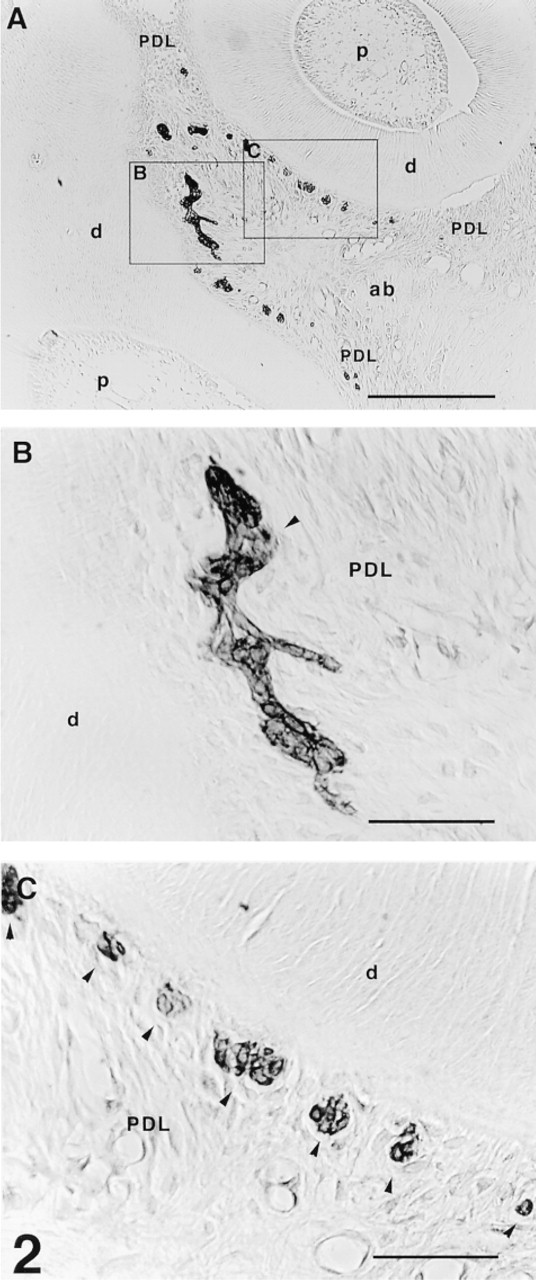
Immunoreactive expression of TrkA in the periodontal ligament in thin horizontal sections (7 μm). (
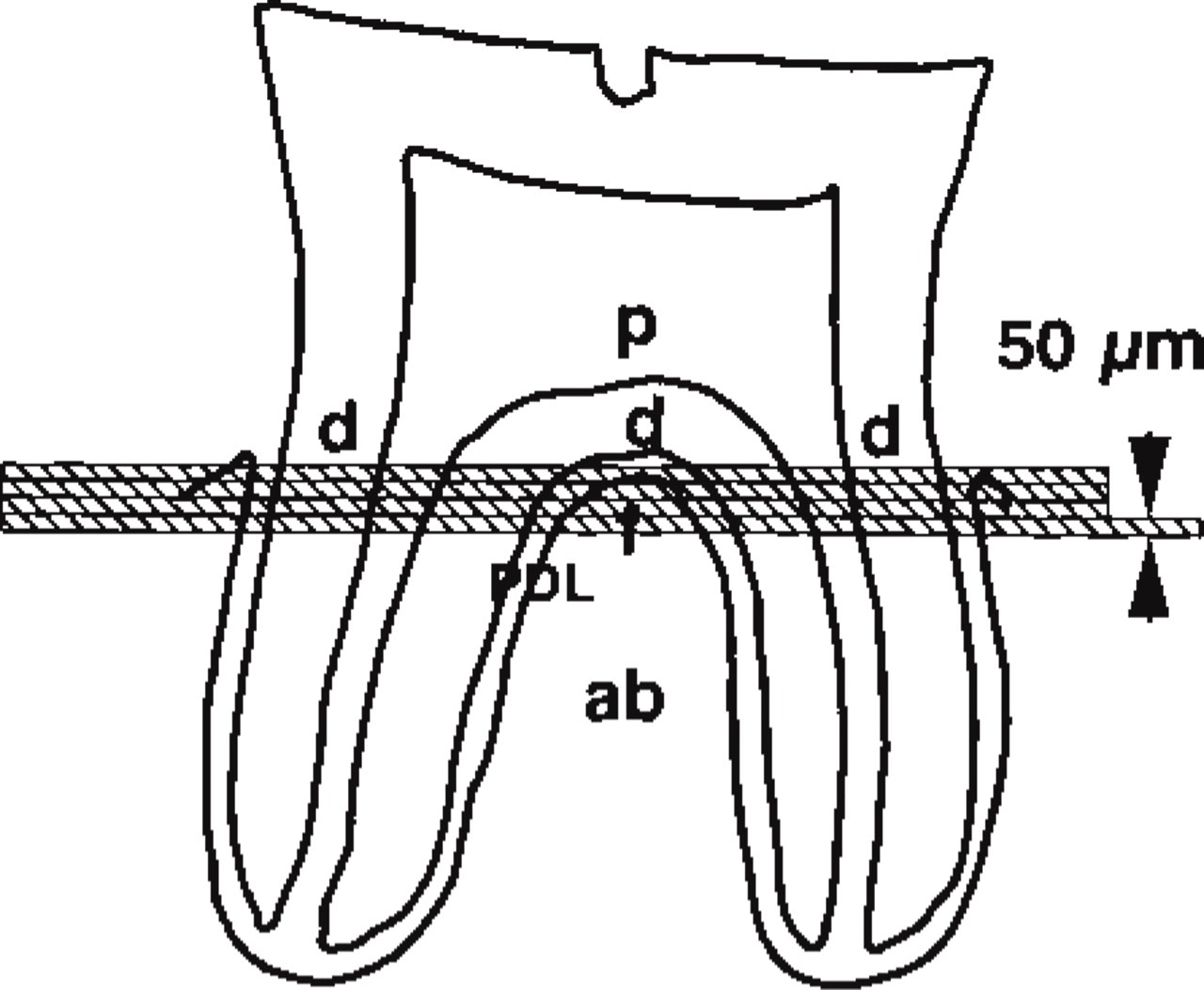
Diagram of a sagittal section of a lower second molar. Serial horizontal sections of 50 μm were obtained near the furcation of the molars. ab, alveolar bone; PDL, periodontal ligament; d, dentine; p, pulp.
Influence of Denervation on Distribution of Immunoreactivity
To evaluate the possible association between sensory nerve denervation and expression of TrkA in epithelial rests of Malassez, we compared the distribution pattern of the immunoreactive cells in the furcation region of lower second molars between the specimens from sham-operated and denervated rats on 50-μm serial sections. Figure 3 shows a diagram of the location of the serial sections shown in Figure 4.
In the sham-operated rats (Figures 4A1–4A4), many TrkA-immunoreactive epithelial rests of Malassez were observed around the furcation regions of the teeth. They were arranged in strands or as networks of strands. In particular, epithelial cells were densely localized near the furcation (Figures 4A2 and 4A3).
In denervated rats, immunoreactivity was also observed in the epithelial rests of Malassez (Figures 4B1–4B4 and 4C1–4C4). However, 1 week after the denervation of inferior alveolar nerve, the size of the immunoreactive cell clusters around all the lower molars was smaller and their distribution area was narrower than those of sham-operated rats (Figures 4B1–4B4 and 5B). These tendencies were more prominent after 3 weeks, and immunoreactive epithelial cells became sparse (Figures 4C1–4C4 and 5C). These changes associated with denervation of the inferior alveolar nerve were observed in all the animals, i.e., 1-week-denervated (n = 5) and 3-week-denervated (n = 5) rats. The adjacent gingival epithelium showed intense TrkA immunoreactivity which was evaluated as a positive control for the immunoreactivity in the denervated specimens (not shown).
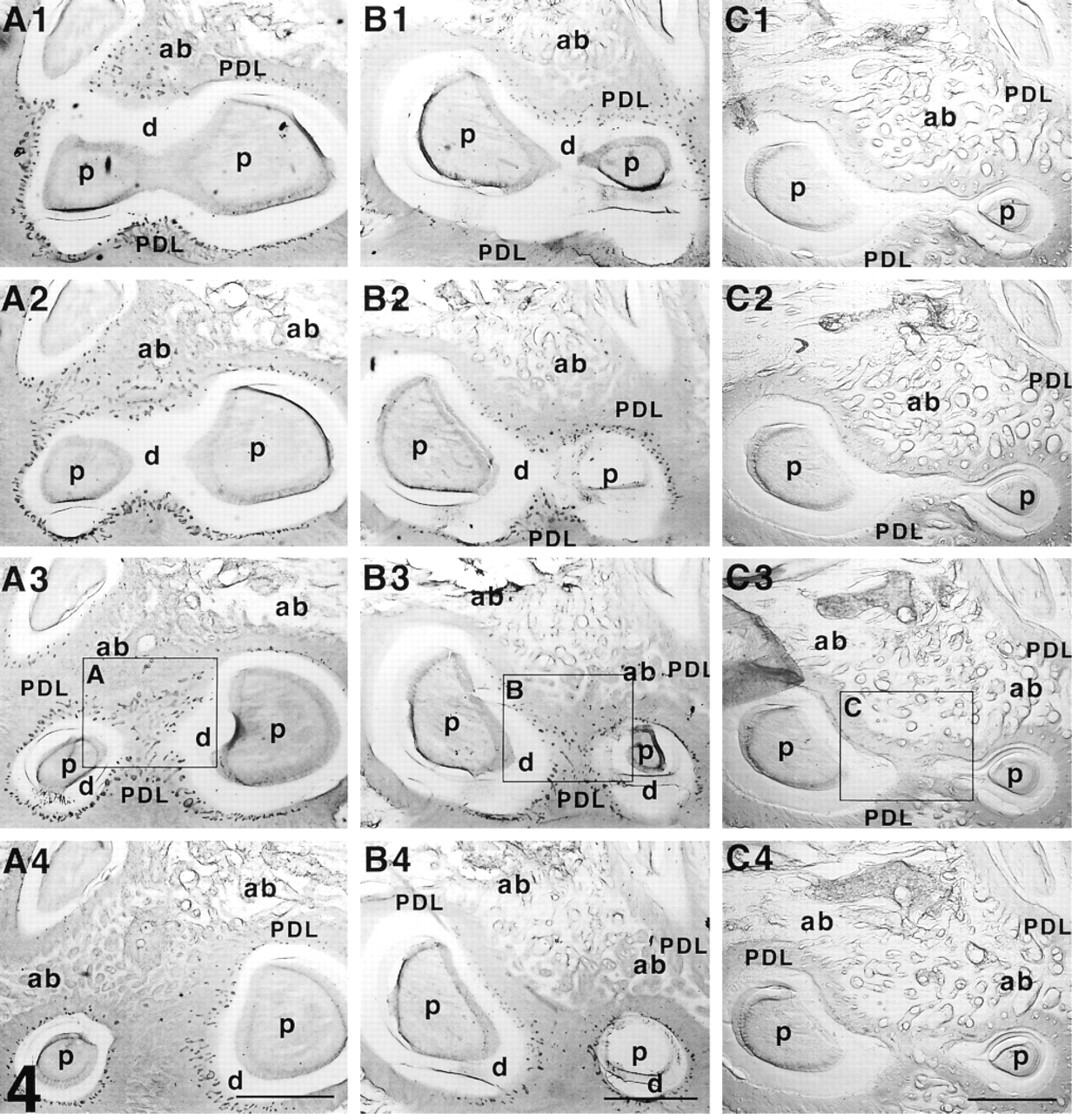
Serial horizontal 50-μm sections around the furcation areas in sham-operated (
Discussion
We found that TrkA immunoreactivity was observed in the epithelial rests of Malassez. They were situated close to the root surface in the horizontal sections and were localized in the cervical and furcation regions of the molars in the rat periodontal ligament in sagittal sections. These findings are in agreement with previous investigations on the distribution pattern of epithelial rests of Malassez (Wesselink and Beertsen 1993). TrkA is expressed in other non-neuronal tissues (Shibayama and Koizumi 1996), such as vascular smooth muscle cells (Nemoto et al. 1998), epithelial subcapsular and medullary cells in the human thymus (Parrens et al. 1998), and dendric cells and blood vessels in human lymph nodes (Garcia et al. 1997), and in the basal layer of the epidermis in human skin (Lopez et al. 1998). Because Trka immunoreactivity was not observed in other non-neuronal cells in the periodontal ligament, TrkA might be used as a marker for the epithelial rests of Malassez in the periodontal ligament.
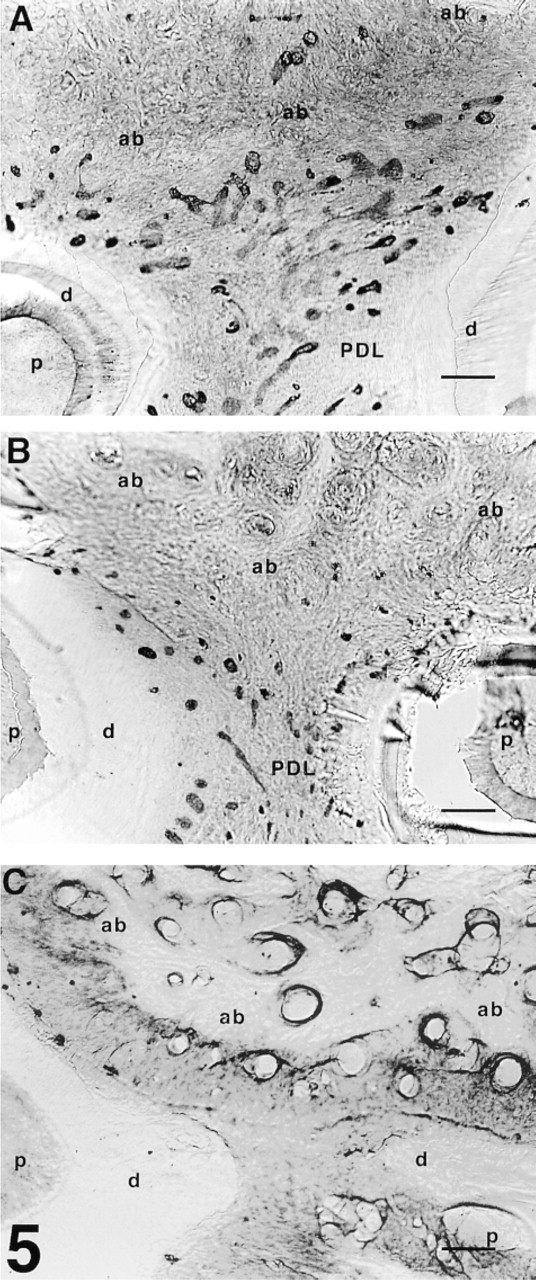
Immunoreactivity in the sham-operated rats (
TrkA is a membrane-bound protein and mediates the functional responses to NGF through activation of tyrosine kinase (Kaplan et al. 1991; Barbacid 1994; Chao and Hempstead 1995). In the present study, Trk A immunoreactivity was localized at the cell surfaces in the epithelial rests of Malassez, and several of these cells were gathered together to form islands or clusters. Electron microscopic observation revealed that these epithelial cells were joined to each other by desmosomes and gap junctions (Lambrichts et al. 1993), which are intercellular channels formed by membrane-spanning proteins that permit exchange of small molecules including second-messenger molecules and ionic species (Christ et al. 1994).
As seen in thin 7-μm sections, epithelial rests of Malassez showed islands of cluster forms, and the distribution pattern of these epithelial cells was in agreement with that found in a previous investigation (Gao et al. 1988). However, observation of the thick horizontal serial sections revealed that the epithelial rests formed a network in the furcation regions rather than being distributed in a scattered pattern. Thus, immunohistological observation of the thick horizontal serial sections has for the first time enabled three-dimensional evaluation of the distribution pattern of the epithelial rests of Malassez expressing TrkA.
The furcation regions of molars are regions at which the alveolar bone and periodontal ligament are remodeled actively, unlike any other regions around the tooth (Vignery and Baron 1980). The periodontal ligament undergoes degeneration and regeneration, and the alveolar bone is remodeled by bone resorption and formation in the growing rats as the result of physiological tooth movement (Vignery and Baron 1980; Takano–Yamamoto et al. 1994). However, there have previously been no investigations into the relationship between these epithelial cells and the remodeling of alveolar bone and/or periodontal ligament. On the other hand, the periodontal ligament is abundantly innervated by sensory nerves, especially in the apical lower third of the ligament (Heyeraas et al. 1993). In the furcation and cervical regions, CGRP-and substance P-immunoreactive fibers were observed only sparsely (Heyeraas et al. 1993). Although epithelial rests of Malassez expressing TrkA are non-neuronal tissues, they have an intimate relationship with peripheral nerve endings, as shown by electron microscopic observation (Lambrichts et al. 1993). On the basis of our present findings, the physiological role of the epithelial rests of Malassez could not be identified. However, they might have a role(s) in alveolar bone remodeling in association with the peripheral nerves in the periodontal ligament.
In the present study, it is noteworthy that inferior alveolar nerve transection caused remarkable changes in the pattern of distribution of TrkA-immunoreactive epithelial cells. Denervation decreased the number of the clusters or islands within 1 week, and they were further decreased in 3 weeks. Moreover, the size of the clusters or islands was also decreased. Transection of the sensory fibers leads to the degeneration of their peripheral portions, with separation from the neurons (Wakisaka et al. 1985). In our study, the edge of the transected nerve was tightly ligated to prevent reinnervation, and the transected nerve endings were confirmed in all the denervated rats when they were sacrificed. Previous ultrastructural findings showed a close association between Ruffini-like and free nerve endings and the basal lamina of the epithelial cell rests (Lambrichts et al. 1993). These findings and ours indicate that sensory nerve innervation may have a regulatory role in maintaining the epithelial rests of Malassez expressing TrkA in the periodontal ligament.
Footnotes
Acknowledgements
Acknowledgments
Supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Science and Culture of Japan (10470446, 10557197, and 11771318).
